Abstract
Keywords
Introduction
Optic disc pits (ODPs) are a congenital anomaly arising from a failure of fetal fissure closure during embryogenesis. The prevalence of the condition is approximately 1 in 11 000. Although the condition is often asymptomatic, it can result in central vision loss from serous macular detachments as well as visual field changes. Untreated OPD maculopathy will progress to a visual acuity (VA) of 20/200 or worse in 80% of cases. 1 The etiology of the subretinal fluid (SRF) associated with macular detachment in ODP maculopathy is unclear. Potential sources include optic nerve blood vessel leakage, leakage of orbital space fluid, the vitreous gel itself, or cerebrospinal fluid efflux.
Multiple treatment options exist to address symptomatic ODPs with submacular fluid. These include laser barricade, macular buckle, and gas tamponade. Because vitreous traction on the optic disc and macula might contribute to egress of fluid into the submacular space via the ODP, vitrectomy with posterior hyaloid release and internal limiting membrane (ILM) peeling—with or without gas tamponade—has also been shown to be useful in these cases.
More recently, other techniques, including an inverted ILM flap technique to cover the pit, inner retinal fenestrations to allow an egress of SRF into the vitreous, glial tissue removal, autologous fibrin, and sealing of the ODP with a biological glue such as Tisseel (Baxter Healthcare), have been tried. 2 The success rates for all methods are variable, with retinal reattachment rates ranging from 50% to 95% and visual improvement rates hovering around 50%.3 –6 Amniotic membrane transplantation (AMT) with a human amniotic membrane graft (AMG) has been used as well; however, reports are somewhat scarce. Because of these limited and variable results, investigation of new therapeutic approaches to address symptomatic ODPs is necessary.
Case Report
A 28-year-old competitive female athlete presented with a best-corrected VA (BCVA) of 20/400 OS associated with ODP maculopathy. Given the severity of the fluid and the patient’s high level of physical activity, the decision was made to proceed with pars plana vitrectomy, posterior hyaloid release, ILM peeling, inner retinal fenestration, SRF drainage through a temporal macular retinotomy, endolaser barricade temporal to the ODP, Tisseel fibrin glue to the ODP, and gas–fluid exchange. The submacular fluid initially resolved with improvement in VA to 20/70 but recurred 3 weeks after resolution of the gas bubble as the patient reengaged in vigorous physical activity. Given the refractory nature of the case, the patient then elected to proceed with an AMT using an AmnioGraft (BioTissue Inc) human AMG to fill in the ODP.
The BCVA was 20/400. Figure 1 shows the ODP in the inferotemporal aspect of the optic nerve with a large cuff of SRF in the macular region and the preoperative optical coherence tomography (OCT) of the nerve and macula, showing extensive intraretinal fluid and SRF.
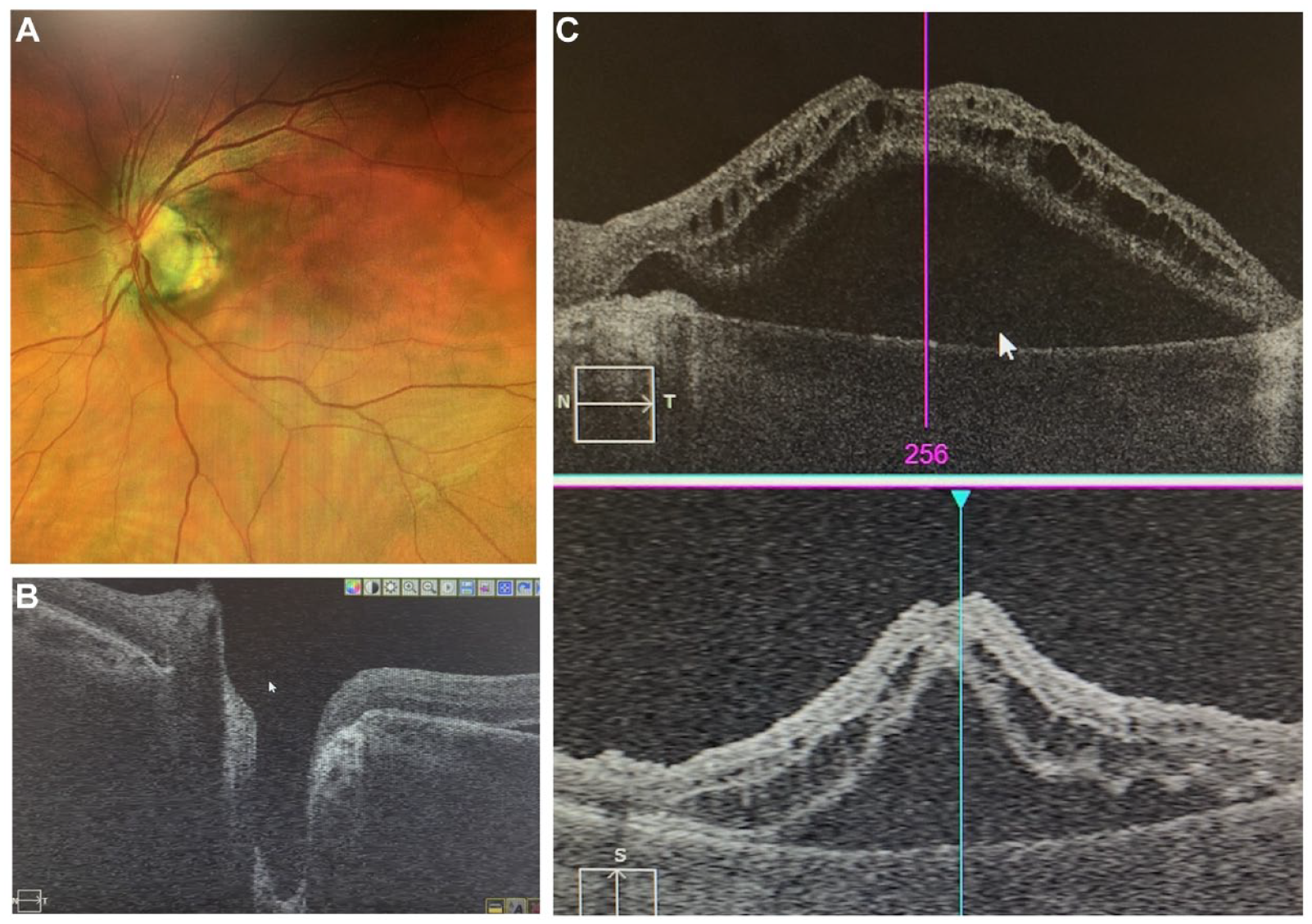
(A) Fundus photograph of the optic disc pit in the inferotemporal aspect of optic nerve with associated cuff of fluid in the macular region. (B) Preoperative optical coherence tomography (OCT) of optic nerve pit (Supplemental Video 1). (C) OCT shows extensive secondary intraretinal and subretinal fluid.
The surgery was performed via a standard 25-gauge 3-port vitrectomy under retrobulbar block using Ngenuity 3D heads-up visualization (Alcon, Inc). Before entering the eye, a 1.5 mm × 1.0 mm segment of the AMG was prepared based on the OCT measurement of the ODP of 1.0 mm × 0.75 mm in basal dimensions. After removal of the graft from the nitrocellulose paper, care was taken to maintain its orientation so that the smooth, shiny epithelial side remained upward facing while the adhesive stromal side abutted the host site (Figure 2).
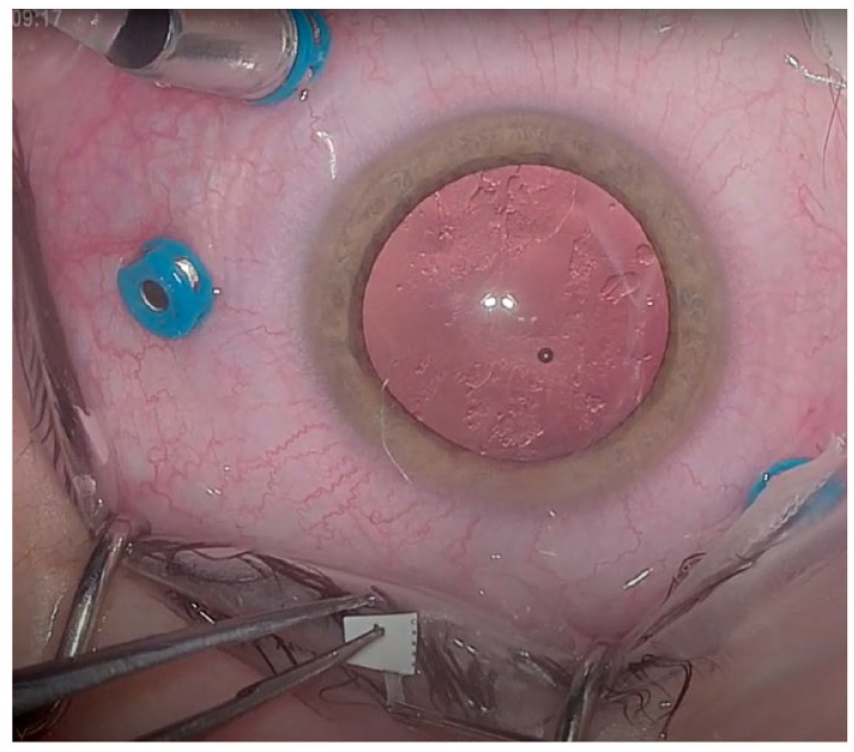
An internal limiting membrane forceps is used to remove the graft membrane (bottom) from the nitrocellulose paper.
After a peripheral vitrectomy with vitreous base shaving to ensure easy introduction of the amniotic membrane, the graft was introduced through a valved port with relative ease using an ILM forceps. Because of the adhesiveness of the graft, it was not possible to effectively place it into the ODP with a single forceps. Therefore, a chandelier light was placed and a MaxGrip endograsping forceps (Alcon Grieshaber) was used as a second instrument to apply the graft to the host site. A full fill of perfluoro-N-octane (PFO) liquid was injected to keep the graft in position (Figure 3). One week later, after confirmation of an intact graft–host interface via OCT scanning and clinical examination, the patient returned to the operating room for PFO removal.
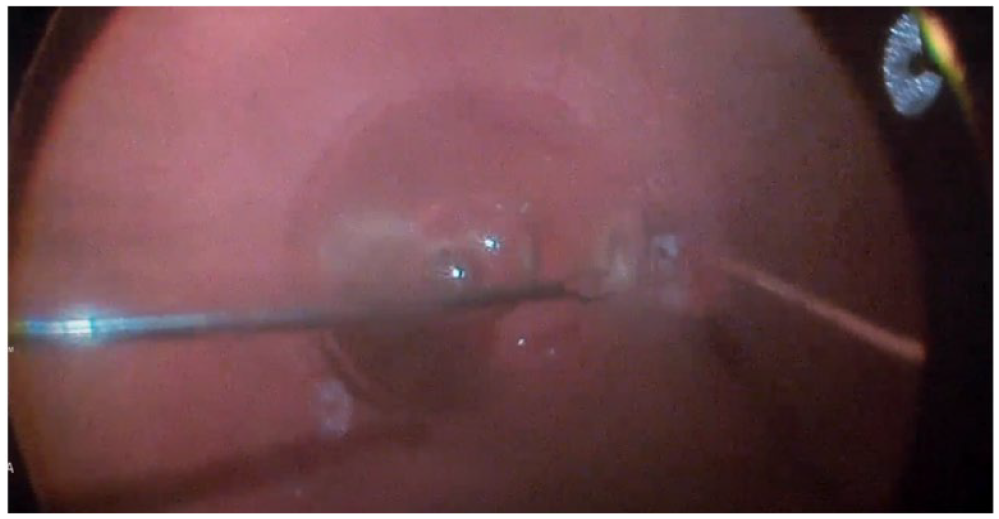
A bimanual technique under chandelier light is used to apply the human amniotic membrane graft to the optic disc pit under perfluorocarbon liquid stabilization. An endograsping forceps was used as the second instrument.
Two weeks after placement of the human AMG, the patient returned for a follow-up. OCT scanning of the macula showed complete resolution of the submacular fluid (Figure 4A), while fundus photography and raster OCT imaging of the optic nerve showed an adequate graft–host interface between the human AMG and the host tissue (Figure 4, B and C). The BCVA had improved to 20/50. These visual and anatomic findings were stable at the 3-month follow-up after the patient had resumed vigorous activity levels. Results were maintained at 6 months. After phacoemulsification with intraocular lens placement at this time, the BCVA returned to 20/25 with minimal symptomatology. The patient reported some mild persistent metamorphopsia that showed gradual subjective improvement over time.
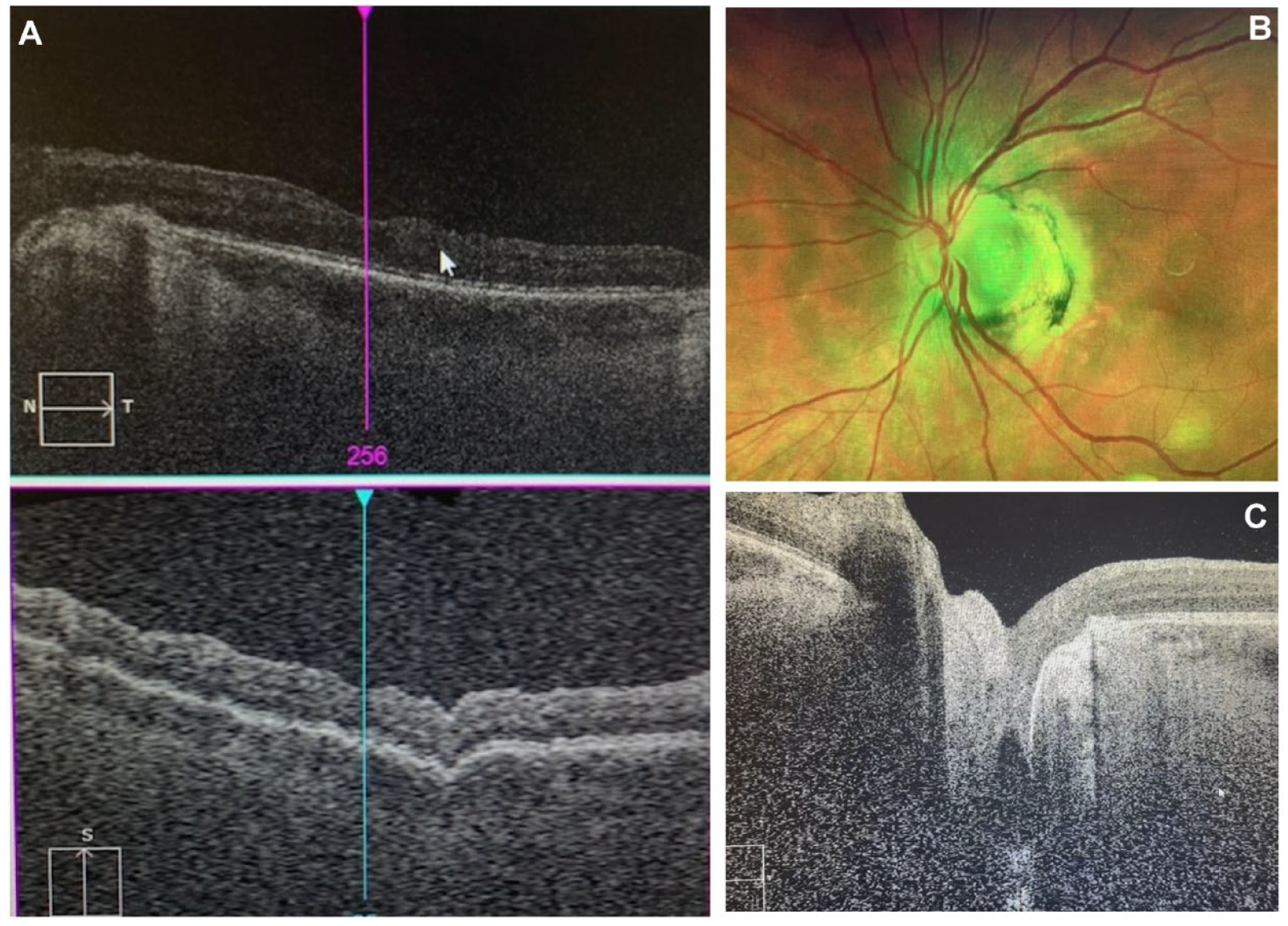
(A) Postoperative optical coherence tomography (OCT) of macula shows resolution of the intraretinal fluid and subretinal fluid. (B) Fundus photograph of an adequate human amniotic membrane graft (AMG)–host interface in the optic nerve pit. (C) OCT of the optic nerve shows an adequate human AMG–host interface in the optic nerve pit (Supplemental Video 2).
Conclusions
Management of ODPs can prove to be challenging to the vitreoretina specialist. Although multiple approaches exist to tackle ODP maculopathy, individual patients can have variable responses to the various modalities available for treatment. This reality can make it difficult to determine which intervention to recommend for a specific case, in particular because no established algorithms exist to guide us. The technique described in this report may add a viable, additional therapeutic option to the clinician’s armamentarium for treating these patients.
AMTs have been successfully used in anterior segment surgery to accelerate healing of surface tissues by serving as an alternative basement membrane and as a transient graft. They have been used in cases such as chemical burns to the cornea and to replace excised tissue when a pterygium or pingueculum is removed. Inherent growth factors and anti-inflammatory effects may accelerate epithelial wound healing. 7
AMT application in posterior segment surgery is less well studied; however, the structural characteristics of the human AMG suggest that it could be useful in a condition such as an ODP in which there is a tissue defect from which additional pathology emanates. If the fluid communication in ODP maculopathy indeed circulates from the vitreous through the ODP and into the subretinal space, occluding that pathway via AMT can limit the fluid egress responsible for the vision-threatening pathology. Human AMGs have been successfully used for macular holes 8 and, in isolated cases, for ODP maculopathy.9,10 The durability of resolution of submacular fluid in ODP maculopathy with this intervention is still in question because recurrence has been noted 1 to 3 years after the procedure. 10 This concern remains for the patient described in this current case report.
The case discussed here represents a situation in which previous intervention with multiple standard-of-care techniques failed. The application of an amniotic membrane in the patient was intended to provide a seal for the ODP because the biosafe glue that had been previously used rapidly degraded, leading to recurrence of the serous macular detachment once the patient resumed athletic activities. The apposition of the stromal side of the human AMG to the optic nerve tissue not only created an effective seal but in addition, the intrinsic growth factors and anti-inflammatory effects associated with it may have allowed for a more effective and permanent scaffolding. Given the refractory nature of this case, a full PFO fill was left in place for 1 week to maximize the likelihood of adherence. This contrasts with other previously described techniques involving simple ILM stripping and gas–fluid exchange or no tamponade.9,10 The combination of steps in this patient may have helped prevent the recurrence of submacular fluid. The persistent resolution of fluid at 3 and 6 months, shown in Figure 4, supports this notion.
Although several challenges and limitations exist with this procedure, it should not be subject to a steep learning curve for the experienced vitreoretina surgeon. Graft preparation, introduction of the graft into the eye, and application of the graft onto the ODP are the 3 components of the procedure that require special attention. Proper preparation of the graft before introducing it into the eye is imperative. In our case, the membrane was intentionally trimmed to a size slightly larger than the ODP, with the intention of providing enough tissue to fully cover and straddle the defect. Failure to fully cover the ODP would likely lead to the recurrence of fluid and maculopathy.
The structural flexibility of the human AMG material presents an additional challenge when introducing it through a valved port system. Entry of the membrane through the valved port with a macular forceps can be facilitated by using a second extraocular instrument, such as a 0.12 mm forceps. This maneuver provides displacement of a port leaflet valve, enlarging the opening and avoiding capture of the human AMG on the valve.
Understanding the difficulty presented by the adhesiveness of the stromal side of the amniotic membrane is the final key to a successful anatomic result. The bimanual technique with the assistance of PFO liquid to stabilize the membrane allows a solid 3-point-fixation maneuver and control of the human AMG during its application to the ODP. The use of a chandelier light is essential to providing the manual freedom necessary to accomplish this. This technique stands in contrast to those previously described, in which a single forceps was used.9,10 Awareness of the orientation of the graft throughout the procedure is a prerequisite for a proper seal given that the stromal side must ultimately face the defect.
Cost is likely the greatest hurdle to this technique, with the price of amniotic membranes ranging between $300 and $900. Given the frequent exclusion of tissue and pharmacologic costs in reimbursement considerations for ocular procedures, the price of a human AMG could limit the ability of a surgery center to perform this procedure and thus patients’ access to it.
Future applications of this modality could involve the use of AMT in addition to drainage of submacular fluid or as the sole therapy, given the apparently effective seal. These approaches could be undertaken with or without endolaser barricade. As with successful cases of fibrin glue application for the ODP, 2 procedures yielding cessation of the leakage from the pit have proven to be a sufficient intervention. This is because submacular fluid reabsorption by retinal pigment epithelium cells occurs spontaneously in patients with healthy subretinal tissue, as is generally the case in the patient demographic suffering from ODP maculopathy. Timely intervention to stop the leak can minimize the damage by the submacular fluid on the neurosensory retina.
The excellent and sustained anatomic results of this procedure coupled with the objective and subjective visual improvement that the patient experienced suggest that AMT is a viable technique for the management of ODP maculopathy. This intervention should be a part of the vitreoretina specialist’s repertoire when evaluating how to treat patients with this condition. Because not every patient is the same and different cases pose different surgical challenges, the bimanual technique with PFO assistance described here provides the vitreoretina surgeon with an additional technical alternative for surgical treatment of ODP maculopathy in difficult or refractory cases, be it the result of patient anatomy, visualization challenges, or varying structural characteristics of the human AMG. The decision to use AMT as a first-line treatment vs reserving it as a rescue procedure should be based on the entire clinical picture and a clear assessment of the patient’s needs and prognosis.
Footnotes
Ethical Approval
Ethics approval for this study was not obtained because of the singular, de-identified nature of the case reported. All methods and data extraction were performed per ICMJE standards.
Statement of Informed Consent
The patient provided written and verbal informed consent before the procedure, including a thorough review of all risks, benefits, and alternatives.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material is available online with this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
