Abstract
Neurorehabilitation is a rapidly developing subspecialty of neurology due to medical advances and growing knowledge on functional recovery from brain injury such as plasticity and regeneration in the nervous system. Furthermore, progress in modern technologies facilitate new therapeutic concepts. Patient-tailored, flexible multimodal neurorehabilitation is essential in neurological diseases due to the diversity of symptoms. In addition, rehabilitative treatment should be realized from disease onset. To fulfill these goals, the neurocenter of the Cantonal Hospital Lucerne established an uninterrupted treatment chain from the emergency stage to the social and occupational reintegration, which is described in this article with a focus on stroke, Parkinson’s disease, and multiple sclerosis patients.
Keywords
Introduction
Neurorehabilitation is a subspecialty of neurology that is rapidly growing in the last few years because medical advances have improved survival and extended life expectancy. Furthermore, the science of functional recovery from brain injury such as plasticity and regeneration in the nervous system has made significant progress.
Patient-tailored neurorehabilitation is essential in numerous neurological diseases. This implies rehabilitative treatment from disease onset (stroke or traumatic brain injury), as well as flexible multimodal rehabilitation in chronic diseases such as Parkinson’s disease (PD) or multiple sclerosis (MS).
To fulfill these goals, the neurorehabilitation unit of the cantonal hospital Lucerne is intimately interconnected to the neurology, neuroradiology, and neurosurgery services. The close collaboration of the neurodiscipline within the neurocenter ensures an uninterrupted patient care from the emergency stage to the social and occupational reintegration (https://www.luks.ch/standorte/standort-luzern/neurozentrum). The continuous treatment chain of neurorehabilitation encompasses several levels: (1) neuropath, (2) inpatient neurorehabilitation, (3) neurorehabilitation day clinic, (4) outpatient neurorehabilitation, and (5) center for professional reintegration for patients with brain damages (ZBA, Zentrum für berufliche Reintegration; Figure 1). This article aims to describe the neurorehabilitatory care of patients in the neurocenter of the Luzerner Kantonsspital including the main research projects, with emphasis on stroke, PD, and MS patients.
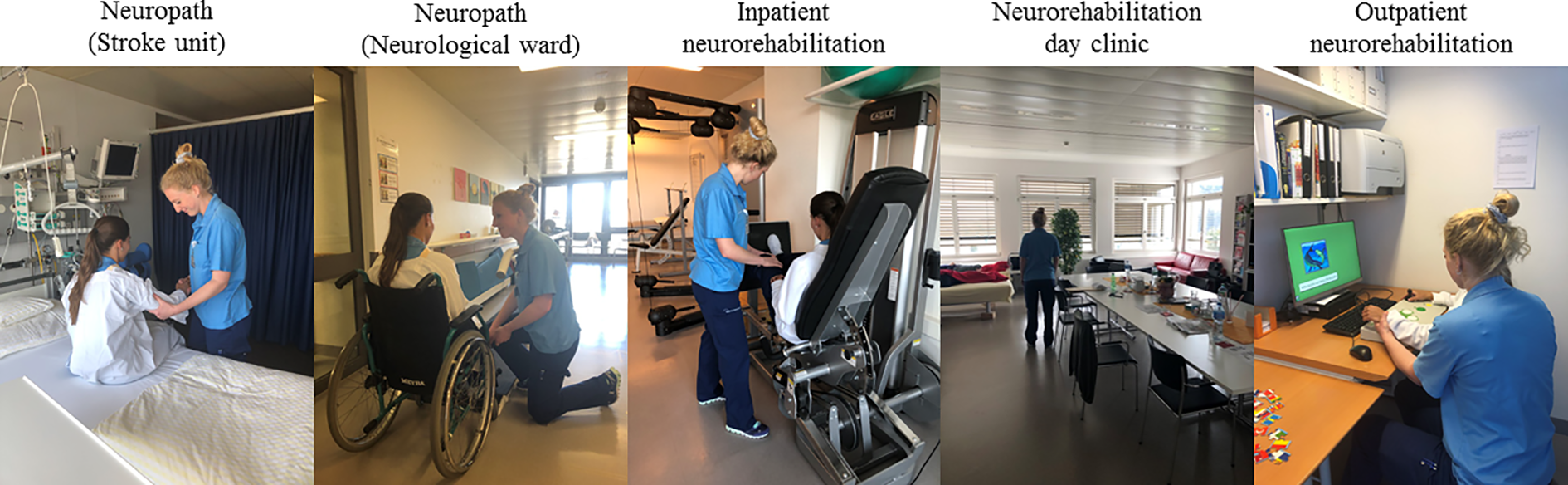
Continuous neurorehabilitatory treatment chain. Neurorehabilitation is offered to patients from day 1 of the hospitalization in different treatment entities by the same team. All shown person are employees of the Luzerner Kantonsspital and gave consent.
Neuropath
For patients with acute neurological disease, most commonly stroke and traumatic brain injury, a “Neuropath” (“Neuropfad”) was implemented. A mobile team of neurorehabilitation specialists (physical therapists, occupational therapists, language therapists, and neuropsychologists) assesses patients with regard to the need for neurorehabilitation at day 1 of the hospital stay. The team triages and organizes further steps to avoid delay and starts neurorehabilitative measures. Patients that are initially hospitalized in acute units of the neurocenter (neurological wards, neurosurgical wards, and stroke unit) are directly transferred to inpatient neurorehabilitation as early as possible. Patients initially hospitalized in another department including the intensive care unit enter the neurorehabilitation directly or via the neurological entities mentioned above. Importantly, patients going through the neuropath constantly get neurorehabilitative therapy by the mobile team.
Inpatient neurorehabilitation
The inpatient neurorehabilitation is located in the main building of the Cantonal Hospital Lucerne, one floor above the acute neurological ward. Besides specialized neurologists and nurses, physical therapists, occupational therapists, language therapists, neuropsychologists, and social workers take care of patients. Patients are screened at entry as to which therapies are necessary and outcome measurements are evaluated depending on the disease. All patients are rated by the Lucerne International Classification of Functioning (ICF)-based multidisciplinary observation scale (LIMOS) described below at admission and weekly until discharge. In addition, a core set of standardized outcome measurements is performed at admission and discharge. 1 On average, patients stay for 32 days ±19 standard deviation (SD), before many of them enter further rehabilitation settings described below. From 2015 to 2018, 1342 patients with a mean age of 61.92 years (±37.11 SD, range: 18–96), 45.67% of them being female, were treated in the inpatient neurorehabilitation: 65% were stroke patients, 9% had PD, 9% traumatic brain injury, 3% brain tumors, 2% MS, and 12% other diseases.
Neurorehabilitation day clinic
The neurorehabilitation day clinic was developed to offer patients high-frequency therapy in an outpatient setting after or as an alternative to an inpatient neurorehabilitation. Based on its intensity, it may also allow earlier discharge from the inpatient stay. Patients come to the day clinic 4–12 weeks depending on the disease and disease course. Therapy is performed 2–5 days a week. Patients stay a minimum of 5 h with at least two therapeutic sessions each day. The day clinic offers a room for relaxation and the possibility to sleep at noon. Qualified nurses take care of patients. Furthermore, besides rehabilitation, nursing, and medical services, social counseling is offered. In 2018, 107 patients with a mean age of 52.96 years (±15.87 SD, range: 18–80), 35.51% of them being female, were treated in the neurorehabilitation day clinic with a mean duration of 55 days ±31 SD: 70% were stroke patients, 14% traumatic brain injury, 7% brain tumors, 5% MS, and 4% PD.
Outpatient neurorehabilitation
In addition, we offer an outpatient neurorehabilitation from one to four times a week (for example, Lee Silverman Voice Treatment). Physical therapy, occupational therapy, language therapy, and neuropsychological therapy can be offered to patients solely or combined, depending on the needs of the patient.
Home-based rehabilitation
To further improve patient-tailored rehabilitation, several home-based rehabilitation programs, currently mostly performed within studies, are offered to patients in different diseases and described in detail below. In general, these programs are supervised by the outpatient neurorehabilitation team. Home-based rehabilitation is a highly promising research field that gets facilitated by tablet- and app-based technologies. It allows frequent training in patients who are not able to attend outpatient programs regularly, due to lack of transportation or work and family duties. Alternatively, it can be implemented in other therapy programs to enhance training frequency and broaden the therapeutic measures. The future goal is to implement home-based training programs in routine care, reimbursed by the insurance companies.
Center for professional reintegration for patients with brain damages (ZBA, Zentrum für berufliche Reintegration für Menschen mit einer Hirnverletzung) (https://www.zba.ch/)
Health-care professionals of the neurocenter also work in the center for professional reintegration for patients with brain damages (ZBA). The ZBA is an independent organization for the reintegration of brain-damaged patients into the professional life. This includes the evaluation of patients with regard to vocational capacities and the supervision of the integration of patients into previous or adapted working environments.
Lucerne ICF-based multidisciplinary observation scale
A valid detection of a patient’s ability to perform activities of daily living (ADL) is crucial in rehabilitation procedures to set measurable treatment goals, to make appropriate discharge arrangements, and to anticipate the need for community support. 2,3 To measure disability in stroke, the Barthel index (BI), the extended BI, and the modified ranking scale or the functional independent measurement (FIM) is often used. These measurements however have drawbacks such as ceiling and floor effects, focus on physical domains, and were not conceptualized to be comprehensive. 4,5
The ICF framework set by the World Health Organization (WHO) was developed to optimize measuring health and disability and has become a standard in neurorehabilitation (WHO, World Health Organization. International Classification of Functioning, Disability and Health: ICF, Geneva, 2001). 6 ICF core-sets were established for stroke, linking ICF domains with existing standardized measurements. 7,8 In addition, self-reported questionnaires and monodisciplinary observation tools incorporating ICF domains were developed. 9 –12 However, self-reported questionnaires strongly depend on preserved cognitive abilities (i.e. insight) of patients and monodisciplinary observation tools do not reflect the whole condition of a stroke patient within a multidisciplinary rehabilitation setting.
Therefore, we developed the LIMOS which is a multidisciplinary observation scale based on the ICF. It includes seven chapters, overall incorporating 45 items, each item is rated on a 5-point scale (1–5) (for more details, see Ottiger et al. 1 and Vanbellingen et al. 13 ) (Figure 2).

Graphical presentation of functional improvement. Spider diagram of the total LIMOS which demonstrates mean admission values (blue) versus discharge mean values (red) of each of the seven chapters (interpersonal interactions and relationships, mobility, self-care, communication, learning and applying knowledge, general tasks and demands, and domestic life). These seven chapters cover the 45 items of the total LIMOS. LIMOS: Lucerne International Classification of Functioning-based multidisciplinary observation scale.
LIMOS is feasible and showed good reliability and validity in stroke patients. 1 In addition, it showed to be more responsive than the FIM and the BI to measure improvements in ADL over time. 13 It covers more cognitive functions and shows neither floor nor ceiling effects and offers the possibility to measure subscales, for instance the functional role of the upper limb in the ADLs. 14
The LIMOS has successfully been used to evaluate the effects of noninvasive brain stimulation on the ADL in stroke patients with neglect. In a randomized controlled trial, continuous theta burst stimulation was shown to significantly improve functional outcome as measured by LIMOS during inpatient neurorehabilitation compared to sham stimulation. 15
Just recently the LIMOS underwent a thorough Rasch-based analysis, which is a more modern psychometric evaluation to standardize scales. 16 The advantage of Rasch-based LIMOS is that items can be ordered on a hierarchical level of difficulty. Besides stroke, the LIMOS can also be performed in all patients treated in inpatient and ambulatory settings and further validations in other diseases are warranted.
As mentioned above, the LIMOS is performed regularly in the inpatient neurorehabilitation and neurorehabilitation day clinic. As shown in Figure 3, the LIMOS improved significantly in both treatment entities if all treated patients are analyzed, indicating that patients did benefit from neurorehabilitation.

LIMOS results at admission and discharge of the inpatient neurorehabilitation and neurorehabilitation day clinic. LIMOS: Lucerne International Classification of Functioning-based multidisciplinary observation scale.
Cognitive neurorehabilitation in stroke patients
When humans acquire knowledge and understanding through sensation, experience, or thought, they do so by performing mental actions or processes denoted by the term “cognition.” Several cognitive domains can be differentiated, for example, attention, memory, language, or executive functions. Patients with stroke often show cognitive impairment, as evident in standardized neuropsychological tests or clinical observation. Cognitive neurorehabilitation aims to remediate or compensate for decreased cognitive abilities. 17 It thus relies on the fundamental principle that the brain has an inherent plasticity that enables it to recover from damage that provoked cognitive impairment. Furthermore, it relies on the possibility that individuals have the capacity to make behavioral adjustments that are necessary due to changing circumstances. Finally, there is a growing appreciation of the influence of contextual variables such as personal, emotional, and social factors.
In our neurorehabilitation, we treat stroke patients with impaired attention, memory, language, or executive functions with different therapeutic approaches. A main focus is on the therapy of spatial hemineglect, defined as the failure to attend, explore, and act upon the contralesional side of space. Spatial hemineglect usually occurs after damage to the language-nondominant hemisphere and is therefore typically found in patients who have suffered right-hemispheric stroke. Traditionally, this disorder of spatial attention is treated with visual exploration techniques, requiring patients to more actively explore the neglected hemispace. We have recently shown that additional noninvasive brain stimulation can improve and accelerate recovery from neglect and—associated with it—can further improve general functional outcome (Figure 4). 15

Continuous theta burst stimulation improves activities of daily living in neglect patients. Significant improvement of ADL in neglect patients after both 8 and 16 trains of cTBS versus sham, as demonstrated by the LIMOS. Total values between admission to and discharge from neurorehabilitation (*p < 0.05). ADL: activities of daily living; LIMOS: Lucerne International Classification of Functioning-based multidisciplinary observation scale; cTBS: continuous theta burst stimulation.
Multiple sclerosis
MS is a chronic inflammatory disease of the central nervous system and the most common cause of non-traumatic disability in young adults in western countries. 18 Despite increasing therapeutic options to ameliorate the disease course, most patients suffer from persistent neurological deficits over time.
Neurologic symptoms can be manifold and highly variable among patients. Therefore, a patient-tailored multimodal treatment is mandatory for the successful neurorehabilitation. 19 To do so, MS patients are treated in all mentioned entities in the neurocenter, depending on the deficits of patients and living conditions. To specifically address the needs of patients in a standardized way, we developed several additional treatment entities for MS patients.
“MS-FIT” is a standardized comprehensive ambulatory neurorehabilitation program that was developed to offer standardized high frequency training in a hospital-based ambulatory setting with the goal to improve disability and consecutively ADL and quality of life (QoL) of patients. Patients perform a circuit training consisting of six workstations (endurance, strength, flexibility, balance, dexterity, and reaction) in groups of three to six patients, 2 h, twice a week for 2 months, supervised by specialized physical therapists. MS-FIT was performed within a clinical study at the Cantonal Hospital Lucerne and University Hospital Bern (ClinicalTrials.gov Identifier: NCT02440516). It was finalized in March 2019 and the results will follow soon. It was feasible and highly accepted by patients and will now be integrated outpatient neurorehabilitation as treatment entity.
Cognitive MS training groups
Cognitive impairment is found in at least 40% of MS patients, during all stages of the disease, and negatively affects QoL as well as vocational capacity. 20 Concerning the rehabilitation of these deficits, a combination of restitutive and compensatory approaches seems most effective. 21 The former directly treats the impaired function (e.g. processing speed and working memory), should be deficit-specific, and can nowadays easily be performed by the patients themselves, for example, on mobile communication devices. On the other hand, learning compensatory strategies is usually taught by therapists. We developed two such compensatory cognitive training protocols in 2017, each incorporated in an occupational group therapy over nine sessions: (a) MS-MEMO is based on well-established (e.g. Chiaravallotti et al. 22 ) visual imagery and storytelling techniques to increase verbal memory performance in everyday situations (e.g. remembering names, shopping items, or passwords) and (b) MS-PLAN adopts goal management training strategies to improve planning and problem-solving capabilities of patients. 23
Home-based training programs specifically developed for MS patient are offered as well, currently mostly within studies. We recently showed that a standardized 4-week home-based dexterity training program with traditional training methods such as “turning metal discs,” “turning nuts on bolts,” and “finger tapping” exercises significantly improved manual dexterity and dexterity-related ADL in MS patients with subjectively and objectively impaired manual dexterity. 24
Currently, we perform as similar study called “App-Based Dexterity Training in Multiple Sclerosis” (ClinicalTrials.gov Identifier: NCT03369470) that investigates if an app-based dexterity training (“Finger Zirkus”; www.fingers-in-motion.de) performed on a tablet computer is effective in improving manual dexterity in MS patients (Figure 5). 25

New technologies for home-based training. Home-based training programs to improve manual dexterity. Left picture: finger circus-app: tablet-based training of several important movements for hand functioning (“Finger Zirkus”; www.fingers-in-motion.de) Right picture: leap motion controller: hand tracking sensors enable virtual reality-based training of finger/hand movements (https://www.leapmotion.com).
As further steps, we perform a feasibility study using a device, which is able to combine gamification and virtual reality (VR). This leap motion controller (LMC) is a low-cost system with camera sensors that track and quantify hand movements and gestures (Figure 5). The aim of the study is to investigate whether VR LMC training, focused on manual dexterity, is feasible in patients with MS. 26 In addition, preliminary data on the impact of the training program on manual dexterity will be collected. If feasibly, bigger controlled studies will be performed with regard to the effectivity of such a training program in improving manual dexterity in MS patients.
In our opinion, expanding ambulatory and home-based therapies in MS is essential to address the needs of patients because modern drug therapies increasingly prevent severe disability minimizing the need of inpatient rehabilitation as shown in our clinic with only 2% inpatient MS patients.
Parkinson’s disease
PD is a progressive neurodegenerative disorder that predominately affects motor and nonmotor basal ganglia circuitry. 27 The degeneration of dopaminergic neurons in the substantia nigra leads to the clinical manifestation of the cardinal motor features of PD: bradykinesia, muscle rigidity, tremor at rest, and impairment of postural reflexes. 27 While dopaminergic therapy improves some symptoms of PD, other symptoms, such as impaired finger dexterity and freezing of gait (FOG), may be less responsive to pharmacological treatment. There is increasing evidence that allied health care is associated with fewer PD-related complications and may improve ADL. 28,29 Our neurocenter offers specific treatments, such as the Lee Silverman voice treatment LOUD and BIG therapy, which are specific high-intensity training protocols to improve speech on the one hand (LOUD) and movement (BIG) on the other hand. 30,31 Furthermore, sensory cueing is applied which has been shown to be effective to initiate, to maintain movements, or to overcome FOG. 32 Additionally, we offer group therapy to improve psychological well-being and consequently QoL. Finally, an important clinical and research focus is impaired manual dexterity in PD. We developed a home-based dexterity training called HOMEDEXT (similar to the program previously published by Kamm et al. 2014 for MS), which proved to be effective in ameliorating manual dexterity in PD. 14 Future research projects will combine repetitive transcranial magnetic stimulation (rTMS) with VR LMC video game-based dexterity training, for example, within a randomized-controlled trial supported by the Jacques and Gloria Gossweiler Foundation. By combining rTMS with VR LMC, we expect to achieve longer lasting effects on dexterous performance. In a recently accepted pilot study, we were already able to demonstrate feasibility and short-term effects on dexterity using VR LMC in PD. 26
Conclusion
Patient-tailored neurorehabilitation is essential in numerous neurological diseases. This implies rehabilitative treatment from disease onset as well as flexible multimodal rehabilitation in chronic diseases. The neurocenter of the Cantonal Hospital Lucerne therefore established an uninterrupted interdisciplinary treatment chain from the emergency stage to the social and occupational reintegration. Within this treatment chain, neurorehabilitation is offered in different entities (neuropath, inpatient neurorehabilitation, neurorehabilitation day clinic, outpatient neurorehabilitation, and home-based training) to ensure an optimal treatment adapted to the needs of patients.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
