Abstract
Introduction
Diabetic retinal disease (DRD) has been regarded as the leading cause of vision loss among working-age adults.1 –3 Visual impairment from DRD manifests as a result of neurovascular complications of diabetes mellitus, including diabetic macular edema (DME) and proliferative diabetic retinopathy (PDR). 4 According to existing estimates in the literature, the rate of blindness (visual acuity [VA] worse than 20/200 in the better-seeing eye) attributable to DRD is between 1.6% and 3.5%.1,3,5,6 However, many estimates are reported from studies that are more than 15 years old and do not capture variation in disease burden and severity across demographic groups.
Indeed, there are several reasons to believe that DRD burden differs across racial and ethnic groups. Currently, Black/African American individuals and Hispanic individuals in the US experience disproportionately higher rates of type 2 diabetes mellitus compared with White, non-Hispanic Americans.7,8 Both Black/African American and Hispanic populations also experience worse glycemic control compared with White, non-Hispanic Americans. 9 Accordingly, both Black/African American and Hispanic individuals have higher rates of diabetic complications, such as diabetic nephropathy, lower limb amputation, and macrovascular conditions. 10 Recent studies have shown a higher prevalence of DRD and DME in Black/African American and Hispanic populations compared with White populations. 11
Care of patients with DM and DRD has improved over recent decades due to pharmaceutical and clinical advances. Despite the advent of a wider range of diabetes medications and enhanced blood glucose management, improvements in glycemic control in patients with diabetes mellitus have stagnated since the early 2010s. 12 Moreover, DRD patients have benefited from improved VA following the introduction of anti–vascular endothelial growth factor (anti-VEGF) agents to treat DME and PDR. Despite these advances, previous research suggests that disparities exist among racial and ethnic minorities with regard to DRD screening rates, severity of DRD, and DRD vision loss.13 –16 While results of earlier studies have suggested that DRD is more prevalent in racial and ethnic minority populations than in non-Hispanic White populations, there is limited data on the extent of visual disability among these communities. This study focuses on the prevalence of severe vision loss (VA of 20/200 [1.0 logMAR], defined as legally blind 17 ) and moderate vision loss (VA of 20/70 [0.54 logMAR], with daytime driving allowed in most states 18 ).
Any policy-based attempts to address disparities in VA outcomes must first be able to accurately measure the current state of visual deficits in the various populations of interest. This study aimed to assess the visual deficits across racial and ethnic groups in patients with DRD, including the vision-threatening forms of DME and PDR.
Methods
Data Source
These analyses rely on data from the Sight Outcomes Research Collaborative (SOURCE) Repository, which contains electronic health records from all patients with eye diseases seen at multiple participating tertiary care health systems across the US. This database includes all medical claims, as well as details from outpatient clinic visits, including care provided, laboratory values, and medications prescribed. SOURCE captures data from clinical encounters with ophthalmologists and optometrists, including refraction and VA measurements obtained at each site. This resource also provides demographic information on all patients within the database. Privacy-preserving software (Datavant, Inc.) deidentifies all patient data prior to integration with the SOURCE repository. The study period includes all patients in participating SOURCE centers from October 2015 to February 24, 2023. Since there is no protected health information present in SOURCE, informed consent was waived, and this study was deemed exempt from Institutional Review Board review.
Cohort
The study cohort consisted of all eyes of all patients diagnosed as having DRD who were included regardless of disease duration at the time of baseline presentation (ie, not all were treatment-naive eyes at baseline). Diagnoses were identified using International Classification of Diseases, Tenth Revision (ICD-10) codes, whose accuracy has been validated for DRD research in previous studies.19 –21 Data were collected from the study cohort at the time of DRD identification. The SOURCE repository uses a best-recorded VA model for assigning the VA, since different methods for measuring VA (such as with or without corrective lens, refraction) may be used during a clinic visit. There were no age restrictions for inclusion in the study cohort. The primary exclusion criterion was the presence of concomitant vision-impairing ocular diagnoses (eg, glaucoma, macular degeneration), except cataracts. Patients with DRD included in the total study cohort were further stratified into subcohorts according to diagnoses of DME or PDR.
Statistical Analyses
The primary outcome was the percentage of eyes whose VA was worse than or equal to 20/200 and worse than or equal to 20/70, with the mean and median VA as secondary outcomes. Other secondary outcomes included a per-patient analysis (vs per-eye). Patients only needed 1 eye to be diagnosed with DRD as the state of interest for inclusion in the analysis. All Snellen VA values were converted to logMAR for statistical analyses. 22 Participants were separated by race and ethnicity into 4 self-described groups: White (non-Hispanic), Black/African American (non-Hispanic), Hispanic, and all other racial/ethnic groupings (Other). Due to the limited number of patients self-reporting other races/ethnicities, those not included in the first 3 categories were grouped as Other.
Univariate and multivariable generalized linear regression models with generalized estimating equations were implemented to estimate differences between descriptive groups while accounting for the non-independence of VA between eyes of the same patient. All statistical analyses were performed using SAS software version 9.4 (SAS Institute, Inc.). The threshold for statistical significance was set at a P value of .05 (2-tailed).
Results
DRD Total Cohort
The total cohort of patients with DRD consisted of 33 888 eyes of 18 426 patients, of whom 53% (17 909 eyes/9762 patients) identified as White (non-Hispanic), 23% (7644 eyes/4162 patients) as Black/African American, 12% (4295 eyes/2303 patients) as Hispanic, and 12% (4040 eyes/2199 patients) as Other race or ethnicity. Table 1 provides baseline characteristics of each study cohort. Black/African American and Hispanic patients were younger and more likely to be female than White (non-Hispanic) patients (each P < .05). Both Black/African American patients and Hispanic patients had higher mean hemoglobin A1c (HbA1c) levels compared with White patients (P < .001).
Baseline Characteristics of Study Patients in the Whole Cohort and Subcohorts, by Race or Ethnicity.
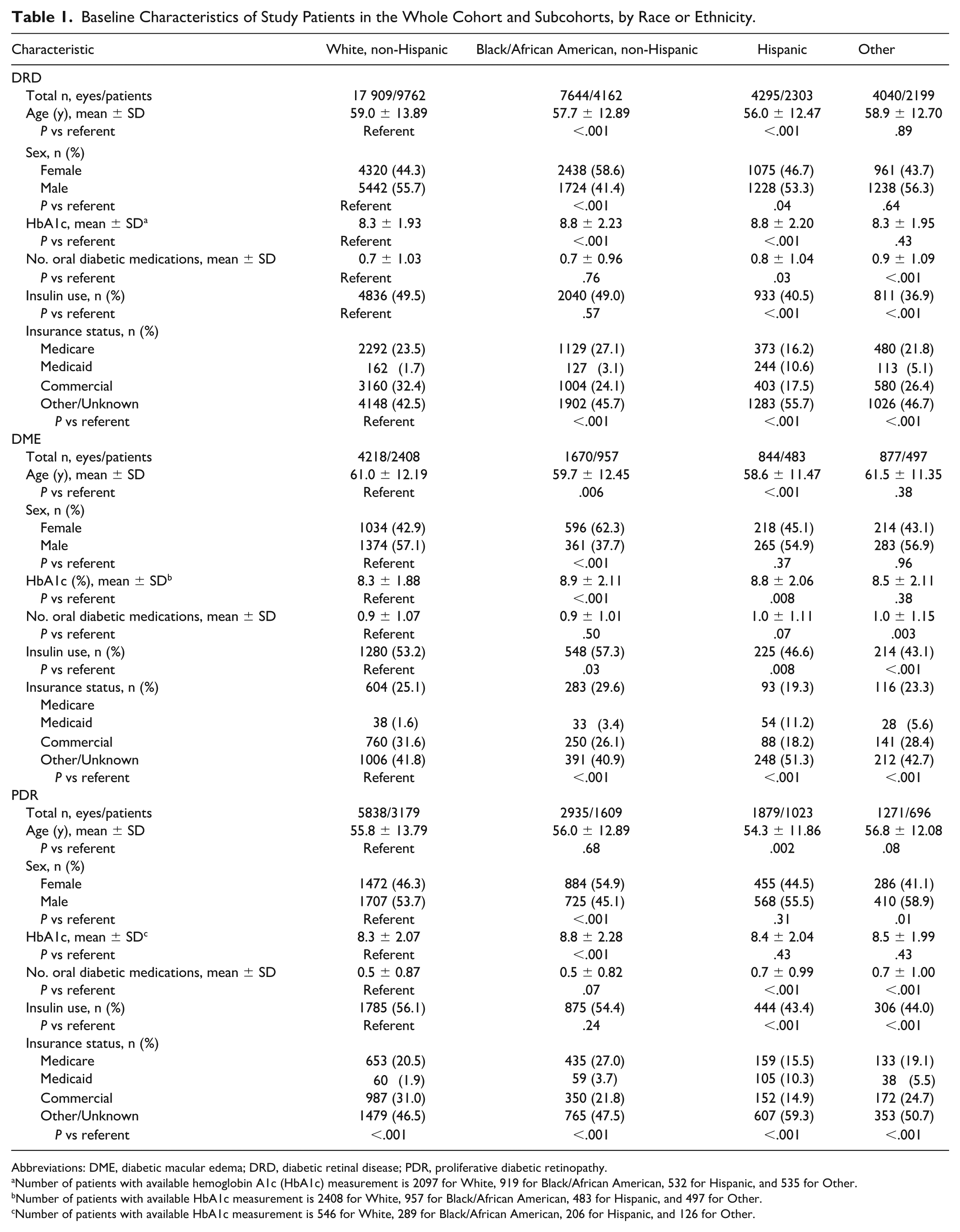
Abbreviations: DME, diabetic macular edema; DRD, diabetic retinal disease; PDR, proliferative diabetic retinopathy.
Number of patients with available hemoglobin A1c (HbA1c) measurement is 2097 for White, 919 for Black/African American, 532 for Hispanic, and 535 for Other.
Number of patients with available HbA1c measurement is 2408 for White, 957 for Black/African American, 483 for Hispanic, and 497 for Other.
Number of patients with available HbA1c measurement is 546 for White, 289 for Black/African American, 206 for Hispanic, and 126 for Other.
When evaluating visual deficits at the patient level (in the better-seeing eye) among all DRD patients, 3.6% of Black/African American patients (n = 151), 4.6% of Hispanic patients (n = 105), and 2.6% of patients reporting Other race or ethnicity (n = 57) all had significantly higher rates of severe vision loss (VA 20/200 or worse) as compared with 1.9% of White (non-Hispanic) patients (n = 188) (each P < .001 in both univariate and multivariable analyses). Similar disparities were seen in patients presenting with moderate vision loss (VA 20/70 or worse in their better-seeing eye), as 10.1% of Black/African American patients (n = 420), 11.9% of Hispanic patients (n = 274), and 8.4% of Other patients (n = 184) all presented at this level of vision loss, compared with only 6.4% of White (non-Hispanic) patients (n = 627) (each P < .001 in both univariate and multivariable analyses) (see Table 2 for patient-level results).
Patients With Visual Deficits Due to DRD and Its Vision-Threatening Forms, by Race or Ethnicity.
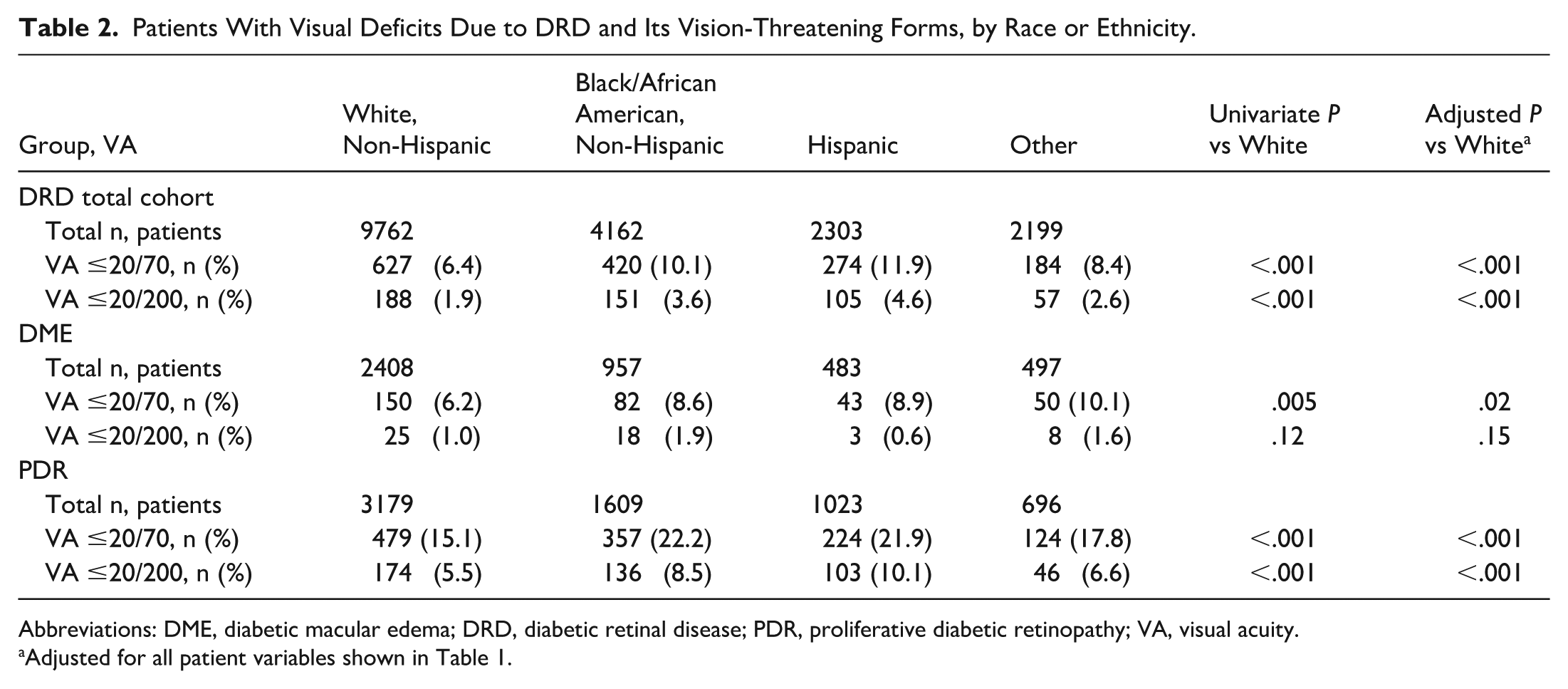
Abbreviations: DME, diabetic macular edema; DRD, diabetic retinal disease; PDR, proliferative diabetic retinopathy; VA, visual acuity.
Adjusted for all patient variables shown in Table 1.
Among all eyes of all patients with DRD, 6.7% (1208 eyes) of White (non-Hispanic) patients were recorded to have a VA worse than or equal to 20/200 at presentation, compared with 10.2% (779 eyes) of Black/African American patients, 11.6% (499 eyes) of Hispanic patients, and 7.8% (314 eyes) of Other patients (each P < .001 in both univariate and multivariable analyses). Similar disparities were seen when eyes presenting with moderate vision loss (VA 20/70 or worse) were evaluated in the DRD cohort, with 14.3% (2561 eyes) of White (non-Hispanic) patients having moderate vision loss, compared with 21.9% (941 eyes) of Hispanic patients, 19.3% (1479 eyes) of Black/African American patients, and 16.7% (676 eyes) of Other patients (each P < .001 in both univariate and multivariable analyses) (see Table 3 for eye-level results).
Eyes With Visual Deficits Due to DRD and Its Vision-Threatening Forms, by Race and Ethnicity.
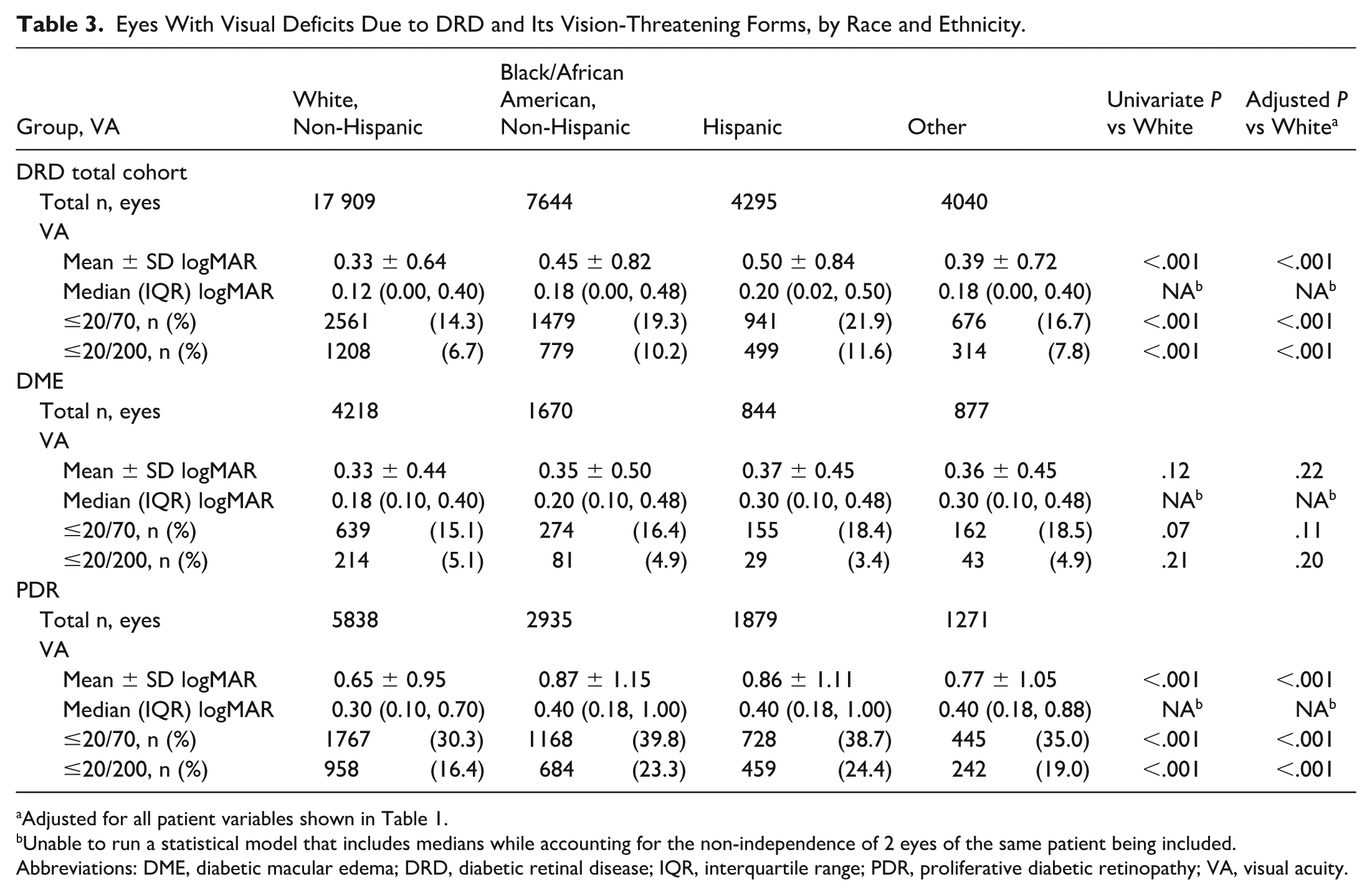
Adjusted for all patient variables shown in Table 1.
Unable to run a statistical model that includes medians while accounting for the non-independence of 2 eyes of the same patient being included.
Abbreviations: DME, diabetic macular edema; DRD, diabetic retinal disease; IQR, interquartile range; PDR, proliferative diabetic retinopathy; VA, visual acuity.
Eyes of White (non-Hispanic) patients had a mean VA of 20/43 (0.33 logMAR) and median VA of 20/26 (0.12 logMAR) at presentation. Eyes of Black/African American patients and Hispanic patients had lower mean and median VA values at presentation: for Black/African American patients, mean VA 20/56 (0.45 logMAR), median VA 20/30 (0.18 logMAR); for Hispanic patients, mean VA 20/63 (0.50 logMAR), median VA 20/32 (0.20 logMAR]; for patients with Other race or ethnicities, mean VA 20/49 (0.39 logMAR), median VA 20/30 (0.18 logMAR) (each P < .001 in both univariate and multivariable analyses).
DME Subcohort
The DME subcohort consisted of 7606 eyes and 4345 patients, of whom 55% (4218 eyes/2408 patients) identified as White (non-Hispanic), 22% (1670 eyes/957 patients) as Black/African American, 11% (844 eyes/483 patients) as Hispanic, and 12% (877 eyes/497 patients) as Other race or ethnicity. Similar to the whole DRD cohort, Black/African American patients with DME were younger on average than White (non-Hispanic) patients and were more likely to be female (each P < .006) (Table 1). Both Black/African American patients and Hispanic patients had higher mean HbA1c levels than White patients (each P < .008).
Unlike the DRD cohort, disparities in the DME cohort were found only when patients with a VA of 20/70 or worse in the better-seeing eye were evaluated. A total of 6.2% of White (non-Hispanic) patients presented with a VA of 20/70 or worse, compared with 8.6% of Black/African American patients, 8.9% of Hispanic patients, and 10.1% of Other patients (univariate P = .005, multivariable P = .02). No differences between groups in the number of patients with 20/200 or worse vision in the better-seeing eye were observed (univariate P = .12, multivariable P = .15) (Table 2). Moreover, no significant differences were identified in the percentage of eyes with VA deficits of 20/70 or 20/200 or worse across racial/ethnic groups (for VA 20/70, univariate P = .07, multivariable P = .11; for VA 20/200, univariate P = .21, multivariate P = .20) (Table 3).
The mean VA in eyes of DME patients was consistent across patients of different races and ethnicities, ranging from 20/43 to 20/47 (logMAR range, 0.33 to 0.37) (P = .12 vs White). The median VA of eyes of White (non-Hispanic) patients was 20/30 (0.18 logMAR), compared with a median VA of 20/32 (0.20 logMAR) in Black/African American patients and 20/40 (0.3 logMAR) in both Hispanic patients and patients of Other race or ethnicity.
PDR Subcohort
The PDR subcohort consisted of 11 923 eyes of 6507 patients, of whom 49% (5838 eyes/3179 patients) identified as White (non-Hispanic), 25% (2935 eyes/1609 patients) as Black/African American, 16% (1879 eyes/1023 patients) as Hispanic, and 10% (1271 eyes/696 patients) as Other race or ethnicity. Hispanic patients were younger on average than White (non-Hispanic) patients (P = .002), had more oral diabetic medications, and were less likely to be taking insulin (each P < .001). Black/African American patients were more likely on average to be female and had higher HbA1c levels than White (non-Hispanic) patients (each P < .001) (Table 1).
White (non-Hispanic) patients with PDR had better vision than the other groups, with only 5.5% of White (non-Hispanic) patients (n = 174) having a VA of 20/200 or worse, compared with 8.5% of Black/African American patients (n = 136), 10.1% of Hispanic patients (n = 103), and 6.6% of Other patients (n = 46) (each P < .001 in both univariate and multivariable analyses). Similar disparities were seen for patients with a VA of 20/70 or worse at presentation, with 15.1% of White (non-Hispanic) patients (n = 479), 22.2% of Black/African American patients (n = 357), 21.9% of Hispanic patients (n = 224), and 17.8% of Other patients (n = 124) each meeting this level of visual disability (each P < .001 in both univariate and multivariable analyses) (Table 2).
At the eye level in patients with PDR, eyes of White (non-Hispanic) patients were less likely to present with a VA of 20/200 or worse (16.4% [958 eyes]) compared with eyes of Black/African American patients (23.3% [684 eyes]), Hispanic patients (24.4% [459 eyes]), and patients of Other race or ethnicity (19.0% [242 eyes]) (each P < .001 in both univariate and multivariable analyses). Similar disparities were seen when eyes with a VA of 20/70 or worse were evaluated in patients with PDR, with the White (non-Hispanic) group having the lowest percentage of eyes meeting that criterion, at 30.3% of eyes (1767 eyes), compared with 39.8% of eyes in the Black/African American group (1168 eyes), 38.7% of eyes in the Hispanic group (728 eyes), and 35.0% of eyes in the Other group (445 eyes) (each P < .001 in both univariate and multivariable analyses) (Table 3).
The mean VA in eyes of White (non-Hispanic) patients was 20/89 (0.65 LogMAR), which was better than the mean VA of 20/148 (0.87 LogMAR) for Black/African American patient eyes, 20/145 (0.86 LogMAR) for Hispanic patient eyes, and 0.77 LogMAR for Other patient eyes (each P < .001). The median VA of eyes of patients with PDR was 20/40 (0.30 LogMAR) in the White (non-Hispanic) group, compared with a median VA of 20/50 (0.40 logMAR) for eyes of Black/African American, Hispanic, and Other patients (Table 2).
Conclusions
Using patient data from numerous large health systems across the US, we examined all eyes of patients presenting with DRD and its vision-threatening subforms of either DME or PDR. We identified clear disparities in visual deficits by racial and ethnic groups. We found that regardless of whether the evaluation focused on single eyes or on patients, Black/African American and Hispanic DRD patients had significantly higher rates of legal blindness (VA 20/200 or worse), with nearly twice and three times as many legally blind patients, respectively, when compared with White (non-Hispanic) patients. Similar disparities were found in evaluating eyes or patients with a VA of 20/70 or worse, with more Black/African American and Hispanic DRD patients than White DRD patients having this level of vision loss. Although these disparities between racial/ethnic groups were either not found or less pronounced in patients with DME, the eyes of patients with PDR showed higher rates of legal blindness and visual disability in Black/African American and Hispanic patients.
While diabetic retinal complications do not necessarily equate to vision loss, several recent studies estimating the prevalence of visually threatening diabetic disease can give context to our results. A survey of the Medical Expenditures Panel Survey from 2009 to 2018 found that among patients with diabetes, Black/African American and Hispanic patients were both about 1.6 times more likely to experience eye complications of diabetes than White patients. 11 Also, a recent study suggested that vision-threatening diabetic retinopathy is 2.5 times more prevalent in the Black/African American population and twice as prevalent in the Hispanic population than in the White, non-Hispanic population. 23 A report analyzing the IRIS Registry found that Black and Hispanic patients were 1.1 times and 1.7 times more likely, respectively, than White, non-Hispanic patients to present with a VA of 20/200 or worse at initiation of anti-VEGF therapy for DME. 24 While our results in patients with DME did not reach statistically significant differences (possibly due to our smaller sample size), the rates of vision loss at this level across the different groups were similar to those found in the IRIS analysis.
Existing research has rarely focused on documenting the prevalence of visual deficits due to DRD across racial groups, leaving few comparable studies for Black/African American or Hispanic patients. Previous studies have estimated the prevalence of legal blindness attributable to DRD in the better-seeing eye as between 1.6% and 4.1%.3,5,6 Compared with previous studies focused on Black/African American patients, our results suggest that racial disparities in visual deficits between White and Black/African American patients may be smaller than previously reported. The Baltimore Eye Study found that visual impairment caused by diabetic retinopathy was over 5 times more prevalent in Black/African American patients than White patients, which is more than double our estimates. 25 However, it should be noted that this comparison is imperfect, as the Baltimore Eye Study used a different definition of visual impairment and included a broader population. The New Jersey 725 Type 1 Diabetes study may provide a lower bound for the prevalence of legal blindness due to DRD. It found that 4.75% of Black/African American patients with diabetes had a VA of 20/200 or worse in their better-seeing eye. 26 It is thus reassuring that our study, in which the cohort included only patients with diabetic eye complications, documented higher rates of legal blindness than the New Jersey 725 study.
The disparities in VA among DRD patients across racial/ethnic groups underscore findings in existing literature indicating racial/ethnic DRD screening-rate disparities. Studies have consistently shown that Black/African American and Hispanic populations are less likely to be screened for DRD than Whites.13 –16,27 Shifts in clinical practice that began during the COVID-19 pandemic, including the increased use of telemedicine, may also exacerbate existing disparities. 28 Public health interventions may seek to alleviate these disparities in outcomes by reducing barriers to screening, such as poor health literacy, lack of insurance coverage, lack of transportation, and mistrust of the healthcare system.27,29 –31 Differentials in screening across cohorts may also have affected our results, as groups with higher screening rates would be more likely to have DRD discovered at less severe stages, impacting who may have been considered truly at risk for vision loss in our study.
Improving screening for DRD alone may not be sufficient, as disparities have been reported across the spectrum of diabetic disease. Racial and ethnic differences in the treatment and management of diabetes mellitus are well documented. Previous investigations of DRD care have found that Black/African American patients are less likely to complete all of the American Diabetes Association recommended processes of diabetes care. 11 In our study, Black/African American patients were found to have higher HbA1c levels than White, non-Hispanic patients across the DRD, DME, and PDR cohorts, indicative of worse systemic disease. Similarly, Hispanic patients in our DRD cohort had higher rates of insulin use than the White, non-Hispanic patients, again suggestive of more severe underlying disease. This has important implications for interpreting our results and understanding that race/ethnicity is likely a surrogate variable for systemic barriers to care, as opposed to a cause of poorer visual outcomes.
Limitations of this study include the study design. Deidentification within the SOURCE database prevented detailed chart review and required reliance on ICD-10 codes for diagnoses. However, past scholarship finds that ICD-10 codes are validated for the identification of DRD, DME, and PDR in research settings.19 –21,32 The SOURCE database covers the subset of patients who seek medical services in tertiary care health systems across the country. Accordingly, our study cohort may oversample uninsured and poorly insured patients relative to the wider population. Additionally, our protocol excluded patients with concomitant non-DRD vision-threatening diseases, except cataracts. The inclusion of patients with cataracts may have impacted VA estimates. Moreover, we were unable to evaluate important factors that could impact health care utilization and visual outcomes, such as health literacy, diet, access to care, and other systemic factors that may be different across racial and ethnic groups. Further research could explore the robustness of our findings to changes in study populations.
DRD remains a significant contributor to visual impairment in the US. While there have been significant advances in the treatment of DRD and DM over the last 2 decades, persistent disparities in visual deficits remain across racial and ethnic groups. Black/African American and Hispanic patients with DRD in our study were more likely to experience visual disabilities, including moderate visual loss and legal blindness, than White, non-Hispanic patients. Future interventions should work to understand the underlying causes to best address these disparities among our patients.
Footnotes
Acknowledgements
No large language models were used to complete the study or write the manuscript.
Ethical Approval
The University of Pennsylvania has deemed all projects performed on the SOURCE database exempt from review due to its deidentified nature.
Statement of Informed Consent
Informed consent was waived due to the deidentified nature of the data set.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Pennsylvania Lions Sight Conservation and Eye Research Foundation. Additional funding was provided by Research to Prevent Blindness.
Data Availability
The data that support the findings of this study can be made available through contracting directly with the SOURCE Repository.
