Abstract
Keywords
Introduction
Geographic atrophy (GA), an advanced form of age-related macular degeneration (AMD), leads to progressive and irreversible visual function impairment owing to permanent destruction of photoreceptors, retinal pigment epithelium, and choriocapillaris in the macula. 1 Multiple ocular and systemic risk factors have been associated with an increased rate of GA lesion growth, including nonsubfoveal GA location, large GA lesion size, multifocal GA, and the presence of exudation or choroidal neovascularization (CNV) in the fellow eye.1–3
In 2023, the first 2 GA treatment options were approved by the US Food and Drug Administration. Pegcetacoplan, a targeted complement inhibitor at the level of C3 and C3b, was approved on February 17, 2023, for patients with GA, with or without subfoveal involvement, with dosing every 25 to 60 days. 2 The safety and efficacy of pegcetacoplan were demonstrated in the OAKS and DERBY trials, which were 2 identical, multicenter, randomized, double-masked, sham-controlled phase 3 trials enrolling patients with GA secondary to AMD. 2 Together, OAKS and DERBY enrolled a heterogenous population of 1258 individuals with subfoveal and nonsubfoveal GA, including those with CNV in the fellow eye, which is often exclusionary in GA clinical trials.
In OAKS, compared with the sham group, pegcetacoplan monthly reduced GA lesion growth at month 12 by 34% (P < .001) among patients with nonsubfoveal GA.2,4 In DERBY, compared with the sham group, pegcetacoplan monthly reduced GA lesion growth at month 12 by 17% (P = .07) among patients with nonsubfoveal GA. In the pooled OAKS and DERBY analysis, compared with the sham group, pegcetacoplan monthly reduced GA lesion growth by 26% (P < .001) among patients with nonsubfoveal GA and by 16% (P = .001) at month 12 across all GA lesion subtypes (nonsubfoveal and subfoveal GA combined). 2 A less frequent dosing regimen of pegcetacoplan every other month also reduced GA lesion growth compared with the sham arm in both trials (OAKS: nonsubfoveal GA subgroup, 21%, P = .02; DERBY: nonsubfoveal GA subgroup, 25%, P = .003; pooled: nonsubfoveal GA subgroup, 23%, P < .001).2,4
Avacincaptad pegol, a complement C5 inhibitor, was approved by the Food and Drug Administration on August 4, 2023, for monthly dosing for up to 12 months in patients with GA. The safety and efficacy of avacincaptad pegol monthly were demonstrated in 2 multicenter, randomized, double-masked, sham-controlled clinical trials: the phase 2b/3 GATHER1 trial (NCT02686658) and the phase 3 GATHER2 trial (NCT04435366). Both avacincaptad pegol trials enrolled patients with nonsubfoveal GA. In addition, CNV in the fellow nonstudy eye was exclusionary.5,6 Compared with the sham group, monthly injections of 2 mg avacincaptad pegol reduced GA lesion growth at month 12 by 17% in GATHER2 (P = .003).5,6
In the absence of a head-to-head clinical trial comparing pegcetacoplan vs avacincaptad pegol in patients with nonsubfoveal GA, it is challenging to assess comparative efficacy. Although direct comparisons of phase 3 clinical trial outcomes suggest that pegcetacoplan may have stronger efficacy than avacincaptad pegol in the nonsubfoveal GA population, these comparisons of top-line, unadjusted data are inherently limited owing to variations in cohort characteristics across trials, potentially introducing confounding factors. Matching-adjusted indirect comparisons are a well-validated analytic tool, recognized by health technology assessment and payer organizations, for conducting adjusted comparisons of treatments across trials when individual patient data are available from 1 trial and only aggregate data are available from another trial.7,8
Matching-adjusted indirect comparisons account for cross-trial differences by applying a propensity score–based approach to balance the distribution of baseline characteristics across trial populations.9,10 This study used anchored matching-adjusted indirect comparison, aligning key ocular characteristics across trials by matching and balancing individual patient data from the less restrictive pegcetacoplan trials to the more restrictive avacincaptad pegol trial. This approach enabled an indirect comparative assessment of pegcetacoplan vs avacincaptad pegol in individuals with nonsubfoveal GA. This study compares GA lesion growth at month 12 with pegcetacoplan monthly vs avacincaptad pegol monthly and with pegcetacoplan every other month vs avacincaptad pegol monthly.
Methods
Data
Data sources included individual patient data from 2 phase 3 pegcetacoplan trials (OAKS and DERBY) and aggregate reported data from the phase 3 avacincaptad pegol trial (GATHER2).5,6 Trial characteristics and inclusion and exclusion criteria are reported in Supplemental Table 1.
OAKS (NCT03525613) and DERBY (NCT03525600) were 2 identical phase 3, multicenter, randomized, double-masked, sham-controlled trials comparing pegcetacoplan with sham injections in patients with GA. There were 1211 patients in the modified intention-to-treat population in OAKS and DERBY. This study used matching-adjusted indirect comparisons with individual patient data from patients randomized to receive 15 mg pegcetacoplan (pegcetacoplan monthly or pegcetacoplan every other month, separately) or sham injections.
GATHER2 (NCT04435366) was a phase 3, multicenter, randomized, double-masked, sham-controlled trial comparing avacincaptad pegol monthly with sham injections in patients with nonsubfoveal GA. There were 447 patients in the modified intention-to-treat population in GATHER2. This study used matching-adjusted indirect comparisons with reported results for patients randomized to receive monthly 2 mg avacincaptad pegol monthly or sham injections.
Study Population
Common inclusion and exclusion criteria across OAKS, DERBY, and GATHER2 included a lower limit of best-corrected visual acuity (BCVA) of 20/320, GA area between 2.5 mm2 and 17.5 mm2, and at least 1 focal lesion of 1.25 mm2 or larger if GA was multifocal (Supplemental Table 1).
Key eligibility criteria from GATHER2 but not OAKS and DERBY included GA lesion borders entirely within 1500 µm from the foveal center, exclusion of fellow nonstudy eye CNV, and vision between 20/25 and 20/320. These GATHER2 eligibility criteria were applied to the OAKS and DERBY individual patient data to select a similar subpopulation across trials. In other words, patients in OAKS and DERBY who did not meet GATHER2 eligibility criteria were excluded from the analyses. Specifically, GATHER2 included patients with nonsubfoveal GA where the GA lesion was located within 1500 µm from the foveal center. Therefore, for this study, patients from OAKS and DERBY with a GA lesion distance from the foveal center greater than 0 to 1500 µm were included, while those with subfoveal GA (lesion distance of 0 µm from the foveal center) and those with a lesion distance more than 1500 µm from the foveal center were excluded.
In addition, patients from OAKS and DERBY with BCVA better than 20/25, who would have been excluded from GATHER2, were excluded from this analysis. Patients with fellow nonstudy eye CNV were also excluded, consistent with GATHER2 criteria. Following these exclusions, the subpopulation from OAKS and DERBY included in this analysis met, to the extent possible, the eligibility criteria reported for GATHER2.
Statistical Methods
The outcome of interest was absolute GA lesion growth from baseline to month 12. Comparisons were made between pegcetacoplan monthly vs avacincaptad pegol monthly (primary analysis) and between pegcetacoplan every other month vs avacincaptad pegol monthly (secondary analysis). Statistical significance was set at P < .05. Mean values are ± SD.
Anchored matching-adjusted indirect comparisons were conducted separately for OAKS vs GATHER2 and for DERBY vs GATHER2. A propensity score–based approach using logistic regression, with enrollment in GATHER2 as the dependent variable, was used to estimate the likelihood of enrollment in GATHER2 vs the pegcetacoplan trials for each patient in OAKS and DERBY, separately. Key variables were selected a priori, including age and ocular characteristics available across trials (baseline square root of GA lesion size, bilaterality of GA, BCVA, low-luminance BCVA, lesion focality [unifocal or multifocal], and intraocular pressure), and were included as independent variables in the logistic regression to ensure balance and reduce bias. Individual patient data from OAKS and DERBY were reweighted using inverse propensity scores to match the means, SDs, and proportions of the baseline characteristics reported in aggregate for GATHER2.5,6
To determine the adequacy of the model, the distribution of the weights for patients in DERBY and OAKS were inspected using histograms. The effective sample size, defined as the equivalent number of independent, nonweighted patients required to yield an estimate with comparable precision to that of the weighted sample estimate, was reported for each analysis.
GA lesion growth from baseline to month 12 with pegcetacoplan vs avacincaptad pegol was compared in OAKS vs GATHER2 and in DERBY vs GATHER2, before and after matching. The results of the separate matching-adjusted indirect comparisons were combined using meta-analysis based on the inverse of the variance of each effect estimate.
Results
Primary Analysis: Pegcetacoplan Monthly vs Avacincaptad Pegol Monthly
OAKS vs GATHER2
The analysis included 103 patients from OAKS (61 received pegcetacoplan monthly; 42 received sham) and 447 patients from GATHER2 (225 received avacincaptad pegol monthly; 222 received sham). After matching, the effective sample size for pegcetacoplan monthly and sham groups in OAKS was 24.47 and 12.16, respectively. Key clinical characteristics, including age, BCVA, and low-luminance BCVA, were balanced across trials (Table 1A).
Baseline Characteristics: Pegcetacoplan vs Avacincaptad Pegol Monthly.
A. OAKS vs GATHER2
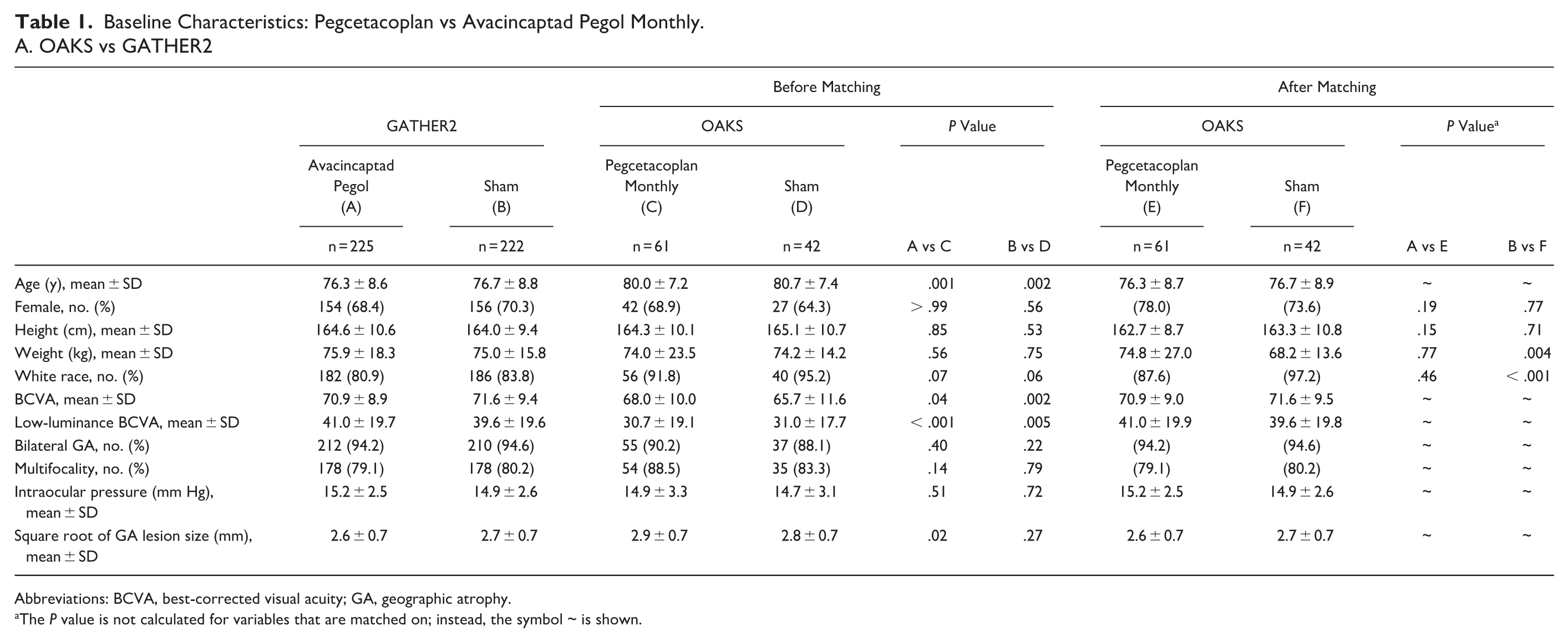
Abbreviations: BCVA, best-corrected visual acuity; GA, geographic atrophy.
The P value is not calculated for variables that are matched on; instead, the symbol ~ is shown.
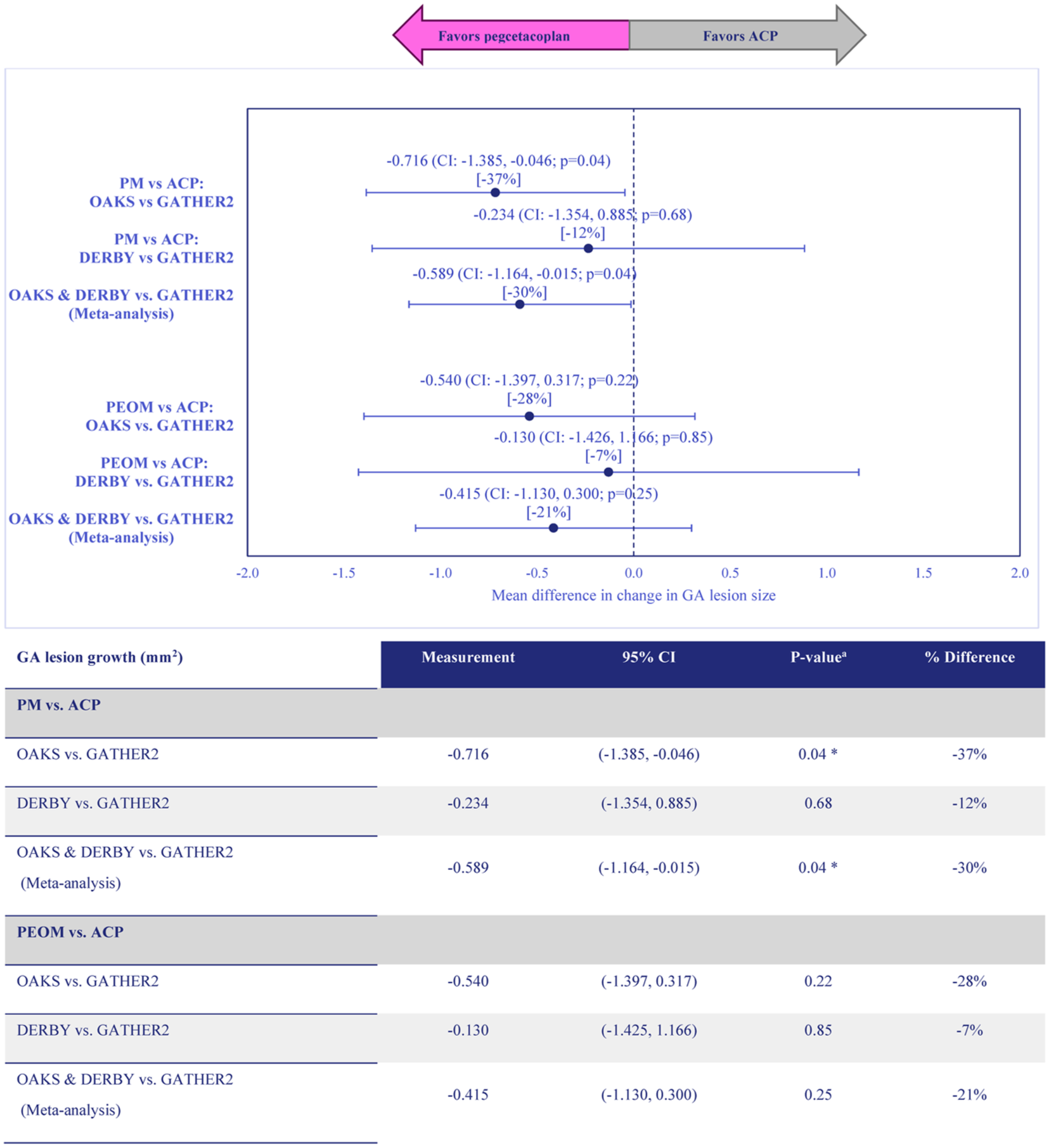
After matching and balancing populations, the difference in nonsubfoveal GA lesion growth at month 12 between pegcetacoplan monthly and sham in OAKS was −1.121 mm2, corresponding to a 42% reduction in GA lesion growth with pegcetacoplan monthly vs sham (Table 2). The difference in GA lesion growth between avacincaptad pegol and sham in GATHER2 was −0.405 mm2, corresponding to a 17% reduction in GA lesion growth with avacincaptad pegol vs sham (Table 2). The anchored difference for GA lesion growth between pegcetacoplan monthly and avacincaptad pegol monthly was −0.716 mm2 (P = .04), corresponding to a significant 37% reduction in GA lesion growth favoring pegcetacoplan monthly vs avacincaptad pegol monthly (Figure 1, Table 3).
Comparisons of geographic atrophy lesion growth at month 12 with pegcetacoplan vs avacincaptad pegol.
DERBY vs GATHER2
The analysis included 102 patients from DERBY (49 received pegcetacoplan monthly; 53 received sham) and 447 patients from GATHER2 (225 received avacincaptad pegol; 222 received sham). After matching, the effective sample sizes for the pegcetacoplan monthly and sham groups in DERBY were 17.43 and 13.95, respectively. Age and all ocular variables were balanced across trials (Table 1B).
B. DERBY vs GATHER2.
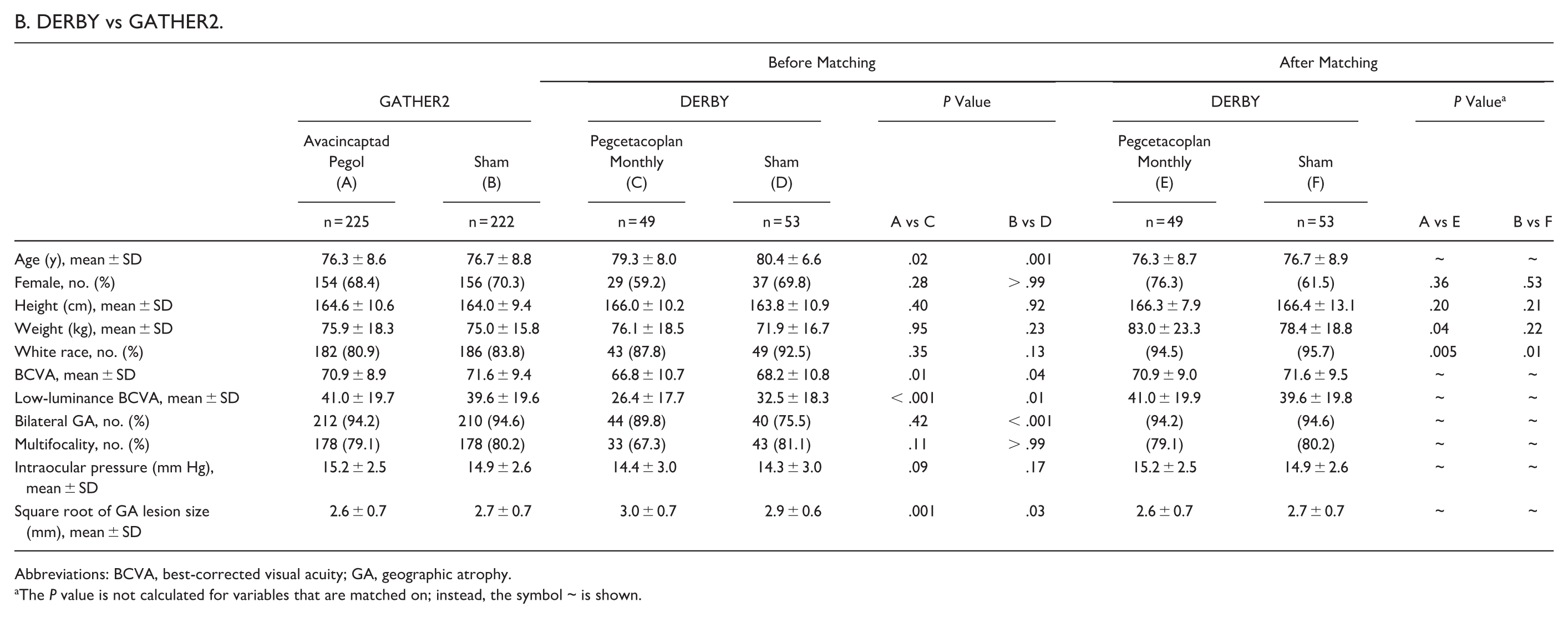
Abbreviations: BCVA, best-corrected visual acuity; GA, geographic atrophy.
The P value is not calculated for variables that are matched on; instead, the symbol ~ is shown.
The difference in GA lesion growth between pegcetacoplan monthly and sham in DERBY was −0.639 mm2, corresponding to a 23% reduction in GA lesion growth with pegcetacoplan monthly vs sham (Table 2). The difference between avacincaptad pegol and sham in GA lesion growth in GATHER2 was −0.405 mm2, corresponding to a 17% reduction in GA lesion growth with avacincaptad pegol vs sham (Table 2). The anchored difference for GA lesion growth between pegcetacoplan monthly and avacincaptad pegol monthly was directionally in favor of pegcetacoplan monthly (−0.234 mm2), corresponding to a 12% reduction in GA lesion growth with pegcetacoplan monthly vs avacincaptad pegol monthly, but not statistically significant (P = .68) (Figure 1, Table 3).
Arm-By-Arm Geographic Atrophy Lesion Growth Results After Matching.
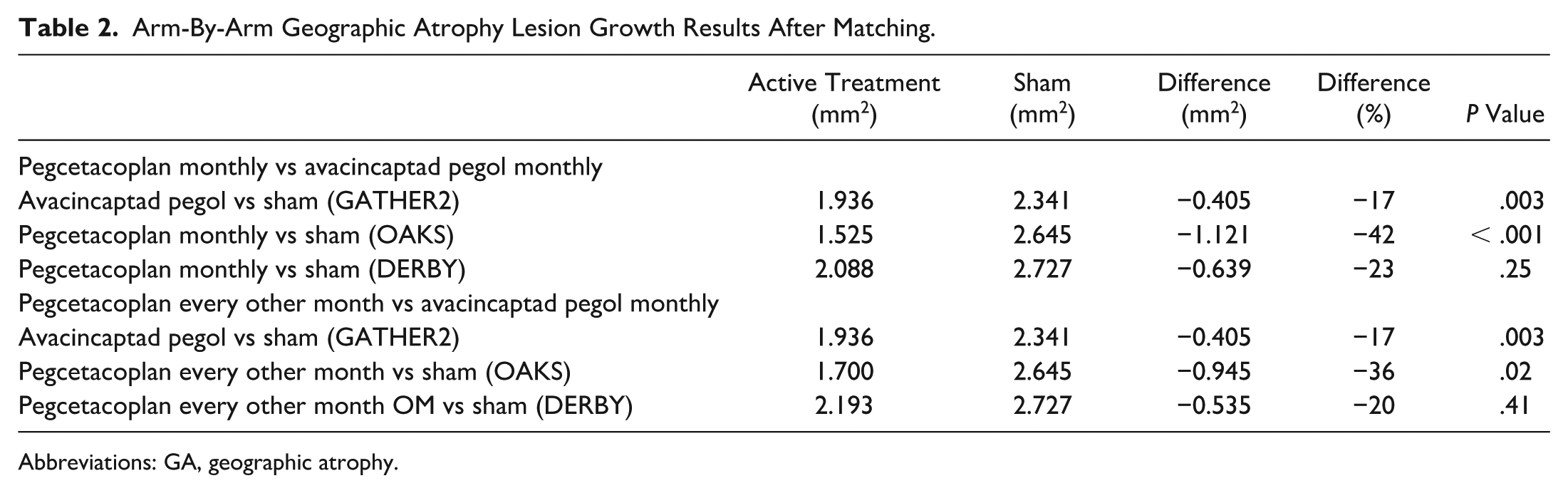
Abbreviations: GA, geographic atrophy.
Comparisons of Pegcetacoplan Monthly vs Avacincaptad Pegol Geographic Atrophy Lesion Growth Results After Matching.
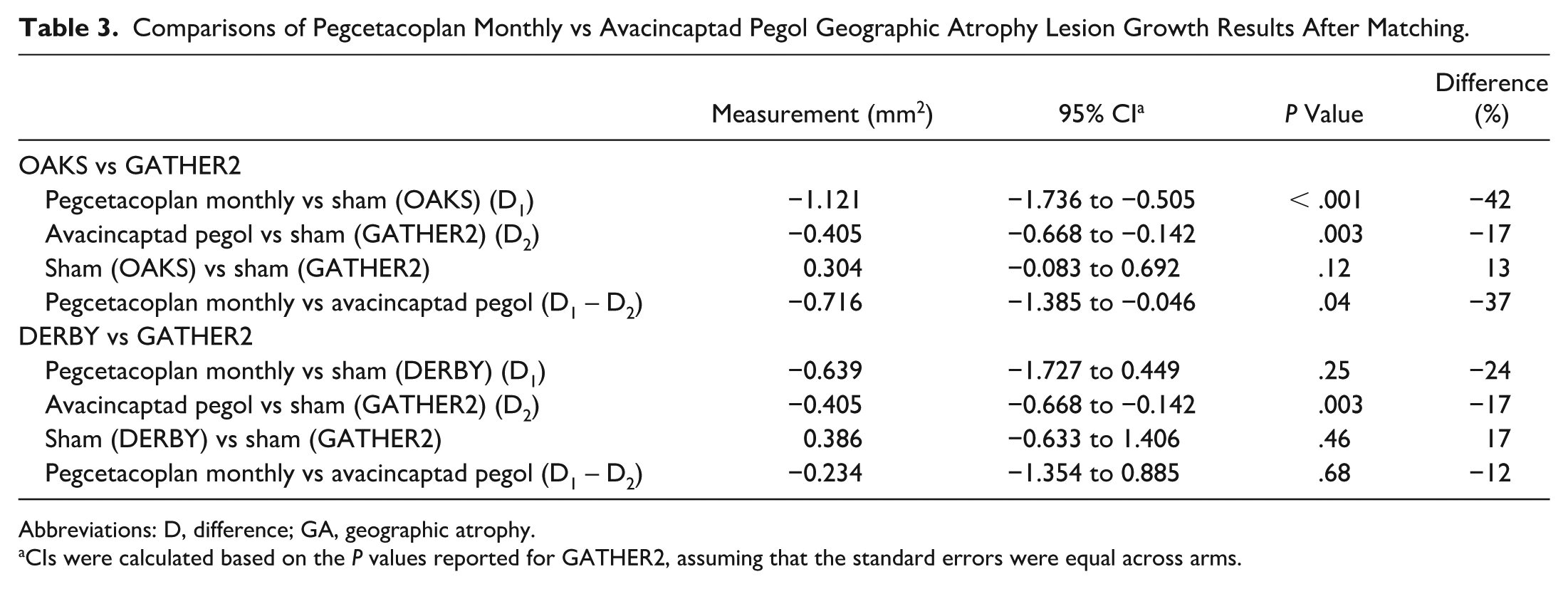
Abbreviations: D, difference; GA, geographic atrophy.
CIs were calculated based on the P values reported for GATHER2, assuming that the standard errors were equal across arms.
Meta-Analysis
In a meta-analysis combining the OAKS vs GATHER2 and DERBY vs GATHER2 analyses, the pooled effect for pegcetacoplan monthly vs avacincaptad pegol monthly was −0.589 mm2 (P = .04), corresponding to a significant 30% reduction in GA lesion growth favoring pegcetacoplan monthly vs avacincaptad pegol monthly (Figure 1).
Secondary Analysis: Pegcetacoplan Every Other Month vs Avacincaptad Pegol Monthly
OAKS vs GATHER2
The analysis included 96 patients from OAKS (54 received pegcetacoplan every other month; 42 received sham) and 447 patients from GATHER2 (225 received avacincaptad pegol monthly; 222 received sham). After matching, the effective sample size for pegcetacoplan every other month and sham groups in OAKS were 5.73 and 12.16, respectively. Age and all ocular variables were balanced across trials (Supplemental Table 2A).
The difference in GA lesion growth between pegcetacoplan every other month and sham in OAKS was −0.945 mm2, corresponding to a 36% reduction in GA lesion growth with pegcetacoplan every other month vs sham (Table 2). The difference between avacincaptad pegol and sham in GA lesion growth in GATHER2 was −0.405 mm2, corresponding to a 17% reduction in GA lesion growth with avacincaptad pegol vs sham (Table 2). The anchored difference for GA lesion growth between pegcetacoplan every other month and avacincaptad pegol monthly was directionally in favor of pegcetacoplan every other month (−0.540 mm2), corresponding to a 28% reduction in GA lesion growth with pegcetacoplan every other month vs avacincaptad pegol monthly, but not statistically significant (P = .22) (Figure 1, Table 4).
Comparisons of Pegcetacoplan Every Other Month vs Avacincaptad Pegol Geographic Atrophy Lesion Growth Results After Matching.
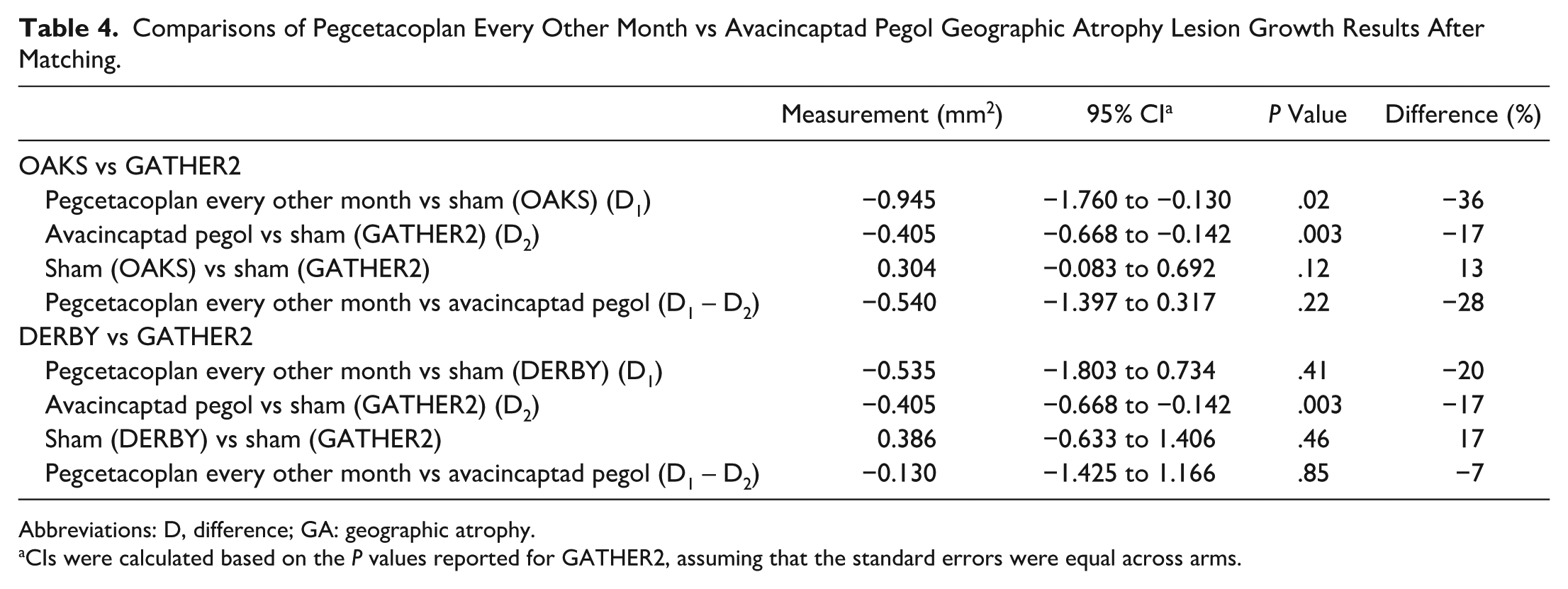
Abbreviations: D, difference; GA: geographic atrophy.
CIs were calculated based on the P values reported for GATHER2, assuming that the standard errors were equal across arms.
DERBY vs GATHER2
The analysis included 108 patients from DERBY (55 received pegcetacoplan every other month; 53 received sham) and 447 patients from GATHER2 (225 received avacincaptad pegol monthly; 222 received sham). After matching, the effective sample size for pegcetacoplan every other month and sham groups in DERBY were 9.63 and 13.95, respectively. Age and all ocular variables were balanced across trials (Supplemental Table 2B).
The difference in GA lesion growth between pegcetacoplan every other month and sham in DERBY was −0.535 mm2 (Table 2). The difference in GA lesion growth between avacincaptad pegol monthly and sham in GATHER2 was −0.405 mm2 (Table 2). The anchored difference for GA lesion growth between pegcetacoplan every other month and avacincaptad pegol was also directionally in favor of pegcetacoplan (−0.130 mm2), corresponding to a 7% reduction in GA lesion growth with pegcetacoplan every other month vs avacincaptad pegol, but not statistically significant (P = .85) (Figure 1, Table 4).
Meta-Analysis
In the meta-analysis, the pooled effect of pegcetacoplan every other month vs avacincaptad pegol monthly in OAKS vs GATHER2 and DERBY vs GATHER2 was directionally in favor of pegcetacoplan every other month (−0.415 mm2), corresponding to a numerically greater 21% reduction in GA lesion growth with pegcetacoplan every other month vs avacincaptad pegol monthly, but was not statistically significant (P = .25) (Figure 1).
Conclusions
In the absence of direct comparative trials between pegcetacoplan and avacincaptad pegol, this study indirectly compared the efficacy of pegcetacoplan vs avacincaptad pegol among patients with nonsubfoveal GA using anchored MAIC and meta-analyses.7,8 OAKS and DERBY were larger trials than GATHER2 with a more heterogeneous population, allowing for the application of the exclusion criteria from the avacincaptad pegol trial to the pegcetacoplan trials’ individual patient data to select a comparable subpopulation.
A propensity score–based approach was used to account for potential cross-study differences in patient characteristics, including age and ocular variables, by balancing the distribution of these variables across the trials.9,10 The sham vs sham comparisons across trials after matching were not statistically significant, indicating that the sham arms were well balanced after adjusting for key variables. For each comparison, results from the matching-adjusted indirect comparisons were combined using a fixed-effects meta-analysis, as OAKS and DERBY were similarly designed trials conducted in the same population.
This study, using matching-adjusted indirect comparisons to adjust for key baseline characteristics, found greater efficacy with pegcetacoplan monthly compared with avacincaptad pegol monthly. The primary analysis demonstrated a statistically significant 30% greater reduction in absolute GA lesion growth at month 12 with pegcetacoplan monthly vs avacincaptad pegol monthly. Pegcetacoplan and avacincaptad pegol have different mechanisms of action—pegcetacoplan inhibits C3 and C3b centrally within the complement pathway, whereas avacincaptad pegol inhibits C5 more downstream. Prior literature has suggested that upstream complement inhibitors may provide greater efficacy than downstream C5 complement inhibitors.11–13 The results of the present primary meta-analysis, comparing monthly dosing of pegcetacoplan and avacincaptad pegol across trials, are generally consistent with these prior findings.
The secondary analysis found a directionally greater, though not statistically significant, reduction in GA lesion growth with less frequent injections of pegcetacoplan every other month vs avacincaptad pegol monthly. The option of a Food and Drug Administration-approved less frequent dosing schedule for pegcetacoplan may have important implications for treatment choice. Patients have reported the burden associated with frequent intravitreal (IVT) injections in other diseases, including neovascular AMD and diabetic macular edema.14–16 In addition, regular clinic visits can be expensive and time-consuming for both patients and their accompanying family members or caregivers, leading to productivity loss and poor compliance with treatment. 14
Patients may also experience discomfort and anxiety related to IVT injections. 17 In this context, a less frequent dosing schedule and fewer clinic visits may be preferred by some patients for the convenience. 18 Thus, our finding that efficacy with pegcetacoplan every other month was not significantly different from avacincaptad pegol monthly could influence treatment choice. Practically, pegcetacoplan every other month may be logistically easier than concurrent monthly dosing of anticomplement agents, particularly if a patient also develops CNV requiring ongoing antivascular endothelial growth factor therapy.
Only the comparable phase 3 trials with pegcetacoplan (OAKS, DERBY) or avacincaptad pegol (GATHER2) were included in this analysis. GATHER1 was not included for multiple reasons. 5 First, GATHER1 used a phase 2b/3 screening trial design that was not comparable to the other phase 3 trials in this study. Second, in GATHER1, patients who developed CNV (wet AMD) in the study eye were excused from the trial and their lesion growth data were not included in efficacy assessments, 5 whereas GATHER2 and the phase 3 pegcetacoplan studies included GA lesion growth data from patients who developed CNV. These differences in study design would not be reconcilable with matching-adjusted indirect comparisons. Finally, GATHER1 did not report key baseline variables such as lesion focality, preventing matching on key baseline variables that are required in the matching-adjusted indirect comparisons.
Altogether, the findings of this study suggest that pegcetacoplan has better efficacy than avacincaptad pegol. The option of a less frequent pegcetacoplan dosing schedule with pegcetacoplan, with efficacy not significantly different from the more frequent dosing schedule with avacincaptad pegol, may have important implications on treatment choice, with the potential to improve patient compliance and treatment outcomes. 19
Limitations
Limitations inherent to the matching-adjusted indirect comparison approach include that individual patient data were available for pegcetacoplan but not for avacincaptad pegol; therefore, patient-level adjustment between trials was not feasible. Although most inclusion and exclusion criteria were consistent across OAKS, DERBY, and GATHER2, there were some differences in study design and enrollment criteria. Many of these differences were accounted for in the matching-adjusted indirect comparisons, as described herein; however, some residual differences remained.
For example, patients in OAKS and DERBY were required to be at least 60 years of age, while patients in GATHER2 were required to be at least 50 years of age. Patients aged 50 to 59 in GATHER2 could not be excluded from the analyses, as only aggregate data were available for GATHER2. To mitigate this concern, age was matched to balance the distribution of baseline characteristics across trials. In addition, the inclusion and exclusion criteria may have been interpreted differently by investigators across trials. In anchored matching-adjusted indirect comparisons, only effect modifiers (ie, variables that alter the effect of the treatment on the outcome such that effectiveness differs by strata) need to be accounted for in the matching-adjusted indirect comparisons weights, as there is expected to be no confounding owing to randomization.
Because not all variables could be simultaneously matched, priority for matching was assigned to variables assessed to be effect modifiers based on clinical input (ie, age and key ocular variables). To avoid bias, these variables were selected a priori. After matching, age and key ocular variables were balanced across trials; however, some differences remained across trial arms in race and weight, which were not expected to have biased the results, as race and weight are prognostic variables rather than effect modifiers. Moreover, baseline matching was only possible for characteristics that were reported for GATHER2.
A common misperception arises from confusion between effective sample size and sample size. As mentioned previously, the effective sample size is defined as the equivalent number of independent, non-weighted patients needed to yield an estimate with comparable precision to that of the weighted sample estimate. In other words, the effective sample size is a statistical measure of the precision of the analysis, distinct from sample size. Small effective sample sizes are common in matching-adjusted indirect comparisons, primarily owing to limited overlap between trials, as matching-adjusted indirect comparisons require a trade-off between the number of adjustment variables and the effective sample size.9,20,21
The matching variables leading to the greatest reductions in effective sample size are those that are most imbalanced across trials, and therefore also the most important to adjust for. 22 To preserve effective sample size in this study, age and ocular variables were prioritized for matching. Other variables, such as race and weight, were considered prognostic variables that were not expected to bias the results. Previous research has reported an average reduction in effective sample size of 70% in matching-adjusted indirect comparisons supporting health technology appraisals, and 80% (up to 98%) in the applied published literature. 23
Generalizability to a broader or different population may be limited. Because reweighting was conducted to match the characteristics of the population in the avacincaptad pegol trial, the population of this analysis is more representative of the narrower GA population included in the avacincaptad pegol phase 3 trial than of the heterogeneous GA population included in the pegcetacoplan phase 3 trials. Future comparison studies between pegcetacoplan and avacincaptad pegol are warranted to address the generalizability limitations of this study.
In these matching-adjusted indirect comparisons among patients with nonsubfoveal GA, pegcetacoplan showed a better efficacy profile than avacincaptad pegol, as evidenced by a significantly greater reduction in GA lesion growth at month 12 with pegcetacoplan monthly vs avacincaptad pegol monthly, and a directionally greater but not statistically significant reduction with pegcetacoplan every other month vs avacincaptad pegol monthly. These findings inform clinical decision-making for patients with GA. Future studies should investigate the comparative effectiveness of pegcetacoplan vs avacincaptad pegol in the real world and assess how differences in efficacy profiles and dosing schedules may influence patient preferences for GA treatments.
Supplemental Material
sj-docx-1-vrd-10.1177_24741264251379842 – Supplemental material for Efficacy of Intravitreal Pegcetacoplan vs Avacincaptad Pegol in Patients With Geographic Atrophy
Supplemental material, sj-docx-1-vrd-10.1177_24741264251379842 for Efficacy of Intravitreal Pegcetacoplan vs Avacincaptad Pegol in Patients With Geographic Atrophy by Paul Hahn, David Eichenbaum, Dilsher S. Dhoot, Charles C. Wykoff, Michael A. Klufas, Michele Intorcia, Daniel Jones, Sujata P. Sarda, Priyanka Bobbili, Rose Chang, Maryaline Catillon, Chunyi Xu, Kirthana Sarathy, Mei Sheng Duh, Caroline R. Baumal and Varun Chaudhary in Journal of VitreoRetinal Diseases
Footnotes
Acknowledgements
Research assistance was provided by Alice Qu, BA, an employee of Analysis Group, Inc.
Ethical Approval
This was a post-hoc analysis of previously collected, anonymized trial data; therefore, ethical review was not required.
Statement of Informed Consent
This was a post-hoc analysis of previously collected, anonymized trial data; therefore, informed consent was not required.
Declaration of Conflicting Interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Dr. Hahn reports serving on the advisory board of Alcon, Apellis, DORC, EyePoint, and Genentech; serving as a consultant for Alcon, Apellis, DORC, EyePoint, and Genentech; serving as a speaker for Eyepoint and Genentech; and receiving research funding from Alexion, Adverum, Apellis, Eyepoint, Genentech, Notal Vision, OcuTerra, Regeneron, Regenxbio, and Samsara. Dr. Eichenbaum reports serving as an investigator for 4DMT, Alexion, Alkahest, Allegenesis, Annexon, AsclepiX, Aviceda, Bayer, Chengdu, EyePoint, Gemini, Genentech, Gyroscope, Ionis, IvericBio, Kodiak, Mylan, NGM, Novartis, Ocular Therapeutix, Opthea, RecensMedical, Regeneron, Regenxbio, and Unity; serving as a speaker for Allergan, Apellis, Bausch + Lomb, Bayer, EyePoint, Genentech, and IvericBio; serving as a consultant for Alimera, Allergan, Apellis, Bausch + Lomb, Coherus, Crinetics, EyePoint, Genentech, Gyroscope, IvericBio, KKR, Kodiak, Novartis, Ocular Therapeutix, Opthea, Outlook, RecensMedical, Regeneron, Regenxbio, ReVive, Samsara, US Retina, and Vial; being a stockholder of Boston Image Reading Center, Hemera Biopharmaceuticals, Network Eye, ReVive, and US Retina; and being the founder of Network Eye. Dr. Dhoot reports serving as a consultant for Apellis Pharmaceuticals, Alimera, Allergan, Coherus, Eyepoint, Genentech, Iveric Bio, Ocular Therapeutix, Oxular, Regeneron, RegenXBio, and Novartis, and being a stockholder of Outlook Therapeutics and Vortex Surgical. Dr. Wykoff reports serving as a consultant for Acucela, Adverum, Aerpio, Alcon, Alimera Sciences, Allergan, Alnylam, Apellis, Arctic Vision, Bausch + Lomb, Bayer, Bionic Vision Technologies, Chengdu Kanghong Biotechnologies, Clearside Biomedical, Corcept Therapeutics, Dutch Ophthalmic Research Center, EyePoint, Genentech, Gyroscope, IVERIC Bio, Kodiak Sciences, Merck, NGM Biopharmaceuticals, Notal Vision, Novartis, OccuRx, ONL Therapeutics, Opthea, Oxurion, Palatin, Polyphotonix, Recens Medical, Regeneron Pharmaceuticals, RegenXBio, Roche, Santen, Takeda, Thea Open Innovation, and Verana Health; receiving personal fees from 4DMT, AbbVie, Aerie, AGTC, Alimera, Allgenesis, Annexon, Arrowhead, Boehringer Ingelheim, Cholgene, Clearside, Curacle, Foresite, Frontera, IACTA, Janssen, Kato, Kiora, Kodiak, Kriya, Nanoscope, Ocular Therapeutix, Ocuterra, OliX, Oxular, PerceiveBio, Perfuse, Ray, Regeneron, Resonance, SciNeuro, Stealth, Surrozen, TissueGen, and Valo; receiving grants for research support from 4DMTAdverum, Aerie Pharmaceuticals, AffaMed, Aldeyra, Allergan, Alexion, Alimera, Alkahest, Allgenesis, Apellis, Amgen, Annexin, Annexon, Asclepix, Bayer, Boehringer Ingelheim, Chengdu Kanghong Biotechnologies, Clearside Biomedical, Curacle, EyePoint, Gemini Therapeutics, Genentech, GlaxoSmithKline, Graybug Vision, Gyroscope, IONIS Pharmaceutical, IVERIC Bio, Kodiak Sciences, LMRI, Mylan, Neurotech Pharmaceuticals, NGM Biopharmaceuticals, Novartis, Nanoscope, Ocular Therapeutix, Ocuphire, OcuTerra, Ophthotech, Opthea, Outlook Therapeutics, Oxurion, Oxular, Oyster Point, PerceiveBio, Recens Medical, Regeneron Pharmaceuticals, RegenXBio, Roche, Samsung Bioepis, Santen, Senju, Taiwan Liposome Company, SamChunDang Pharm, Sandoz, UNITY, Verily, and Xbrane BioPharma; receiving speaker fees from Regeneron Pharmaceuticals; and receiving other support from ONL, PolyPhotonix, RecensMedical, TissueGen, Visgenx, and Vitranu outside the submitted work. Dr. Klufas reports serving as a consultant for Genentech and Allergen and as a lecturer for Genentech and Regeneron. Drs. Jones and Baumal are employees of Apellis Pharmaceuticals, Inc., Michele Intorcia and Sujata P. Sarda were employees of Apellis Pharmaceuticals, Inc. at the time of this study. Priyanka Bobbili, Rose Chang, Maryaline Catillon, Chunyi Xu, Kirthana Sarathy, and Mei Sheng Duh are employees of Analysis Group, Inc., a consulting company that has provided paid consulting services to Apellis Pharmaceuticals, Inc. Dr. Chaudhary reports serving as an advisory board member for Alcon, Roche, Bayer, Novartis, Apellis, and Boehringer Ingelheim, and receiving grants from Bayer, Novartis, and Roche. The authors certify that this manuscript reports the secondary analysis of clinical trial data that were shared with them, and that the use of these shared data is in accordance with the terms agreed upon their receipt. Data for the pegcetacoplan trials were provided by the trial sponsor, Apellis Pharmaceuticals, Inc. Data for the comparator trials were publicly available on the ClinicalTrials.gov website and the respective trial publications cited in this manuscript.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by Apellis Pharmaceuticals, Inc.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
