Abstract
Keywords
Introduction
As more patients have cataract surgery and the global aging population continues to grow, the burden of capsular bag failure and the need for secondary intraocular lens (IOL) placement is rising. 1 This is the result of a wide range of underlying causes, including but not limited to complicated cataract surgery, ocular trauma, conditions with inherent zonular instability (eg, pseudoexfoliation syndrome), and connective tissue disorders such as Marfan syndrome. 2
Secondary IOL placement can be successfully achieved with anterior chamber IOLs, iris-fixated IOLs, or scleral-fixated IOLs. IOL placement with scleral sutures has become a popular secondary IOL technique, in particular when a pars plana vitrectomy (PPV) is necessary for IOL removal or lensectomy. 3 Favorable outcomes and low rates of complications with scleral-sutured IOL implantation are well documented in the literature.4 –7 Scleral-sutured IOL placement can occur simultaneously at the time of the PPV or the patient can be left aphakic with staged IOL fixation at a later date. The decision to stage scleral-sutured IOL placement is largely surgeon and patient dependent. However, it is unclear how staging scleral-sutured IOL placement ultimately affects visual recovery and complications.
To our knowledge, no previous study has directly compared the outcomes of staged vs simultaneous scleral-sutured IOL surgery. The aim of this study was to compare the visual outcomes and complication rates of staged scleral-sutured IOL surgery with those of simultaneous surgery.
Methods
Institutional review board approval was obtained, and the study adhered to the tenets of the Declaration of Helsinki. Cases of scleral-sutured placement of an Akreos AO60 IOL (Bausch + Lomb) performed by 7 different surgeons between July 1, 2016, and December 31, 2022, were reviewed. The patients were identified using Current Procedural Terminology codes 66985 for secondary insertion of an IOL prosthesis and 66986 for exchange of an IOL prosthesis.
The following were recorded: the patient’s age, sex, and race; the surgical indication(s); the relevant ophthalmic history; the preoperative and postoperative Snellen visual acuity (VA); and the intraoperative and postoperative complications. When the PPV and lensectomy or removal of the IOL occurred at the same time as fixation of the secondary IOL, the procedure was considered simultaneous. The decision to stage or perform simultaneous surgery was surgeon dependent based on the individual circumstances of the patient. A case was considered staged if an initial PPV was performed for lensectomy or removal of an IOL without placement of a secondary IOL. A history of previous PPV for an unrelated ocular comorbidity, such as a retinal detachment (RD) or vitreous hemorrhage, was not considered a staged surgery.
Snellen VA was converted to logMAR notation for statistical analysis. Counting fingers VA was recorded as 20/2000 Snellen equivalent, with a corresponding conversion to 2.0 logMAR. 8 Hand motions VA corresponded to 3.0 logMAR. 8
Intraocular pressure (IOP) was measured with a Tono-Pen (Medtronic). Using previously published criteria,9,10 hypotony was defined as an IOP of 5 mm Hg or less. Ocular hypertension was defined as an IOP of 25 mm Hg or more at any postoperative visit.
Postoperative corneal edema and cystoid macular edema (CME) were recorded if they lasted longer than 30 days postoperatively. CME was confirmed by the presence of intraretinal or subretinal fluid on spectral-domain optical coherence tomography of the macula. The outcome measures were the change in VA and the incidence of intraoperative and postoperative complications.
Surgical Technique
Each identified case involved completion of a PPV using the 25-gauge Constellation vitrectomy system (Alcon) and scleral fixation of an Akreos AO60 IOL using polytetrafluoroethylene (Gore-Tex) sutures. The same procedure (Supplemental Video) was performed for each case. First, 2 localized conjunctival peritomies were created at 3 o’clock and 9 o’clock. Light cautery was applied to the sclera. An infusion cannula was placed inferotemporally 3.0 to 4.0 mm posterior to the limbus. The 3 o’clock and 9 o’clock positions were marked on the cornea. Four sclerotomes were made 2.5 to 3.0 mm posterior to the limbus and 5.0 mm apart. A complete PPV was performed with thorough shaving of the vitreous base.
A 3.5 mm clear corneal shelved incision was made using the keratome blade. Externally, the 7-0 polytetrafluoroethylene sutures (CV-8) were looped through both ipsilateral eyelets in an anterior–posterior, posterior–anterior fashion. The IOL was folded and passed through the corneal incision into the sulcus after injection of viscoelastic. The ends of the polytetrafluoroethylene sutures were externalized through the sclerotomies. The IOL was centered before the sutures were tied down and buried. The corneal incision was typically closed with 10-0 nylon suture. The conjunctival peritomies were then closed. No intraocular gas or air was introduced to minimize the risk for IOL opacification.
Statistical Analysis
Statistical analysis was performed using SPSS Statistics software (version 28, IBM) and the Wilcoxon signed rank test, Pearson χ2 test, and t test of independent samples when appropriate. Statistical significance was set at P < .05.
Results
This study included 98 eyes of 92 patients, 82 eyes (83.7%) that had simultaneous surgeries (simultaneous group) and 16 (16.3%) that had staged surgeries (staged group). Table 1 shows the patients’ demographic characteristics, relevant ophthalmic history, and surgical indications. Of the patients, 57 (58.2%) were men and 84 (85.7%) were White. The demographic characteristics, relevant ophthalmic history, and surgical indications were similar between the simultaneous group and staged group except the rate of trauma involving globe rupture (disussed below).
Patient Characteristics.
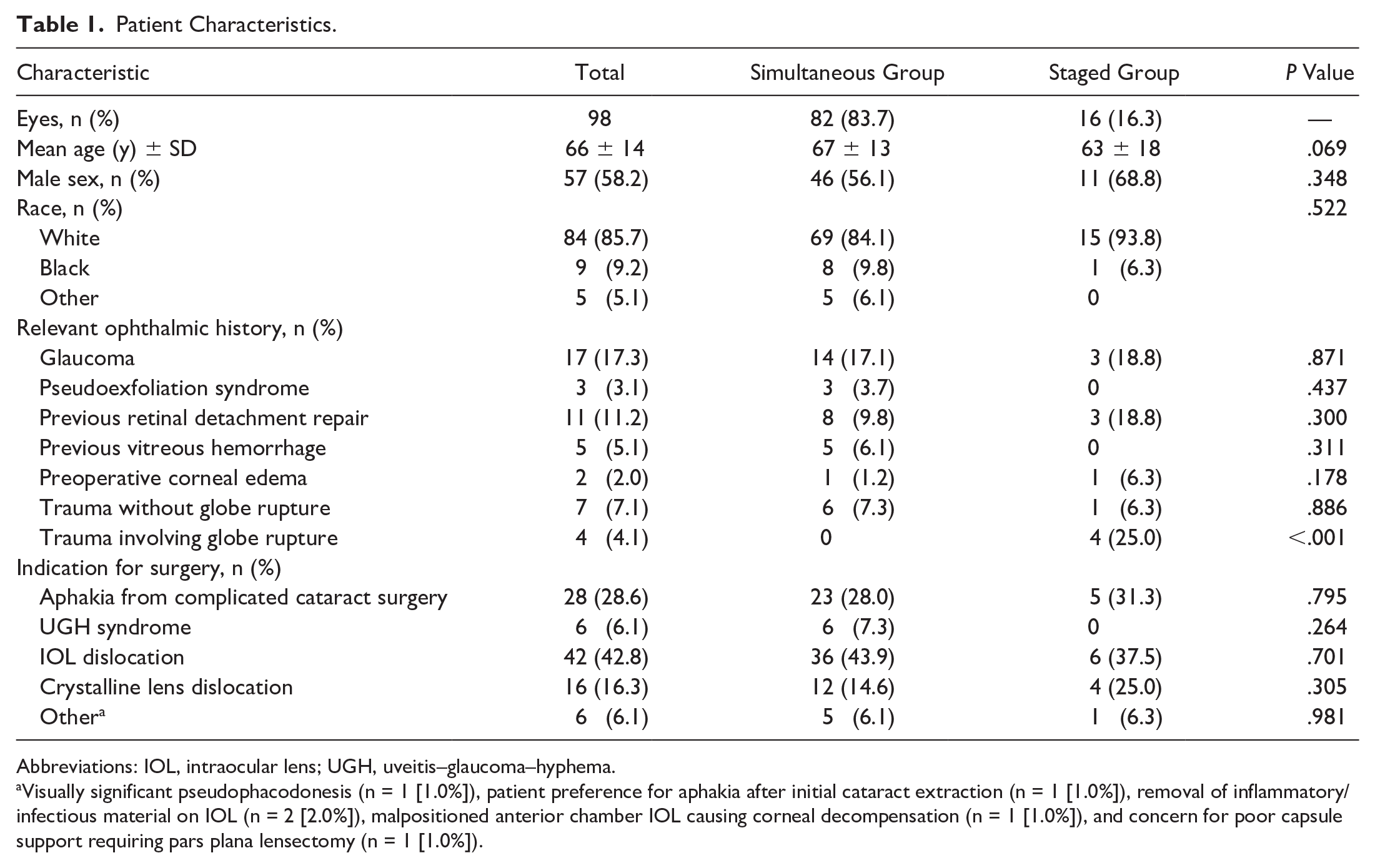
Abbreviations: IOL, intraocular lens; UGH, uveitis–glaucoma–hyphema.
Visually significant pseudophacodonesis (n = 1 [1.0%]), patient preference for aphakia after initial cataract extraction (n = 1 [1.0%]), removal of inflammatory/infectious material on IOL (n = 2 [2.0%]), malpositioned anterior chamber IOL causing corneal decompensation (n = 1 [1.0%]), and concern for poor capsule support requiring pars plana lensectomy (n = 1 [1.0%]).
Regarding the ophthalmic history, the 2 most common findings were glaucoma (17.1%, simultaneous group; 18.8%, staged group) and previous RD repair (9.8%, simultaneous group; 18.8%, staged group). There was no significant difference between the simultaneous group and staged group in the number of eyes with previous RD repair (P = .300) or with coexisting preoperative corneal edema (P = .178) (Table 1).
All 4 eyes with history of open-globe trauma had staged surgery, and none had simultaneous surgery; the difference was statistically significant (P < .001) (Table 1). Three of the open-globe trauma cases involved an intraocular foreign body that was removed at the time of the initial PPV and lensectomy. One case had severe corneal scarring and a history of ocular hypertension before scleral-sutured IOL placement, while another involved commotio retinae that was self-limited. Another case had macular scarring with poor VA unrelated to the trauma that occurred before simultaneous surgery. The mean follow-up was 346 days (median, 240; range, 26-2616).
The 2 most common indications for scleral-sutured IOL placement were IOL dislocation or subluxation (42.8%) followed by aphakia from complicated cataract surgery (28.6%). The indications were similar between the simultaneous group and staged group.
In the staged group, the mean time between the initial PPV and scleral-sutured IOL placement was 511 days (median, 184; range, 15-2462). Three cases in that group experienced significantly longer times to scleral-sutured IOL placement after the initial PPV. Two of these eyes were of 1 patient with poor visual potential resulting from an optic nerve coloboma; the time between PPV and scleral-sutured IOL placement was 2254 days and 2462 days, respectively. The longer lapse was because IOL removal was performed previously and the patient initially chose to remain aphakic. The third eye was of a patient who chose to remain aphakic for 1107 days after concurrent complicated cataract surgery and successful macular hole repair. When these 3 outliers are excluded, the mean time between the initial PPV and scleral-sutured IOL placement was 167 days (median, 140; range, 49-388).
Eyes in the staged group with a previous open-globe injury had mean delay to scleral-sutured IOL placement of 237 days (median, 232; range, 96-388). Two cases with persistent corneal edema after the initial PPV experienced delays to scleral-sutured IOL placement of 184 days and 203 days, respectively. There were no cases of CME after the initial PPV with planned staged surgery.
Table 2 compares the visual outcomes between the simultaneous group and staged group. The mean preoperative baseline VA was worse in the staged group (1.8 logMAR; Snellen equivalent, 20/1262) than in the simultaneous group (1.3 logMAR; Snellen equivalent, 20/400), although the difference was not statistically significant (P = .104). The Snellen equivalent VA improved to 20/191 in the staged group and to 20/63 in the simultaneous group. The between-group difference in the mean VA improvement was not statistically significant (P = .49).
Visual Outcomes.
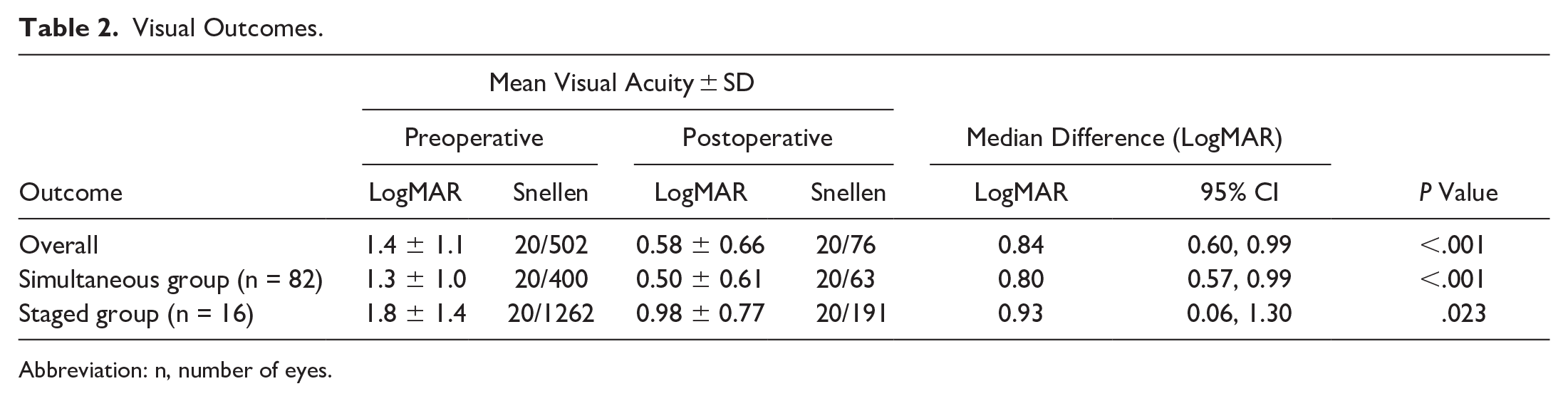
Abbreviation: n, number of eyes.
Table 3 compares the postoperative complications between the simultaneous group and staged group. There were no intraoperative complications. The most common complications were postoperative CME (9.2%) followed by hypotony (8.2%). All cases of hypotony were managed nonsurgically, with 2 cases treated with pressure patching and 6 cases with medication including steroid and atropine drops. The median duration of hypotony was 17 days (range, 5-47). Three cases of hypotony resulted in transient choroidal detachments postoperatively; all cases resolved with medical management. There was 1 case of postoperative endophthalmitis (culture negative) in the simultaneous group, which resolved after intravitreal injection of ceftazidime, vancomycin, and moxifloxacin. No additional surgical intervention was performed.
Postoperative Complications.
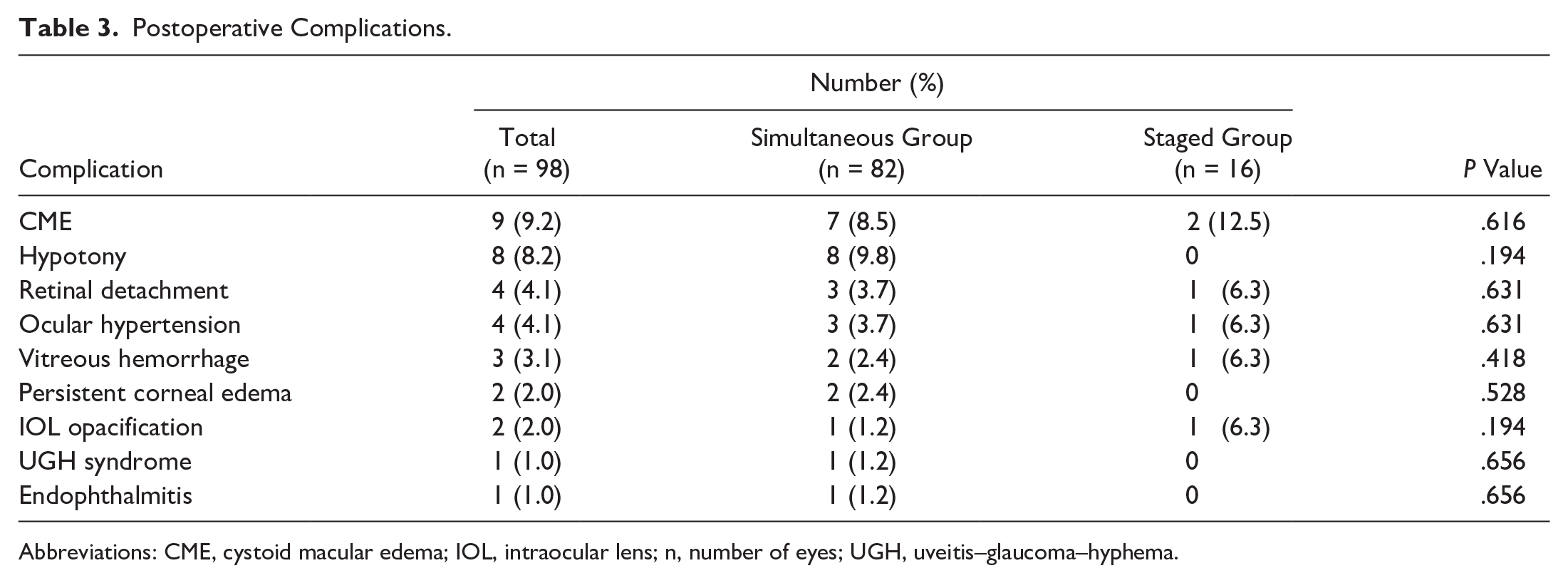
Abbreviations: CME, cystoid macular edema; IOL, intraocular lens; n, number of eyes; UGH, uveitis–glaucoma–hyphema.
Four eyes (4.1%) had an RD after scleral-sutured IOL placement, 1 (6.3%) in the staged group and 3 (3.7%) in the simultaneous group. One eye with previous RD repair had IOL opacification after silicone oil tamponade for a recurrent RD. Another eye had IOL opacification after sulfur hexafluoride (SF6) gas was used to re-tamponade a partially detached corneal graft from Descemet-stripping automated endothelial keratoplasty (DSAEK). In this case, the keratoplasty followed staged PPV and scleral-sutured IOL placement for persistent corneal edema after complicated cataract surgery that was initially noted preoperatively. There were no additional cases of persistent corneal edema requiring corneal transplantation.
Two cases of RD occurred after staged surgery, at which time the eyes had RD repair; both eyes had previous RD repair. The overall transient postoperative hypotony rate was 8.2%, with all cases occurring in the simultaneous group (P = .194); the between-group difference was not statistically significant (P = .194). There were no cases of hypotony maculopathy. All cases of CME and ocular hypertension resolved within 90 days. No eye had suture erosion or persistent idiopathic postoperative inflammation. All cases of postoperative vitreous hemorrhages resolved without surgical intervention. No definitive cases of IOL tilt or dislocation were identified; however, there was 1 case of uveitis–glaucoma–hyphema (UGH) syndrome thought to be caused by a floppy iris or more anterior placement of the IOL; the UGH syndrome resolved after a laser peripheral iridotomy was performed over an area of suspected iris–haptic chaffing.
Table 4 shows the effects of pars plana lensectomy with a fragmatome on the rates of complications in the simultaneous group. The rate of hypotony was higher when a pars plana lensectomy was performed (23.8%) than when it was not (4.9%). The rate of postoperative CME, RD, vitreous hemorrhage, and persistent corneal edema was not significantly affected by the use of the fragmatome during simultaneous surgery (Table 4).
Effect of Fragmatome on Complication Rates After Simultaneous PPV and Scleral-Sutured-IOL Placement.
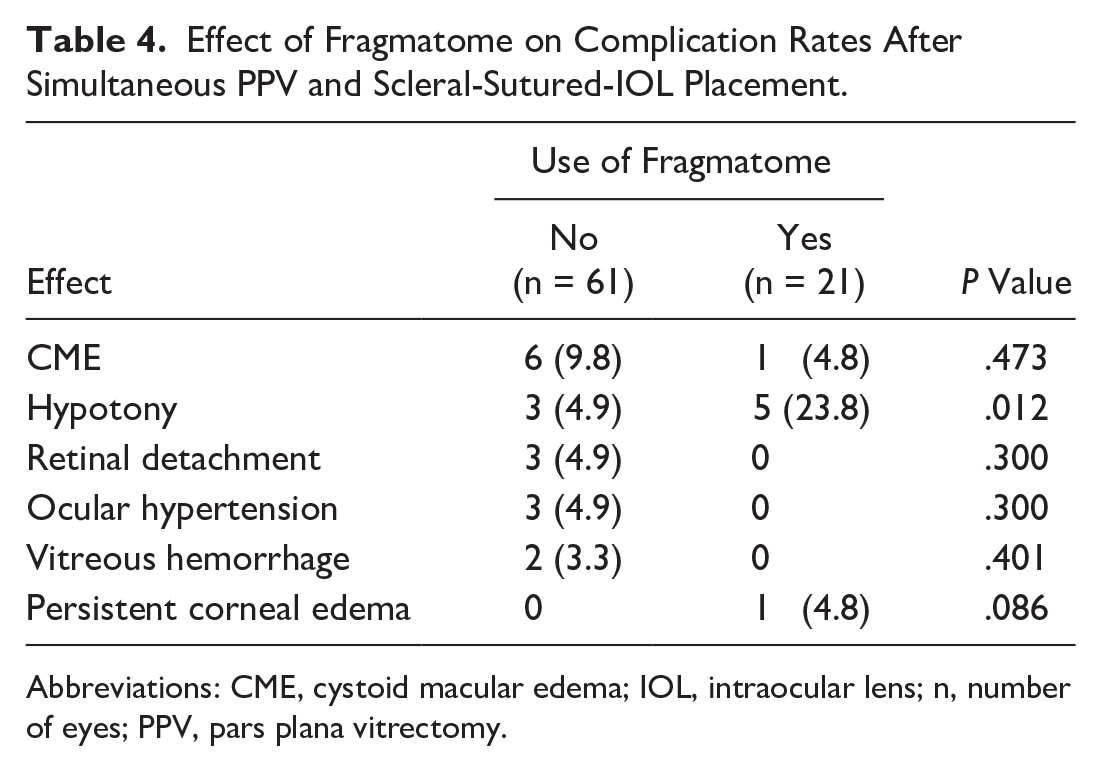
Abbreviations: CME, cystoid macular edema; IOL, intraocular lens; n, number of eyes; PPV, pars plana vitrectomy.
Conclusions
When the capsular bag is lost, the decision to stage or simultaneously combine PPV and scleral-sutured IOL implantation depends on several factors. The data in the literature comparing the effect of surgical staging on the visual outcomes and complication rates of PPV and scleral-sutured IOL placement are insufficient. To our knowledge, our study is the first to help close this gap by directly comparing outcomes in patients who had staged surgery with those who had simultaneous PPV and scleral-sutured IOL placement.
The visual outcomes in both the simultaneous group and the staged group were favorable. Most procedures in this study were simultaneous because this technique may result in faster visual rehabilitation and potentially limit the number of sequential procedures. However, staged placement of a scleral-sutured IOL after the initial PPV can be performed to minimize tissue manipulation, operative time, and postoperative inflammation, in particular in “sicker” eyes that have had multiple previous surgeries, have an insufficient view because of corneal edema resulting from the previous complicated surgery, or had previous trauma.
Comparing eyes that had staged surgery with those that had simultaneous surgery revealed several subtle differences that may affect patient selection. Patients in the staged group tended to have worse preoperative VA, although the sample size in this study did not suggest a statistical difference. This may indicate a surgeon preference to stage procedures in eyes deemed “sicker” given our finding that eyes with previous open-globe trauma were more likely to receive staged surgery than those scheduled for staged surgery. The rate of previous RD repair was higher in the staged group; however, the sample was not large enough to achieve statistical significance. The rate of previous RD repair in the staged group may explain the worse preoperative VA. These eyes may benefit from a staged procedure, which allows the eye to recover from recent complicated surgeries before an IOL is placed and may result in a decreased rate of corneal decompensation if there is notable preoperative corneal edema.
The overall rate of complications after scleral-sutured IOL placement was similar to that in previous studies,9 –12 and in general the complications were nonpersistent, resolving within 90 days. The most common complication of scleral-sutured IOL placement continues to be CME, with a similar postoperative rate between staged surgery and simultaneous surgery.
Hypotony, although transient after treatment with pressure patching and topical steroids/atropine, seemed to occur at a higher rate in the simultaneous group than in the staged group; the rate of hypotony in our study is similar to that in previous studies.4,13 Repeated maneuvers through the sclerotomies to perform a thorough vitrectomy and to then place a scleral-fixated IOL during simultaneous surgery may explain this transient hypotony. In addition, pars plana lensectomy increases the rate of hypotony after simultaneous surgery. The creation of an additional larger wound for the fragmatome likely places the eye at a higher risk for postoperative hypotony. We advise close monitoring for signs of hypotony at the end of the case and careful placement of a suture to close leaking sclerotomies. It is reassuring that all cases of hypotony resolved spontaneously, with no sight-threatening complications such as choroidal detachment.
Persistent corneal edema occasionally requiring corneal transplantation has been reported after scleral-sutured IOL placement 13 ; however, this complication was rare in our study. Theoretically, simultaneous surgery may increase the risk for corneal edema as a result of the more prolonged operating time and increased maneuvers within the anterior chamber; however, our study did not confirm this trend. It is more likely that the presence of corneal edema is related to ocular comorbidities, including previous complicated cataract surgery, that were present before scleral-sutured IOL placement.
The 2 cases of postoperative IOL opacification in our study occurred after 1 simultaneous surgery and 1 staged surgery. In 1 case, the opacification occurred in relation to SF6 gas in the anterior chamber that was placed after DSAEK. In the other case, silicone oil tamponade was used to address an RD, a previously reported phenomenon. 14 Thus, it may be prudent to consider a staged approach if complications are anticipated in a higher risk eye when using the Akreos IOL and simultaneous surgery. Although subconjunctival sutures for IOL fixation have previously raised concerns about conjunctival erosion and infection, 11 there were no suture-related complications in either group in our study. We recommend burying the polytetrafluoroethylene suture knot to limit the risk for a subconjunctival knot.
The strengths of our study include a large sample of patients with a variety of surgical indications. Each surgery was performed in a similar manner with little variability. Preoperative patient counseling is critical when choosing a surgical strategy on a case-by-case basis. Patients should be educated about the expected outcomes and potential complications.
All cases in this cohort had placement of an Akreos AO60 IOL, which limits the generalizability of our results to other surgical techniques of secondary IOL placement. Additional limitations of the study include its retrospective design, leading to possible selection bias, with a larger number of “healthier” eyes than “sicker” eyes having simultaneous surgery. The differences between the 2 groups in this study should be interpreted with caution given the small number of staged surgeries. To reduce these biases, a future large multicenter randomized prospective study comparing staged vs simultaneous vitrectomy and scleral-sutured IOL placement is necessary to determine whether a staged procedure offers a decreased risk for postoperative complications. Furthermore, future studies should also include refractive data to augment the analysis of visual outcomes between these 2 techniques.
In conclusion, our results suggest that the difference between simultaneous surgery with PPV and scleral-sutured IOL implantation and staged placement of a scleral-sutured IOL after PPV is minimal. Staged surgery may be reserved for eyes with preoperative or intraoperative complications; however, the data suggest that other than in these cases, simultaneous surgery should be performed for quick visual rehabilitation and to reduce the overall cost to the patient.
Footnotes
Authors’ Note
Presented at the annual meeting of the Retina Society, October 11–14, 2023, New York, NY, USA.
Ethical Approval
This study was conducted in accordance with the Declaration of Helsinki. The collection and evaluation of all protected health information were performed in a US Health Insurance Portability and Accountability Act–compliant manner.
Statement of Informed Consent
All patients provided informed consent, including permission for publication of all photographs and images included herein.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of the article: Dr. Nair is on the advisory board of EyePoint. Dr. Finn is on the advisory board of Allergan, Apellis, EyePoint, and Iveric Bio and is a consultant to and on the advisory board of Genentech. Dr. Day declared no potential conflicts of interest with respect to the research, authorship, and/or publication of the article.
Funding
Supported by an unrestricted grant to the Department of Ophthalmology & Visual Sciences, Vanderbilt University Medical Center, from Research to Prevent Blindness, which had no role in the design and conduct of the study; collection, management, analysis, and interpretation of the data; preparation, review, or approval of the manuscript; or decision to submit the manuscript for publication.
Supplemental Material
Supplemental material is available online with this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
