Abstract
Introduction
Blindness and visual impairment are significant and growing public health concerns as the US population ages. An estimated 7 million Americans are visually impaired, with 1 million meeting the definition of legal blindness, and this number is expected to double by 2050. 1 In older adults, age-related macular degeneration and glaucoma are among the leading causes of permanent vision loss, whereas diabetic retinopathy is the major cause in the working-age population.2–4 Loss of vision has profound functional consequences as it diminishes the ability to perform activities of daily living such as reading, driving, and self-care, which can lead to loss of independence.
There is also mounting evidence that vision loss is associated with adverse mental health outcomes. A recent study found that 31% of adults with ocular disease experience anxiety symptoms. 5 Similarly, a meta-analysis found that patients with chronic eye diseases are 1.6 times more likely to suffer depression than those without, with an estimated 25% prevalence of depression among eye clinic patients with visual impairments. 6 The mechanisms linking vision loss to mental health problems are multifactorial. Vision loss often leads to loneliness, social isolation, and feelings of fear and worry about the future. 7 Patients with significant vision loss often lose the ability to perform everyday tasks and develop a loss of confidence in navigating the world, which can result in diminished self-esteem and social withdrawal. 8 Additional contributors to depression in older adults include the burden of ongoing treatments, uncertainty about prognosis, and the lack of social support networks, all of which can contribute to anxiety and depression. 7
Older adults are already a vulnerable group with respect to mental health. Late-life depression is common in the geriatric population, with an estimated 10%–20% of adults over the age of 60 years reporting experiencing depression. 9 Furthermore, depression in the elderly is often underdiagnosed and is associated with significant disability and mortality. 9 One particularly tragic outcome of untreated depression is suicide. Older adults have a disproportionately high suicide rate. A total of 22% of suicide-related deaths in the US have been found to occur in individuals older than age 65 years, despite this age group accounting for only 17% of the population.10,11 Moreover, suicide attempts in the elderly tend to be more lethal, with approximately 1 in 4 attempts resulting in death, compared with only 1 in 200 attempts resulting in death in young individuals. 11 Given that the loss of vision could further compound mental health and hopelessness in an older patient, there is a need for large-scale quantitative data to understand the extent to which vision loss may be associated with the risk of depression, anxiety, and suicidal behavior.
To fill this gap, we conducted a retrospective cohort study identifying patients who received their first diagnosis of blindness, due to any cause, at age ≥60 years, and compared the risk of depression, anxiety, and suicide in this group with that in a matched control group of subjects with normal vision. By quantifying these risks and their timing, our aim is to highlight the mental health impact of blindness in the elderly and underscore the importance of screening and supporting patients who lose vision.
Methods
Study Design
The study was designed as a retrospective cohort study, with data obtained using the TriNetX Analytics Network, a federated network of de-identified electronic health records from numerous healthcare organizations. This platform enabled the aggregation of clinical data across diverse institutions in real time. The study was exempt from institutional review board approval, as only de-identified data were analyzed. The study was conducted in accordance with the principles of the Declaration of Helsinki and follows the Strengthening the Reporting of Observational Studies in Epidemiology guidelines for cohort studies.
Study Population
Data for this study were collected on June 27, 2025, from TriNetX, with a study period spanning from 2006 to 2024. Diagnoses are represented in TriNetX using the International Classification of Diseases, Tenth Revision, Clinical Modification (ICD-10-CM) code set and Current Procedural Terminology (CPT) codes. Detailed coding information is provided in Supplemental Table 1.
The exposure cohort consisted of patients ages ≥60 years with a diagnosis of blindness, as defined by ICD-10-CM codes designating blindness of both eyes (H54.0X), blindness of 1 eye with low vision in the fellow eye (H54.1X), blindness of 1 eye (H54.4X), and legal blindness (H54.8X). These codes are only used in patients who meet category level 3 or above blindness (visual acuity worse than 20/200 Snellen). To ensure that the diagnosis was attributable to permanent vision loss, a second instance of the code was required at least 1 year after the initial use of the code. The control cohort was selected from among patients who underwent routine eye examinations and whose electronic health records reported CPT codes of 92002 or 92004, followed by a second ophthalmic visit with recorded CPT codes of 92002, 92004, 92012, or 92014 at 1 year after the initial visit. This ensured that the control subjects were followed up by an eye care provider who could document whether they had reached the blindness threshold. The control cohort could not have any ICD-10-CM codes designating legal blindness or low vision (codes H54.0X, H54.1X, H54.4X, H54.8X, H54.2X, and H54.5X). A flow chart depicting cohort selection can be found in Figure 1.
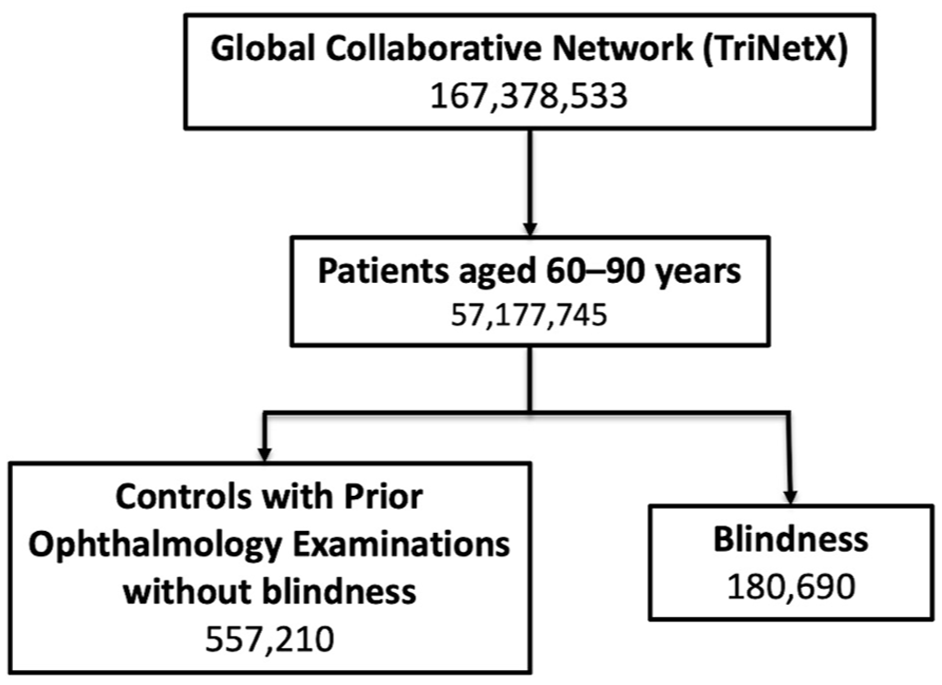
Flow chart depicting cohort selection using de-identified TriNetX Analytics Network electronic health records data from 2006 to 2024, selecting patients ages ≥60 years with a diagnosis of blindness based on International Classification of Diseases, Tenth Revision, Clinical Modification codes. Patients were propensity score matched with a control group of subjects who underwent routine ophthalmology examinations and had normal vision.
Covariates
Baseline demographic variables, including age, race, ethnicity, and gender, classified according to available data in the electronic health records, were obtained. Additionally, diagnoses that could potentially be associated with depression, anxiety, or suicide were queried in both cohorts. These diagnoses, based on ICD-10-CM codes, included history of depressive disorders (F32, F33, F34, F39), anxiety disorders (F41), bipolar disorder (F31), and relevant comorbid conditions such as sleep disorders (G47), chronic pain (including fibromyalgia [M79.1] and chronic pain [G89.2]), schizophrenia (F20–F29), neurodegenerative diseases (Parkinson’s disease [G20]), and substance use (Z72.0, F17, F10, F11). Additionally, Systemized Nomenclature of Medicine (SNOMED) codes were assessed to document the use of antidepressant medications (CN609) and tricyclic antidepressants (CN601).
Propensity score matching (PSM) was performed to match patients with control subjects while controlling for potential confounders such as age, gender, race, ethnicity, and the presence of relevant comorbidities. PSM was conducted using the TriNetX built-in analysis platform, employing 1:1 matching with a nearest-neighbor greedy algorithm and a caliper of 0.25 standard deviations.
Outcomes
Primary outcomes included the incidence and relative risk (RR) of major depressive episodes (ICD-10-CM code F32), persistent major depression (ICD-10-CM codes F33, F34), anxiety disorders (ICD-10-CM code F41), and suicide attempts (ICD-10-CM code X71–X83, T14.91X) at multiple time points after the initial diagnosis of blindness according to ICD-10-CM codes.
Secondary Subgroup Analysis
The study further explored the primary outcomes by stratifying the cohorts into 2 groups: those with a history of depression (ICD-10-CM codes F32, F33, F34) and those without a history of depression. In this stratified analysis, only anxiety disorders and suicide rates were considered as outcomes.
Validation Analysis
To enhance the rigor of the study, a second validation analysis was performed. This analysis utilized a control group of patients with astigmatism (ICD-10-CM code H52.2), ensuring that the same outcomes, covariates, and statistical methods were applied to this group. The results from the validation analysis were compared with those from the primary analysis to assess consistency and reliability.
Statistical Analysis
Statistically significant differences between groups were determined using 2-sided paired t-tests. A P value less than .05 was considered statistically significant. The RRs with 95% confidence intervals (95% CIs) were calculated for all primary and secondary outcomes.
Results
Baseline Characteristics
Before matching, inclusion criteria were met by 168 399 patients in the blindness cohort and 484 416 in the control cohort. After matching, both cohorts included 143 668 patients each. The mean (±SD) age after PSM was 70.9 ± 11.4 years in the blindness cohort and 70.4 ± 10.8 years in the control cohort. Males comprised 49.2% of the blindness cohort and 50.9% of the control cohort. Racial distribution in the blindness cohort was 62.8% White, 16.7% Black or African American, and 3.6% Asian, while the control cohort showed a similar racial distribution, with 60.4% White, 18.4% Black or African American, and 3.4% Asian. Ethnically, 64.5% of patients with blindness self-reported as not Hispanic or Latino, while 7.0% identified as Hispanic or Latino. Similarly, 63.0% of the control cohort identified as not Hispanic or Latino, and 7.2% identified as Hispanic or Latino.
Regarding comorbidities, 13.8% of both cohorts had sleep disorders, 13.3% of the blindness cohort and 13.4% of the control cohort had depressive episodes, 11.2% of the blindness cohort and 11.1% of the control cohort had other anxiety disorders, and 10.3% of the blindness cohort and 10.2% of the control cohort had chronic pain. In the blindness cohort, 20.6% of patients reported use of antidepressant treatments and 4.1% reported use of tricyclic antidepressants; similar frequencies were seen in the control cohort, with 21.1% reporting use of antidepressants and 4.1% reporting use of tricyclic antidepressants. Full characteristics before and after matching are shown in Table 1.
Baseline Characteristics of Patients Ages ≥60 Years With Blindness and Control Subjects With Normal Vision, Before and After Propensity Score Matching.
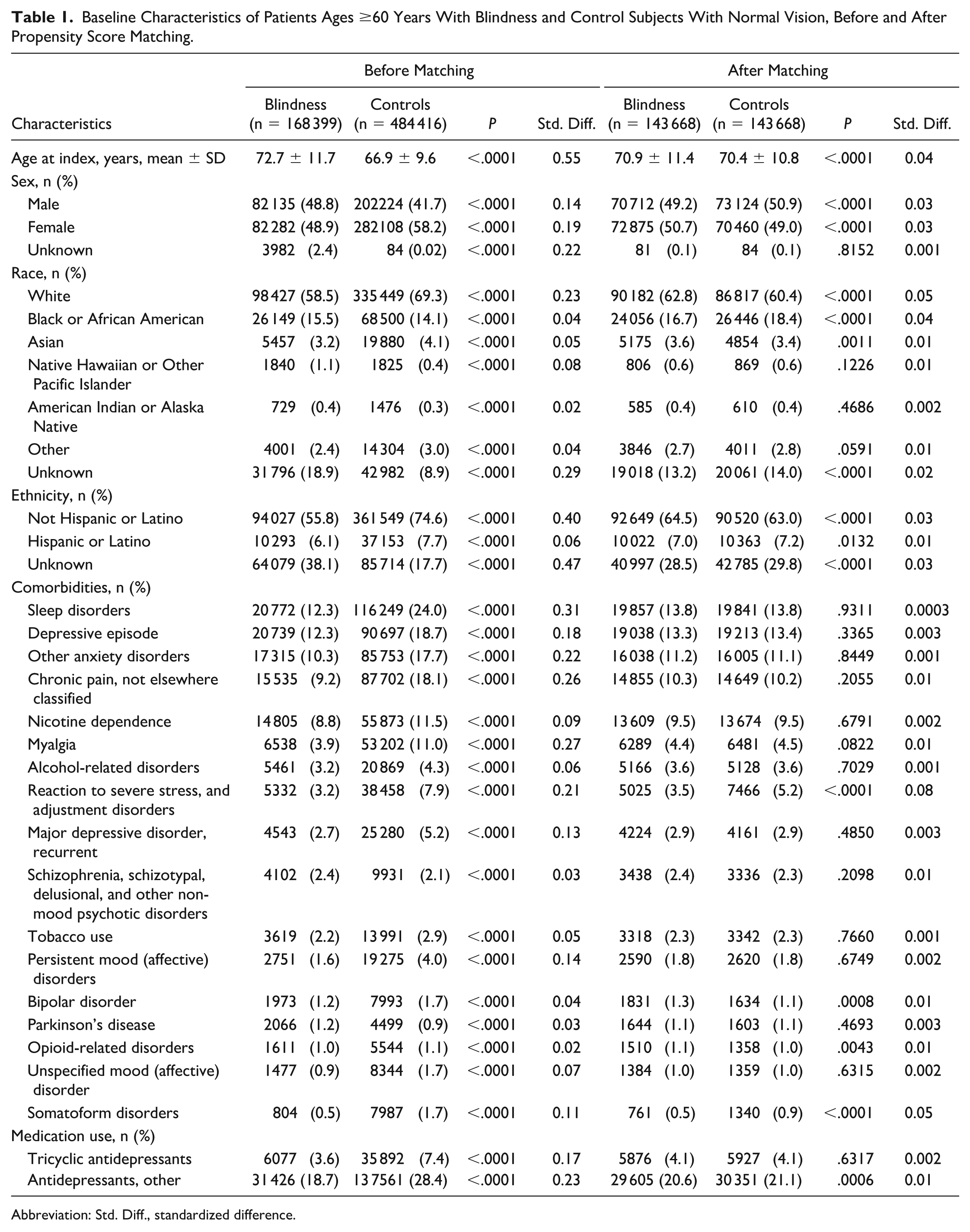
Abbreviation: Std. Diff., standardized difference.
Risk of Depression, Anxiety, and Suicide Attempts
The cumulative rates of depressive episodes, major depression, anxiety, and suicide attempts were recorded over the 1-, 3-, and 5-year follow-up periods for patients with blindness compared with matched controls (Table 2). After PSM, both groups included 143 668 patients at each time point.
Risk of Depressive Episodes, Major Depression, Anxiety, and Suicide Attempts in Patients Ages ≥60 Years With Blindness Compared With Control Subjects With Normal Vision.
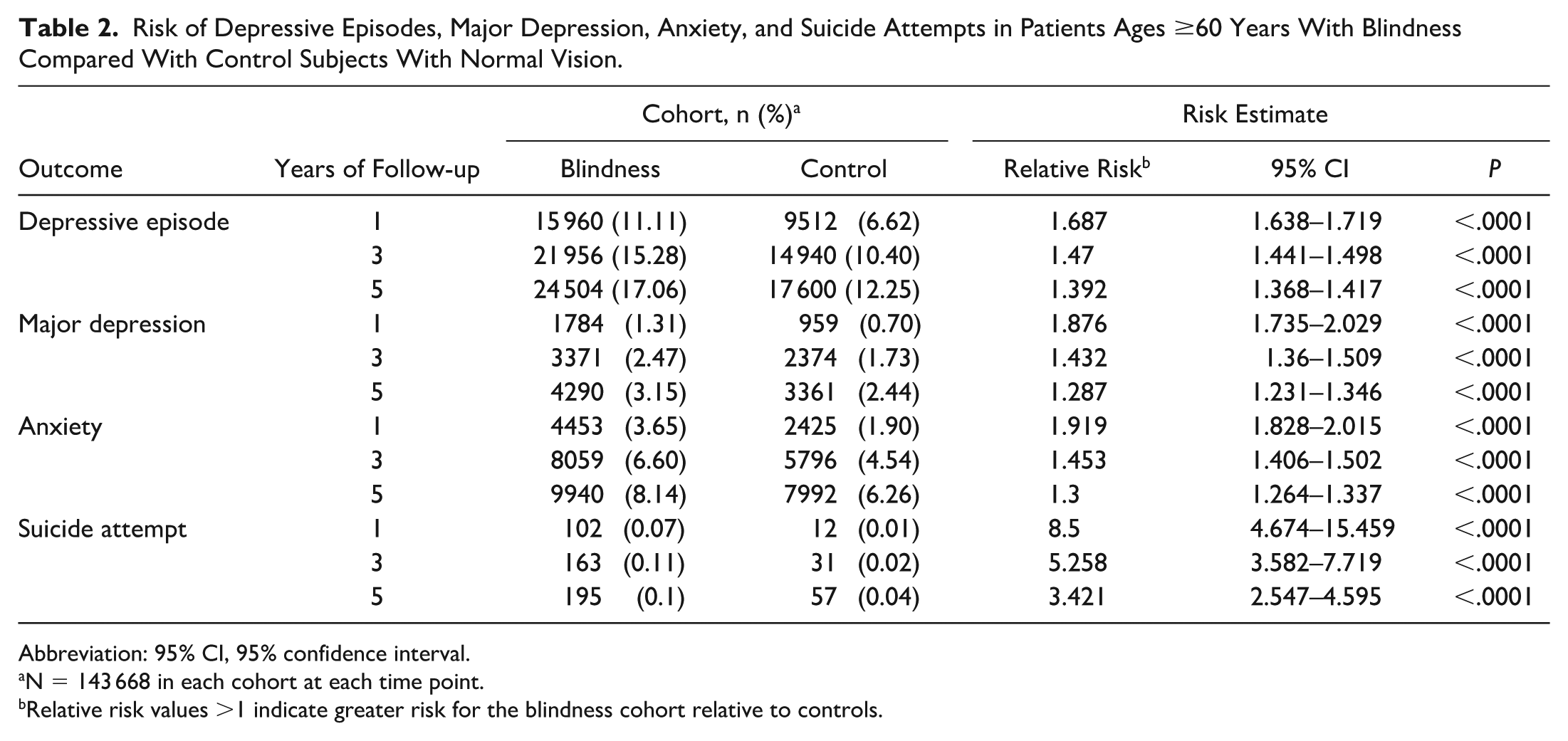
Abbreviation: 95% CI, 95% confidence interval.
N = 143 668 in each cohort at each time point.
Relative risk values >1 indicate greater risk for the blindness cohort relative to controls.
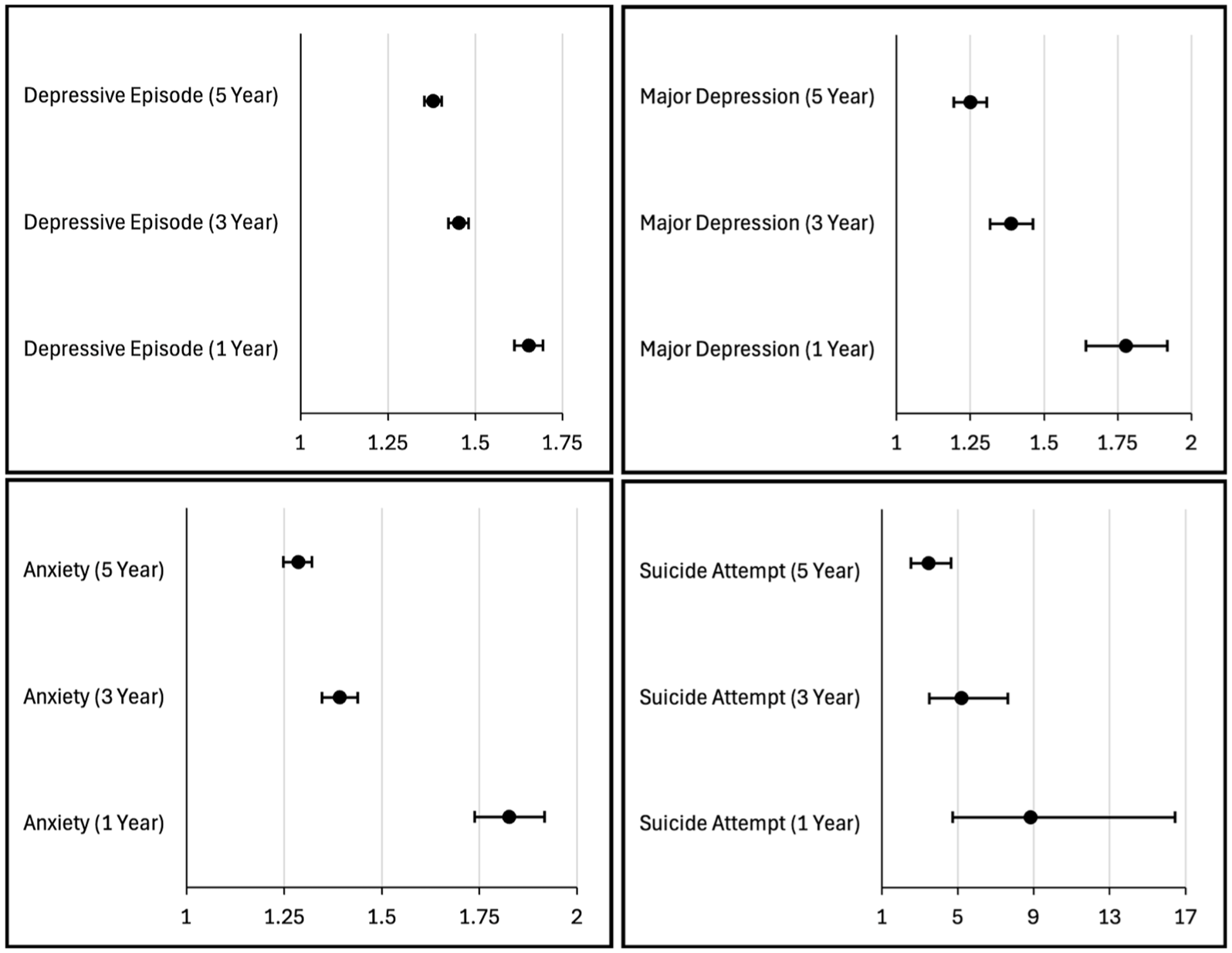
Patients with blindness were found to have a higher risk of major depressive episodes compared with controls at all time points. Incidence of depressive episodes at 1 year was 11.11% in the blindness cohort compared with 6.62% in the controls (RR, 1.69; 95% CI, 1.64–1.72), with similar trends in increased risk of depressive episodes at 3 years (RR, 1.47; 95% CI, 1.44–1.50) and 5 years (RR, 1.39; 95% CI, 1.37–1.42). The risk of a new diagnosis of persistent major depression was also more common in the blindness cohort, with an increased risk observed at 1 year (RR, 1.88; 95% CI, 1.74–2.03), 3 years (RR, 1.43; 95% CI, 1.36–1.51), and 5 years (RR, 1.29; 95% CI, 1.23–1.35).
A higher incidence of anxiety was noted in the blindness cohort compared with controls, including increased risk at 1 year (RR, 1.92; 95% CI, 1.83–2.02), 3 years (RR, 1.45; 95% CI, 1.41–1.50), and 5 years (RR, 1.30; 95% CI, 1.26–1.34). Moreover, suicide attempts were significantly more common in the blindness cohort, with an increased risk at 1 year (RR, 8.50; 95% CI, 4.67–15.46), 3 years (RR, 5.26; 95% CI, 3.58–7.72), and 5 years (RR, 3.42; 95% CI, 2.55–4.60). These results are summarized in Figure 2.
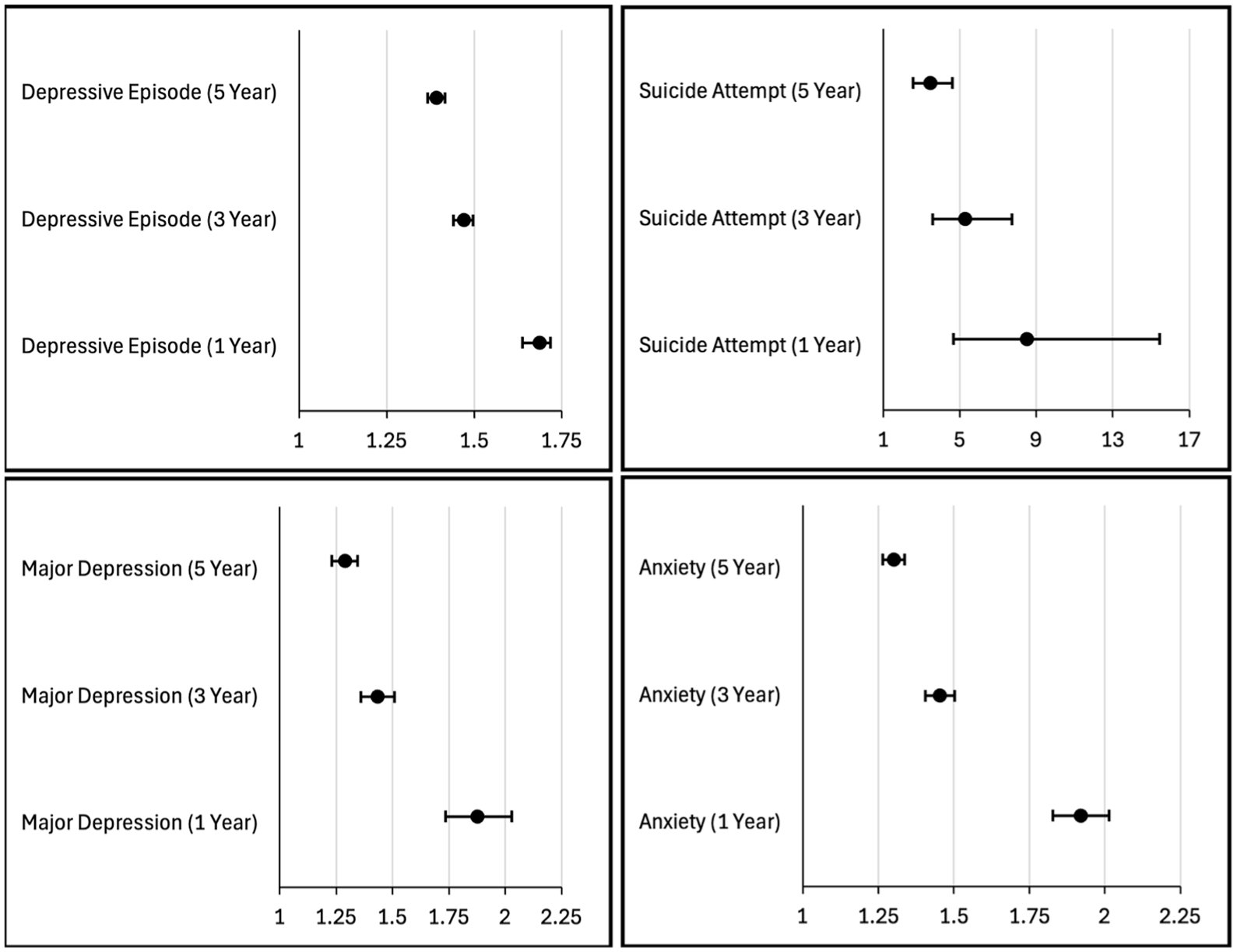
Risk of depressive episodes, suicide attempts, major depression, and anxiety recorded over the 1-, 3-, and 5-year follow-up periods for patients with blindness compared with matched controls with normal vision. Values are the relative risk with 95% confidence intervals for each outcome in the blindness group relative to controls.
Outcome Risks in Patients With and Those Without a History of Depression
A secondary analysis was performed comparing both cohorts according to those with a history of depression (N = 27 337) (ICD-10-CM codes F32, F33, F34) and those without a history of depression (N = 20 503). In the subgroup with a history of depression, patients with blindness had higher rates of anxiety compared with controls at 1 year (RR, 1.46; 95% CI, 1.33–1.59), 3 years (RR, 1.17; 95% CI, 1.11–1.25), and 5 years (RR, 1.06; 95% CI, 1.01–1.12). Suicide attempts were also significantly more common in the subgroup of patients with blindness who had a history of depression, with an increased risk of suicide attempts at 1 year (RR, 8.00; 95% CI, 5.07–15.72), 3 years (RR, 4.33; 95% CI, 2.70–6.96), and 5 years (RR, 3.64; 95% CI, 2.40–5.53).
In patients without a history of depression, blindness was associated with significantly higher rates of anxiety compared with controls at all time points. The rate of anxiety at 1 year was 2.33% in the blindness cohort compared with 1.36% in controls (RR, 1.71; 95% CI, 1.60–1.83), with similar trends in increased risk of anxiety at 3 years (RR, 1.23; 95% CI, 1.17–1.28) and 5 years (RR, 1.09; 95% CI, 1.05–1.13). In those without a history of depression, suicide attempts were more common in the blindness cohort than in the control cohort at 3 years (RR, 3.40; 95% CI, 1.68–6.88) and 5 years (RR, 2.35; 95% CI, 1.39–3.97), but not at 1 year (P = .068). These results are summarized in Figure 3.
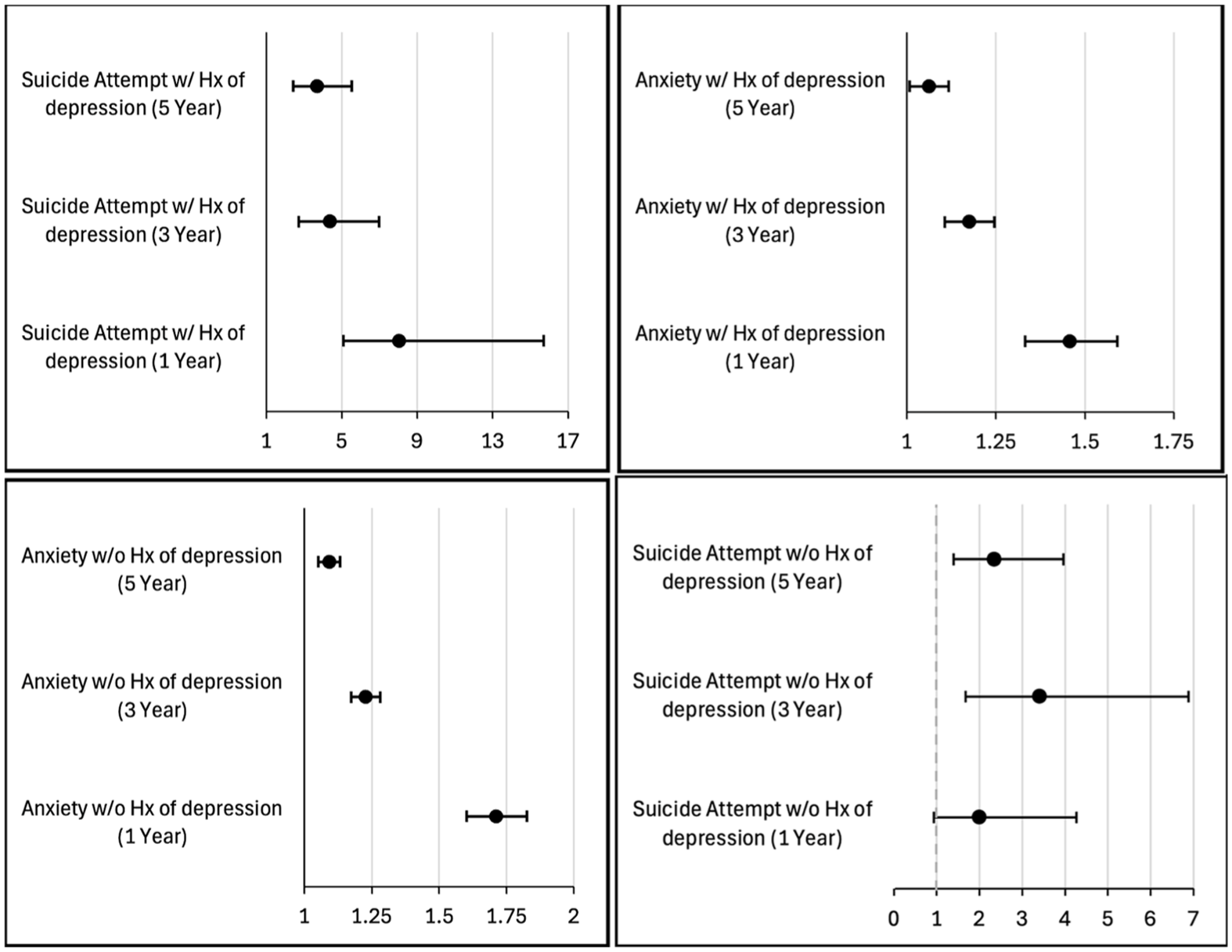
Risk of suicide attempts and anxiety recorded over the 1-, 3-, and 5-year follow-up periods in patients with blindness compared with matched controls with normal vision according to subgroups of those who had a history (Hx) of depression and those who did not have a history of depression. Values are the relative risk with 95% confidence intervals for each outcome in the blindness group relative to controls.
Outcome Risks in Validation Analyses
A secondary validation analysis using a control group of patients with astigmatism (N = 284 518) was performed to enhance the rigor of the study. PSM was performed to ensure balance in comorbidities between these controls and those in the blindness cohort (Table 3). The validation analysis showed that the blindness cohort remained at elevated risk of depressive episodes, major depression, anxiety, and suicide attempts at all time points, with RRs that overlapped those found in the primary analysis (Table 4). These results are summarized in Figure 4.
Risk of Anxiety and Suicide Attempts in Patients Ages ≥60 Years in the Blindness and Control Cohorts With or Without a History of Depression.
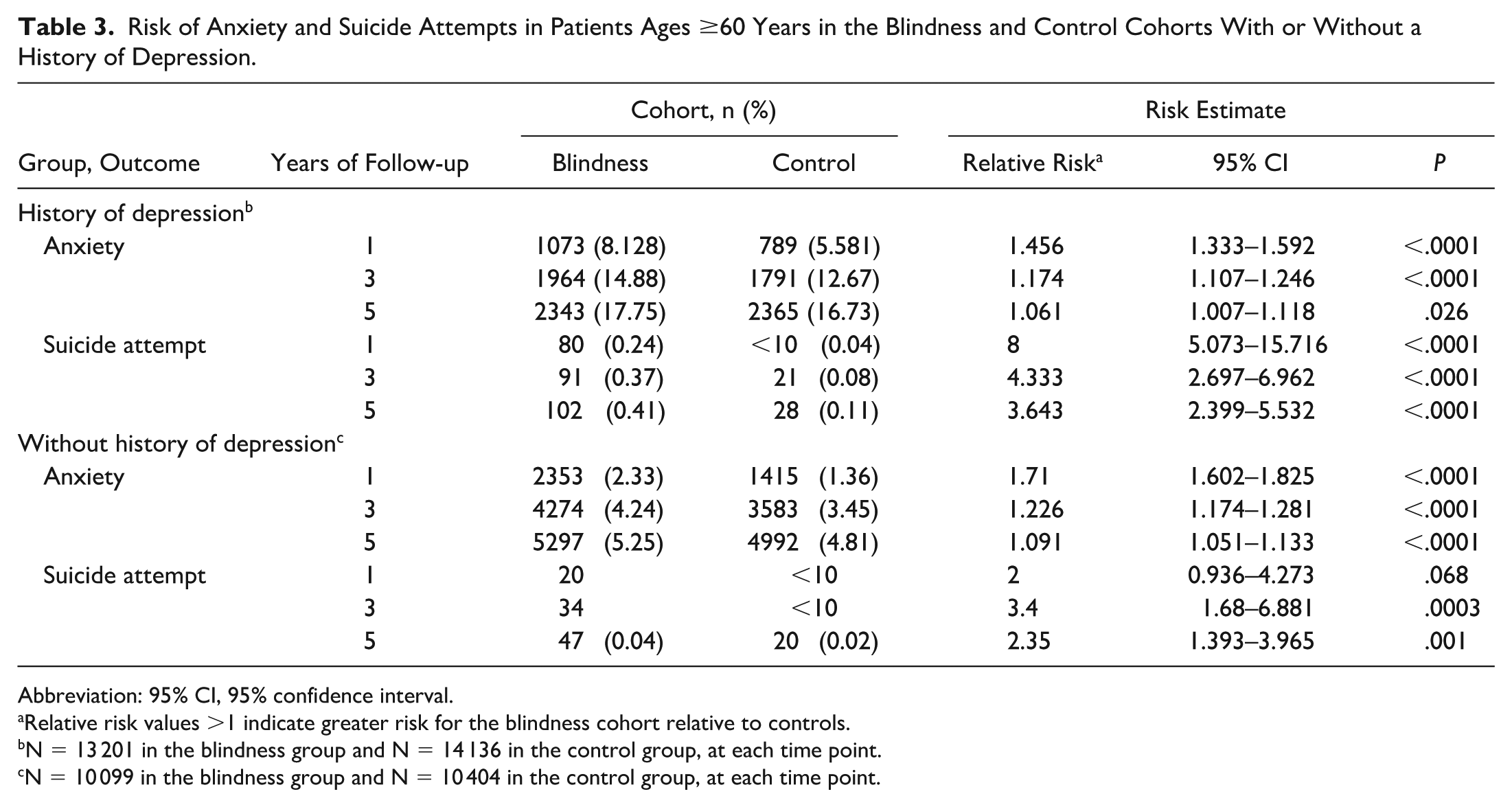
Abbreviation: 95% CI, 95% confidence interval.
Relative risk values >1 indicate greater risk for the blindness cohort relative to controls.
N = 13 201 in the blindness group and N = 14 136 in the control group, at each time point.
N = 10 099 in the blindness group and N = 10 404 in the control group, at each time point.
Risk of Depressive Episodes, Major Depression, Anxiety, and Suicide Attempts in Patients Ages ≥60 Years in the Blindness Cohort Compared With a Validation Control Cohort of Patients With Astigmatism.
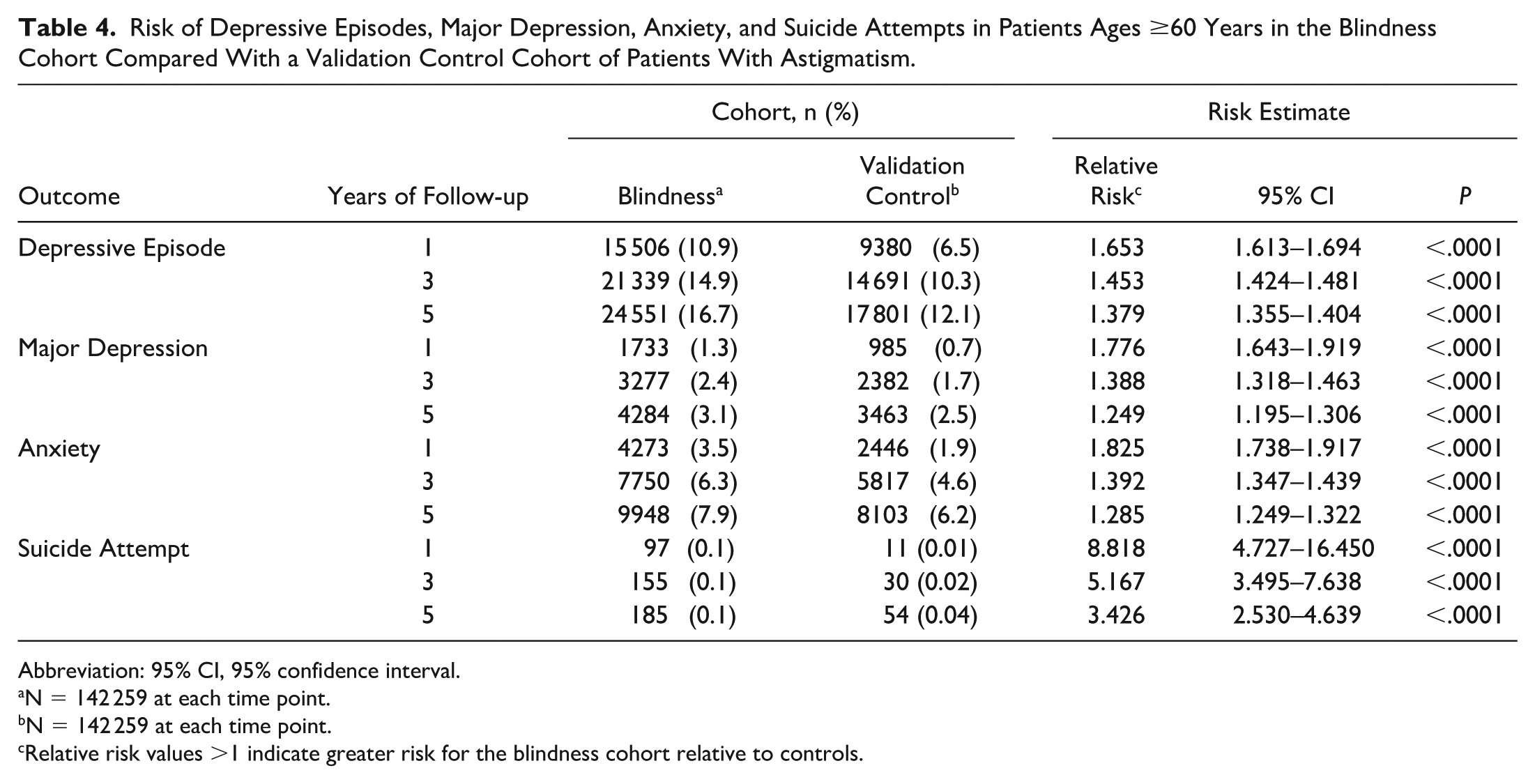
Abbreviation: 95% CI, 95% confidence interval.
N = 142 259 at each time point.
N = 142 259 at each time point.
Relative risk values >1 indicate greater risk for the blindness cohort relative to controls.
Risk of depressive episodes, major depression, anxiety, and suicide attempts recorded over the 1-, 3-, and 5-year follow-up periods in the validation analysis of patients with blindness compared with matched control subjects with astigmatism. Values are the relative risk with 95% confidence intervals for each outcome in the blindness group relative to controls.
Discussion
The current study demonstrated that patients ages ≥60 years who were diagnosed as having blindness in 1 or more eyes had significantly higher rates of depression, anxiety, and suicidal behavior compared with matched controls. These differences were greatest in the first year after diagnosis but remained elevated up to 5 years after diagnosis. Although the absolute rates of suicidal behavior were low, at 0.07%, in patients with blindness, this represented an 8-fold risk compared with matched controls.
These results align with and extend the existing literature on vision impairment and mental health. Previous cross-sectional studies have found associations between depression and visual impairment, with 1 meta-analysis indicating that the prevalence of a depression diagnosis in patients with blindness was 25%, 6 which is similar to the rate reported in the present study, at 22%. A longitudinal study from Korea similarly reported on the effect of visual impairment in increasing the risk of depression, with an overall hazard ratio of 1.19, which was even higher in patients who were legally blind (hazard ratio 1.31). 12 The RRs for a diagnosis of depression were higher in the present study than in the study from Korea, varying between 1.29-fold and 1.88-fold increased risk relative to controls. These differences may highlight other contributing factors in the development of depression that cannot be captured in billing database studies, such as social support, which is often more common in Asian countries and lacking in older adults. 13
The current study also found high rates of development of new anxiety disorders following the diagnosis of blindness, for which prior data have been less abundant. This is important, as studies have found a compounding negative effect on patients with multiple comorbid psychiatric conditions. 14 Most importantly, the current study found evidence of an increased suicide risk after diagnosis of blindness. Even though the absolute incidence of suicide was low, at 102 cases or 0.07% at 1 year, it must be interpreted in the context of lethal outcomes of suicide attempts in the geriatric population. Older adults have the highest suicide completion rates of any age group, at 25%, meaning that this uptick represents a serious precipitating factor for life-threatening behavior.10,11
The current study also found that the increased risk of depression, anxiety, and suicide attempts was most pronounced in the first year after diagnosis of blindness, with the RRs still significantly different but tapering by 5 years. This pattern suggests that there is an acute psychological impact immediately following the loss of vision. The first year likely represents a critical adjustment period during which patients grapple with their vision loss. Prior research has described a similar phenomenon in which depression was more common in patients in ophthalmology after receiving an irreversible eye diagnosis compared with those in long-term vision rehabilitation services. 15 It is likely that the decreases in the risks of depression, anxiety, and suicide attempts over time in the current study are indicative of patients adapting to their vision loss, receiving rehabilitative treatment, and/or social support. Studies have demonstrated that engaging in low-vision rehabilitation can improve the patient’s ability to cope with their reduced vision, leading to lower rates of depression, highlighting the importance of such interventions.16,17 In 1 study, a nonsignificant improvement in depression scores was observed following low-vision rehabilitation. 16 Nonetheless, even at 5 years, the depression and anxiety rates in patients with blindness remained significantly higher than in the controls, indicating that the mental health disparity does not fully resolve.
The findings of the current study have significant implications from a public health and clinical standpoint. At present, mental health screening is not uniformly standard in eye care settings, but there is growing recognition that such screening may be beneficial. The United Kingdom’s National Institute for Health and Care Excellence recommends that all patients with visual impairment be screened with 2 questions for identifying whether depression and anxiety are present. Research has shown that even using simple brief screening tools within ophthalmology clinics is feasible and can uncover a substantial number of unrecognized cases of depression. 18 In 1 study of older patients with age-related macular degeneration, up to 44% had significant symptoms of depression warranting intervention, with many having never been diagnosed or treated for depression with a screening intervention. 19 The results of the current study suggest that the period soon after a blindness diagnosis is the critical window in which such screening and intervention should occur. By proactively addressing mental health, many studies have demonstrated better patient outcomes as well as lower rates of suicide. 20 By combining approaches with the visual rehabilitation that ophthalmology provides can ensure the best possible outcomes for patients with low vision.
Finally, the secondary analysis examining the impact of a history of depression revealed that both cohorts remained at elevated risk for developing chronic anxiety and suicidal behavior. As expected, patients with a preexisting diagnosis of depression exhibited higher overall rates of these outcomes, consistent with prior literature.21–23 Previous studies have demonstrated that individuals with a history of psychiatric illness are more likely to develop a new mental health disorder than controls. 24 While these findings underscore the importance of screening all patients for emerging psychiatric symptoms, they also highlight the particular need for enhanced monitoring and support for individuals with a known history of depression. Notably, the observed rate of suicide attempts of 0.24% in patients with a history of depression in the first year after diagnosis of blindness is particularly high and nearly 9 times higher than the rates reported among individuals with a new diagnosis of terminal cancer. 25
Although the current study sheds light on many important topics, it has several limitations. First, as an observational retrospective analysis using electronic health records, the findings may be limited by the level of coding accuracy, as TriNetX does not have access to visual acuity data. The accuracy of this study depends on consistent use of ICD-10-CM billing codes, which define legal blindness as visual acuity of 20/200 or worse or a visual field of 20 degrees, category 3 blindness as worse than 20/200, category 4 as worse than 20/1200, and category 5 as no light perception. However, some patients who met these criteria may have been coded only for their underlying ocular disease (eg, subfoveal geographic atrophy) rather than with a blindness-specific code. In addition, many institutions tend to apply these codes primarily when patients require documentation for disability benefits or other vision-related services, which may limit their capture in routine clinical practice.
Similarly, depression and anxiety may be undercoded. Many older adults with depressive symptoms do not seek mental health care and may misattribute symptoms to normal aging, and many studies have demonstrated a high rate of underdiagnosis. 26 This potential underestimation would bias the results toward the null, so that the true mental health impact could be even greater than that reported in the current study.
Furthermore, the current study also did not differentiate between different causes of blindness. It is possible that the psychological effect of sudden, traumatic vision loss due to a retinal detachment or retinal vascular occlusion might differ from that of a gradually progressive loss, such as macular degeneration or glaucoma. The aggregate approach used in this study could mask such nuances.
Finally, there is the possibility of attrition and selection bias due to loss to follow-up, especially in the latter time points. Patients with severe depression have been found to engage less with the healthcare system, and it is possible they may die, which could lead to an underestimation of the rate of long-term psychiatric incidence in the blindness group. Despite these limitations, the consistency of the results, including those observed in a validation cohort, and the strong effect sizes, enhances confidence in the robustness of the core findings of this study.
In conclusion, the current study provides compelling evidence that older adults who experience significant vision loss have higher rates of developing a major depressive episode, a new diagnosis of persistent major depression, a new diagnosis of anxiety, and suicidal behavior compared with controls with normal vision. These risks are most elevated in the first year after blindness, highlighting the critical window for intervention. As such, there is a critical need to integrate mental health screening and support into the care of patients with serious vision impairment.
Supplemental Material
sj-docx-1-vrd-10.1177_24741264261432179 – Supplemental material for Risk of Depression, Anxiety, and Suicidal Behavior in Older Adults With Severe Vision Loss
Supplemental material, sj-docx-1-vrd-10.1177_24741264261432179 for Risk of Depression, Anxiety, and Suicidal Behavior in Older Adults With Severe Vision Loss by Charles Zhang, Georges AbouKasm, Jonathan B. Lin, Kisha Kalra, Joaquín Sosa Lockward and Audina Berrocal in Journal of VitreoRetinal Diseases
Footnotes
Ethical Approval
The study was exempt from institutional review board approval, as only de-identified data were analyzed. The study was conducted in accordance with the principles of the Declaration of Helsinki and follows the Strengthening the Reporting of Observational Studies in Epidemiology guidelines for cohort studies.
Statement of Informed Consent
Only de-identified data were analyzed in this study, and therefore, informed consent was not required.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material is available online with this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
