Abstract
Keywords
Introduction
On February 17, 2023, pegcetacoplan 15 mg (Syfovre, Apellis Pharmaceuticals) was approved by the US Food and Drug Administration (FDA) to treat geographic atrophy (GA) secondary to age-related macular degeneration (AMD). Pegcetacoplan is a polyethylene-glycolylated (PEGylated) peptide that blocks complement factor C3, which has been implicated in the development of both the exudative and advanced atrophic forms of AMD. 1 The phase 3 DERBY and OAKS studies found that over 24 months, intravitreal pegcetacoplan slowed the growth of GA by 21% when given monthly and by 17% when given every other month (EOM) compared with sham injections. 2 During the studies, there were no reported cases of retinal vasculitis or retinal occlusive vasculitis.
Beginning in April 2023, after FDA approval of intravitreal pegcetacoplan, the American Society of Retina Specialists (ASRS) Research and Safety in Therapeutics (ReST) Committee began receiving reports of intraocular inflammation after intravitreal pegcetacoplan administration. In addition to cases of intraocular inflammation without retinal vascular involvement, several reported cases included retinal vasculitis resulting in occlusive retinal vasculopathy and significant vision loss.
The purpose of this study was to analyze the characteristics of retinal vasculitis cases after intravitreal pegcetacoplan administration that were voluntarily reported to the ASRS ReST Committee following post-marketing approval of pegcetacoplan.
Methods
The ASRS ReST Committee hosts a reporting system through which ASRS members can submit cases of adverse events believed to be associated with retina drugs or devices. Adverse events reports were submitted directly to the ASRS ReST Committee or through the ASRS Adverse Event reporting website (https://www.asrs.org/forms/4/asrs-adverse-event-report-form). From these sources, the committee followed up with the reporting physicians to gather additional clinical information and ophthalmic images. Clinical and imaging data from all cases of physician-reported pegcetacoplan-associated retinal vasculitis cases submitted by October 12, 2023, were collected and reviewed.
All patient information and images were de-identified by the submitting physicians before data were submitted to the ASRS. This study was completed under the tenets of the Declaration of Helsinki and the US Health Insurance Portability and Accountability Act of 1996 and was deemed exempt from institutional review board (IRB) oversight by the IRB at Tufts Medical Center, Boston, MA.
De-identified original imaging, including color photographs, fluorescein angiography (FA) images, and optical coherence tomography (OCT) images, were collected when available. The images were graded independently by 3 retina and uveitis specialists (G.J.J., S.K.S., J.L.D.) for the presence or absence of findings (listed below). If 1 of the image reviewers disagreed with the other 2 on a particular feature, the final grade was the grade on which 2 of the 3 reviewers agreed. If the quality of an image was insufficient to assess a given finding, the image was deemed ungradable for that finding. In addition, the images were evaluated by all members of the ReST Committee for final classification of the presence or absence of vasculitis and presence or absence of occlusive retinal vasculitis.
The definitions of retinal vasculitis, retinal vasculopathy, occlusive retinal vasculitis, and occlusive retinal vasculopathy are not standardized. According to a 2005 report from the Standardization of Uveitis Nomenclature for Reporting Clinical Data Study, there was disagreement among study members on how to best define retinal vasculitis. In the study, there was consensus that “it is a descriptive term for those situations in which there is evidence of ocular inflammation and retinal vascular changes.” In addition, “Although the group provisionally agreed to consider perivascular sheathing and vascular leakage or occlusion on fluorescein angiogram as evidence of retinal vascular disease for the classification of retinal vasculitis, there was consensus that the definition of retinal vasculitis required more work.” 3 In the present study, imaging criteria to evaluate for possible signs of retinal vasculitis and/or vasculopathy were derived from studies of the diagnosis of retinal vasculitis and retinal vascular occlusion secondary to brolucizumab.4 –7 In addition, other examination findings were taken into consideration, including the presence of intraocular inflammation and signs of anterior segment neovascularization.
Color photographs were assessed for vascular sheathing, intraretinal hemorrhages, retinal whitening and/or cotton-wool spots, boxcarring, and optic nerve head edema. FA images were assessed for vascular leakage, optic nerve leakage, and vascular occlusion. Macular OCT images were assessed for inner retinal edema with hyperreflectivity (suggestive of acute ischemia) and macular swelling in the absence of hyperreflectivity of other retinal layers. Other imaging features that were graded as present or absent across imaging modalities included arterial involvement, venous involvement, macular ischemia, and peripheral ischemia. Graders were asked to determine the presence or absence of all these imaging markers and to assess for the presence or absence of retinal vasculitis and occlusive retinal vasculopathy. Cases that were confirmed to have imaging signs of retinal vasculitis were included in the analysis. Cases that had intraocular inflammation but that were not graded as having definitive findings consistent with retinal vasculitis were excluded from the study analysis, as were cases the ReST Committee deemed to be most likely related to infectious endophthalmitis.
The ReST Committee also collected a standardized clinical dataset, including patient sex, age at time of injection, eye, race, medical history (including any history of autoimmune disease), allergy history, COVID-19 vaccine status and type (if known), most recent COVID-19 infection (if known), ocular history, previous number and type of antivascular endothelial growth factor (anti-VEGF) injections (including days between most recent anti-VEGF injection and pegcetacoplan injection), history of inflammation associated with anti-VEGF, number of pegcetacoplan injection(s), lot number of the causative pegcetacoplan injection, filter needle type used to draw up medication, presence or absence of intraocular inflammation at the time of injection, days between injection and subsequent follow-up, symptoms at adverse effects (AEs) presentation, presence or absence and location of intraocular inflammation, presence or absence of vasculitis as determined by the treating physician, confirmation of retinal vasculitis by imaging as read by the treating physician, presence or absence of vascular occlusion as determined by the treating physician, treatment modalities, workup of patient after vasculitis diagnosis, number of days between pegcetacoplan and final follow-up, presence or absence of intraocular inflammation at the final follow-up (and time to resolution if applicable), residual examination findings, and residual symptoms. The visual acuity (VA) and intraocular pressure (IOP) were recorded from baseline, at AE presentation, and at the final follow-up visit.
Data were tabulated with Excel software (Microsoft Corp). Snellen VA was converted to logMAR equivalents for the purpose of analysis.
Results
Demographics
Between April 25, 2023, and October 12, 2023, 32 eyes of 28 patients with intraocular inflammation after pegcetacoplan injection were reported to the ReST Committee. Of these, 17 eyes of 15 patients were reported to have retinal vasculitis by the submitting physicians. The first reported case of retinal vasculitis was received by the ReST Committee on July 3, 2023. After expert image analysis, 3 eyes of 2 patients were deemed not to have retinal vasculitis on imaging. The remaining 14 eyes of 13 patients had confirmed retinal vasculitis based on expert review and were included for further analysis in this study (Figures 1 to 4).
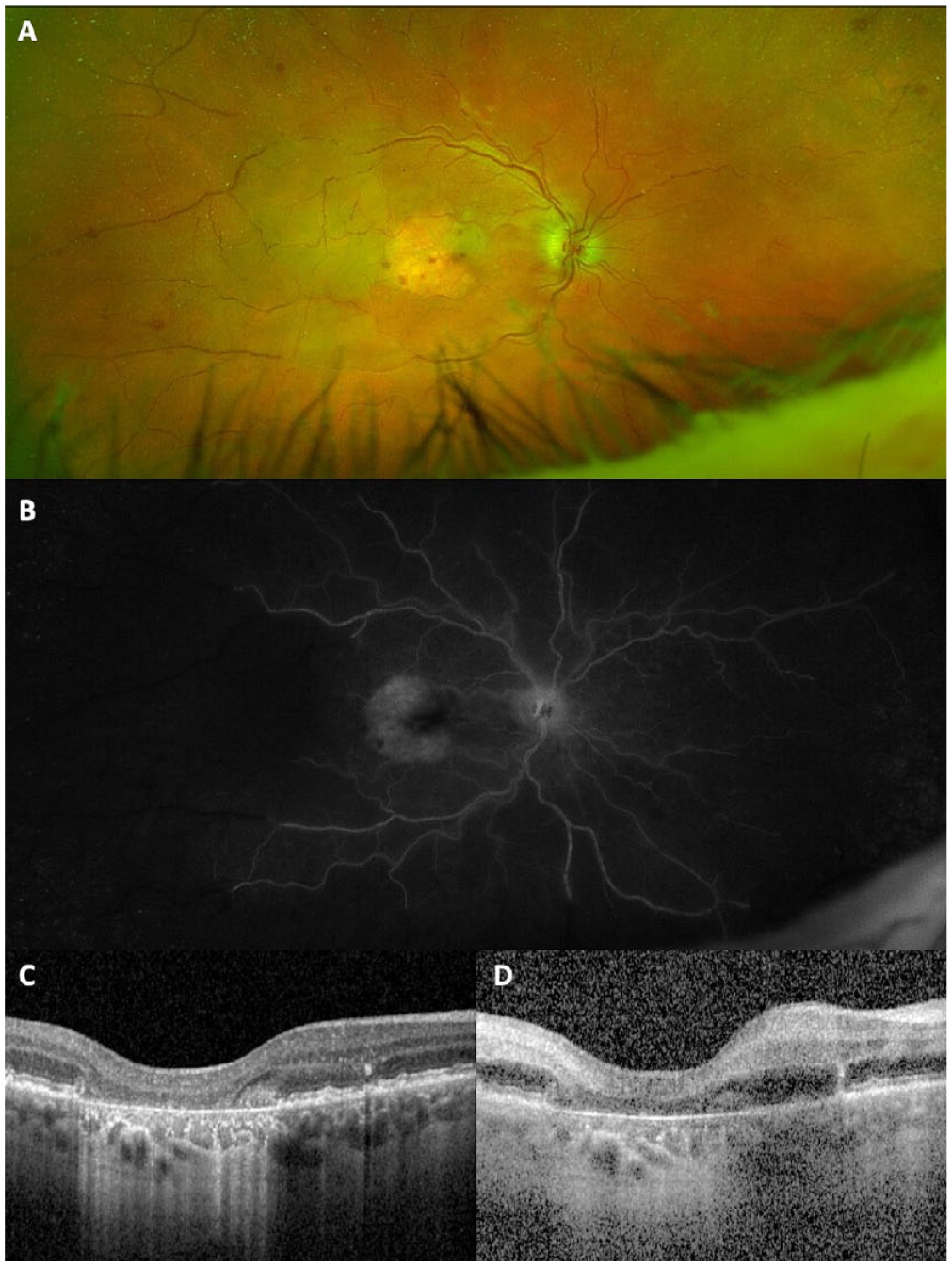
A 70-year-old man with bilateral GA received a pegcetacoplan injection in the right eye (Patient 10, Table 2). A 19-gauge filter needle was used to draw up the medication. In the right eye, the VA was 20/30 and the IOP was 16 mm Hg. Ten days after injection, he presented with blurry vision, eye pain, and redness in the right eye, which had a VA of CF and an IOP of 27 mm Hg. (A) A fundus photograph of the right eye taken 14 days after pegcetacoplan injection shows GA in the macula, scattered retinal hemorrhages, vascular sheathing, boxcarring in the peripheral retinal vessels, and retinal whitening in the macula and temporal retina. (B) FA shows diffuse and profound occlusion of the peripheral arterioles and venules in the peripheral retina and leakage from the venules and optic nerve. (C) An OCT image taken at the time of pegcetacoplan injection shows complete RPE and outer retina atrophy and a normal inner retinal layers architecture. (D) A matched OCT image taken 14 days after pegcetacoplan injection shows thickening and increased hyperreflectivity of the inner retinal layers, consistent with acute retinal ischemia.
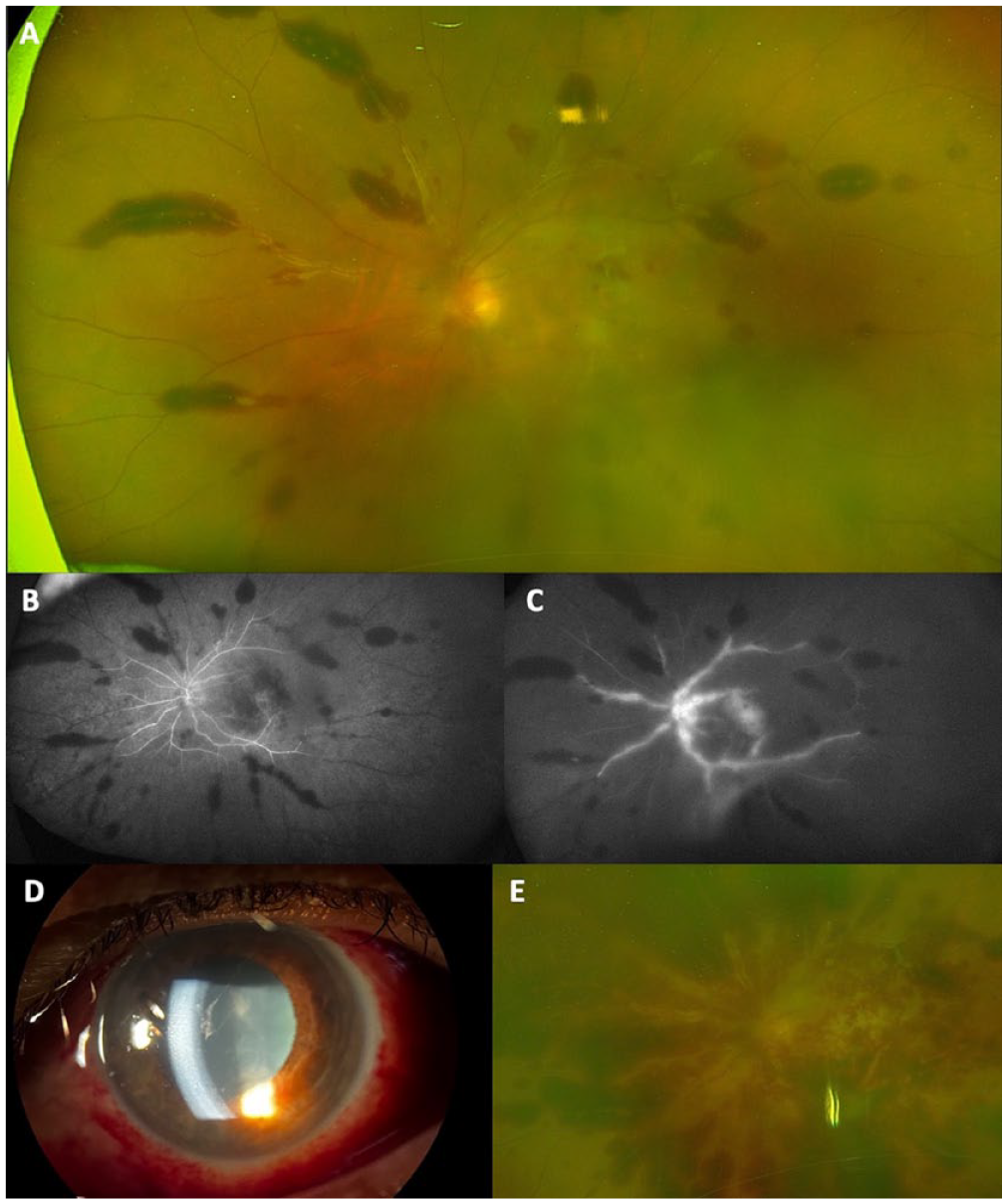
A 67-year-old woman with bilateral GA received a pegcetacoplan injection in the left eye (Patient 1, Table 2). At the time of injection, the VA was 20/100 and the IOP was 10 mm Hg in the left eye. Nine days after injection, she presented with left eye pain and decreased vision. The VA was 20/400 and the IOP was 10 mm Hg in the left eye. By day 17 after the pegcetacoplan injection, evidence of anterior segment neovascularization had developed and the VA had declined to LP. By day 112 after pegcetacoplan injection, the eye was enucleated. Pathology results were pending at the time of this report. (A) A fundus photograph of the left eye 9 days after pegcetacoplan injection shows hazy media. There were large peri-venous retinal hemorrhages, venous sheathing, optic nerve edema, and macular whitening. (B) Early FA shows diffuse and profound occlusion of the peripheral arterioles and venules in the peripheral retina. (C) Late FA shows leakage from the venules and optic nerve and macular leakage. (D) An anterior segment examination showed 2+ conjunctival injection and conjunctival hemorrhages, mild corneal edema, and 1+ cells as well as fibrin in the AC. (E) A fundus photograph from day 17 after pegcetacoplan injection shows much more extensive retinal hemorrhages and macular whitening.
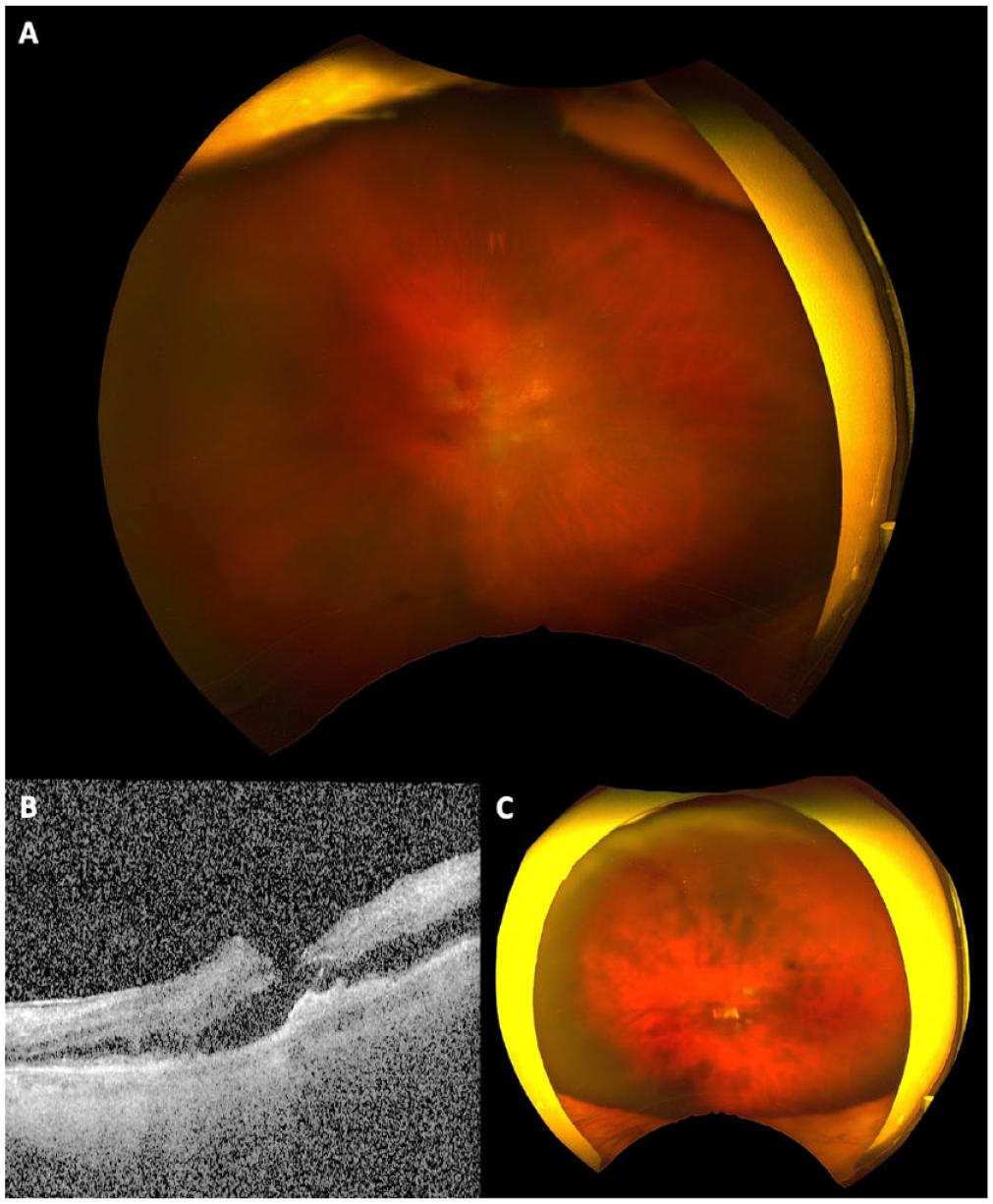
A 96-year-old man with a history of GA received a pegcetacoplan injection in the left eye (Patient 3, Table 2). At the time of injection, the VA was 20/60 and the IOP was 10 mm Hg in the left eye. Eight days after injection, he presented with left eye pain and blurry vision. His VA was HM, and the IOP was 32 mm Hg. FA could not be performed because of poor venous access. By 3 weeks after pegcetacoplan injection, the VA had declined to NLP, and evidence of anterior segment neovascularization had developed. (A) A fundus photograph of the left eye 8 days after pegcetacoplan injection shows hazy media, scattered retinal hemorrhages, venous sheathing, and macular whitening. (B) Macular OCT shows thickening and hyperreflectivity of the inner retina, suggesting acute inner retinal ischemia. (C) A fundus photograph taken 3 weeks after pegcetacoplan injection shows diffuse intraretinal hemorrhages, many of which are peri-venular.
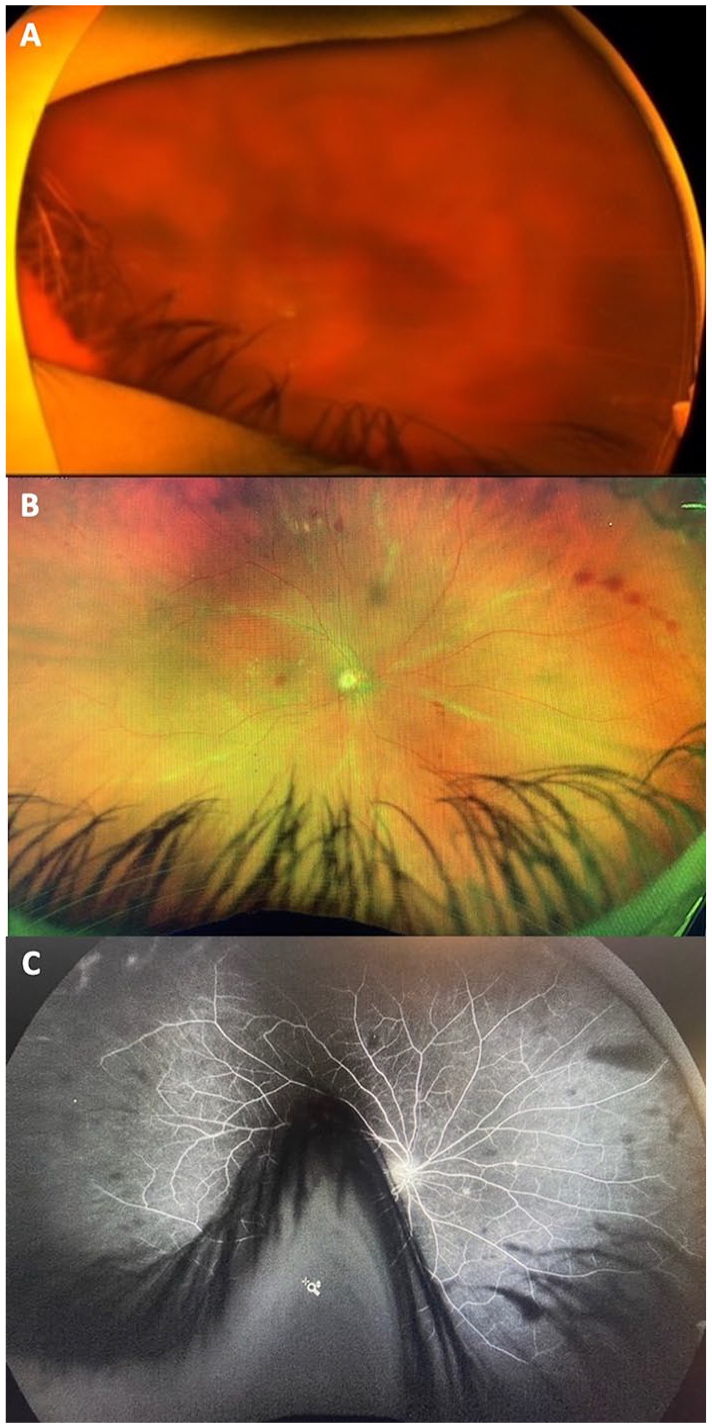
A 43-year-old woman with a history of bilateral macular atrophy secondary to Stargardt disease received a pegcetacoplan injection in the left eye (Patient 13, Table 2). An 18-gauge filter needle was used to draw up the medication; 19-gauge filter needles had been removed from the clinic by this time. Four days later, the patient received an injection of avacincaptad pegol in the right eye. At the time of the injections, the VA was 20/100 OD and 5/200 OS and the IOP was 17 mm Hg in both eyes. Nine days after pegcetacoplan injection, the patient presented with decreased vision and pain in the left eye. The VA was NLP and the IOP was 40 mm Hg in the left eye. (The right eye was quiet at this time.) Two days later, the right eye had a decrease in vision and pain. On that day, the VA was HM and the IOP was 40 mm Hg in the right eye. The patient subsequently developed neovascular glaucoma in the left eye, and on day 40 after pegcetacoplan injection, the left eye was enucleated. Pathology results were pending at the time of this report. (A) A fundus photograph of the left eye 9 days after pegcetacoplan injection shows hazy media. There were diffuse retinal hemorrhages and vascular sheathing. (B) A fundus photograph of the right eye 7 days after avacincaptad pegol injection shows hazy media, scattered retinal hemorrhages, and diffuse vascular sheathing. (C) Midphase FA shows disc leakage, mild vascular leakage, and peripheral nonperfusion.
Table 1 shows the patients’ demographics. There was no identifiable pattern related to previous medical history, ocular history, or drug allergies. None of the patients had any known recent exposure to the COVID-19 vaccine. Two patients had recent COVID-19 infections; 1 had the COVID-19 infection 3 weeks previously, and another had the COVID-19 infection 3 months before presentation. The other patients had no known recent COVID-19 infections. Five patients (36%) had a possible history of ocular or systemic inflammatory disease, 4 with hypothyroidism and 1 with a history of uveitis (Patient 4, Table 2), although no patient had active uveitis at the time of pegcetacoplan treatment.
Patient Characteristics (14 Eyes; 13 Patients).

Abbreviation: anti-VEGF, antivascular endothelial growth factor.
For neovascular age-related macular degeneration.
Individual Patient Data.
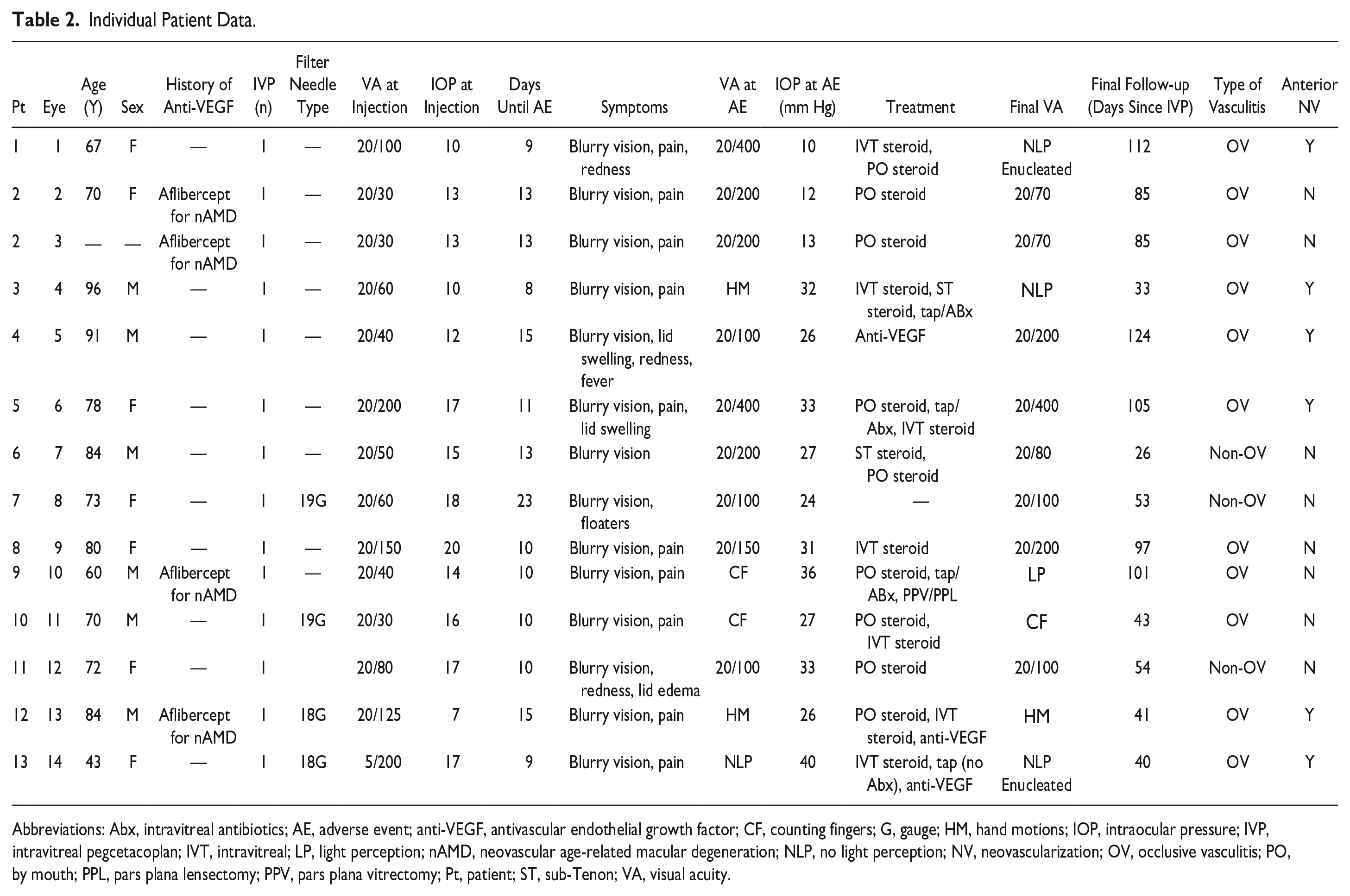
Abbreviations: Abx, intravitreal antibiotics; AE, adverse event; anti-VEGF, antivascular endothelial growth factor; CF, counting fingers; G, gauge; HM, hand motions; IOP, intraocular pressure; IVP, intravitreal pegcetacoplan; IVT, intravitreal; LP, light perception; nAMD, neovascular age-related macular degeneration; NLP, no light perception; NV, neovascularization; OV, occlusive vasculitis; PO, by mouth; PPL, pars plana lensectomy; PPV, pars plana vitrectomy; Pt, patient; ST, sub-Tenon; VA, visual acuity.
Thirteen eyes were treated with pegcetacoplan for macular atrophy in the setting of AMD, and 1 patient (Patient 13, Table 2 and Figure 4) was treated with pegcetacoplan for macular atrophy secondary to Stargardt disease. Four eyes (29%) of 3 patients (23%) were receiving anti-VEGF treatment with intravitreal aflibercept for neovascular AMD (nAMD) on an ongoing basis in the same eye before receiving pegcetacoplan. One eye with nAMD received both aflibercept and pegcetacoplan (as well as an anterior chamber [AC] paracentesis) on the same day, while the others received the pegcetacoplan injection on a different day than the anti-VEGF treatment.
All AEs arose after the first pegcetacoplan injection. The most recent pegcetacoplan injection that was given in this series was on September 22, 2023. The mean time from the most recent pegcetacoplan injection to presentation was 12 days (median, 10.5 days; range, 8-23 days). There was no pattern that implicated specific lot numbers (there were 7 different lot numbers provided from 12 injecting physicians) or filter needles (the type of filter needle was known and reported in 4 eyes of 4 patients; 2 of these were 19-gauge needles and 2 were 18-gauge needles). No pegcetacoplan injection was given in an eye with intraocular inflammation as noted by the reporting physician. An AC paracentesis was performed in 2 eyes (14%) at the time of injection. The 1 patient with bilateral vasculitis had same-day bilateral pegcetacoplan injections. Another patient with pegcetacoplan-associated retinal vasculitis in 1 eye had retinal vasculitis in the fellow eye after treatment with intravitreal avacincaptad pegol (Izervay, Astellas Pharma Inc); the injections were spaced 4 days apart between the 2 eyes with these 2 different agents (Patient 13, Table 2). Table 2 shows the individual patient data.
Symptoms and Examination Findings
Symptoms at AE onset included blurry vision (100%), eye pain in 10 eyes (71%), redness in 4 eyes (29%), eyelid edema in 3 eyes (21%), and floaters in 1 eye (7%). One patient had reported a fever (8%). The median VA was 20/300 (range, 20/100-no light perception [NLP]) at AE presentation and 20/60 (range, 20/30-5/200) at baseline before the event. AC inflammation was present in all eyes, and vitreous inflammation at AE presentation was reported in 12 eyes (86%). The median AC cell grade was 2+ cells (mean, 1.8+ cells; range, 0.5+ to 3+ cells); a grade for vitreous cells was not collected. None of the eyes presented with a hypopyon, 1 eye presented with AC fibrin, and another eye developed AC fibrin 2 days after the initial presentation. Six eyes (43%) presented with corneal edema. The median IOP was 15 mm Hg (range, 7-20 mm Hg) at the time of pegcetacoplan injection and 27 mm Hg (range, 10-40 mm Hg) at AE presentation; 10 eyes (71%) had an increase of more than 10 mm Hg from baseline at AE presentation. Table 3 shows a summary of the examination and imaging findings.
Summary of Examination and Imaging Findings.
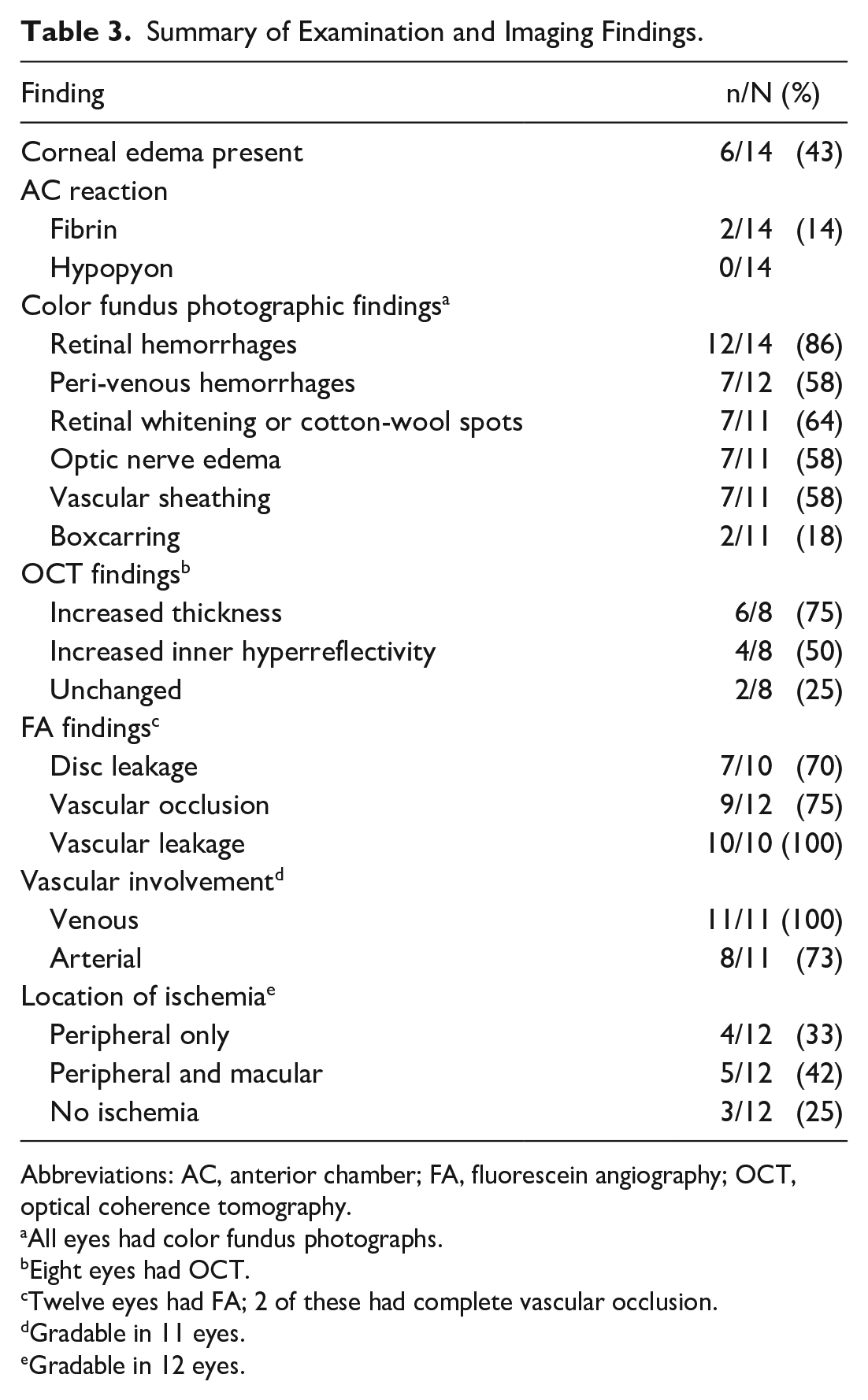
Abbreviations: AC, anterior chamber; FA, fluorescein angiography; OCT, optical coherence tomography.
All eyes had color fundus photographs.
Eight eyes had OCT.
Twelve eyes had FA; 2 of these had complete vascular occlusion.
Gradable in 11 eyes.
Gradable in 12 eyes.
Vasculitis Features
Imaging was available for all eyes with retinal vasculitis. These included color photographs of all eyes, FA of 12 eyes (86%), and OCT of 8 eyes (57%). A spectrum of vasculopathy was seen, ranging from minimal to severe. Retinal venules were affected in all 11 eyes that could be graded for this finding. Arterioles were affected in 8 of 11 eyes that could be graded for this finding; all 8 of these eyes were also graded as having occlusive retinal vasculopathy. Retinal vasculitis and the subcategory of occlusive retinal vasculopathy were defined using specific imaging criteria as summarized above. After image analysis, 3 eyes (21%) were determined to have retinal vasculitis without apparent occlusion, while 11 eyes (79%) were determined to have occlusive retinal vasculopathy. There was peripheral ischemia in all 9 eyes with occlusive retinal vasculopathy that had FA; ischemia also involving the macula was observed in 5 (55%) of these eyes. Table 3 shows additional features on FA and OCT.
Outcomes
The most recent follow-up visit was a mean of 59 days (median, 58 days; range, 15-109 days) since initial presentation for the AE and 71 days (median, 69.5 days; range, 26-124 days) since the pegcetacoplan injection. At the most recent follow-up visit, the median VA was 20/200 (range, 20/70-NLP). Eight eyes (57%) had a more than 3-line decrease in VA at the final follow-up, while 6 eyes (43%) had a more than 6-line decrease in VA (Table 2). Six eyes (42%) had developed signs of anterior segment neovascularization (hyphema and/or neovascularization of the iris) by the most recent follow-up visit, and 2 eyes were enucleated after they became blind and painful. (Pathology results were pending at the time of this report.)
Treatment approaches were varied. Some eyes appeared to have reduced inflammation after the use of corticosteroids, yet no trends were identifiable that could predict greater success with any specific approach. Twelve eyes (86%) were treated with topical corticosteroids, 8 patients (62%) received systemic corticosteroids, 8 eyes (57%) received intravitreal corticosteroid injections, 2 eyes (14%) received sub-Tenon corticosteroid injections, and 1 eye (6%) had a pars plana vitrectomy. Three eyes (21%) received intravitreal antibiotics, 2 patients received systemic antibiotics, and 1 of these patients also received systemic antivirals. Sampling of intraocular fluid was performed for 4 eyes, including the 3 eyes that received intravitreal antibiotics. An AC paracentesis was performed in 3 eyes, and a vitreous tap was performed in 1 eye. In all 4 of these eyes, cultures and Gram stain were negative for any organisms. Polymerase chain reaction testing in 1 of these eyes was also negative.
Conclusions
Over the course of 24 months in the phase 3 OAKS and DERBY trials of pegcetacoplan for GA secondary to dry AMD, 419 participants were treated in the monthly arm and 420 participants in the EOM arm. During the trials, 3.8% and 2.1% of participants developed intraocular inflammation in the monthly arm and EOM arm, respectively. Other notable AEs included 4% and 5% of participants with retinal hemorrhages in the monthly arm and EOM arm, respectively, and 10% and 7% with vitreous floaters in the monthly arm and EOM arm, respectively. Ischemic optic neuropathy was reported in 1.7% and 0.2% of participants treated monthly and EOM, respectively, and in none of the participants assigned to sham. 2 There were no reported cases of retinal vasculitis or occlusive retinal vasculopathy in the clinical trials. Notably, however, there was no defined protocol in these studies to obtain angiography in cases of intraocular inflammation.
Since FDA approval of pegcetacoplan on February 17, 2023, and up to October 12, 2023, the ASRS ReST Committee and an independent expert panel of uveitis specialists received and analyzed data from 14 eyes of 13 patients with retinal vasculitis occurring after intravitreal pegcetacoplan injection. After independent image analysis, 11 eyes (79%) were determined to have occlusive retinal vasculopathy, while 3 eyes (21%) had retinal vasculitis without apparent vascular occlusion. Notably, there were disagreements between graders for some of these cases, highlighting the lack of consensus regarding the optimum terminology for drug-related intraocular inflammation, retinal vasculitis, and/or retinal vascular occlusion. We discuss the possible etiologies, epidemiology, study limitations, and recommendations below.
The etiology of retinal vasculitis after pegcetacoplan injection is still unknown. Intraocular inflammation was an associated factor with all cases and is likely associated with the etiology of the retinal vasculitis and occlusion in these patients. Pegcetacoplan has a novel mechanism of action compared with other intravitreal medications: It inhibits complement factor 3. The direct inhibition of the complement cascade opens the possibility of a number of novel mechanisms for these adverse events given that complement defects are associated with an elevated risk for a broad spectrum of infectious as well as noninfectious inflammatory disorders. 8 A discussion of these potential etiologies is included below.
Intraocular inflammation is a potential risk with intravitreal injection of any medication. Certainly, manufacturing impurities may be one possibility with any new medication. However, it is notable that pegcetacoplan is a synthetic PEGylated peptide and not a biologically manufactured compound. a On July 29, 2023, Apellis Pharmaceuticals reported no manufacturing-related issues affecting product quality, no contaminants in post-marketing distributed vials, and no indication of drug-related immunogenicity in the clinical trial. 9 Another possible cause of intraocular inflammation could be the interaction between syringes, needles, and the drug. On August 22, 2023, Apellis Pharmaceuticals reported that “internal structural variations were identified in the 19-gauge filter needle included in certain injection kits; Apellis recommends use of kits with the 18-gauge filter needle, which are already in distribution.” 10 The company also noted that “a causal relationship has not been established between the structural variations in this 19-gauge filter needle and the rare events of retinal vasculitis in the real world.” In our series, the type of filter needle was known in 4 of 14 eyes. Two were performed with 19-gauge needles, while the other 2 developed retinal vasculitis with occlusive vasculopathy after the use of 18-gauge filter needles, after all 19-gauge filter needles had been definitively removed from the clinic (Patients 12 and 13, Table 2).
Intraocular inflammation has long been known to be a risk of intravitreal anti-VEGF medications and has been associated with bevacizumab (Genentech/Roche), ranibizumab (Genentech/Roche), and aflibercept (Regeneron Pharmaceuticals, Inc) at rates between 0.033% and 2.9% per injection. 11 More recently, the combination of intraocular inflammation, retinal vasculitis, and retinal vascular occlusion has been most notably observed in association with brolucizumab (Novartis).6,7 A secondary review of the HAWK/HARRIER studies by a scientific review committee found intraocular inflammation in 50 (4.6%) of 1088 eyes. Of the 50 eyes with intraocular inflammation, 36 (3.3%) had concomitant retinal vasculitis, of which 23 (2.1%) had concomitant retinal vascular occlusion.4,12,13 There have also been rare post-marketing reports of retinal vasculitis associated with other anti-VEGF medications. The FDA collects these reports through the FDA Adverse Event Reporting System. This publicly available database contains (unverified) reports of retinal vasculitis that have been associated with bevacizumab (Genentech/Roche), ranibizumab, aflibercept, brolucizumab, and faricimab (Genentech/Roche). 14
The mechanism of intraocular inflammation after anti-VEGF injection remains unknown. Suggested mechanisms include an immune response to the drug itself, other protein contaminants within the medication, or differences in pH, while mechanisms of inflammation clusters have been attributed to silicone oil residues, silicone/protein aggregates, or endotoxins.15 –18 In the case of brolucizumab, much higher rates of anti-drug antibodies (ADAs) at baseline as well as induced or boosted during clinical trials have been reported compared with ranibizumab and aflibercept.19 –22 It is possible that the higher rates of preexisting and treatment-emergent anti-brolucizumab antibodies may help explain the higher rates of inflammation with brolucizumab than with the other anti-VEGF drugs; however, it is unclear what their role is in cases of intraocular inflammation. Of note, with pegcetacoplan, the incidence of ADAs was relatively low in clinical trials; they were detected in 2.5% to 4% of participants in the OAKS and DERBY trials. 2
Another possible immune mechanism to explain cases of intraocular inflammation associated with pegcetacoplan could be related to anti-PEG antibodies. PEG is a bio-inert, hydrophilic molecule that comes in a variety of lengths and forms. The attachment of PEG chains to other molecules (ie, PEGylation) leads to substantial improvements in the pharmacokinetic properties and efficacy of various pharmaceutical agents (eg, pegcetacoplan and avacincaptad pegol). Recently, immune reactions and hypersensitivity reactions have been reported as side reactions to PEGylated therapeutics. This has been thought to be due in part to both associations with preexisting as well as treatment-induced anti-PEG antibodies. It is thought that an increasing prevalence of preexisting anti-PEG antibodies in the population may be the result of increasing exposure to PEG in cosmetics, household products, processed foods, and pharmaceutical products. 23 It is possible that an immune or hypersensitivity reaction from preexisting or treatment-induced PEG antibodies could explain the appearance of intraocular inflammation as well as retinal vasculitis in some eyes treated with pegcetacoplan and could also explain the appearance of the bilateral reaction to 2 separate PEGylated pharmaceutical compounds (pegcetacoplan and avacincaptad pegol) in 1 patient in this series (Patient 13, Table 2).
Pegcetacoplan-associated retinal vasculitis has a resemblance to hemorrhagic occlusive retinal vasculitis (HORV), which is another delayed-onset occlusive retinal vasculitis that has been associated with intraocular injection of vancomycin. 24 In vancomycin-associated HORV, the most characteristic examination findings included diffuse retinal hemorrhages (often along the venules) and venous sheathing, similar to many patients in the present series. Many patients (56%) with vancomycin-associated retinal vasculitis developed rapid-onset neovascular glaucoma, similar to 42% of patients in this pegcetacoplan series. The etiology of HORV remains unclear; however, authors have suggested it may be related to a type III (immune complex–mediated) or a type IV (T cell–mediated) hypersensitivity reaction to vancomycin.24,25 The similarities in the clinical appearances of these 2 diseases suggest there may be a shared immunologic mechanism to the retinal vasculitis in vancomycin-associated HORV and pegcetacoplan-associated retinal vasculitis.
Of note, 3 patients in this series received intravitreal vancomycin. In 2 eyes, vancomycin was injected after the appearance of occlusive vasculitis had occurred, making the likelihood that this represents (vancomycin-associated) HORV unlikely in these patients. In another patient (Patient 9, Table 2), the retina was poorly visualized until after a tap/inject with vancomycin and a subsequent vitrectomy with lensectomy, and it is possible that vancomycin could have played a role in this patient’s occlusive vasculitis.
Regarding endophthalmitis, a potential concern with broad-spectrum complement inhibition is the increased susceptibility to bacterial infections. Complement C3 deficiency leads to impaired immune complex solubilization, altered immune responses, defective phagocytosis, and absent complement-dependent bactericidal activity. Typically, infections that affect C3-deficient individuals (including those undergoing systemic complement inhibition) are recurrent and severe and are caused by encapsulated bacteria such as N meningitidis, S pneumoniae, and H influenza.26 –28 With the use of intravitreal pegcetacoplan, it is plausible that the eye may be more susceptible to infectious endophthalmitis; however, the cases presented here did not present with typical signs of infectious endophthalmitis (ie, hypopyon, dense vitritis, severe AC inflammation), did not have evidence of any organisms on Gram stain, and did not grow any organism on intraocular cultures in the 4 cases in which Gram stain and cultures were performed.
Retinal vasculitis has been reported in association with bacterial endophthalmitis. In these reports, patients presented with retinal hemorrhages, periphlebitis, and vascular sheathing, which are similar to the findings seen in cases of retinal vasculitis in this report. However, endophthalmitis-related retinal vasculitis cases also typically had whitish exudates along the retinal vessels, and often these eyes quickly progressed to more fulminant endophthalmitis with hypopyon and dense vitritis.29 –33 Some of the retinal findings in the more severe cases in our series of pegcetacoplan-associated retinal vasculitis could be consistent with vasculitis related to early bacterial endophthalmitis; however, we believe that bacterial infection is unlikely in these eyes for the following reasons:
The timing of presentation of the patients in this series is not typical for postinjection endophthalmitis, which most commonly appears within the first week after an injection and is more commonly associated with pain, conjunctival injection, hypopyon, and dense vitritis with a minimal view to the retina. 34
If endophthalmitis were to occur in the setting of complement factor C3 inhibition, bacterial growth in the eye would be expected to be more fulminant because of a lack of complement factor C3 as the opsonization of the bacteria would be limited or eliminated, which would allow for faster bacterial overgrowth. Because of an expected higher number of bacteria in this scenario, one would expect there to be a higher chance for cultures to be positive.
The inflammation in many of these cases improved with corticosteroid treatments and without antibiotic treatments.
There were 2 cases of bilateral involvement, 1 of which developed retinal vasculitis in the fellow eye after avacincaptad injection given on a different day than pegcetacoplan (Patient 13, Table 2), which would be highly unlikely if the cause were an exogenous infection.
Endophthalmitis secondary to pegcetacoplan has been reported in the OAKS/DERBY trials and after FDA approval of pegcetacoplan, and as far as is known those cases presented similarly to other cases of exogenous bacterial endophthalmitis (with hypopyon and dense vitritis).
With all of this being said, the ReST Committee would like to remind readers that infectious endophthalmitis can rarely cause the appearance of retinal vasculitis after intravitreal injection of any agent. It is important to keep this etiology on the differential diagnosis and to treat with intravitreal antibiotics if infection is suspected.
Regarding complement dysregulation, immune complexes comprise antibodies bound to target antigens. Unhindered, immune complexes grow by aggregation, eventually becoming large, insoluble aggregates that can lodge in capillary beds and trigger inflammation, vascular damage, and tissue damage. In healthy individuals, immune complexes activate the classic pathway and become coated with C1 and fragments of C4 and C3. These proteins mask antigens in the immune complex and thereby limit growth of aggregates. Simultaneously, the complement coating of immune complexes provides ligands for receptors on erythrocytes, which sequester immune complexes, and for receptors on phagocytic cells, which engulf and destroy immune complexes.
Patients with deficiencies in complement components (in particular classic pathway components) may present with immune complex disease that closely resembles systemic lupus erythematosus, itself a disease of complement dysregulation.26,27 The injection of pegcetacoplan inherently dysregulates the complement system in the eye, and this may theoretically lead to immune complex aggregation and vascular damage. Indeed, a similar mechanism (type III hypersensitivity) has been suggested as a possible etiology in vancomycin-associated HORV, as mentioned previously, and this mechanism could explain the similarity in appearance between pegcetacoplan-associated retinal vasculitis and HORV. In addition, that Patient 13 developed retinal vasculitis in both eyes after receiving different intravitreal complement inhibitors in each eye (avacincaptad pegol is an inhibitor of complement factor 5) suggests that the shared mechanism of complement inhibition may be a possible explanation for the development of bilateral retinal vasculitis in this patient. Another explanation could be that a shared, possibly PEGylated, target between the 2 drugs caused a shared immune-mediated reaction, as discussed previously. However, if immune complex aggregation were an effect of pegcetacoplan, one would expect systemic immune complex deposition in patients who are given systemic pegcetacoplan for paroxysmal nocturnal hemoglobinuria, which is not a known AE of this drug or other systemic complement inhibitors. 35
Although Apellis released that as of October 5, 2023, more than 100 000 vials (commercial and sample) of intravitreal pegcetacoplan had been distributed, the incidence of retinal vasculitis after pegcetacoplan remains unclear for several reasons. 36
Voluntary reporting of adverse events is inherently subject to underreporting, and it is possible that some cases of retinal vasculitis have not been reported to the ReST Committee, the company, or the FDA.
Some cases of retinal vasculitis may be mild and difficult to detect without a careful dilated fundus examination and/or FA, and these cases may not have been identified by the treating physicians.
The incidence of retinal vasculitis may change with repeated injections and/or evolve with further experience with pegcetacoplan. Pegcetacoplan had only been on the market for approximately 8 months at the conclusion of the data collection period for this study, and many patients may not have received multiple injections yet.
Although the number of vials distributed can be determined, it is currently not known how many of these vials have been used for treatment.
The appearance of retinal vasculitis in this series only occurred after the first pegcetacoplan injection. Therefore, the incidence of retinal vasculitis might be more appropriately calculated as “per patient” rather than “per injection,” and the number of patients treated may be more pertinent than vials distributed or used. Analysis of completed, ongoing, and future clinical and post-marketing trials as well as vigilant assessment and reporting of AEs by treating physicians will be important to better estimate the incidence of pegcetacoplan-associated retinal vasculitis.
This study has several limitations. The information is limited to data that were voluntarily submitted to the ASRS ReST Committee. Follow-up was limited to the termination date of data collection, and some patients had ongoing inflammation and retinal vascular occlusion that may change with longer follow-up. It is possible that VA results may have also been affected by the lack of long-term follow-up. Without long-term follow-up and with a limited number of cases with wide-ranging severity and treatment approaches, this study was unable to determine optimum treatment modalities. In addition, at the time of this analysis, pegcetacoplan had been on the market for approximately 8 months. In this series, all cases of retinal vasculitis arose after 1 pegcetacoplan injection; however, it is possible that many patients in the US were not yet treated with more than 1 injection in that time frame. Thus, careful examination after any pegcetacoplan injection and before administering any additional injections is suggested.
The ReST Committee has several recommendations. First, a careful evaluation of the anterior segment and posterior segment with dilation should be performed to detect any signs of active inflammation before any pegcetacoplan injection. Appropriate discussion should be performed with the patient before initiation of the drug, and informed consent should be obtained. Patients should be educated on the symptoms of intraocular inflammation and be advised to return for prompt evaluation if these symptoms occur. Because retinal vasculitis in this series occurred after the first injection in all eyes, the ReST Committee suggests waiting for at least 1 month after the first eye is treated with pegcetacoplan before considering anti-complement treatment in the fellow eye. In eyes that develop intraocular inflammation with or without signs of retinal vascular involvement, imaging, including OCT, widefield or ultra-widefield fundus photography, and widefield or ultra-widefield FA, can be helpful in visualizing the presence or absence as well as the spectrum of vasculopathy in these patients.
The data from this series were not sufficient to make any conclusive statements regarding the best treatment strategies. If infectious endophthalmitis is suspected, appropriate antibiotic treatment should be strongly considered. Inflammation after pegcetacoplan should be followed closely with an assertive approach to management. Corticosteroid treatments may reduce the level of inflammation in these patients, although their ultimate impact on visual results is unknown. Patients with peripheral ischemia are at high risk for developing neovascular glaucoma, and intravitreal anti-VEGF treatment and/or panretinal photocoagulation may be warranted in these patients. In eyes with retinal vasculitis after pegcetacoplan, retreatment should be avoided or carefully considered only after the intraocular inflammation has completely resolved. Because of the potentially severe nature of the consequences of retinal vasculitis secondary to pegcetacoplan, caution is advised when considering injection of pegcetacoplan in monocular patients or when bilateral injections are being contemplated.
Four eyes in this series were in patients who had developed macular atrophy in the setting of previous anti-VEGF injections for nAMD. These patients would have been excluded from the OAKS/DERBY trials for pegcetacoplan; therefore, it is unclear what the risk–benefit profile of intravitreal pegcetacoplan is in these eyes. Future studies of pegcetacoplan and other anti-complement drugs in the setting of patients with active nAMD may elucidate a role for these medications in this patient population. In addition, 1 patient in this series received a pegcetacoplan injection for macular atrophy secondary to Stargardt disease, which also has not been adequately studied for this indication, and the benefits of this drug for any indication other than GA secondary to nonexudative AMD remain unclear.
The ASRS recommends that physicians continue to report any adverse events to the Society through the ASRS Adverse Event reporting website (https://www.asrs.org/forms/4/asrs-adverse-event-report-form). In addition, reports of adverse events should be made to the pharmaceutical company, which is required to report them to the US FDA. The company may also be able to track lot numbers and other equipment (eg, needles or syringes) through supply chain tracking. With post-marketing pharmacovigilance, especially with new pharmaceutical products, we can aim to protect the sight of our patients.
Footnotes
Acknowledgements
The ASRS ReST Committee would like to thank Daniel F. Martin, MD (no financial interests to disclose), for assistance in independent grading of the images during the early phase of data collection. The ReST Committee would also like to recognize the vigilance and reporting of the following physicians (in alphabetical order) as well as those who prefer to remain anonymous who assisted in this report: Christopher Aderman, MD; Thomas Albini, MD; Kyle Alliman, MD; Mark Barakat, MD; Courtney Crawford, MD; Elizabeth Dao; Stella Douros, MD, FACS, FASRS; Jordana Fein, MD; Ron Gallemore, MD, PhD; William Johnson, MD; Abdul-Hadi Kaakour, MD; Alexander Port, MD; Austin Strohbehn, MD; Stephen Schwartz, MD, MBA; Kristine Traustason, MD.
Correction (February 2024):
Article updated to correct a sentence in the Conclusions section; see in-text footnote a for details.
a
The original text has been revised, as avacincaptad pegol was inaccurately classified as a synthetic PEGylated peptide.
ReST Committee (Alphabetical)
Fehrina S. Ali, MD, MPH, FASRS, Westchester Medical Center, Westchester, NY, USA
J. Fernando Arevalo, MD, PhD, FACS, FASRS, Wilmer Eye Institute, Baltimore, MD, USA
Netan Choudhry, MD, FRCS, FASRS, Vitreous Retina Macula Specialists of Toronto, Toronto, ON, Canada
Nieraj Jain, MD, FASRS, Emory Eye Center, Atlanta, GA, USA
Peter K. Kaiser, MD, FASRS, Cole Eye Institute, Cleveland Clinic, Cleveland, OH, USA
Henry Leder, MD, Leder Retina and Uveitis, Jacksonville, FL, USA
Eric Schneider, MD, FASRS, Tennessee Retina, Nashville, TN, USA
Lejla Vajzovic, MD, FASRS, Duke University, Durham, NC, USA
Robin Vora, MD, Kaiser Permanente, Oakland, CA, USA
Andre J. Witkin, MD, FASRS, Tufts Medical Center, Boston, MA, USA
Yoshihiro Yonekawa, MD, FASRS, Wills Eye Hospital, Philadelphia, PA, USA
Ethical Approval
This study was completed under the tenets of the Declaration of Helsinki and the US Health Insurance Portability and Accountability Act of 1996 and was deemed exempt from institutional review board (IRB) oversight by the IRB at Tufts Medical Center, Boston, MA, USA.
Statement of Informed Consent
Informed consent was not required for this study.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article:Dr. Witkin is a principal investigator for Regeneron and Apellis.
Dr. Kim is on the advisory board of Amgen, Apellis, Bausch + Lomb, and Coherus and is a consultant to Bayer, Genentech/Roche, and Regeneron.
Dr. Jaffe is a consultant to Roche/Genentech, Annexon, Regeneron, and Boehringer-Ingelheim.
Dr. Srivastava has received research funding from Regeneron and is a consultant to Regeneron, Bausch + Lomb, Eyepoint, Allergan, Adverum, Novartis, and RegenxBio.
Dr. Davis has received grants from the National Eye Institute ADVISE and ADJUST trials and Gyroscope and is a consultant to Kodiak Science, 4D Molecular Therapeutics, and Aura Biosciences.
Dr. Kaiser has received research grant support from Apellis (Digital Angiography Reading Center), has stock options with Ocular Therapeutix, and is a consultant to Allergan/Abbvie, Stealth Biotherapeutics, Bausch + Lomb, Novartis, Bayer, Regeneron, Ocular Therapeutix, Allegro, Alzheon, Annexon Biosciences, Beohringer Ingelheim, Carl Zeiss Medtec, Clearside Biomedical, Complement Therapeutics, DelSiTech, Galimedix, and Iveric Bio.
Dr. Ali is on the speakers’ bureau of Apellis and Iveric Bio, is on the advisory board of OcuTerra, and is a consultant to Allergan/Abbvie, Apellis, Genentech, Iveric Bio, Optomed, and Regeneron.
Dr. Arevalo has received research grant support from Topcon Medical Systems Inc and is a consultant to THEA Laboratories, Alimera Sciences Inc, and Iveric Bio.
Dr. Choudhry is on the advisory board of Novartis, Topcon Medical Systems Inc, Bayer, Apellis, and Optos PLC and is a consultant to Novartis, Roche Pharma, Bausch + Lomb, Alcon, J&J, Apellis, Allergan/Abbvie, and Viatris.
Dr. Jain is a principal investigator for Janssen Pharmaceuticals and Ocugen Inc.
Dr. Schneider is a principal investigator for Carl Zeiss Meditec, Oxurion, and Regeneron and is a consultant to Carl Zeiss Meditec and Notal Vision.
Dr. Vajzovic is a principal investigator for AGTC, Alcon, Aldeyra, Gyroscope, Heidelberg Engineering, Janssen, Novartis, Ocular Therapeutix, RegenexBio, and Roche/Genentech; is on the advisory board of Clearside Biomedical; and is a consultant to Alcon, Alimera Sciences, Allergan/Abbvie, Apellis, Bausch + Lomb, Iveric Bio, Ocugen, and Roche/Genentech.
Dr. Vora is on the speakers’ bureau of Iveric Bio, Disease State Education (nonpromotional), and is a consultant to Outlook Therapeutics and Paradigm Biopharmaceuticals.
Dr. Yonekawa is a primary investigator for Genentech, Kodiak, and Regeneron and is a consultant to Alcon, EyePoint, and Versant Health.
Dr. Leder declared no potential conflicts of interest with respect to the research, authorship, and/or publication of the article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
