Abstract
Keywords
Introduction
In August 2023, the US Food and Drug Administration (FDA) announced the approval of avacincaptad pegol for the treatment of geographic atrophy (GA) secondary to age-related macular degeneration (AMD). Avacincaptad pegol modulates the complement cascade system, namely C5, a key component related to the innate immune system and cell death. 1 The FDA approved avacincaptad pegol based on the findings of the GATHER1 and GATHER2 clinical trials. GATHER1 was an international, prospective, randomized, double-masked, sham-controlled phase 2/3 clinical trial that found a reduction in GA growth over 12 months. 2 GATHER2 was a 24-month, international, randomized, double-masked, phase 3 clinical trial that showed slower GA lesion growth with avacincaptad pegol injections compared to sham. No cases of intraocular inflammation (IOI), endophthalmitis, or ischemic optic neuropathy were reported in the GATHER1 or GATHER2 trial.
Real-world outcomes of intravitreal therapies often differ from clinical trial findings due to differences in patient characteristics, implementation, adherence, and other factors. In addition, due to the novelty of complement inhibition for GA, as well as prior reports of IOI associated with its use, clinicians may be hesitant to implement these therapies until further real-world data have been gathered. The purpose of this study is to assess the safety of avacincaptad pegol in a real-world dataset with a sample size larger than that of the GATHER1 and GATHER2 trials.
Methods
Study Design
This was a retrospective, observational cohort study describing clinical outcomes in patients with GA treated with intravitreal avacincaptad pegol 2 mg (0.1 mL of 20 mg/mL). A retrospective chart review was conducted using data from several retina practices within the PRISM Vision Group (Retina Group of Washington, Retina Care Center, and New Jersey Retina); eligible patients were identified by querying the electronic health record system for all encounters during which Healthcare Common Procedure Coding System (HCPCS) J-codes for avacincaptad pegol were noted during the period from August 3, 2023, to October 10, 2024. The study adhered to the tenets of the Declaration of Helsinki and received approval from the WCG Institutional Review Board.
Outcome Measurements
Data collected included patient demographics, AMD history, and treatment details, including age, sex, race, laterality of injected eye(s), number of injections for each eye, duration of treatment, and number of visits. Information gathered at each of the follow-up visits included best-corrected Snellen visual acuity and intraocular pressure (IOP). Adverse events recorded included IOP elevation necessitating anterior chamber paracentesis, endophthalmitis, IOI, vasculitis, ischemic optic neuropathy, conversion to exudative AMD, acute posterior vitreous detachment, and rhegmatogenous retinal detachment.
Statistical Analysis
Descriptive statistical analysis was used to assess baseline demographics and rates of adverse events following injection of avacincaptad pegol. A paired t test was used to compare the frequency of intravitreal injections of anti-vascular endothelial growth factor (anti-VEGF) agents before and after initiation of avacincaptad pegol therapy.
Results
Baseline Demographics
A total of 461 eyes of 335 patients with GA were treated with intravitreal injections of avacincaptad pegol 2 mg (0.1 mL of 20 mg/mL) from August 3, 2023, to October 10, 2024. Table 1 displays the baseline demographic characteristics. Of these patients, 127 were male (38%) and 208 were female (62%), with a mean age of 82 years. The majority of patients were White (89%). With respect to their AMD history, 100% of eyes had a diagnosis of advanced nonexudative AMD with GA, 238 (51.6%) had extrafoveal atrophy, and 223 (48.4%) had subfoveal atrophy. Sixty-seven eyes (15%) also had a history of exudative AMD. A total of 125 patients received injections in both eyes, and of these, 55 patients received bilateral same-day avacincaptad pegol injections. The mean follow-up time was 120 days (median, 125).
Baseline Demographic Characteristics (N = 335 patients).

Safety Profile
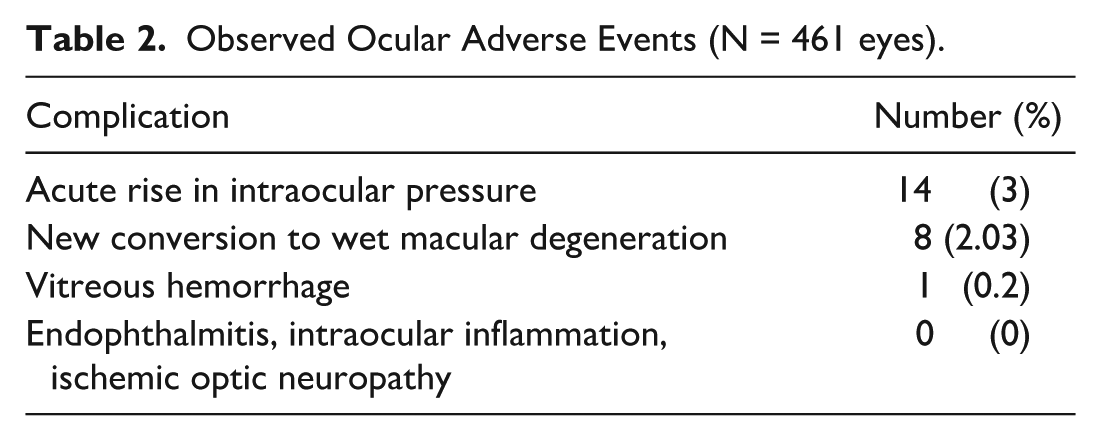
The mean injection interval for avacincaptad pegol was 52 days, and the mean number of avacincaptad pegol injections per eye during the follow-up period was 3.33. Table 2 delineates the observed ocular adverse events. Throughout the follow-up period, there were no reports of endophthalmitis, IOI, or ischemic optic neuropathy in the cohort.
Observed Ocular Adverse Events (N = 461 eyes).
Rise in IOP
Immediately postinjection, 14 eyes (3%) developed an acute rise in IOP manifesting as no light perception (NLP) immediately following intravitreal injection (mean, 46 mm Hg; range, 38-55 mm Hg). Three of the 14 eyes that developed NLP underwent anterior chamber paracentesis for acute IOP reduction, while the remainder soon recovered vision without requiring paracentesis. None of the patients who developed an acute rise in IOP causing NLP had a preexisting history of glaucoma. None of them developed or demonstrated clinical signs of glaucomatous optic neuropathy during the follow-up period, and none of the affected eyes required treatment with topical antihypertensive medications or incisional glaucoma surgery. One patient who developed an acute IOP elevation received a modification to their treatment protocol in which a half dose of avacincaptad pegol (0.05 mL) was administered during subsequent visits.
Neovascular Conversion
Eight eyes (2.03%) with initially nonexudative AMD developed a new diagnosis of exudative, neovascular AMD (nAMD) following initiation of avacincaptad pegol treatment, with a mean of 2 avacincaptad pegol injections (range, 1-3) prior to conversion. One patient developed nAMD in both eyes, with each converting after the first respective avacincaptad pegol injection in each eye, which were performed at separate visits. Two of the 6 patients who developed nAMD after avacincaptad pegol had preexisting nAMD in the contralateral eye. All of the conversions to nAMD were discovered incidentally at scheduled follow-up visits that took place, on average, 43 days after injection. None of the patients who converted to nAMD during treatment developed noticeable symptoms and, therefore, did not require emergency visits for evaluation between scheduled appointments. Five of the 8 eyes that converted to nAMD continued receiving intravitreal avacincaptad pegol injections, on average, 20 days following treatment for their newly diagnosed nAMD, with the time between intravitreal anti-VEGF injection for nAMD and resumption of intravitreal avacincaptad pegol injections ranging from 10 to 49 days. In the 5 eyes that resumed avacincaptad pegol therapy, the intravitreal avacincaptad pegol injections and intravitreal anti-VEGF agent injections were performed on different days.
Neovascularization Disease Activity
A total of 67 eyes (15%) had a prior history of nAMD requiring anti-VEGF injections before initiation of intravitreal avacincaptad pegol injections. In eyes that continued to receive intravitreal anti-VEGF injections following the initiation of avacincaptad pegol therapy, the average intervals between anti-VEGF injections before and after avacincaptad pegol initiation were 57.5 weeks and 62 weeks, respectively. This was not found to be a statistically significant increase in anti-VEGF frequency (P = .55).
Conclusions
This study describes the real-world safety profile of avacincaptad pegol and may be helpful to clinicians as they decide how to incorporate complement inhibitor therapies in their clinical practice. In this study, avacincaptad pegol treatment led to a 2.03% rate of new nAMD conversion and a 3% rate of acute postinjection IOP elevation resulting in transient NLP. The treatment was not associated with IOI, nonarteritic anterior ischemic optic neuropathy, or endophthalmitis.
Our study revealed a lower rate of new nAMD conversion with avacincaptad pegol treatment than in the GATHER2 trial, in which there was a 5% rate of nAMD conversion in the avacincaptad pegol group and 3% in the sham group. 3 In GATHER2, follow-up duration was 12 months, while in our study, the mean follow-up time was 120 days. Using this mean follow-up of 120 days and the rate of nAMD conversion of 2.03% seen in our study, the predicted annualized rate of nAMD (over 365 days) in our population is approximately 6.17%. This is similar, albeit slightly higher, than the GATHER2 rate. In contrast, pegcetacoplan, a C3 inhibitor that was the first FDA-approved complement inhibitor in the United States, was shown to be associated with a 12% risk of conversion to nAMD when administered monthly and 7% when administered every other month. 4 While the mechanism underlying the conversion to nAMD is not fully understood, it is thought that complement modulation may alter microglial cells to a more proangiogenic state with a resulting downstream increased risk of neovascularization. 5
Sterile IOI is a known, rare complication associated with various intravitreal injections, including antibiotics, anti-VEGF medications, steroids, ocriplasmin, gene therapy, and complement inhibitors, specifically, pegcetacoplan.6 –11 In December 2023, the ReST Committee presented 14 eyes associated with retinal vasculitis after the first injection of intravitreal pegcetacoplan. 12 The report also described 1 patient with macular atrophy secondary to Stargardt disease who developed retinal vasculitis in 1 eye after receiving intravitreal pegcetacoplan, followed by retinal vasculitis in the fellow eye after intravitreal avacincaptad pegol. All eyes had anterior chamber inflammation, 86% of the eyes had vitritis, and 2 eyes required enucleation. The etiology of IOI associated with pegcetacoplan remains unknown. In contrast, there were no cases of IOI identified with avacincaptad pegol in the current study or the GATHER1 and GATHER2 clinical trials for avacincaptad pegol.2,3 Despite our relatively large cohort of 461 eyes, this sample size may still be too small to capture the rare and potentially vision-threatening adverse effects of IOI and occlusive vasculitis, which should remain key concerns for clinicians and patients deciding whether to use this medication.
Avacincaptad pegol requires administration of a larger volume (0.1 mL) compared with other intravitreal medications, which typically require an injection of 0.05 mL. This has led to concern about the potential risk for elevated IOP. IOP elevation is a common occurrence after intravitreal injection and may be influenced by factors such as injection volume, viscosity, composition, and patient characteristics such as history of glaucoma, systemic blood pressure, and translaminar pressure gradient. Zhou et al demonstrated a 2-fold increase in the risk of sustained IOP elevation in eyes that underwent repeated intravitreal injections. 13 Furthermore, a greater volume of intraocular injection has been associated with a statistically significant increase in IOP. 14 In our cohort, 14 eyes (3%) developed an acute rise in IOP associated with NLP, with 3 eyes requiring AC paracentesis. In comparison, in the GATHER2 trial for avacincaptad pegol, 9% of eyes developed increased IOP. However, the modality of IOP measurement, severity of IOP elevation, and method of treatment were not disclosed in the original randomized controlled trials. 3 Further studies are needed to assess the long-term consequences of avacincaptad pegol–associated IOP elevation.
The main strength of this study is its large cohort size. However, its limitations include its retrospective design and a follow-up period of 120 days. At the time of data collection, avacincaptad pegol had only been approved in the United States for 1 year. Therefore, the long-term effects of the medication and potential adverse events after additional injections remain unknown. Additional research with larger sample sizes and longer follow-up durations will help to provide a better understanding of the medication’s long-term safety profile.
Footnotes
Ethical Approval
The study adhered to the tenets of the Declaration of Helsinki.
Statement of Informed Consent
This is an institutional review board–approved retrospective study that received a waiver for informed consent.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
