Abstract
A unique case of patient with primary essential CVG is presented. The patient has no underlying medical conditions. A series of hyaluronidase injections spaced out in 6 week intervals was injected into the subcutaneous tissues overlying the scalp deformities. The end result after 3 sessions of injections demonstrated significant improvement which has been maintained over the 12 month follow up period. This is the first case report of the successful use of hyaluronidase injections to treat CVG in Canada.
Introduction
Cutis Verticis Gyrata is a rare disorder which is characterized by the formation of thickened scalp skin which resembles the cerebral cortex gyri. It has been divided into 3 forms; primary essential, primary nonessential, and secondary. 1,2 It is a benign disorder, although it is still important to pursue investigations to exclude local or systemic underlying disorders. 3 The primary nonessential variety makes up 0.5% of cases. Men are most commonly affected by the primary essential type; usually coming into view at the time of or after puberty. 90% of cases are diagnosed after 30 years old. 4 Secondary forms affect both men and women almost equally. The presentation is mainly dependent on the underlying disorder. 5
There have been associations between primary nonessential CVG and mental retardation, cerebral palsy, epilepsy, cataract, and blindness. Primary essential CVG is not associated with any comorbidities. 6 Secondary disorders causing CVG include but are not limited to acromegaly, pachydermoperiostosis, psoriasis, chronic pemphigus, myxoedema, intradermal amyloidosis, intradermal cerebriform naevus, congenital melanocytic naevi, naevus lipomatosus, neurofibroma and insulin resistance syndrome. Histological hypertrophy of sebaceous structures is seen with without collagen thickening. 7 Prevalence of CVG in males is 1 in 100 000 and 0.026 in 100 000 in females. 6
We present a case of primary essential CVG in a male patient without underlying medical conditions. A course of hyaluronidase injections was injected locally to his scalp deformity showing a significant improvement over the period of treatments. This is the first case report of CVG treatment using hyaluronidase injections in Canada.
Discussion
Cutis verticis gyrata was reported originally by Alibert in 1837, however Robert was the first to describe this condition in 1843. 8 This particular medical condition was addressed using differing nomenclature such as bulldog scalp, cutis sulcata and cutis capitus strata. 9 In 1907, the now accepted term, Cutis Verticis Gyrata (CVG) was first used by Unna. 10
Cutis verticis gyrate (CVG) is a scalp condition reflected in the development of deep grooves and folds that resemble cerebral cortex gyri. 1 The parietal and occipital lobes are the main affected scalp areas. 2 CVG is divided into primary and secondary variants based on their associated diseases. Secondary classification is consistent with other scalp diseases or systemic illnesses. 9 Garden and Robinson further divided primary CVG into essential and nonessential subtypes. Primary essential has no association with any irregularities, while neurological and psychiatric involvement is present with the primary nonessential variant. Despite the asymptomatic aspect of the scalp appearance, the accumulation of cutaneous secretions within the grooves can cause a certain degree of discomfort presented by an offensive odour, burning sensation and pruritus. The hair of an affected patient remains intact but there have been reports of textural changes. 4 CVG is estimated to affect 1/100,000 men and 0.26/100,000 women in the general population. 6
In order to prevent infections and maceration, an increased level of hygiene in the localized region is recommended. For patients dealing with major psychological and cosmetic concerns or have complications with maceration and infections, surgery is the best option. Scalp reduction surgery is the main surgical approach. 3 Welborn et al. of the University of Texas McGovern Medical School first reported an improvement of CVG based on the use of hyaluronidase injections as a less invasive treatment for CVG, with minimal side effects. 11 There have been only a limited number of treatment options available for the management of CVG and they are mainly invasive techniques. Based on our research in the medical literature, we decided to also try the hyaluronidase technique in order to confirm its effectiveness and avoid a surgical procedure for our patient. As seen in the Welborn case report, our patient also showed a significant improvement of his scalp deformity with 3 treatments of hyaluronidase injections (Figures 1 and 2).
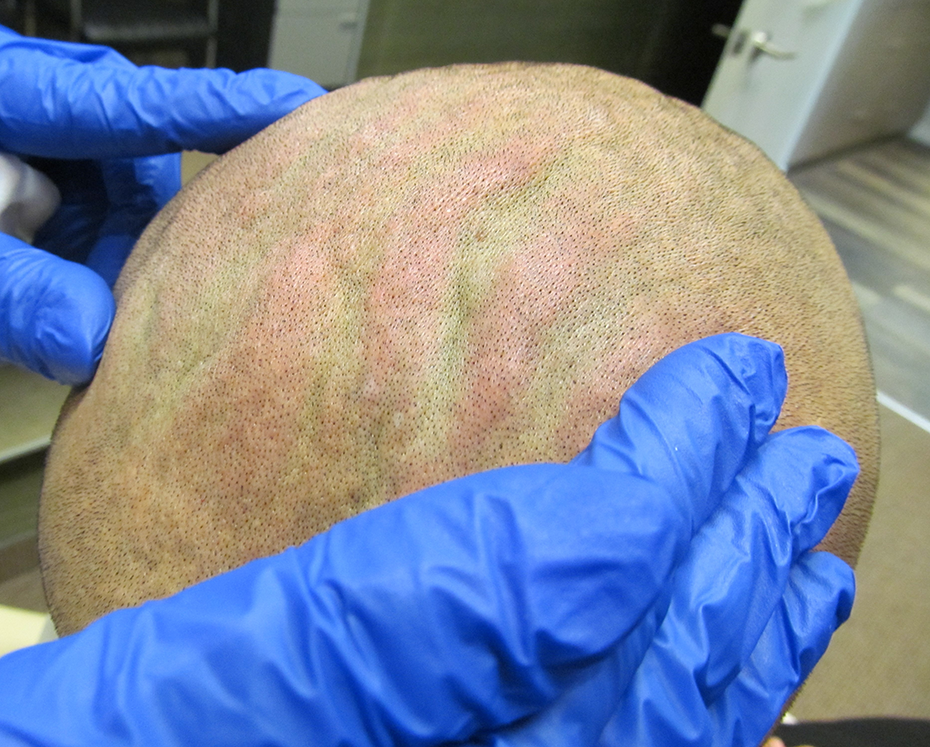
Initial presentation of patient with CVG of the scalp.
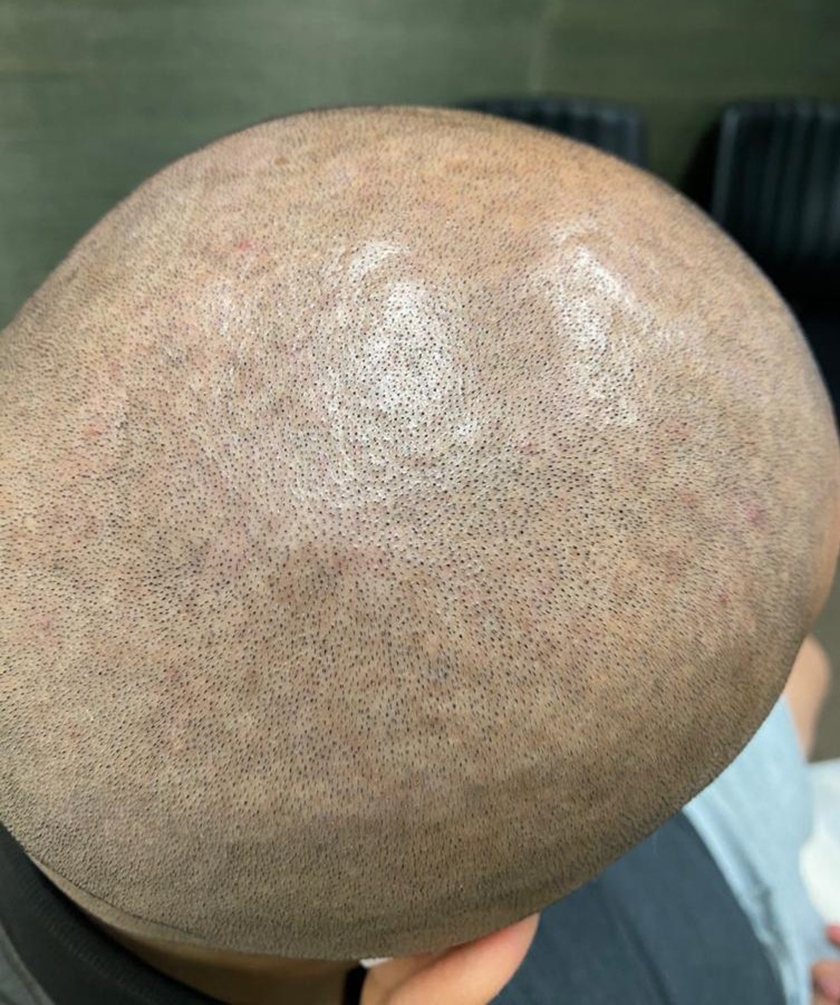
Presentation after 3 injections of hyaluronidase for CVG of the scalp.
Local injections of hyaluronidase can cause side effects such as local pruritis and allergic reactions. The incidence of allergic reactions is reported to be 0.05% to 0.69% 12,13 . Urticaria and angiodema have also been reported to occur at low frequency (less than 0.1%). 13 -15 In our patient, no side effects occurred with the hyaluronidase injections.
Hyaluronan, also known as hyaluronic acid (HA), is a polysaccharide with a high molecular mass located in the extra cellular matrix. It is the main component of the connective tissue. 15 HA is highly hydrophilic and has an extraordinary hydration capacity. This aids the viscoelastic properties of the skin. 14 Hyaluronidase is a non-ATP-requiring enzyme and breaks down HA. Hyaluronidase has been used in the practice of medicine for decades and has been successful in enhancing subcutaneous fluid infusion, as an adjuvant to accelerate the absorption and dispersion of drugs in subcutaneous tissue or to manage extravasation. 8,11,14 In plastic surgery and local operative procedures, hyaluronidase is used as a local adjuvant in order to increase diffusion capacity for local anesthetics and to improve the analgesic efficacy with reduced intraoperative and postoperative pain. The primary use of hyaluronidase in contemporary plastic surgery and aesthetic injectable practices is its ability to break down hyalorinic acid fillers to correct complications and unsatisfactory results after filler injections. 12,13 The therapeutic affect of hyaluronidase is such that hyaluronic acid is broken down into monosaccharides by cleaving its glycosidic bonds and other acid mucopolysaccharides in the connective tissue. 11,13 -15 . It appears that this breakdown process is responsible for the improvements that are see in the appearance of the scalp after subcutaneous injection of hyaluronidase for CVG patients.
Conclusion
Subcutaneous hyaluronidase injections directed towards the deformity manifested by CVG appears to offer an alternative treatment option for these patients. The current treatment options that have successfully treated this condition involve surgical procedures that are inherently more invasive. We present a less invasive alternative that effectively treats the condition in a non invasive, relatively painless procedure spaced out over three 6-week intervals. Our protocol, based on a previous case report, involved the subcutaneous injection of 200 IU of hyaluronidase at 6 week intervals. Three treatments were given. Our patient demonstrated significant improvement over the course of treatment and has remained asymptomatic in the follow up period of over 1 year. Our case is the second reported successful CVG treatment using hyaluronidase injections, and the first in Canada. We suggest that further research is still required in order to explore its mechanism of action and if repeated success could be achieved with other CVG cases.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Statements and Informed Consent
Verbal consent has been obtained from the patient to utilize their photographs documented in the patient chart.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Statement of Human and Animal Rights
The procedures followed were in accordance with the ethical standards of the responsible committee on human experimentation (institutional and national) and with the Helsinki Declaration of 1975, as revised in 2000 (5).
Statement of Informed Consent
Informed consent to participate in the study was obtained from the participant as well as consent for publication of individual patient data including details, images, or videos relating to the individual participants and a written informed consent for the publication of these was obtained from the participants (or their parent or legal guardian in the case of children under 16) and a statement to this effect should appear in the manuscript.
