Abstract
Introduction
Secondary intraocular lens surgery plays an important role in restoring vision for patients with aphakia, dislocated intraocular lens, or complications after cataract surgery.1,2 Secondary intraocular lens surgery carries substantial societal value, in addition to restoring visual acuity. Patients who regain vision often experience improved independence, are able to return to the workforce, and have an enhanced quality of life. The impact extends beyond individual outcomes, as untreated aphakia or intraocular lens dislocation can lead to functional disability, higher rates of falls, and greater reliance on caregivers and social support systems.3,4
These procedures are often complex and require careful intraoperative techniques to achieve favorable visual outcomes. With an aging population, the demand for cataract surgery is projected to increase by at least 72% by 2036, yielding a corresponding rise in intraocular lens-related complications. 5 Throughout the past 30 years, rates of pseudophakic intraocular lens exchange have increased, with lens dislocations representing a common indication. 6 As healthcare costs continue to escalate, however, questions about whether these complex surgeries are adequately reimbursed persist.
Trends in ophthalmology have shown declining Medicare payments, raising concerns about financial sustainability. Between 2000 and 2021, inflation-adjusted reimbursement for a sample of common retina procedures declined by an average of 32%, despite increasing procedural complexity and a growing patient population. The financial pressures stemming from these cuts may sway practice patterns, particularly for time-intensive procedures that are inadequately reimbursed. Recent time-driven activity-based cost studies in ophthalmology have highlighted disparities between operative costs and reimbursement, particularly for pars plana vitrectomy (PPV) for retinal detachment (RD) repair, scleral buckle surgery, and panretinal photocoagulation.7–10 These studies have shown net financial losses for retinal surgeries performed under current reimbursement figures.
Despite the growing literature evaluating cost-reimbursement imbalances in ophthalmology, secondary intraocular lens surgery remains understudied from a financial perspective. In this context, “secondary intraocular lens surgery” includes 3 scenarios: (1) repositioning a malpositioned or dislocated intraocular len, (2) removing and exchanging a malfunctioning or malpositioned intraocular len, and (3) placing a new intraocular lens in an aphakic eye. A recent study by Pan et al 11 assessed the costs and reimbursement for secondary intraocular lens procedures, suggesting that these surgeries frequently led to net negative margins. While that study quantified financial losses, it did not determine the operative time threshold at which these procedures become unprofitable. Without such analysis, it is unclear how often secondary intraocular lens procedures exceed financially sustainable operative times and whether adjustments to reimbursement rates are needed.
This study evaluates the operative times, costs, and break-even thresholds for secondary intraocular lens surgeries performed with concurrent PPV at a high-volume academic center. By defining the time thresholds necessary to maintain financial viability, these findings may offer insights to support reimbursement advocacy efforts.
Methods
Study Design
We queried the Massachusetts General Brigham Research Patient Data Registry to obtain a retrospective cohort of 97 patients who underwent secondary intraocular lens procedures with PPV at the Massachusetts Eye and Ear between January 1, 2023, and December 31, 2023. Patients suitable for inclusion in this study were identified using Current Procedural Terminology (CPT) codes for secondary intraocular lens repositioning (66825), secondary intraocular lens insertion only (66985), and secondary intraocular lens exchange (66986) (Supplemental Table 1).
Exclusion criteria included patients with aborted procedures, had missing operative notes, or underwent concurrent keratoplasty or glaucoma shunt placement. Patients who received PPV in conjunction with their secondary intraocular lens procedure were identified via retrospective chart review of their operative notes. This study was approved by the Massachusetts General Brigham Institutional Review Board and adhered to the tenets of the Declaration of Helsinki.
Cost Estimation and Profitability Calculations
Maximum reimbursement for each procedure was calculated by summing the physician fee and facility fee disbursed by Medicare for the corresponding CPT code (Supplemental Table 3). In clinical practice, PPV performed in conjunction with secondary intraocular lens implantation is reimbursed at a 50% rate for the lower-paying procedure for both professional and facility fee. We therefore calculated the maximum reimbursement rate for joint secondary intraocular len/PPV cases to reflect this reality (Supplemental Table 3, Appendix). As all patients in our cohort underwent their procedures in an academic hospital setting, we used Medicare hospital reimbursement rates for all calculations.
Cost estimates and procedure break-even times were calculated as previously reported. 7 In brief, hospital costs for operating room personnel, materials, and overhead were collected from Massachusetts Eye and Ear in the year 2021 and adjusted to account for 12.4% inflation between 2021 and 2023 (Supplemental Table 2). The intraocular lens itself was assumed to incur a cost of $0 to the hospital, given separate reimbursement and is therefore not included in the overall cost calculation. Of note, anesthesia costs were calculated strictly based on the duration of anesthesia time for each procedure, regardless of whether the procedure was performed under monitored anesthesia care, general anesthesia, or total intravenous anesthesia.
Total preoperative and postoperative costs were calculated as previously. 7 Costs for pre- and postoperative clinic visits were estimated using previously published studies of similar ophthalmic surgeries. Total day-of-procedure costs for each patient were calculated based on the operative time of their respective procedure, and median costs were then calculated for each secondary intraocular lens procedure category. A full list of costs used in this analysis, and formulas used to calculate total cost per operation, can be found in the Appendix.
Where applicable, costs were adjusted for 12.4% inflation between 2021 and 2023 based on the United States Consumer Price Index. Break-even times for each procedure category were calculated as the time at which the total costs of an operation equaled the estimated Medicare reimbursement for that operation (Appendix). Excess costs per procedure were calculated by subtracting the total procedure cost from expected Medicare reimbursement.
Statistical Analysis
All statistical analyses were performed using Python (version 3.9.13). Data visualization was performed using seaborn (version 0.11.2).
Results
Demographics
We identified 97 secondary intraocular lens procedures performed in 97 unique patients at Massachusetts Eye and Ear in 2023. The most common indications for secondary intraocular lens surgery in our cohort were intraocular lens dislocation (54.4%) and aphakia (40.4%). The median age of patients within this cohort was 67.5 years (IQR, 51.5-83.5 years; range, 23-91 years), and 62.8% of patients were men.
The majority of patients (66.1%) were assigned an American Society of Anesthesiologists Physical Classification score of 2, indicating preoperative diagnosis with 1 or more mild, systemic medical comorbidities. Monitored anesthesia care was utilized for 80 cases (83.3%), general anesthesia for 9 cases (9.4%), total intravenous anesthesia for 7 cases (7.3%), and unspecified for 1 case (1.0%).
Within this cohort, 83 procedures were performed by 1 surgeon, and 14 were performed by 2 surgeons. Within single-surgeon procedures, 8 patients underwent intraocular lens repositioning with PPV, 41 patients had intraocular lens insertion with PPV, and 34 patients received intraocular lens exchange with PPV. Within double-surgeon procedures, 2 patients underwent intraocular lens repositioning with PPV, 5 patients received intraocular lens insertion with PPV, and 7 patients had intraocular lens exchange with PPV.
Cost, Reimbursement, and Break-even Times
Total variable cost per intraocular lens procedure was calculated to be $54.16 per operating room minute. Total materials cost per intraocular lens procedure was calculated to be $1258.46. Total preoperative and postoperative costs of ophthalmologic procedures at Massachusetts Eye and Ear were previously calculated to be $94.66 and $547.55, respectively (Supplemental Table 2). 7
We first calculated the total costs for each secondary intraocular lens procedure and surgeon number category. Median total costs for secondary intraocular lens repositioning were found to be $7081.40 (single-surgeon) and $9399.45 (double-surgeon) (Table 1). Median day-of-surgery costs for secondary intraocular lens insertion were found to be $8348.18 (single-surgeon) and $8794.43 (double-surgeon) (Table 1). Median day-of-surgery costs for secondary intraocular lens exchange were found to be $9055.23 (single-surgeon) and $9686.04 (double-surgeon) (Table 1).
Comparison of Median Costs and Profitability at Median Operative Times for All Single- and Double-Surgeon Secondary Intraocular Lens Procedures, Including Profitable and Unprofitable Cases.

Abbreviation: PPV, pars plana vitrectomy.
Medicare reimbursement rates were found to be $3032 for secondary intraocular lens repositioning alone, $3096 for secondary intraocular lens exchange alone, and $2698 for secondary IOL insertion alone (Supplemental Table 3). PPV performed in conjunction with secondary intraocular lens procedures was reimbursed at 50% proration for both provider and facility fee for the lower reimbursed procedure (Supplemental Table 3).
Based on these cost and reimbursement estimates, we calculated break-even times for each intraocular lens procedure type to maintain profitability. The break-even times for single-surgeon procedures were: secondary intraocular lens repositioning, 71.54 minutes; secondary intraocular lens insertion, 71.00 minutes, and secondary intraocular lens exchange, 72.08 minutes (Table 2). The break-even times for these procedures with 2 surgeons were: secondary intraocular lens repositioning, 66.19 minutes; secondary intraocular lens insertion, 65.68 minutes; and secondary intraocular lens exchange, 66.69 minutes (Table 2).
Break-even Times for Secondary Intraocular Lens Procedures With Concurrent PPV.

Abbreviation: PPV, pars plana vitrectomy.
Next, we calculated the differences between total operative time and break-even time for each secondary intraocular lens procedure category. The median time overages for single-surgeon intraocular lens cases with PPV were: intraocular lens repositioning, 13.96 minutes; intraocular lens insertion, 36.00 minutes; intraocular lens exchange, 46.91 minutes. For double-surgeon cases, the median time overages were: intraocular lens repositioning, 49.31 minutes; intraocular lens insertion, 40.31 minutes; and intraocular lens exchange, 53.31 minutes.
We next calculated the excess cost of each procedure by subtracting the expected Medicare reimbursement from the total operative costs. We found that hospital costs exceeded reimbursement for 85.56% of all procedures in our cohort (Figure 1). Single-surgeon procedures produced median excess costs of −$822.40 for intraocular lens repositioning with PPV (13.14% over reimbursement), −$2121.18 for intraocular lens insertion with PPV (34.10% over reimbursement), and −$2764.23 for intraocular lens exchange with PPV (43.94% over reimbursement) (Table 1). Double-surgeon procedures produced median excess costs of −$3140.45 (50.17% over reimbursement), −$2567.43 (41.23% over reimbursement), and −$3395.04 (53.97% over reimbursement) for intraocular lens repositioning, insertion, and exchange, respectively (Table 1).
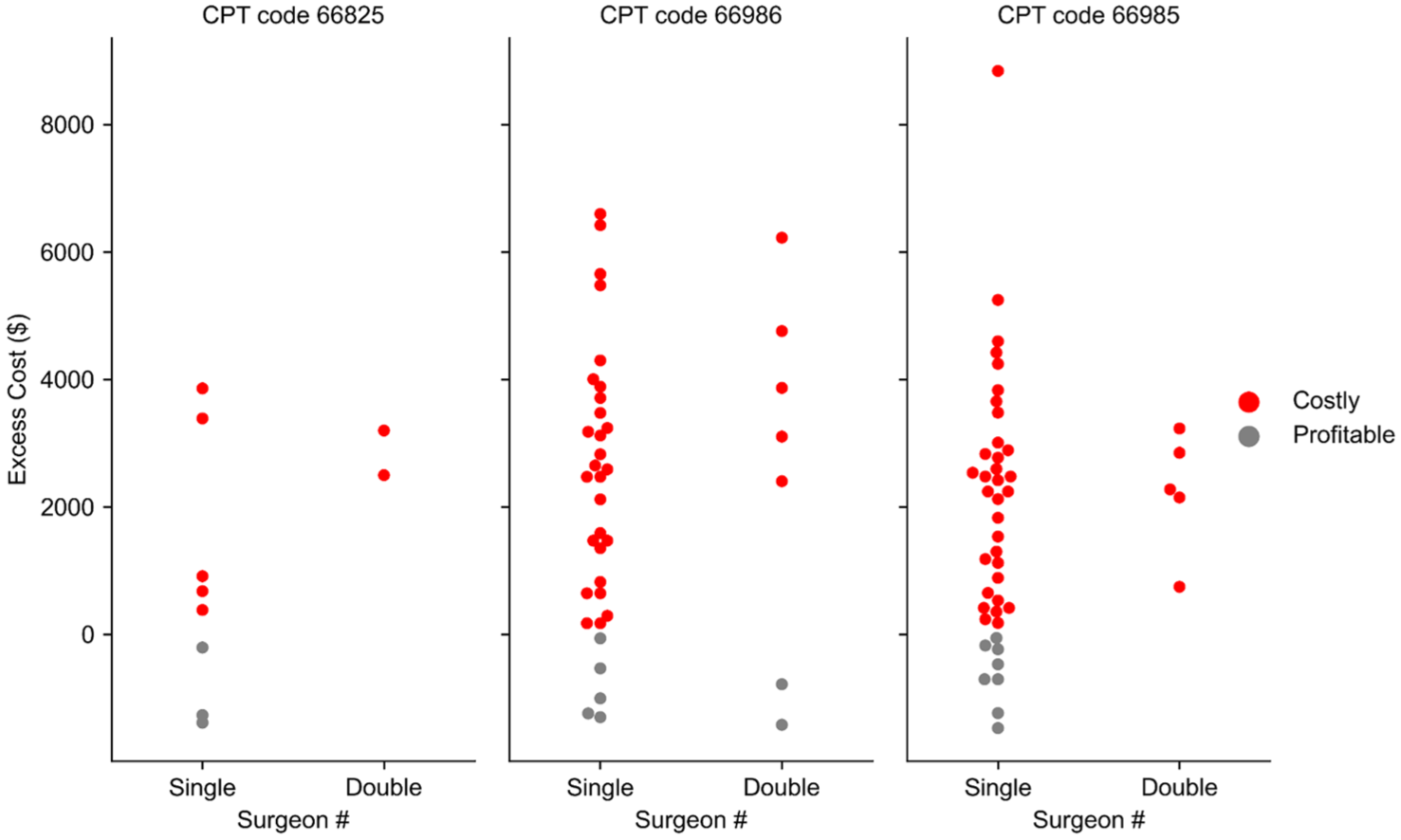
Excess cost in dollars for secondary intraocular lens procedures with pars plana vitrectomy, grouped by surgeon number. Each point represents an individual procedure. Point color reflects whether cases produced net negative costs (red) or net positive earnings (grey) at expected Medicare reimbursement rates.
Conclusions
Our study of secondary intraocular lens procedures performed at a large academic center in 2023 showed that the break-even times to maintain profitability with PPV were approximately 75 minutes. More than 85% of all secondary intraocular lens procedures evaluated exceeded the Centers for Medicare & Medicaid Services reimbursement rate, with both single-surgeon and double-surgeon procedures being performed at a net financial loss. No clear unifying factor (eg, patient demographics, number of surgeons, type of procedure) was found among the small proportion of profitable cases.
Intraocular lens exchange with PPV was associated with the greatest excess costs, reaching nearly −$2800 in single-surgeon cases and −$3400 in double-surgeon cases. Importantly, the median operative times for secondary intraocular lens surgery across procedure types and number of surgeons exceed the corresponding break-even times by tens of minutes—a time deficit that implies an imbalance between expected operative times and reality.
Recent studies have begun to characterize the time-based costs and associated reimbursements of various retina procedures, including PPV for RD repair,8–10 scleral buckle surgery,7,12 and panretinal photocoagulation, 13 among others.14–17 Such investigations are important, as retina surgeons are referred complex patients who require procedures that often entail substantial operative times.
In the past 2 decades, inflation-adjusted Medicare reimbursements for common retina procedures decreased significantly. A recent report by Li and Adelman 18 showed a decline in reimbursement in 29 of 38 retina procedures between 2000 and 2021, with an average decrease of 32% after adjusting for inflation. Procedure volume over the same period decreased in 20 of 38 retina procedures, despite a growing and aging patient population.
Inadequate reimbursement carries the potential of decreasing access to certain vision-saving surgeries, especially for vulnerable patients, as community retina specialists are forced to refer complex, time-intensive cases to academic medical centers to remain viable. We have previously shown a trend toward more complex, longer RD repair surgeries being performed by an academic medical center’s retina practice when compared with community surgeon cases. 19
To our knowledge, there is only 1 published time-based cost analysis of secondary intraocular lens surgery. Pan et al 11 reviewed 391 secondary intraocular lens cases (CPT codes 66985 and 66986) performed during a 10-year period (2014-2023), analyzing day-of-surgery costs, operative times, and associated Medicare reimbursement. Costs were greater in our study compared with their reported day-of-surgery costs for intraocular lens insertion plus PPV and intraocular lens exchange plus PPV, which totaled $5519 (vs our $8348) and $5272 (vs our $9055), respectively. These findings may stem from differences in operative times seen in our studies, their use of state-specific Centers for Medicare & Medicaid Services rates, and variable exclusion criteria. Pan et al reported that all secondary intraocular lens subsets “result in net negative margins based on Medicare reimbursement.”
It is important, however, to consider that while the mean net margins were negative in their study, there are indeed secondary intraocular lens surgeries, albeit few, that break even or are profitable based on the allotted Medicare reimbursement. This is an advantage of our study, which is the first to report break-even times for secondary intraocular lens surgeries and to report the proportion of cases occurring at our academic referral center that exceed the break-even time. Our study also included intraocular lens repositioning procedures (CPT 66825), which their study did not investigate.
Time-cost analyses in retinal surgery reveal significant financial constraints, with most procedures incurring costs that exceed reimbursement. Blumenthal et al 7 analyzed operative times and costs in scleral buckle surgery, finding that the median cost per case was $7674.64, while the Medicare reimbursement for CPT 67017 was $4961.00, resulting in a $2713.64 loss per case (a 55% higher cost than reimbursement). The study also calculated a break-even operative time of 54.43 minutes, yet 99.1% of cases exceeded this threshold.
Pan et al 8 assessed the reimbursement adequacy of standard and complex PPV for RD repair and found that both consistently resulted in financial losses. After accounting for operative costs, the net margin for standard PPV was −$976.93 per case, while complex PPV had a greater net loss of −$3271.10 per case. Despite higher reimbursement for complex PPV, the cost-to-reimbursement gap was even wider, underscoring the financial challenges associated with these cases.
Haliyur et al 9 expanded on these RD repair data, reporting that only the lowest 50th percentile of standard PPV cases and 10th percentile of complex PPV cases by cost were adequately reimbursed. These findings highlight a significant reimbursement shortfall for both standard and complex PPV, with complex cases incurring substantially greater financial deficits despite higher total reimbursement amounts. Hwang et al 10 further substantiated these reimbursement shortfalls, demonstrating that the average total cost of PPV for primary rhegmatogenous RD was $6247.17, while Medicare reimbursement covered only $5442.91 (net loss of $804.26 per procedure). They reported a break-even time of 64.90 minutes, yet the actual average operative time in their study was 90.49 minutes, such that 100% of the cases exceeded the break-even threshold.
These findings collectively show that current reimbursement models do not adequately cover the true costs of common vitreoretinal surgeries, raising concerns about the financial viability of performing these procedures in both academic and private practice settings.
Implications
Our study adds to the growing literature underscoring a systemic shortcoming in reimbursement policies, where key complex procedures are not adequately supported in the payment models. Ensuring adequate reimbursement for secondary intraocular lens surgery is not only a matter of clinical quality but also of public health and economic policy, as timely visual rehabilitation can reduce disability, caregiver reliance, and productivity loss.20,21 While prior studies have demonstrated substantial reimbursement gaps in RD repair and other vitreoretinal surgeries, our findings extend these concerns to secondary intraocular lens procedures. Without reform, these financial disincentives may contribute to declining surgeon participation in such procedures, limiting access to care for patients requiring secondary intraocular lens placement.
These concerns assume further importance considering the Medicare reimbursement cuts for 2025, which reduce the average payment rates under the Physician Fee Schedule by 2.93%. 22 Addressing these reimbursement inadequacies will benefit from ongoing coordinated advocacy efforts involving the American Academy of Ophthalmology, the American Medical Association, and the American Society of Retina Specialists to push for policy changes that more accurately reflect the current and future costs of retina surgeries. The recent surge in published time-based cost studies may serve to provide further concrete data for use by the Specialty Society Relative Value Scale Update Committee when making recommendations to the federal government, potentially presenting an argument for higher Medicare Ambulatory Payment Classifications for complex retina diagnosis/procedure codes, especially when accounting for inflation.
Study Strengths and Limitations
Our findings delineate real-world economic considerations of secondary intraocular lens implantation with or without PPV as performed in a tertiary care center. These data demonstrate the current insufficiency of Medicare reimbursement for these procedures and thus the need for reimbursement reform.
As our analysis is based on single-institution, single-year data, the exact costs and profitability margins presented here may not generalize to all institutions. Specifically, given that ambulatory surgery centers receive different Medicare reimbursement than hospitals for the same procedures, we expect that our findings will differ from the same analysis performed in an ambulatory surgery center setting. Given trends toward higher facility fees in a hospital setting, we anticipate greater cost pressures associated with inadequately reimbursed secondary intraocular lens procedures performed in ambulatory surgery centers.
Future work should investigate differences in profitability in these different practice settings. Additionally, greater clarity would be achieved by future studies exploring specific surgical characteristics associated with greater surgical length, especially in secondary intraocular lens procedure categories that were underrepresented in our cohort and that would thus benefit from more powered analysis. Time-cost analyses by intraocular lens type, and that investigate whether operative times vary significantly between anterior chamber, sulcus, scleral-fixated, and scleral-sutured intraocular lenses, are warranted to better understand intraocular lens type-specific variability in operative time and costs. Future work should also assess the cost-effectiveness of secondary intraocular lens surgery, incorporating patient-centered outcomes and cost per quality-adjusted life-years to better inform policy and reimbursement decisions.
The majority of secondary intraocular lens procedures performed with vitrectomy are not profitable under current Medicare reimbursement rates for hospital centers. Increased reimbursements for these complex procedures must be considered to achieve economic sustainability and maintain quality standards.
Supplemental Material
sj-docx-1-vrd-10.1177_24741264261428752 – Supplemental material for Time-Driven, Activity-Based Cost Analysis of Secondary Intraocular Lens Implantation
Supplemental material, sj-docx-1-vrd-10.1177_24741264261428752 for Time-Driven, Activity-Based Cost Analysis of Secondary Intraocular Lens Implantation by Ryan S. Meshkin, Samantha E. Hoffman, Celine Chaaya MD, Srujay Pandiri, Nicolas A. Yannuzzi, Hasenin Al-Khersan, Ghassan Ghorayeb, Ella H. Leung, Miguel A. Busquets, Dan A. Gong, Anton M. Kolomeyer, Philip I. Niles, Michael M. Lai, Dilraj S. Grewal and Nimesh A. Patel in Journal of VitreoRetinal Diseases
Supplemental Material
sj-docx-2-vrd-10.1177_24741264261428752 – Supplemental material for Time-Driven, Activity-Based Cost Analysis of Secondary Intraocular Lens Implantation
Supplemental material, sj-docx-2-vrd-10.1177_24741264261428752 for Time-Driven, Activity-Based Cost Analysis of Secondary Intraocular Lens Implantation by Ryan S. Meshkin, Samantha E. Hoffman, Celine Chaaya MD, Srujay Pandiri, Nicolas A. Yannuzzi, Hasenin Al-Khersan, Ghassan Ghorayeb, Ella H. Leung, Miguel A. Busquets, Dan A. Gong, Anton M. Kolomeyer, Philip I. Niles, Michael M. Lai, Dilraj S. Grewal and Nimesh A. Patel in Journal of VitreoRetinal Diseases
Supplemental Material
sj-docx-3-vrd-10.1177_24741264261428752 – Supplemental material for Time-Driven, Activity-Based Cost Analysis of Secondary Intraocular Lens Implantation
Supplemental material, sj-docx-3-vrd-10.1177_24741264261428752 for Time-Driven, Activity-Based Cost Analysis of Secondary Intraocular Lens Implantation by Ryan S. Meshkin, Samantha E. Hoffman, Celine Chaaya MD, Srujay Pandiri, Nicolas A. Yannuzzi, Hasenin Al-Khersan, Ghassan Ghorayeb, Ella H. Leung, Miguel A. Busquets, Dan A. Gong, Anton M. Kolomeyer, Philip I. Niles, Michael M. Lai, Dilraj S. Grewal and Nimesh A. Patel in Journal of VitreoRetinal Diseases
Footnotes
Appendix
Variables definitions:
Authors’ Note
Drs. Meshkin and Hoffman are co-first authors.
Ethical Approval
This study was conducted in accordance with the Declaration of Helsinki. The collection and evaluation of patient health information was performed in a Health Insurance Portability and Accountability Act-compliant manner.
Statement of Informed Consent
The need for informed consent has been waived by the institutional review board.
Declaration of Conflicting Interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Dr. Patel serves as a consultant for Alcon, Alimera, Allergan, Apellis, Atheneum, Biogen, Dutch Ophthalmic Research Center (DORC), EyePoint Pharmaceuticals, Genentech, Gerson Lehrman Group, Guidepoint, Kyoto Drug Company, Lifesciences, Regenxbio, and Regeneron. Dr. Yannuzzi serves as a consultant for EyePoint Pharmaceuticals, Genentech, and Regenxbio. Dr. Busquets serves as a consultant for Alcon and Notal Vision. Dr. Al-Khersan serves as a consultant for Dutch Ophthalmic Research Center (DORC) and Genentech. Dr. Kolomeyer serves as a consultant for Alimera, Allergan (AbbVie), Apellis, Biogen, Genentech, Inc. (Roche), and Regeneron; and as a speaker for Biogen, Genentech, Inc. (Roche), and Iveric Bio (Astellas).Dr. Niles serves as a consultant for Regeneron. Dr. Lai serves as a consultant for Genentech. Dr. Grewal serves as a consultant for ANI Pharmaceuticals, Apellis, Astellas, Bausch & Lomb, Dutch Ophthalmic Research Center (DORC), EyePoint Pharmaceuticals, Genentech/Roche, Priovant, and Regeneron. The remaining authors have no relevant financial disclosures. There are no conflicts of interest.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: John and Michele Simourian, as well as the Saint Vincent de Paul Foundation, support the Retina Innovation Fund, Massachusetts Eye and Ear, Boston, MA. The funding organization had no role in the design or conduct of this research. The project described was supported by award number T32GM144273 (S.E.H.) from the National Institute of General Medical Sciences. The content is solely the responsibility of the authors and does not necessarily represent the official views of the National Institute of General Medical Sciences or the National Institutes of Health.
ORCID iDs
Supplemental Material
Supplemental material is available online with this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
