Abstract
Cutis verticis gyrata (CVG) is characterized by extensive formation of scalp skin that mirrors the folds of the cerebral gyri. Giant CVG secondary to cerebriform intradermal nevus (CIN) has been rarely reported, and its management mainly involves surgical excision. In certain cases of giant CVG, however, surgical excision is dilemmatic. This case report describes a giant CVG secondary to CIN that was successfully managed with scalp expansion and surgical excision. Previously published studies are also reviewed.
Keywords
Introduction
Cutis verticis gyrata (CVG) is a rare congenital or acquired scalp disorder that manifests as scalp skin redundancy that mirrors the folds of the cerebral gyri. It was classified as primary (essential/nonessential) and secondary CVG by Polan and Butterworth. 1 CVG has a definite male predilection with an estimated prevalence of 1/100,000 in the male population and 0.026/100,000 in the female population. 2 Primary essential CVG is an extremely rare isolated and idiopathic condition, and primary nonessential CVG is associated with neuropsychiatric or ophthalmological disorders. Secondary CVG is more common and often occurs as a response to inflammatory or neoplastic processes. Cerebriform intradermal nevus (CIN) is an uncommon cause of secondary CVG, representing approximately 12.5% of all cases of CVG. 3 Management requires surgical excision of the overlapping skin folds to provide a flat and smooth cosmetic appearance. Giant CVG secondary to CIN has been rarely reported, and surgical management is somewhat dilemmatic. The purpose of this case report is to present a case of giant CVG secondary to CIN in a 27-year-old woman that was successfully managed with surgical excision after scalp expansion and to review previous studies involving surgical management of CVG published in the literature.
Case report
A 27-year-old woman was admitted to the hospital for evaluation of a huge black scalp macule (20 × 18 cm) with thinning of the overlying hair (Figure 1). The asymptomatic scalp macule first appeared upon her birth and gradually increased in size. On physical examination, the scalp macule was characterized by brain-like sulcus and gyrus formation, which is a typical feature of CVG, without other abnormalities. Normal results were obtained for all laboratory tests, including routine blood, urine, and stool analyses; hepatorenal function; blood glucose and lipid concentrations; and electrolyte concentrations. The patient’s medical and familial histories were unremarkable. The patient was diagnosed with secondary CVG. Because tumoral lesions are the most common cause of secondary CVG and because of the aesthetic concerns associated with the disease, surgical management was considered the optimal treatment.

(a) Occipital view of the cutis verticis gyrata (CVG) before scalp expansion, measuring 20 × 18 cm. (b) Lateral view of the CVG before scalp expansion.
We were concerned that the patient did not have enough hair-bearing tissue for scalp reconstruction after surgical excision because of the extensive scope of the lesion. Therefore, a two-stage surgery with reconstruction was finally planned. During the first stage, four rectangular inflatable tissue expanders (300-, 300-, 100-, and 80-mL) were placed under the scalp galea on both sides of the forehead and mastoid regions (Figure 2). Over a 6-month period, the expanders were inflated twice weekly until they reached a total volume of 1400, 1400, 450, and 400 mL, respectively. During the second stage, full-thickness excision of the lesion with a 0.5-cm skin margin was performed without disturbing the periosteum, and closure was achieved with rotation advancement flaps from the adjacent expanded scalp without perioperative complications (Figure 3). The patient was diagnosed with CIN according to postoperative pathological findings. During the 6-month postoperative follow-up, there was no evidence of recurrence; only a scar was present under the coverage of the hair.

Four rectangular inflatable tissue expanders (300-, 300-, 100-, and 80-mL) were placed under the scalp galea on both sides of the forehead and mastoid regions and inflated twice weekly until they reached a total volume of 1400, 1400, 450, and 400 mL, respectively. (a) Occipital view of the cutis verticis gyrata (CVG) after scalp expansion. (b) Lateral view of the CVG after scalp expansion.

(a) Occipital view of the operation area 1 month after excision. (b) Lateral view of the operation area 1 month after excision.
Discussion
As an uncommon cause of CVG, CIN typically presents as a slowly enlarging, asymmetric skin-colored macule of the parietal or occipital scalp with female predominance. The diagnosis is based on histopathologic findings. 4 As its size increases, the lesion becomes more prominent and may exhibit separation by furrows, forming CVG. The management of giant CVG secondary to CIN is mainly surgical excision, not only for aesthetic and psychological reasons but also because of the 4.5% risk of malignancy transformation. 5 Surgical management mainly involves excision and reconstruction using local flaps, free flaps, skin grafts, or scalp expansion according to the size of the lesion. Scalp reconstruction is deemed the most important concern.
The application of local flaps and skin grafts with different shapes and sizes is the traditional method of scalp reconstruction. Flaps and grafts can be used in most patients, but they cannot provide an optimal aesthetic outcome when the scalp defect after excision is extremely large. Moreover, the risk of relapse may increase when the excision is incomplete, and a second surgery might be required for complete correction. A free flap (e.g., a free latissimus dorsi musculocutaneous flap) is another feasible method of scalp reconstruction because of its large surface, long vascular pedicle, and reliably well-vascularized tissue. Similarly, serratus anterior, rectus abdominis, omental, and radial forearm flaps are also candidates during scalp reconstruction. Although a free flap reduces the number of operations and is an alternative in patients with a large scalp defect after excision or without enough hair-bearing tissue for reconstruction, use of such a flap can result in an unaesthetic hairless region in the surgical area and scar formation at the donor site, resulting in an unsatisfactory cosmetic appearance. Scalp expansion provides optimal aesthetic and functional outcomes with maximum efficiency by expanding the remaining parts of the scalp, making the scalp flap design during surgery simple and flexible. For reconstruction of large scalp defects, the most appropriate donor remains the scalp itself, which makes scalp expansion the optimal treatment option for giant CVG.
All cases of surgically managed CVG published in the literature according to our PubMed search are listed in Table 1.2,4,6–24 Thirty-five patients (11 female, 24 male; mean age, 26.9 years) with CVG (maximum lesion diameter, 8–32 cm) were successfully treated with scalp excision followed by direct suturing (n = 9), scalp subcision (n = 1), local flaps (n = 2), free flaps (n = 2), skin grafts (n = 3), and scalp expansion (n = 18). Various excision patterns have been reported, such as a Y-shaped excision, T-shaped excision, fleur-de-lis pattern, and butterfly-shaped excision. Scalp reconstruction with local flaps and skin grafts has mainly been used in patients with relatively small scalp defects; the largest reported defect was 20 × 15 cm. For giant CVG with an expected large resultant scalp defect, a free flap is an alternative and scalp expansion is the favorable option, with a reported maximum corrected scalp defect of 32 × 27 cm.
Cutis verticis gyrate managed with surgical options published in the literature.
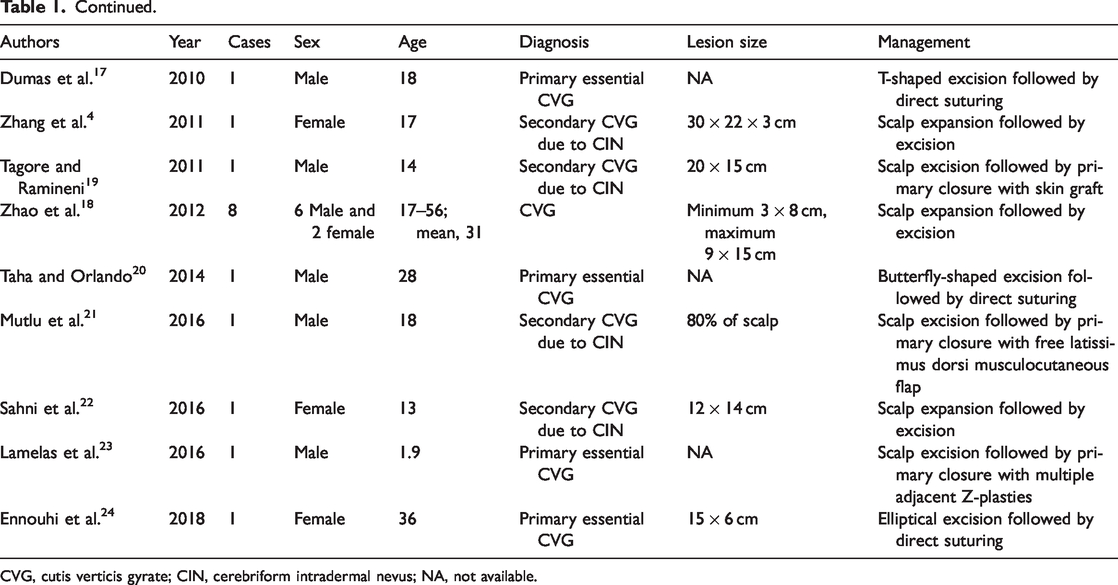
CVG, cutis verticis gyrate; CIN, cerebriform intradermal nevus; NA, not available.
Conclusion
Giant CVG secondary to CIN may be excised with scalp expansion in patients without enough hair-bearing tissue for scalp reconstruction.
Footnotes
Acknowledgement
We would like to thank the patient for providing consent for publication of this case.
Declaration of conflicting interest
The authors declare that there is no conflict of interest.
Ethical approval
All procedures involving this patient were performed in accordance with the ethical standards of the institutional and national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards.
Funding
This work was supported by the CAMS Innovation Fund for Medical Sciences (Grant No. CAMS-12M-1-007).
Informed consent
Informed consent was obtained from the patient described in this report.
