Abstract
Keywords
Introduction
Down syndrome was first described by John Langdon Down in 1862 1 and is one of the most prevalent genetic disorders worldwide. Three types of Down syndrome have been identified. The most common form, trisomy 21, results from a nondisjunction event during cell division and accounts for approximately 95% of cases. This error occurs primarily during maternal meiosis I (66%) or II (21%), with smaller contributions from paternal meiosis I (3%) and meiosis II (5%). Consequently, the embryo carries 3 copies of chromosome 21 instead of the usual 2.
A second form, present in about 3% of cases, arises from Robertsonian translocation, in which a full or partial copy of chromosome 21 becomes attached to another chromosome, typically t(14:21). The least common form, mosaicism, occurs in about 2% of cases and is characterized by the presence of 2 cell lines, with some cells having the normal complement of 46 chromosomes and others having 47.2,3
The incidence of Down syndrome is approximately 1 in 643 live births, with an estimated 5713 neonates diagnosed each year.4,5 Clinically, individuals with Down syndrome exhibit short stature with short fingers, straight hair, prominent epicanthal folds, a flat nasal bridge, low-set ears, upturned palpebral fissures, small mouths, relative macroglossia, a sandal gap between the first and second toes, and generalized hypotonia. 6 Associated systemic health issues in individuals with Down syndrome include polycythemia, myelodysplastic syndrome, leukemia, congenital heart defects, hypothyroidism, diabetes mellitus, obesity, and intellectual disabilities. 7
Down syndrome has several ophthalmic manifestations, such as strabismus (2%-5%), amblyopia (36.4%), nasolacrimal duct obstruction (membranous type), keratoconus (71.3%), Brushfield spots (13%-70%), refractive errors, and retinal abnormalities (1.7%-40%) such as lattice degeneration, chorioretinitis, and retinal detachment. Lens abnormalities, including congenital and acquired cataracts, affect approximately 5% to 50% of individuals with Down syndrome.8,9
Klinefelter syndrome, first described in 1942 by Harris F. Klinefelter, 9 results from the presence of 2 or more X chromosomes in a phenotypic male, with the most common karyotype being 47,XXY, accounting for approximately 80% to 90% of cases. This chromosomal abnormality arises primarily from nondisjunction during meiotic divisions but can also occur during early embryonic mitosis.10,11 The remaining 10% to 20% of cases include higher-grade aneuploidies (48,XXY; 49,XXXXY; or 48,XXYY), mosaicisms (47,XXY/46,XY), or structurally abnormal chromosomes (47,XqY). Klinefelter syndrome affects approximately 1 in 500 newborn males. More than 95% of affected individuals present with small testes (<4–6 mL), over 99% have infertility, more than 95% exhibit azoospermia, 60% to 80% show decreased facial hair, and 30% to 60% have decreased pubic hair. Additional associated conditions include type 2 diabetes mellitus (10%-39%), osteopenia, and osteoporosis (40%). Ocular abnormalities are rare but include microspherophakia, lens subluxation, and congenital cataracts.12 –14
Double aneuploidy refers to the presence of 2 chromosomal aberrations in a single individual and is thought to arise from meiotic nondisjunction or a single mitotic error, possibly originating from 1 or both parents. It was first reported in 1959 by Ford et al 15 in a case involving trisomy 21 and Klinefelter syndrome, and its estimated incidence ranges from 0.4 to 0.9 per 10 000 male births, although many such conceptions result in spontaneous abortions.15–20 We present the first reported case of a 2-month-old infant with Down–Klinefelter syndrome (48, XXY, +21) harboring a novel MAPKAPK3 mutation.
Case Report
A 2-month-old male infant with Down–Klinefelter syndrome was born via vaginal delivery at 39 weeks and 1 day to a 40-year-old mother and a 40-year-old father. The mother had a history of 1 spontaneous abortion and 1 preterm birth. The parents were nonconsanguineous. The mother had a history of subclinical hyperthyroidism and denied exposure to alcohol, medications, cigarettes, or other teratogenic substances during pregnancy. Screening for infectious etiologies was negative for human immunodeficiency virus, chlamydia, syphilis, hepatitis B and C, and toxoplasmosis. The father’s medical history was unremarkable. Prenatal ultrasonography in the first trimester revealed increased nuchal translucency and a small nasal bone. Despite these findings and the prior obstetric history, the parents declined genetic counseling and did not pursue follow-up with a geneticist or obstetrician.
On physical examination, the newborn’s birth weight was 3.04 kg (10th–25th percentile according to World Health Organization growth standards), length was 46 cm (approximately 10th percentile; World Health Organization 3rd–97th percentile range, 44.9–54 cm), and head circumference was 32.5 cm (approximately 7th percentile; World Health Organization 5th–95th percentile range, 32.1–38.5 cm). The anterior fontanelle was soft, flat, and open. The infant exhibited dysmorphic features, including brachycephaly, a flat facial profile, low-set ears, upslanting palpebral fissures, epicanthal folds, and a small nose. Additional findings included a sandal gap, generalized hypotonia, and an undescended right testicle.
All the routine laboratory tests were reported to be normal, except for hyperbilirubinemia (neonatal total bilirubin 8.0 mg/dL; reference range, 0.1–5.0 mg/dL) and hypothyroidism (thyroid-stimulating hormone 24.370 µIU/mL and free thyroxine 1.73 ng/dL). The hearing screening test was abnormal, prompting referral to a pediatric audiologist to determine the type and extent of the hearing impairment. Given the pregnancy history, abnormal findings of prenatal ultrasound, and dysmorphic features, a genetic specialist was consulted for further evaluation because of high suspicion for Down syndrome. Comprehensive evaluation, including karyotyping with mosaic studies, revealed an abnormal karyotype of 48,XXY,+21, consistent with Down–Klinefelter syndrome.
The thyroid abnormality was treated with levothyroxine 10 µg/kg/day. Thyroid ultrasonography demonstrated increased vascularity within the thyroid tissue. Renal and bladder ultrasonography findings were normal. Echocardiography revealed an atrial septal defect, a ventricular septal defect, and patent ductus arteriosus. The consulting cardiologist noted that these findings were not affecting the patient’s health, as his vital signs were stable and supplemental oxygen was not required.
During newborn examination, the patient was noted to have absent red reflexes. An examination under anesthesia was therefore performed to fully investigate any ophthalmic abnormalities. On examination, both eyes demonstrated prominent epicanthal folds. Intraocular pressure was 21 mm Hg in the right eye and 20 mm Hg in the left eye. The conjunctiva and sclera were white and quiet. The corneas were clear, with diameters of 11 mm in both eyes. The anterior chambers were deep, the iris showed no nodules, and the pupils were round and reactive to light. Both eyes had posterior polar cataracts in the lens centered on the optical axis (Figure 1). B-scan ultrasonography revealed no abnormalities of the vitreous or retina. Given the high risk of amblyopia, the patient underwent immediate sequential bilateral pars plana vitrectomy and lensectomy, while preserving the lens capsule for future secondary intraocular lens implantation. Based on the examination findings and family history, genetic testing was pursued, and the results identified a MAPKAAPK3 mutation (c.1039G>A; p.Asp347Asn).
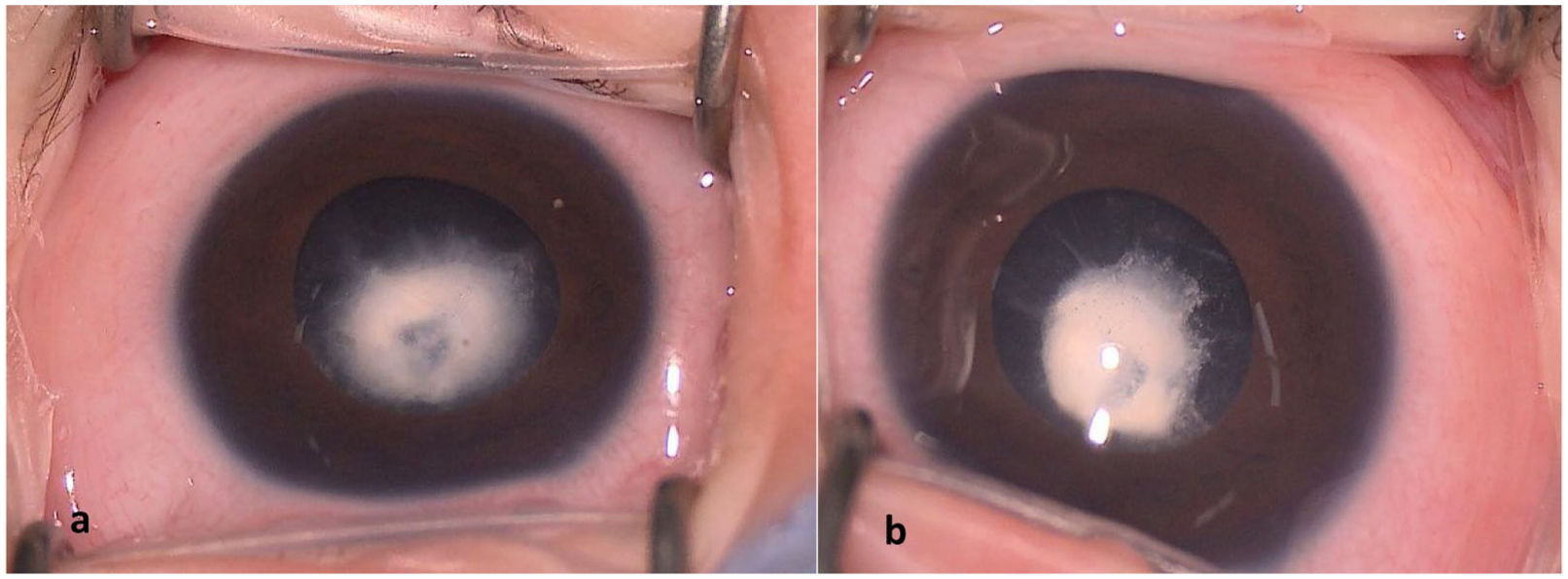
Anterior segment of the (A) right eye and the (B) left eye. The conjunctiva and sclera are white and quiet. The corneas are clear, and posterior polar cataracts are present.
Conclusions
While there are numerous pathological genetic conditions affecting humans, a few involve abnormalities in the number of chromosomes within cells. Both Down syndrome and Klinefelter syndrome result from errors in chromosomal segregation and may coexist as double aneuploidy, a condition characterized by an abnormal number of chromosomes, typically involving an extra sex chromosome and an additional autosomal chromosome (eg, trisomy 13, 18, or 21). 21 Risk factors for double aneuploidy include advanced maternal and paternal ages (over 33 and 38 years, respectively), abnormal prenatal ultrasonographic findings, and early pregnancy loss.22–24 Known karyotypes include 48,XXY,+21 (Klinefelter–Down Syndrome) and 48,XXY,+18 (Klinefelter–Edwards Syndrome). More complex aneuploidies involving 3 or more additional chromosomes are associated with low neonatal survival rates. Notably, although advanced parental age is a risk factor for double aneuploidy, having a previous child with trisomy increases the risk of aneuploidy in subsequent pregnancies regardless of parental age. 25
In our patient, clinical features of Klinefelter syndrome were not evident because of his young age. The characteristic phenotypic features of Klinefelter syndrome typically appear after puberty and include gynecomastia, small penis size, hypogonadism, sparse body hair, low muscular development, metabolic disturbances, low libido, and infertility related to low testosterone. Although Klinefelter syndrome lacks distinctive ocular features, some authors have reported decreased corneal thickness and volume, reduced retinal thickness, and a smaller foveal avascular zone in affected patients.26,27
In patients with Down–Klinefelter Syndrome, neonatal features of Down syndrome are evident, as seen in our patient, who exhibited brachycephaly, flat facial features, upslanting palpebral fissures, epicanthal folds, a small nose, a sandal gap, hypotonia, an undescended testicle, hypothyroidism, and impaired auditory function. Our patient also had cardiac defects similar to those reported by Shen et al, 27 including a large ventricular septal defect, atrial septal defect, and patent ductus arteriosus.28,29
With regard to ocular findings, congenital cataracts are not typically associated with Down–Klinefelter syndrome, although a prevalence of approximately 3% to 5% has been reported. Congenital cataract, defined as a lens opacity present at birth, accounts for 5% to 20% of cases of childhood blindness worldwide.30–32 Haargaard and Fledelius 31 estimated a frequency of 1.4% for congenital cataracts, which may be unilateral (47%) or bilateral (61%). Additionally, 25% of bilateral cases are linked to systemic diseases, and 50% have a genetic basis. Congenital cataracts can also result from intrauterine infections (such as TORCH) or exposure to teratogenic agents.32,33
In patients with Down syndrome, Tătaru et al 18 identified the most common morphopathological forms as lamellar, posterior polar, nuclear, cerulean, and total cataracts. In contrast, cataracts in patients with Klinefelter syndrome are rare and typically result from early-onset diabetes mellitus. 34 Only 2 case reports have described bilateral congenital cataracts in patients with Klinefelter syndrome. The first involved a 3-month-old infant who underwent cataract extraction with anterior vitrectomy for bilateral anterior polar cataracts. The second described a 2-month-old male infant with microcornea, malformed pupils, and bilateral anterior polar cataracts who also underwent bilateral cataract extraction, anterior vitrectomy, and pupiloplasty. Both patients developed nystagmus resulting from delayed treatment.
Our patient underwent similar surgical procedures and demonstrated a favorable outcome after 3 months; however, long-term follow-up remains necessary.35,36
Two key attributes of our case are that our patient was genetically diagnosed with both Down–Klinefelter Syndrome (48,XXY,+21) and a novel MAPKAPK3 mutation. The literature suggests that his bilateral posterior polar cataracts could stem from altered gene expression associated with trisomy 21. Puri and Singh 4 reported that elevated superoxide dismutase levels in individuals with Down syndrome may increase reactive oxygen species and, consequently, the risk of cataract formation.37,38
Second, the MAPKAPK3 mutation identified in our patient has been associated with Martinique crinkled retinal pigment epitheliopathy. The MAPKAPK3 gene encodes an enzyme that regulates key biochemical pathways, particularly those related to cell growth and stress responses. Our patient had a p.Asp347Asn mutation affecting acetylation, ubiquitylation, and phosphorylation sites, which may impair P38 protein binding and alter protein function. This is the first reported instance of this mutation in a patient with Down–Klinefelter syndrome. Although the clinical implications of this mutation are unknown owing to the patient’s young age, it presents a unique opportunity to explore the impact of genetic conditions on organ development and function. The MAPKAPK3 protein is vital for retinal pigment epithelium physiology owing to its higher expression in this tissue. Typically, macular conditions associated with MAPKAPK3 variants manifest after 25 to 30 years of age.39–41
Treatment of bilateral congenital cataracts depends on lens density and involvement of the visual axis. Observation with refractive correction, patching, and cycloplegic penalization is ideal for congenital cataracts smaller than 3 mm that have a good red reflex and are not associated with strabismus or nystagmus. In the absence of these criteria, surgical intervention is warranted. Lambert et al 41 found that surgery for bilateral congenital cataracts should be performed before 10 weeks of age to prevent amblyopia. Long-term follow-up is crucial for visual development and to enable early detection of complications such as secondary glaucoma and retinal detachment. Our patient exhibited absent red reflexes owing to bilateral congenital cataracts greater than 3 mm in diameter and involving the central visual axis, prompting immediate sequential bilateral vitreoretinal surgery.42–44
In summary, Down–Klinefelter syndrome (48,XXY, +21) is a rare cytogenetic disorder characterized by the coexistence of 2 chromosomal aberrations within the same individual. In our case, the patient showed clinical features of Down syndrome, including bilateral congenital cataracts, whereas the characteristic features of Klinefelter syndrome are expected to appear later in life.
To our knowledge, this is the first report describing the coexistence of a MAPKAPK3 variant with double aneuploidy. Although the clinical implications of this finding are not yet known, a close and long-term follow-up is essential to better understand its impact.
Footnotes
Acknowledgements
We extend our gratitude to the nurses and technicians who collaborated during patient attention.
Ethical Approval
This study was conducted following the principles outlined in the Declaration of Helsinki.
Statement of Informed Consent
Written informed consent was obtained from the patient’s parents for the publication of this case and the accompanying image.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by the NIH Center Core Grant (P30EY014801) and an unrestricted grant from Research to Prevent Blindness (GR004596-1).
