Abstract
In the initial assessment of the patient with acute vertigo or dizziness, both structured history-taking and a targeted bedside neuro-otological examination are essential for distinguishing potentially life-threatening central vestibular causes from those of benign, self-limited peripheral labyrinthine origin and thus for deciding on further diagnostic testing. In this article, the key elements of the vestibular and ocular motor examination, which should be obtained at the bedside in these acutely dizzy patients, will be discussed. Specifically, this will include the following five domains: ocular stability for (I) nystagmus and for (II) eye position (skew deviation), (III) the head-impulse test (HIT), (IV) postural stability, and (V) ocular motor deficits of saccades, smooth pursuit eye movements, and optokinetic nystagmus. We will also discuss the diagnostic accuracy of specific combinations of these bedside tests (i.e. HIT, testing for nystagmus and vertical divergence, referred to as the H.I.N.T.S. three-step examination), emphasizing that the targeted neuro-otological bedside examination is more sensitive for identifying central causes in acute prolonged vertigo and dizziness than early MRI of the brain.
Introduction
Acute vertigo or dizziness is one of the most frequent single causes for patients to present to the emergency department (ED), representing between 2.1% and 4.4% of all consultations 1 –4 and causing annual costs of about 9 billion dollars. 5,6 The differential diagnosis of acute vertigo and dizziness is broad and extends over many different specialties, increasing the risk of both inappropriate diagnostic testing and missed diagnoses. No single diagnosis accounts for more than 5–10% of all cases and more than 15% of all patients presenting to the ED with vertigo or dizziness suffer from a dangerous cause. 1 In this setting, the initial clinical assessment (including systematic history-taking and a targeted bedside examination) is the key to narrowing down the differential diagnosis and to deciding on further diagnostic testing and treatment. 7
In this article, the key elements of the vestibular and ocular motor exam and the correct interpretation of deficits in the acutely dizzy patient will be described. Thus, its aim is to provide tools to the clinician that facilitates the distinction between acute central and peripheral vestibular causes and non-vestibular causes of vertigo and dizziness at the bedside. This is especially true for acute prolonged vertigo or dizziness in association with nausea/vomiting, gait imbalance, motion intolerance, and nystagmus (i.e. meeting the diagnostic criteria for an acute vestibular syndrome according to the International Classification of Diseases 11th Revision. 8 ) Here the distinction between dangerous, potentially life-threatening central causes such as vertebrobasilar stroke on the one hand and benign, self-limiting peripheral causes such as acute peripheral vestibulopathy on the other hand is essential. Thus, based on the clinical examination skills demonstrated here, the clinician should be able to decide which patients require an expedited diagnostic workup including neuroimaging and auxiliary vestibular testing and which patients should be monitored on a stroke unit.
For the bedside examination of vestibular and ocular motor functions, the following five domains should be assessed: ocular stability for (I) nystagmus and (II) skew deviation, (III) the HIT, (IV) postural stability, and (V) ocular motor deficits. 9,10
Testing for possible benign paroxysmal positional vertigo has been discussed in a recent publication in this journal by Straumann and Brandt. 11
Ocular stability—identifying spontaneous or GEN
Normally the eyes are kept stable in the desired position. This is true for looking straight-ahead and eccentric gaze. However, damage to the peripheral or central structures of the vestibular or ocular motor system may result in eye drift (“slow phase”), usually interrupted by compensatory saccadic eye movements (“fast phase”), bringing the eyes back to the desired position. When evaluating such spontaneous jerk nystagmus (i.e. nystagmus elicited while looking straight-ahead), several elements should be assessed, including the main beating direction of the nystagmus (horizontal vs. vertical vs. torsional vs. diagonal), the effect of fixation and its suppression (typically a peripheral-type nystagmus can be suppressed or diminished on fixation—thus is increased on fixation suppression—while lacking visual fixation suppression favors a central cause) and its modulation depending on eye position (i.e. whether it follows Alexander’s law—see further below). While predominantly horizontal spontaneous nystagmus (fast nystagmus phase beating away from the lesioned side) is common in both peripheral and central vestibular disorders, pure vertical or pure torsional spontaneous nystagmus suggests a central origin. Removal of visual fixation can be achieved either by the use of Frenzel’s goggles or during ophthalmoscopy if the fixating eye is transiently covered. Alternatively, if no Frenzel’s goggles and ophthalmoscope are available, this can be achieved also by the penlight-cover test, that is, by shining a penlight in one eye while intermittently occluding the other eye. 12
In the next step, the effect of gaze on the nystagmus should be assessed. Varying nystagmus intensity (being higher when looking into the direction of the quick phases, i.e. away from the lesioned side) but unchanged beating direction of the fast phase is the characteristic of peripheral-type spontaneous nystagmus and is referred to as Alexander’s law. In contrast, a gaze-dependent change of beating direction (i.e. left beating on left gaze and right beating on right gaze, termed gaze-evoked nystagmus (GEN)) is of central origin (for a detailed illustration see Figure 1).
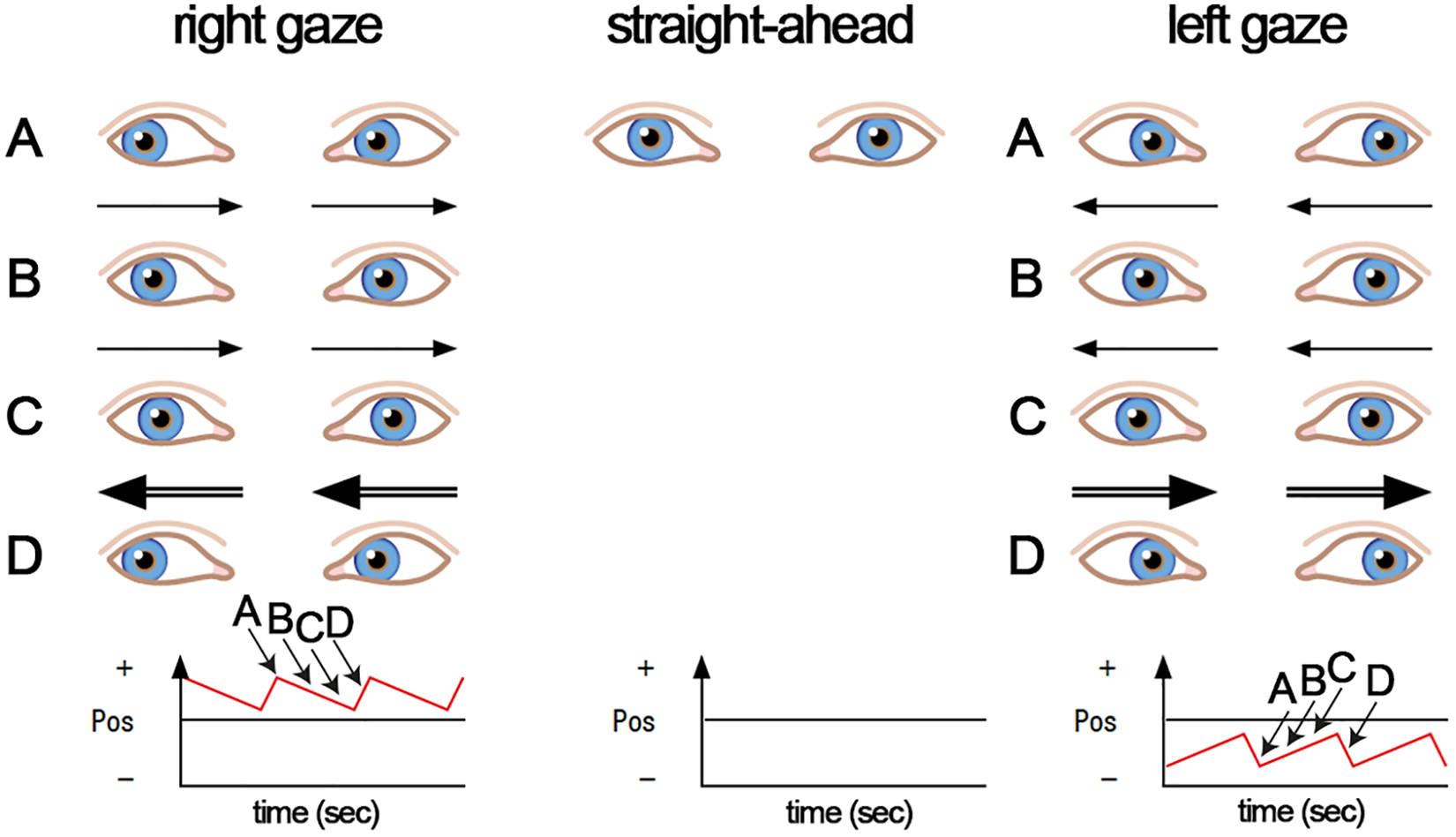
Testing for ocular stability at eccentric gaze. In healthy human subjects, gaze remains stable in the eccentric position. In patients with brainstem or cerebellar deficits, when in an eccentric position (position “A”), the eyes may show centripetal drift (i.e. towards primary gaze, indicated by the thin horizontal arrows) at right and left gaze (positions “B” and “C”), which is then compensated by a centrifugal correction saccade (thick horizontal arrows), bringing the eyes back to the eccentric position (position “D”). This results in a jerk nystagmus that changes its beating direction depending on the direction of gaze is shown in the bottom line. Source: Modified and reprinted with permission. © Rheuma Schweiz 2019.
For vertical nystagmus, a varying intensity depending on gaze can be depicted as well, with downbeat nystagmus becoming more intense on downward gaze but less intense on upward gaze and upbeat nystagmus being most prominent on upward gaze.
The GEN is also tested without spontaneous nystagmus in the primary gaze position. During lateral gaze to the right and left side in the end position, it is necessary to be sure that the patient is able to fixate the target with both eyes (being too lateral means that only the abducting eye is able to see the target, while the adducting eye is behind the nose). In the correct lateral end position, a GEN of a few beats is normal; if a persisting nystagmus occurs, this is pathological and points to a central origin.
Static vestibular function after unilateral peripheral–vestibular deficits usually shows good recovery by central compensation. Thus, spontaneous nystagmus disappears but can be uncovered by horizontal head-shaking (at about 2 Hz) or vibration (at a frequency between 30 Hz and 60 Hz) to the mastoid bone (both with Frenzel’s goggles on to suppress visual fixation) for approximately 10 s. Alternatively, applying the Rose positional test (also referred to as straight head-hanging test) by asking the patient to hyperextend the head and neck while lying in supine position may unmask an underlying vestibular imbalance, as head-hanging is a nonphysiologic and “uncalibrated” head position. These techniques may show a mostly horizontal nystagmus and thus are useful to identify compensated asymmetries in the angular vestibulo-ocular reflex (VOR; e.g. in case of a chronic unilateral vestibular deficit), with the nystagmus always beating away from the lesioned side. If the direction of the elicited nystagmus is vertical or torsional (termed “perverted nystagmus”), this points to a central pathology.
Other far less commonly encountered nystagmus patterns of central origin include pendular nystagmus (i.e. nystagmus without a clear distinction between slow and fast phases), periodic-alternating nystagmus (i.e. a horizontal nystagmus that changes its beating direction periodically about every 80–120 s) and see-saw nystagmus (i.e. a nystagmus with changing elevation and intorsion of one eye and at the same time depression and extorsion of the other eye).
Ocular stability—Testing for skew deviation
Vertical ocular drift on alternating cover test is another sign that should be tested at the bedside. Therefore, the patient is asked to fixate a small visual target (e.g. the tip of a pen) while the examiner is alternating covering one eye after the other. The examiner then looks for compensatory vertical movements of the eye that was just uncovered. Typically, one eye deviates upward and the other deviates downward on a pathological alternating cover test. Presence of such vertical skew deviation (i.e. vertical divergence of the eyes) in the absence of a trochlear nerve palsy suggests a central origin and is part of the H.I.N.T.S. (Head Impulse, Nystagmus, Test of Skew) bedside examination (see below). It should be noted that skew deviation may be accompanied by static binocular cyclorotation (no bedside testing possible) and head-tilt, a triad referred to as ocular-tilt reaction (OTR). 13 While an OTR may be of peripheral–vestibular origin also on rare occasions, skew deviation will be smaller than in central causes. An important differential diagnosis of vertical skew is a trochlear nerve palsy. For distinction, application of the Bielschowsky head-tilt test (see Lee et al. 14 for more detail) and testing for vertical ocular deviations both in upright and in supine position is recommended, as skew deviation modulates with head-orientation relative to gravity, becoming minimal in the supine position, while in the case of a trochlear nerve palsy the deviation of the eyes is body-position independent. Another means to assess the integrity of the peripheral–vestibular and central–vestibular pathways is by the use of the Bucket test. Thereby patients are asked to align a visual line along perceived direction of gravity (referred to as the subjective visual vertical). 15 Lateralized lesions will result (depending on the lesion location) in either ipsilesional or contralesional roll-tilts of the perceived vertical. 13
HIT—Assessing the integrity of the VOR
The VOR compensates for head rotations and thus enables stable gaze on an object in space during head movements. Applying high peak velocities (above 120–150° s−1) and high accelerations (usually in the range of 1000–4000 s−2), low-amplitude (10–15°) head impulses along the planes of the different semicircular canals allows side-specific testing of the integrity of the VOR for a single semicircular canal at the bedside in acutely dizzy patients. Thereby the examiner asks the subject to fixate an object in space (usually the examiner’s nose) while applying the head rotations, as illustrated in Figure 2. Importantly, the direction of head rotation should be unpredictable. As one of the fastest brainstem reflexes with a duration of approximately7–13 ms, compensating for head rotation is finished before it is perceived by the human eye. Thus, for the examiner, the subject’s gaze remains stable. In contrast, interruptions of the VOR will result in a delayed correction for head rotation triggered by retinal slip, thus such late (latency about 100 ms) compensatory eye movements can be visually perceived by the examiner as “overt catch-up saccades.” It is important to apply the HIT with sufficiently high peak velocities (>150° s−1) in order to achieve a side-specific (ipsilateral) assessment of peripheral–vestibular function. The goal is to drive the frequency of the inhibitory vestibular nerve fibers of the contralateral labyrinth to zero to isolate the contribution of the excitatory pathway of the ipsilateral labyrinth. While bedside testing of the horizontal canals can be reliably performed with little training, assessing the vertical canals requires more practice and is often assessed only with video-oculography. Thus, in clinical bedside examination, the horizontal HIT is tested routinely. This is sufficient in most cases because the horizontal canals are those which are affected regularly in peripheral–vestibular disorders. While the bedside HIT reliably identifies acute unilateral peripheral–vestibular deficits, patients with a chronic vestibular deficit might be well compensated. Specifically, a large proportion of corrective saccades might be invisible or covered because of a very short latency after onset of the head movement (the so-called “covert catch-up saccades”). Thus, in such patients, the sensitivity of the bedside HIT is only about 63–72%. 16 This can be overcome by the use of portable quantitative (usually video-based) head-impulse testing devices. 17

Head-impulse test (HIT) to the right, showing a normal (negative) response in the top row and an impaired (positive) HIT in the bottom row. While the eyes remain stable in space in a normal (negative) HIT, they deviate in the direction of the head rotation in an abnormal HIT, resulting in delayed (catch-up) saccades (indicated by the arrows). Source: Modified and reprinted with permission. © Rheuma Schweiz 2019.
Note that the absence of deficits in the efferent portion of the VOR (e.g. an abducens nerve palsy) is a prerequisite for interpreting vestibular function by the HIT. If ocular motility is fully preserved in one eye only, it is recommended to cover the other eye while performing the HIT. While an abnormal HIT is a characteristic finding of peripheral vestibular deficits, also central lesions along the pathway of the VOR may result in an abnormal HIT, referred to as vestibular “pseudoneuritis.” This includes lesions located at the root entry zone of the vestibulocochlear nerve and lesions involving the vestibular nuclei presenting as dorsolateral medullary syndrome (Wallenberg syndrome). Thus, the HIT alone does not always allow a reliable distinction between peripheral and central causes and should be combined with other ocular motor signs.
A three-step bedside testing battery called H.I.N.T.S. includes the head impulse test, testing for GEN and skew deviation (see Table 1 for details). This has been shown to distinguish peripheral from central causes with high sensitivity and specificity in patients with acute prolonged vertigo or dizziness accompanied by either spontaneous or GEN. 18,19 Adding a fourth sign (ipsilateral new-onset hearing loss) increases the sensitivity of H.I.N.T.S. (termed “H.I.N.T.S. plus”) for detecting central (usually ischemic) lesions even further. 20 Likewise, additional testing of the saccade and smooth pursuit systems (see below) also increases the sensitivity for detecting a central origin.
H.I.N.T.S. plus bedside testing battery.a

H.I.N.T.S.: Head Impulse, Nystagmus, Test of Skew; VOR: vestibulo-ocular reflex; HIT: head-impulse test.
a Teaching videos can be found at http://novel.utah.edu/Newman-Toker/collection.php.
Postural testing
Postural control (with eyes closed) relies on both vestibular and proprioceptive input. Damage to peripheral or central vestibular pathways may impair postural control, resulting in body lateropulsion and imbalance. By asking the patient to stand with the feet together, hands by the side and with eyes closed (termed “Romberg test”), postural stability is assessed. Increased sway in the Romberg test indicates either vestibular or proprioceptive impairment (or may be functional if it improves when the patient is distracted). However, for the correct differential diagnosis, it is important to look for signs of peripheral neuropathy in the clinical examination (reflexes and sensory deficits including malleolar vibration sense). Asking the patient to stand on a rubber foam pad with the eyes closed in a next step allows selective testing of vestibular function as proprioceptive input is canceled. While increased sway is normal even in healthy controls, falls during this test are highly suggestive for bilateral vestibular impairment. If patients are unable to stand without support, postural stability should be assessed in an upright sitting position (with arms crossed). Inability to sit upright without support favors a central cause. 21
Ocular motor testing for identifying central signs
Shifting gaze onto an object of interest and subsequently keeping the gaze on this object is provided by both VORs and visually mediated reflexes. Applying slow (0.5–1 Hz) head oscillations while the patient is fixating a stationary target evokes redundant activity of the VOR and the smooth-pursuit system. Thus, if the patient cannot keep the eyes on the target, both mechanisms are deficient. Targeted testing of the VOR is described above.
Since the smooth pursuit system, the saccade system, and the optokinetic reflex are mediated by different neuronal networks in the brainstem, deficits in testing these systems point to central ocular motor signs. A simple rule is that horizontal types of eye movements are generated in the pontomedullary brainstem, whereas vertical and torsional eye movements are generated in the midbrain.
To assess the smooth pursuit system, the patient is asked to follow a moving target along either the vertical or the horizontal plane with constant velocity (at about 20°s−1). If other eye movements (e.g. GEN) interfere with smooth pursuit testing, visual suppression of the VOR (also termed VOR cancellation) can be used to assess the integrity of the smooth pursuit system while the eyes remain centered and are not moving during sinusoidal head movements. Therefore, the patient is asked to look at a head-fixed target (e.g. his/her thumb that is rotating at the same speed as the patient’s head and body). Assuming an intact VOR, the inability to suppress nystagmus during head oscillations and simultaneous fixation of a head-fixed target suggests an impaired smooth pursuit system.
Optokinetic nystagmus can be triggered by a moving visual pattern or a handheld rotating drum. It allows an assessment of the conjugacy of pursuit eye movements (slow phase of nystagmus) and saccades (fast phase of nystagmus).
To test fast, saccadic eye movements, the patient is asked to shift the gaze quickly between different earth-fixed targets (e.g. the examiner’s nose and the tip of a pen). This is usually assessed in both the horizontal and the vertical plane. Saccades are then evaluated regarding their latency, velocity, and accuracy. Slow, delayed, or dysmetric (hypo- or hypermetric) saccades point to a central pathology but have limited lesion-localizing value. While hypermetric saccades are most often seen in cerebellar pathologies, hypometric saccades are more characteristic of brainstem lesions but may be observed in cerebellar pathologies as well.
It should be noted that the brainstem nuclei involved in generating vertical and torsional saccades are located in the midbrain, whereas those responsible for triggering horizontal saccades can be found in the pons. Thus, the pattern of saccadic impairment (predominantly horizontal vs. predominantly vertical) may provide useful information for lesion location.
Visual fixation may also be interrupted by saccadic intrusions or oscillations, that is, back-and-forth saccadic eye movements. These include square-wave jerks, ocular flutter, and opsoclonus and point to a central pathology. While brief pauses between single saccades (i.e. the presence of an intersaccadic interval) are characteristic for square-wave jerks (typically seen in the horizontal plane), ocular flutter (saccades restricted to a single plane) and opsoclonus (defined as random saccades in any plane) do not show inter-saccadic intervals.
Conclusions
In order to narrow down the differential diagnosis and to distinguish dangerous from benign conditions in the acutely dizzy patient, a targeted bedside vestibular and eye movement examination is highly recommended. Specifically, the H.I.N.T.S. bedside testing battery has been shown to be more sensitive for detecting central causes than an early (i.e. obtained within the first 24–48 h) MRI including diffusion-weighted imaging. 19 By performing all five examination steps demonstrated here, the sensitivity for finding a central origin of vertigo or dizziness will even be higher than that of the traditional three-step bedside HINTS examination.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
