Abstract
Autologous skin grafts (autografts) remain the gold standard in the treatment of skin loss. For extensive wounds or burns, however, identifying adequate donor sites can be the limiting factor. Additionally, donor sites are associated with pain, risk of infection, and poor cosmetic outcomes. Many skin substitutes have been engineered as alternatives to traditional autografts. These substitutes, however, all leave something to be desired either functionally or cosmetically. This report describes the use of a new technology, autologous homologous skin constructs, to regenerate full-thickness skin grafts that maintain functional polarity, allowing important components of skin such as glands and hair follicles to regenerate. These grafts only require small samples of full-thickness skin from the patient, decreasing issues of donor site availability.
Introduction
In the treatment of large skin defects not amenable to secondary healing, primary closure, or local tissue rearrangement, split and full-thickness autologous skin grafts remain the gold standard of care. 1 For extensive wounds or burns, however, identifying adequate donor sites can be the limiting factor. Additionally, donor sites are associated with pain, risk of infection, and poor cosmetic outcomes. Various skin substitutes and technologies have been engineered over the recent years; however, each lack ideal functional or cosmetic characteristics when compared with traditional autografts. 2 A new technology, autologous homologous skin constructs (AHSC), offers the possibility of full-thickness skin regeneration from a small full-thickness tissue sample, providing a new solution for challenging cases of soft tissue loss. 3 In contrast to its predecessors, AHSC reconstitute autologous cells that remain “functionally polarized,” thus allowing important components of skin such as glands and hair follicles to regenerate. Here, we present 2 cases of large pediatric wounds in which AHSC was successfully used to regenerate functional full-thickness skin, including hair and glands, with minimal donor site morbidity.
Cases
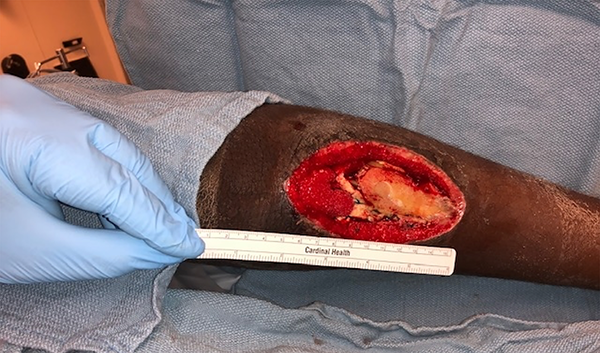
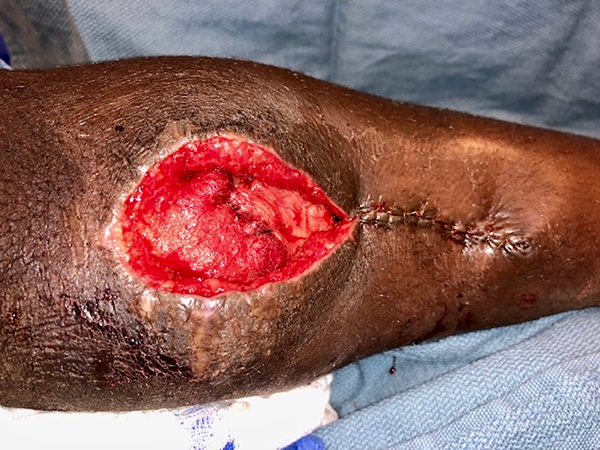
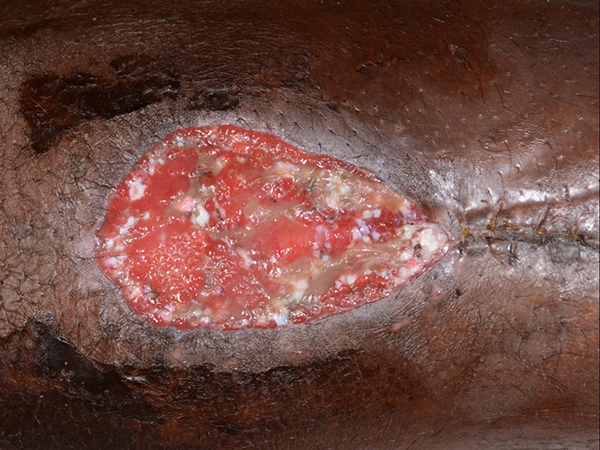
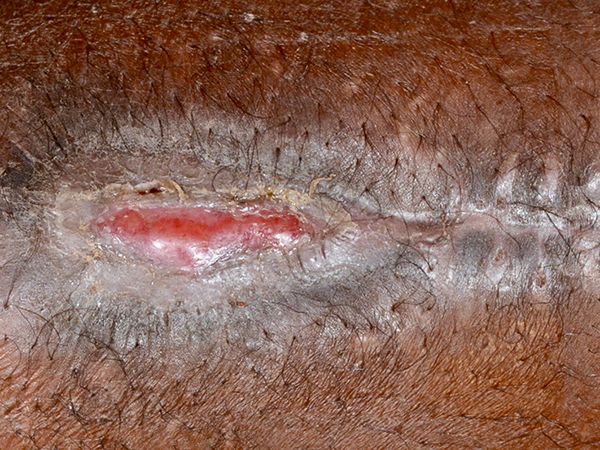
An 18-year-old female with no past medical history presented after ejection from the back seat of a vehicle. She was noted to have an extensive degloving injury to her left dorsal foot and additional left knee wound. After the remainder of her trauma workup was negative, she was taken urgently to the operating room (OR) for washout and debridement of left dorsal foot and knee wound; post-operatively her foot wound measured 20 cm2. She returned to the OR for repeat debridement 2 days later after which the wound volume was measured to be 51.59 cm3 by 3-D volumetric analysis (Vectra (Cleveland, OH); Canfield Scientific, Inc (Parsippany, NJ)) (Figure 1). At that time, a small full-thickness skin graft was harvested from her lower abdomen and sent for processing. Five days after her initial injury, the AHSC product arrived and she underwent application to both the foot (Figure 2) and knee injuries. A silicone gel membrane was then applied over the skin graft per manufacturer recommendations and a negative pressure dressing (wound VAC) was applied. On post-operative day 5, she underwent a dressing change under anesthesia and exchange of the silicone gel layer. At that time, the graft site was observed and there were no signs of dehiscence or infection. On post-operative day 16, she was seen in clinic, at which time it was noted that she had granulation tissue to the center of the wound, new skin growth, and epithelialization around the lateral edges (Figure 3). She was observed on an outpatient basis. Her smaller knee defect was nearly healed by 4 weeks post-operatively and hair growth through the area of skin grafting was observed. By 8 weeks, she was noted to have nearly full epithelialization of her dorsal foot wound (Figure 4). At 9 months after her initial injury, the wound was fully healed with a 3-D volumetric analysis of the wound totaling 19.29 cm3 due to scar contraction.
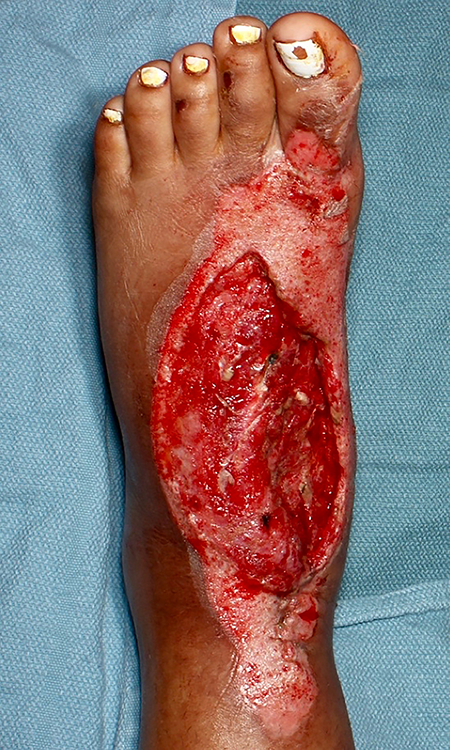
Before autologous homologous skin construct (AHSC) application.
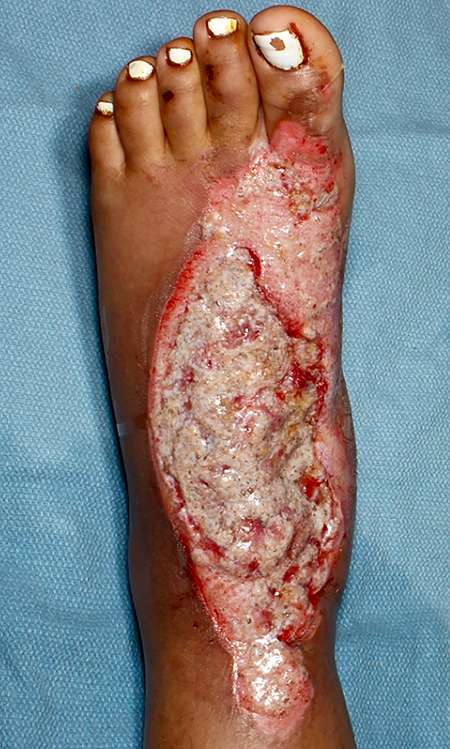
After autologous homologous skin construct (AHSC) application.
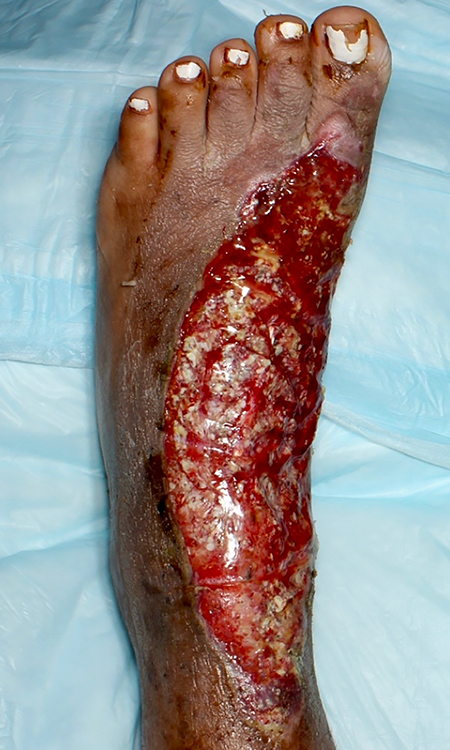
Postoperative day 16.
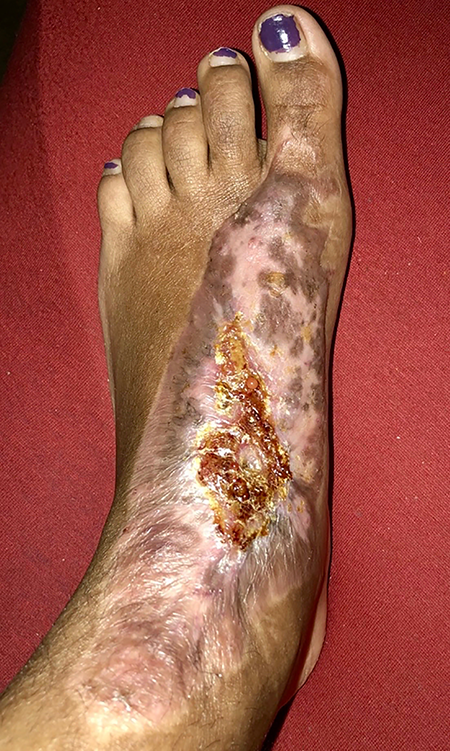
Postoperative day 58.
The second patient is a 10-year-old female with cerebral palsy and spastic diplegia who presented with a surgical site infection after an orthopedic procedure for leg length discrepancy. She was initially taken to the OR by orthopedics for debridement, which left her with a substantial soft tissue defect with exposed hardware (Figure 5). Plastics was consulted and took the patient back to the OR for further debridement and subsequent coverage of the hardware with pedicled medial and lateral gastrocnemius flaps. After flap reconstruction, an approximately 20 cm2 wound remained (Figure 6). At this time, a 6 cm2 full-thickness skin graft was harvested from the ipsilateral thigh; she returned to the OR 5 days later for application of AHSC with placement of a silicone gel membrane layer and wound VAC. Unfortunately, wound volume analysis was not performed preoperatively. On postoperative day 7, the patient underwent a dressing change and exchange of silicone gel membrane layer; at this time, no granulation tissue was noted but the wound appeared clean and free of infection. On postoperative day 14, superficial epithelium formation was noted and 3-D photos were taken with a wound volume of 0.63 cm3 (Figure 7). At 6 weeks after her surgery, her skin graft site was healing well and she was only noted to have a 1-cm linear defect where the skin did not regenerate but that was healing spontaneously (Figure 8). At 7 weeks, repeat 3D imaging showed a wound volume of 0.04 cm3.
Before flap reconstruction.
After flap reconstruction.
Postoperative day 14.
Postoperative day 48.
Discussion
In the realm of wound care, autologous skin grafts (autografts) remain the gold standard in the treatment of skin loss. 1,4 In the case of extensive skin defects secondary to burns, trauma, or oncologic resection, healthy donor skin can be scarce; this poses a challenge to reconstructive surgeons, especially in the pediatric population where surface area is already limited. 5 In addition, split-thickness skin grafts (STSGs) often create an undesirable cosmetic result, contracture limits their use on or near joints, and the donor site often causes more discomfort than the graft itself. 6,7 In response to the need for an alternative to autologous skin grafting, various skin substitutes have been engineered as alternatives to traditional autografts. These substitutes aim to restore function and provide durable barrier protection to underlying structures, all while appearing cosmetically acceptable with appropriate pigmentation. 4
The acellular skin substitutes are likely the most well-known and widely used in the treatment of soft tissue defects. Examples include Integra, which uses silicone, collagen, and shark chondroitin-6-sulphate, and Alloderm, created from decellularised cadaveric dermis. 2,5,7 Both are used as dermal substitutes that can provide coverage to decrease fluid loss and allow for revascularization and dermal cell repopulation. 2,4 These dermal substitutes prepare the wound bed to later allow autologous split-thickness skin grafting. Without cells, however, these skin substitutes provide suboptimal barrier function and inadequate longevity. 2 In response to the shortfalls of decellularised options, tissue engineers have experimented with the addition of neonatal fibroblasts or cultured autologous keratinocytes to existing matrices to create cellular skin substitutes that promote regeneration through secretion of cellular growth factors. 8 The use of cultured autologous keratinocytes, however, has declined due to issues with poor barrier function and unpredictability compared to STSGs. 9 A newer option for epidermal regeneration is cell-spray grafting technologies which allows for a small sample of donor tissue that is used to form a suspension of dermal and epidermal cells that can be sprayed over an area of full-thickness injury. 4,10 One drawback to this strategy, however, is that the skin appendages such as hair follicles and glands are lost in the process.
Autologous homologous skin construct technology offers a means of regenerating full-thickness skin that is functionally polarized. Functional polarity allows cells to reorient themselves into the correct layers (epidermis, dermis, and hypodermis) and allows regeneration of appendages such as hair follicles and glands. 3,11 Autologous homologous skin construct is an autologous, homologous product derived from a small full-thickness sample of the patient’s skin. This sample is manufactured into AHSC and arrives back at the original hospital 48 to 72 hours later. The product is delivered in a syringe containing a suspension of polarized skin cells that can then be used in the place of or in addition to skin grafts to regenerate fully functional skin over the defect site. 3 Here, we have presented 2 cases of pediatric injuries in which AHSC was used to avoid donor site morbidity and regenerate functional skin. Although early case reports such as these are encouraging, additional studies are needed to determine the barrier function, longevity, and long-term cosmetic outcome of this product.
Footnotes
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: The senior author, Dr Anand Kumar, has a reportable conflict of interest as past clinical advisor to PolarityTE, Inc, from February 2017 to February 2019, with stock option compensation of >10 000. Nicholas Sopko is a developmental officer for PolarityTE who played a limited advisory role in the study.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Statement of Human and Animal Rights
All procedures followed were in accordance with the ethical standards of the responsible committee on human experimentation (institutional and national) and with the Helsinki Declaration of 1975, as revised in 2008 (5).
Statement of Informed Consent
Informed consent was obtained from all patients for being included in the study.
