Abstract
Neurofibromatosis type 1 (NF1) is an autosomal dominant disorder characterized by the formation of benign nerve sheath tumours known as neurofibromas. A 28-year-old female presented with a large neurofibroma extending from the elbow of the right arm into the axilla and across the right breast. A mastectomy was performed to excise 4 kg of tissue with an immediate flap reconstruction. Post-operatively, the patient’s hemoglobin levels dropped significantly due to excessive blood loss, but the patient was discharged by post-operative day 5 with no additional complications. This study raises awareness for NF1, describes possible complications that may arise following excision of highly vascularized neurofibromas, and outlines possible techniques for breast reconstruction.
Introduction
Neurofibromatosis type 1 (NF1), previously known as von Recklinghausen disease, is a rare genetic condition with a prevalence of 1 in 3000. 1 The disease involves an autosomal dominant mutation on chromosome 17, which leads to a loss of function of neurofibromin, a tumour suppressor protein. This results in uncontrolled proliferation and the formation of neurofibromas, an abnormal growth of the nerve sheath. Typical presentation of NF1 includes café-au-lait macules, axillary freckling, and iris lesions called Lisch nodules. 2 There is no cure for neurofibromatosis; treatment is largely targeted toward symptom management. 3
Here, we present a rare case of NF1 associated with unilateral gigantomastia. Gigantomastia is characterized by excessive breast tissue growth of over 1.5 kg per breast. 4 The patient was treated with a right mastectomy and immediate flap reconstruction. We conducted a literature review regarding the presentation and long-term complications of NF1, the classification systems of gigantomastia, current breast reconstruction techniques, and compare our results with a similar case of NF1-associated gigantomastia.
Case Presentation
A 28-year-old Caucasian female with no family history of NF1 presented with a large neurofibroma tumour extending from the right arm into the axilla and across the right chest (Figures 1 –3). This tumour had been growing in size since birth. Preoperatively, the tumor was found to extend 46 cm below the inframammary fold, covering the right side of the abdomen, and was shown to infiltrate the nerves of the brachial plexus. The patient consented for right mastectomy, neurofibroma tumour axillary excision, and immediate thoracoabdominal flap reconstruction.
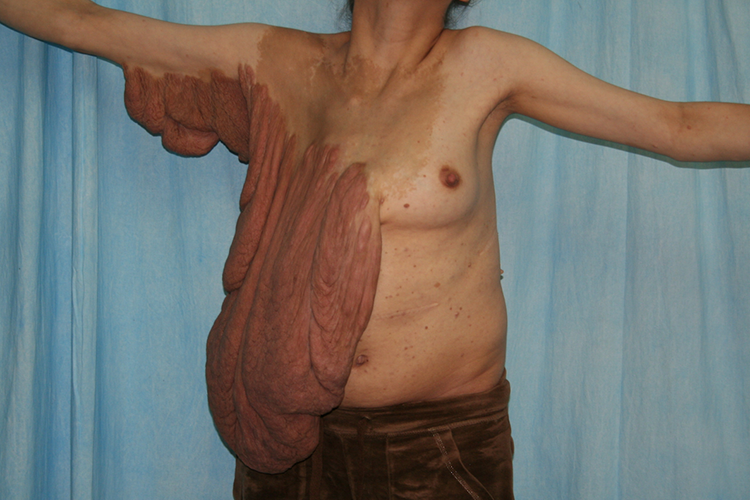
Preoperative anterior view of neurofibroma tumour.
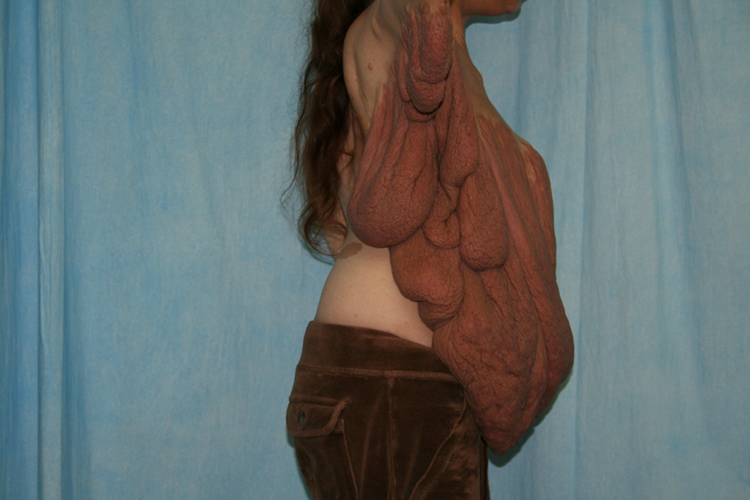
Preoperative lateral view of neurofibroma tumour.
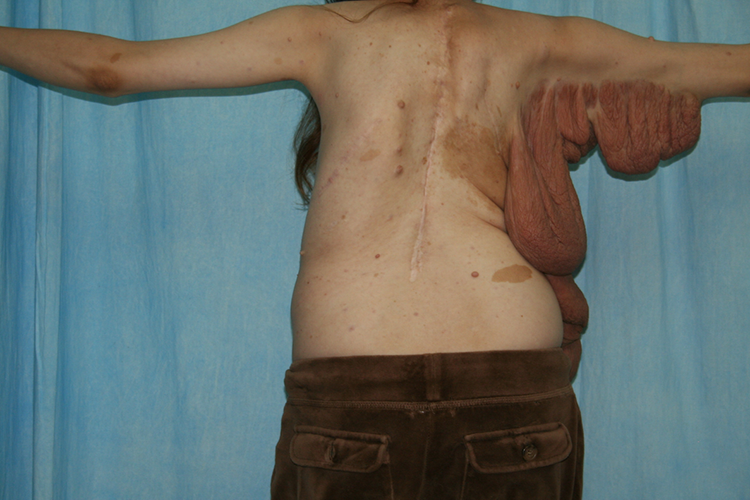
Preoperative posterior view of neurofibroma tumour.
The neurofibroma lesion was found to be highly vascular and there was significant bleeding that occurred during the first incision. To achieve hemostasis, 2L of tumescent solution containing 40 mL of xylocaine and 4 mL of adrenaline was infiltrated across the patient’s right chest, axilla and arm. The tumour was removed across the right chest with a mastectomy down to the pectoralis major. No nipple could be identified. The excision went over the sternum across the inframammary fold and around to the back.
Tumours were identified around the medial and lateral cord of the brachial plexus and along the intercostal brachial nerve. Medial cord tumours were incorporated directly into the nerves and thus could not be fully removed. Lateral cord tumours were integrated with the pectoralis major muscle, which needed to be split for tumour excision. Intercostal brachial nerve tumours were highly vascularized, and so arteries and veins entering the tumours were dilated and clipped.
The abdominal skin was elevated and brought up to the clavicle where it functioned as an advancement flap for right breast reconstruction. This abdominal tissue was also used as a transposition flap to cover the axilla. Additional tissue from the lateral thoracic area was used to cover deficits across the posterior arm and the remainder of the axilla. Prior to anesthesia reversal, it was observed that the tumour was ripping through the flap edges in the axilla, which required appropriate re-suturing. The patient weighed 43 kg preoperatively and the total weight of tumour excised was over 4 kg. Tumour samples from the chest, brachial plexus, and axilla were sent for pathology examination and returned negative for malignancies.
Postoperative Care
The patient’s hemoglobin (Hb) dropped from 104 g/L preoperatively to 46 g/L postoperatively. Following surgery (post-operative day [POD] 0), she was immediately admitted to the intensive care unit. The patient continued to bleed from the wound site, seeping through the dressing. The patient was pale and showed dizziness upon sitting up. The patient was transfused with 1 unit of packed red blood cells. Following transfusion, her Hb rose to 58 g/L. An additional unit of red blood cells was given and the Hb stabilized at 84 g/L. Intravenous (IV) piperacillin–tazobactam, and IV iron sucrose was also started.
On POD 2, chest X-ray indicated air space disease in the lung bases, and mild crackles were also heard in these areas. The patient developed shortness of breath and was started on furosemide. It was noted that bleeding from the wound site stopped. From POD 3 to 5, no major complications were noted and the patient stabilized. On POD 5, the patient was discharged home. The patient returned to the emergency department on POD 7 with possible cellulitis at IV site on her left arm and was treated with cephalexin. No additional complications were reported.
On 2.5-year follow-up, our patient had no further complications and is pleased with the aesthetic result (Figures 4 –6). No new major neurofibromas had been identified. The patient also recently had a second child following the surgery with no complications.
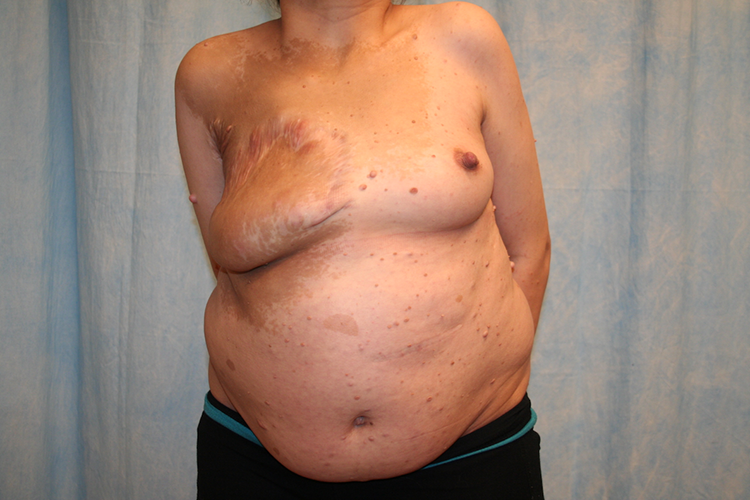
Postoperative 2.5-year follow-up anterior view.
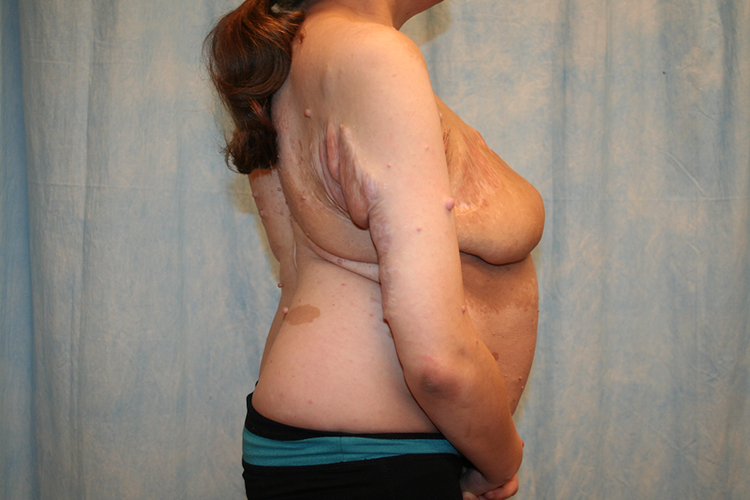
Postoperative 2.5-year follow-up lateral view.
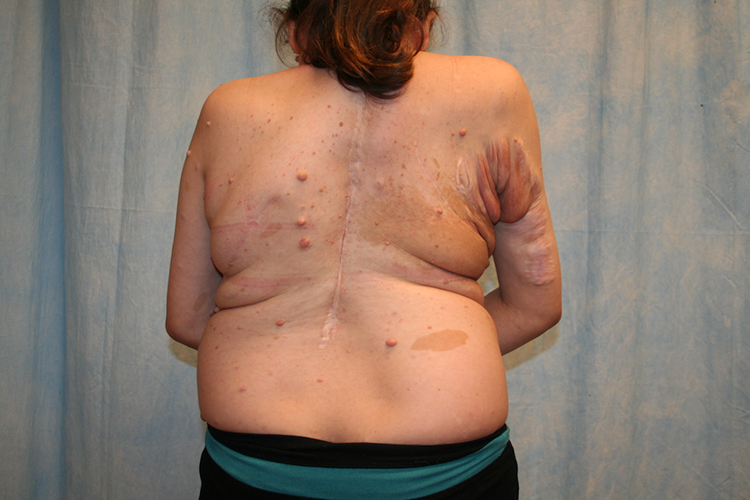
Postoperative 2.5-year follow-up posterior view.
Discussion
Neurofibromatosis can be divided into type 1, type 2, and schwannomatosis, with the most common being NF1. Neurofibromatosis type 1 tumours can be classified into 3 main categories: cutaneous, located on or within the dermis; nodular, found below the dermis; and plexiform, surrounding multiple nerves. 5 Plexiform neurofibromas have an increased risk of developing into malignant peripheral nerve sheath tumours and are highly vascular, making them a significant challenge for surgical resection. 5,6
Children with NF1 can present with bone deformities resulting in short stature, scoliosis, and osteoporosis. These children may also have neurological deficits, learning disabilities, and visual defects associated with optic glioma development. Adults with NF1 often present with café-au-lait macules, Lisch nodules, axillary freckling, and hypertension. 5 Long-term complications for adults with NF1 include increased risk for breast cancer in women less than 50 years old and increased likelihood of headaches. 7,8
The patient is a 28-year-old female who presented with numerous café-au-lait macules and a large neurofibroma that extended across her right chest, axilla, right arm, and surrounded the nerves of her brachial plexus. The patient has a history of scoliosis which was corrected surgically during childhood. She also exhibits short stature characteristic of NF1, standing at 4 feet 7 inches.
Breast reconstruction post-mastectomy can be performed using autologous tissues, implants, or a combination of both. 9 Implant-based techniques offer faster recovery times, shorter operative times, and eliminate the risk of donor site complications. However, implant-based techniques may require multiple follow-up visits, to progress with tissue expansion, and carry the risk of implant rupture or infection. Autologous tissue methods, on the other hand, may provide more aesthetically cohesive breast mound reconstructions and can serve a dual purpose of removing undesired tissue. Autologous methods include using flaps from the latissimus dorsi, transverse rectus abdominis, gluteus, or thighs. When selecting the method of breast reconstruction, it is important to consider the patient’s body habitus, desired aesthetic outcome, and anatomical variations in flap perforation. 9,10
The most commonly used flaps include the transverse rectus abdominis musculocutaneous (TRAM) flap and the deep inferior epigastric perforator (DIEP) flap, both of which arise from the rectus abdominis. 10 A TRAM flap has robust vascular supply but requires removal of at least a portion of the rectus abdominis muscle, which may result in abdominal weakness and bulging. In contrast, the DIEP flap spares the rectus abdominis but may have less blood supply. 9 A less common technique for breast reconstruction is the thoracoabdominal flap, a fasciocutaneous flap that uses tissue from the upper abdomen and relies on segmented perforating vessels. 11,12 This flap may carry the advantage of better contouring, colour matching, and minimal donor site complications compared to the musculocutaneous TRAM flap. Disadvantages of the thoracoabdominal flap include unpredictable variations in distal flap blood supply which may result in slower healing of the wound site. 12
After noticing increased neurofibroma growth during her first pregnancy, a common occurrence in patients with NF1, 13 the patient elected for a right mastectomy and immediate thoracoabdominal-based flap reconstruction.
Gigantomastia is a clinical condition of excessive breast tissue growth, which can lead to patients presenting with mastalgia, back pain, and loss of nipple sensation due to chronic nerve traction. 4,14 Although there is no universally accepted definition for gigantomastia, much of the literature classifies the disorder as breast growth requiring tissue removal of at least 1.5 kg per breast. 4 More recently, Dafydd et al proposed an alternative definition for gigantomastia which accounted for the patient body mass index. They stipulate that gigantomastia should be defined as breast tissue exceeding 3% of an individual’s total body weight. 15 The tumour excised from the patient in this case report weighed more than 4 kg and accounted for over 10% of her body weight. This makes her eligible for either classification of gigantomastia.
To the authors’ knowledge, there is only one other case report found in the literature of NF1 affecting the breast tissue. Velchuru et al presented a 36-year-old patient with café-au-lait macules, Lisch nodules, and extensive neurofibromas on the lower portion of her left breast. Flap reconstruction of the left breast was considered but their patient ultimately elected for a minimalistic approach, and so a modified wise excision with mastopexy was performed. No major complications were identified. 16 A modified wise pattern breast reduction was contraindicated for our patient due to the minimal breast tissue remaining post neurofibroma excision.
Conclusion
Overall, the successful treatment and reconstruction of this rare case of unilateral gigantomastia associated with NF1 required the use of a less common thoracoabdominal flap. Tumour resection proved difficult due to tumour infiltration in the brachial plexus and the highly vascular nature of the neurofibroma. This study raises awareness for NF1, describes possible complications that may arise following excision of highly vascularized neurofibromas, and outlines possible techniques for breast reconstruction. Patient provided informed consent for information and images included in this case report.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Statement of Human and Animal Rights
This article does not contain any experimental studies with human or animal subjects.
Statement of Informed Consent
All persons gave their informed consent prior to their inclusion in the study.
