Abstract
Keywords
Introduction
Nearly 100 million people worldwide have diabetic macular edema (DME). It is the leading cause of vision loss among people with diabetes, affecting more than 75 000 Americans annually. 1 Currently, the standard-of-care treatment for patients with DME is intravitreal (IVT) antivascular endothelial growth factor (anti-VEGF) therapy. 2 Although effective, short-term therapies such as anti-VEGF injections, which can require monthly administration, are associated with increased retinal thickness variability (RTV). These fluctuations are correlated with inconsistencies in the amount of disease control possible between injections and may be related to worse visual acuity (VA) outcomes over time. 2
Studies in patients receiving anti-VEGF therapy document significant retinal thickness fluctuations as measured by changes in central subfield thickness (CST) between treatments, 2 which then correlate with worse visual outcomes.2 –5 Use of a sustained-delivery IVT corticosteroid implant may offer longer lasting and more consistent treatment effects when compared with anti-VEGF therapy. This more consistent drug delivery may reduce RTV and, therefore, result in better patient outcomes. 6
The 0.19 mg fluocinolone acetonide (FAc) IVT implant (Iluvien, Alimera Sciences) is indicated for the treatment of DME in patients who have been previously treated with a course of corticosteroids and did not have a clinically significant rise in intraocular pressure (IOP). 7 Previous work has demonstrated efficacy and a favorable safety profile for the 0.19 mg FAc implant.6,8 The PALADIN study (IOP Signals Associated With ILUVIEN; ClinicalTrials.gov identifier NCT02424019) was a phase 4, nonrandomized, open-label observational study that evaluated the safety and functional outcomes of the 0.19 mg FAc IVT implant. 6 While the initial PALADIN analysis demonstrated the beneficial effects of the 0.19 mg FAc implant on the mean CST and best-corrected VA (BCVA), 6 the present analysis expanded on those findings, exploring the effects of consistent corticosteroid delivery via the 0.19 mg FAc implant on the magnitude of RTV and its subsequent ability to impact visual and treatment burden outcomes in patients with DME.
Methods
In the PALADIN study, 202 eyes from 159 patients with DME received the 0.19 mg FAc implant and were observed for up to 36 months. PALADIN was conducted at 41 sites across the United States. 6 The study protocol received institutional review board approval. The current analysis is a post hoc analysis based on previous studies of the 0.19 mg FAc implant.4,9
To receive the 0.19 mg FAc implant, all included patients must have previously received ocular corticosteroids without having an increase in IOP that the investigator judged as significant. 6 Details about the patient demographics, study design, methods, and inclusion criteria have been published previously.6,8
The eye receiving the 0.19 mg FAc implant was considered the study eye; in patients who had 2 study eyes, each eye was treated as a separate study eye. Additionally, the protocol mandated that the second study eye be discontinued from the study as soon as the first study eye completed 36 months.
Retinal thickness was measured by CST. RTV was assessed as CST area under the curve (CST-AUC), retinal thickness amplitude (RTA), and retinal thickness standard deviation (RTSD). CST-AUC was equivalent to the degree of dryness over a given period. For CST-AUC analysis, eyes were required to have at least 3 CST measurements before and after FAc implant, and for RTA and RTSD, eyes were required to have at least 3 CST measurements before FAc implant and at least 3 CST measurements starting 6 months after FAc implant. This was based on previous evidence that stable pharmacologic effects of the 0.19 mg FAc implant begins at approximately 6 months.4,9,10 Thus, in some analyses, eyes that did not meet these criteria were excluded, so not all analyses included all eyes. CST-AUC evaluated the entire patient treatment journey, whereas in the post-FAc period RTA and RTSD evaluated the journey’s post-pharmacologic effect.
RTA was the highest retinal thickness minus the lowest and was equivalent to the maximum range of retinal stretch over a given time period. RTSD was equivalent to the variations from the mean in retinal thickness over time. Methods for calculating CST-AUC, RTA, and RTSD are shown in Figure 1. Additionally, treatment burden was assessed as the number of yearly supplemental treatments of DME.
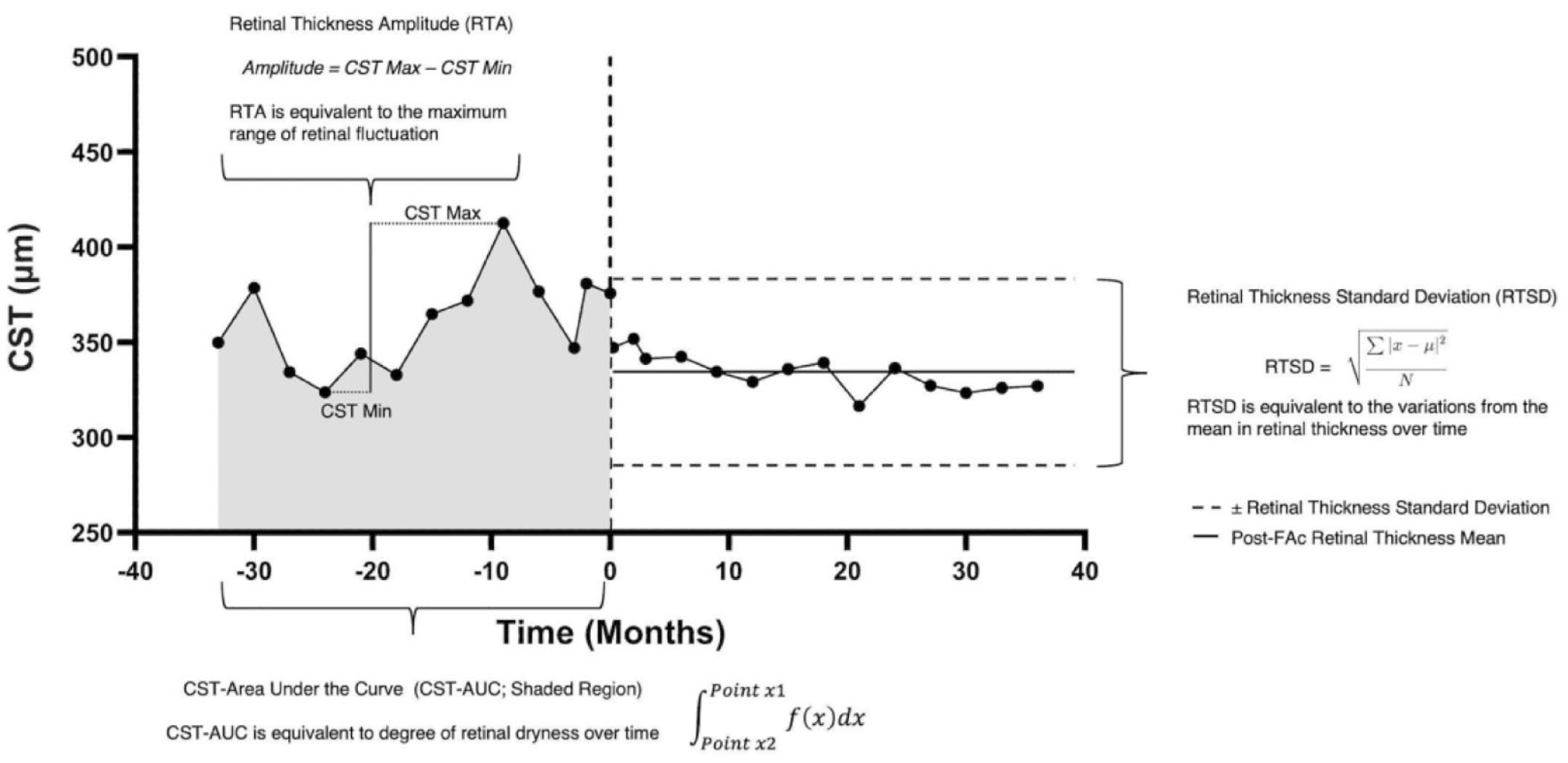
Calculation methods for RTV (CST-AUC, RTA, and RTSD).
The last-observed BCVA was used to generate the greatest sample size, as using specific timepoints would have excluded those eyes that did not have follow-up to 12, 24, or 26 months. Also, RTV was calculated over the same period that would correspond to the last-observed BCVA. For the study, the mean follow-up was >2 years.
General linear models were fit to understand the association between the last-observed BCVA and 3 different outcomes of interest: CST-AUC, RTA, and RTSD. The way the data were plotted resulted in a negative slope in the association between the variables, meaning R2 of −1 indicated a perfect fit and R2 of 0 indicated no correlation or fit at all. The last-observed BCVA was included as a covariate.
The baseline BCVA was included as an additional covariate to account for differences prior to administration of the 0.19 mg FAc implant. The coefficient of determination (R2 value) was the proportion of the variation in the outcome that was predictable by the covariates, with an R2 value close to 1 indicating high predictability and an R2 value close to 0 indicating low predictability.
Based on a 1-sample, 2-sided chi-square test, a sample size of 150 patients provided approximately 90% power to detect a difference of 10% between the historical and anticipated incidence rates of IOP >30 mm Hg. The observed incidence rate of IOP >30 mm Hg in the FAME (Fluocinolone Acetonide in Diabetic Macular Edema) trials was 20% 11 ; it was anticipated that the incidence rate in the present trial would be 10% or less.
Results
Retinal Thickness
At baseline, 32.9% of eyes had a CST ≤300 µm; the percentage of eyes with CST ≤300 µm fluctuated throughout the 36 months after FAc implant but was largely increased relative to baseline. At its peak, 55.4% of eyes had a CST ≤300 µm, which occurred at month 21. At month 36, 46.8% of eyes had a CST ≤300 µm (Figure 2).
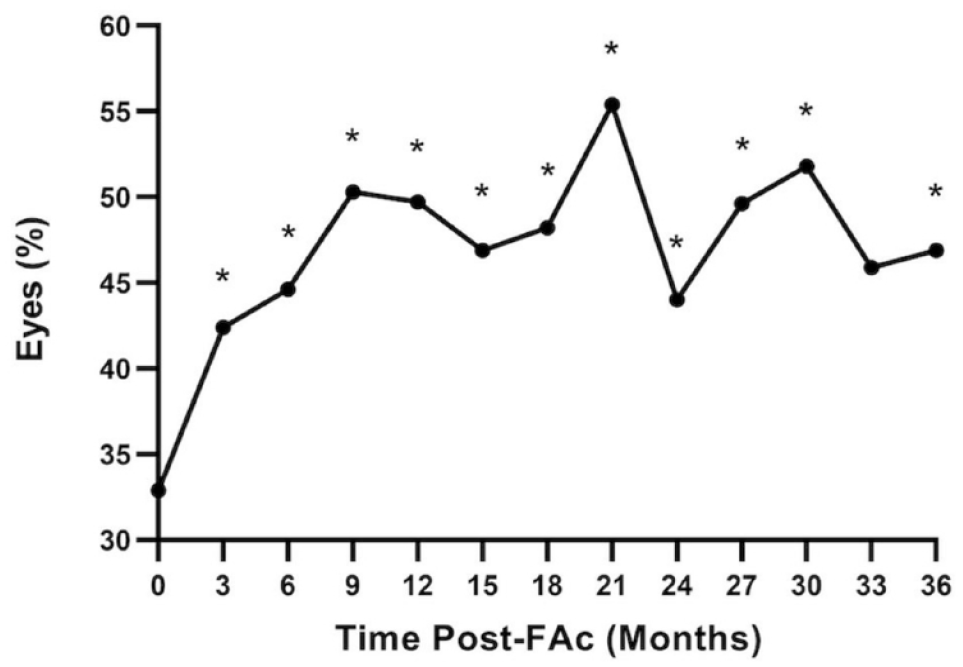
Percentage of eyes with CST ≤300 µm over 36 months (n = 202). Data were analyzed using the McNemar test to determine significance. *P < .05 compared with baseline.
RTV Before and After FAc Implant
In the PALADIN study, FAc significantly reduced RTV in all measures, including CST-AUC, RTA, and RTSD, compared with pre-FAc administration (P < .0001) (Figure 3).
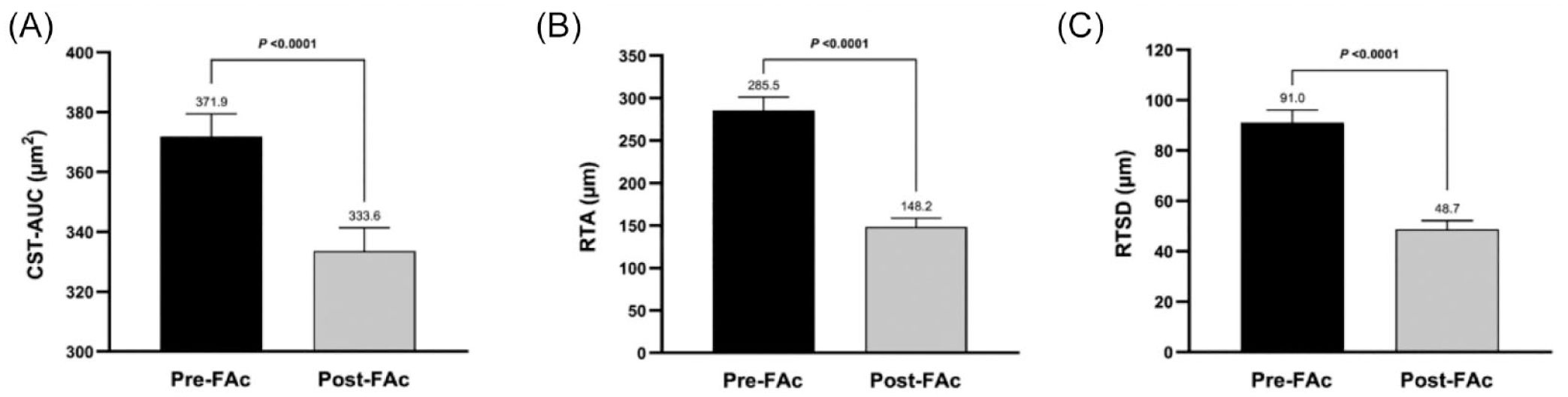
RTV before and after FAc implant as measured by OCT (n = 151). Assessments include (A) CST-AUC, (B) RTA, and (C) RTSD. Results were expressed as mean µm ± SEM, and data were analyzed using a paired t test to determine significance.
Visual Acuity and Treatment Burden by RTV Quartiles
Eyes were split into quartiles (Qs) based on RTV outcomes (CST-AUC, RTA, and RTSD), with Q1 associated with the lowest magnitude variability and Q4 associated with the highest magnitude variability. Measurements of RTV were then broken down into quartiles and analyzed for visual-related and treatment burden–related outcomes. Quartiles were defined as CST-AUC (Q1, 168.3-263.3 µm; Q2, 263.8-315.5 µm; Q3, 316.3-380.0 µm; Q4, 381.3-720.5), RTA (Q1, 1.0-43.0 µm; Q2, 44.0-96.0 µm; Q3, 98.0-188.0 µm; Q4, 190.0-548.0 µm), and RTSD (Q1, 0.7-16.2 µm; Q2, 16.3-31.7 µm; Q3, 31.8-63.0 µm; Q4, 65.1-211.2 µm). Across all RTV metrics, eyes in Q1 and Q2 saw the greatest improvement in BCVA, while Q3 and Q4 either maintained or lost BCVA (Figure 4).
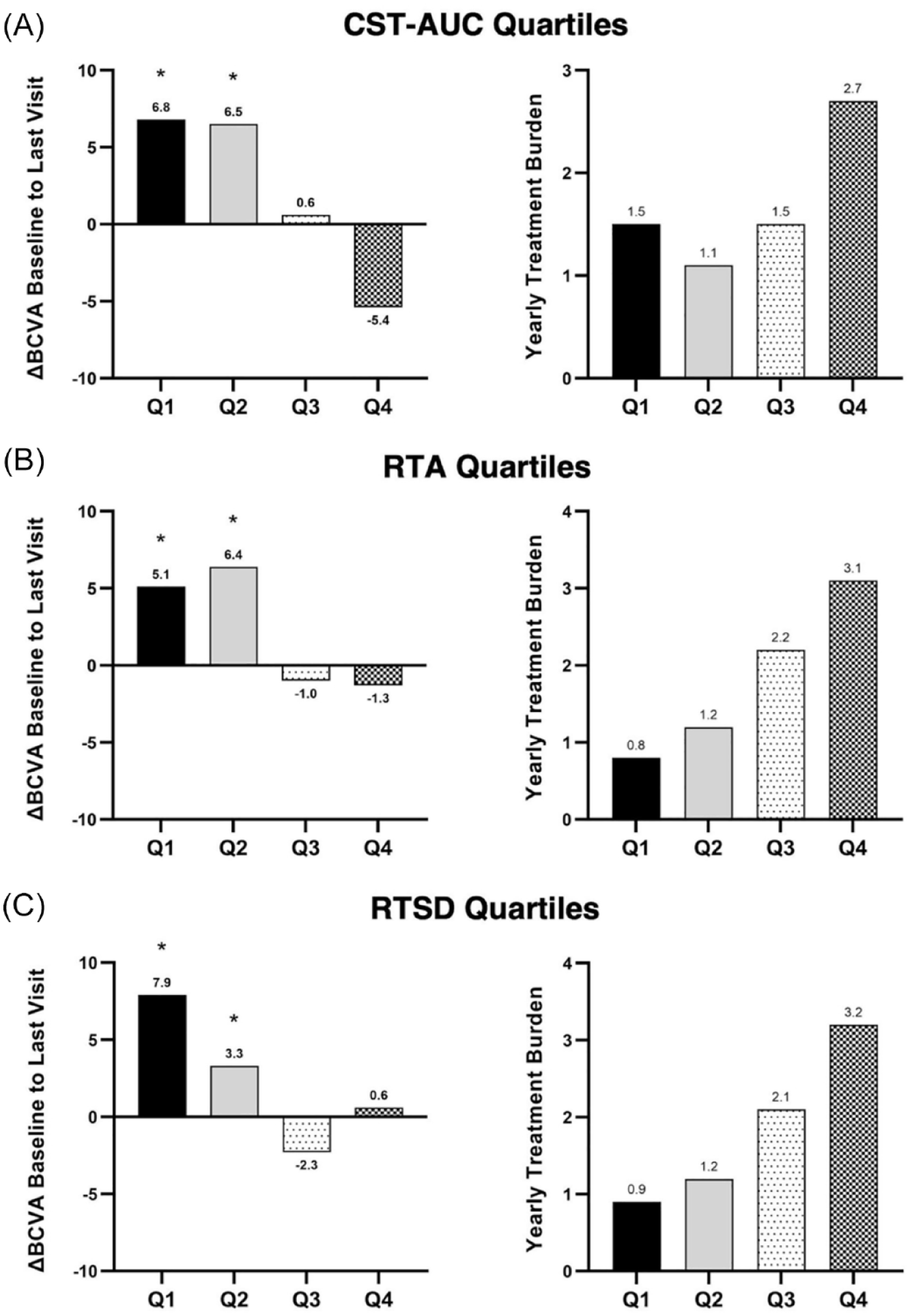
Change in BCVA from baseline to the last study visit and average yearly treatment burden (as measured by the number of yearly supplemental treatments of diabetic macular edema) after FAc implant by (A) CST-AUC quartiles (Q1, n = 50; Q2, n = 50; Q3, n = 50; Q4, n = 50), (B) RTA quartiles (Q1, n = 45; Q2, n = 44; Q3, n = 46; Q4, n = 46), and (C) RTSD quartiles (Q1, n = 45; Q2, n = 45; Q3, n = 45; Q4, n = 46). Q1 represents the quartile with the smallest respective RTV values, while Q4 represents the quartile with the largest values. Results were expressed as the change in mean BCVA letters from baseline and were analyzed using a paired t test to determine significance. *P < .05.
Regarding cataract surgery, 29 of 202 eyes were phakic at baseline, and 18 of those 29 eyes underwent cataract surgery while on study, the majority of which were in Q3 and Q4. Quartiles were also linearly related to yearly treatment burden, as measured as the number of yearly supplemental treatments of DME, with R2 values of 0.69, 0.93, and 0.97 for CST-AUC, RTA, and RTSD, respectively.
Relationship Between RTV and Visual Outcomes
To assess whether RTV closely correlated with visual outcomes, each RTV measurement (CST-AUC, RTA, and RTSD) was plotted against the last-observed BCVA and fit to a simple linear regression. The last-observed BCVA letter score exhibited linear correlations with CST-AUC (R2 = −0.100), RTA (R2 = −0.125), and RTSD (R2 = −0.162) (Figure 5; Table 1). A multivariate linear regression was performed with baseline BCVA as a covariate, which improved correlations between the last-observed BCVA and RTV metrics (Table 1).
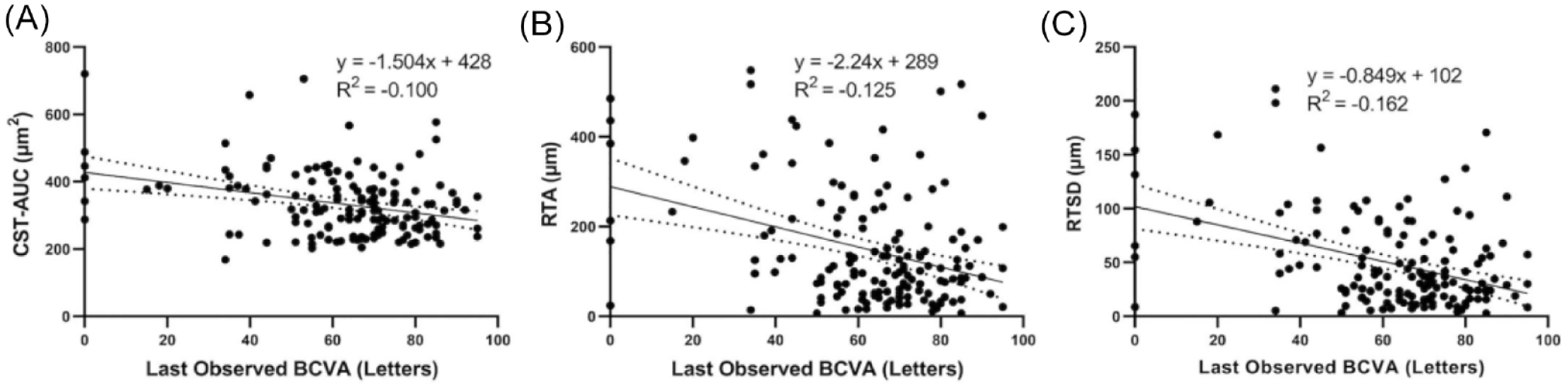
Correlation of RTV as measured by (A) CST-AUC (n = 151), (B) RTA (n = 151), and (C) RTSD (n = 151) with last-observed BCVA. Data were fit into a simple linear regression.
Correlation of RTV Metrics to Last-Observed BCVA for Simple and Multivariate Linear Regression.

Abbreviations: BCVA, best-corrected visual acuity; CST-AUC, central subfield thickness-area under the curve; RTA, retinal thickness amplitude; RTSD, retinal thickness standard deviation; RTV, retinal thickness variability.
Conclusions
In its primary analysis, data from PALADIN, a 3-year, phase 4, observational study, supported the safety and efficacy of the 0.19 mg FAc implant in the real world. 6 The 0.19 mg FAc IVT implant, a long-acting therapeutic option that treats DME for up to 36 months, provides consistent control of RTV with continuous microdosing of the retina. Other PALADIN analyses demonstrated that the 0.19 mg FAc implant significantly reduces the mean CST concurrent with increased mean BCVA and better disease control as measured by yearly treatment burden.6,12 To expand on these reports, the present analysis examined the impact of RTV, a known predictor of poor visual outcomes,3,5 on anatomic and functional measures. In this post hoc analysis of the PALADIN study, the 0.19 mg FAc implant demonstrated an ability to significantly reduce RTV through 36 months, which was associated with an increase in the percentage of patients with a dry retina. Additionally, reduced RTV was directly correlated with functional outcomes: lower RTV was associated with better BCVA gains and reduced treatment burden.
High RTV is a known complicating factor in the management of DME that often leads to poor patient outcomes.3,5 Cyclic stretching of retinal pigment epithelium cells, on which photoreceptors rely for metabolic support, can induce apoptosis. 13 Mechanical stretching can also cause retinal ganglion cell death, directly inhibiting phototransduction. 14 These cellular effects translate to functional outcomes. In a post hoc analysis of Diabetic Retinopathy Clinical Research (DRCR) protocols T and V, eyes experiencing larger CST fluctuations, and therefore greater mechanical stress on retinal cells, lost significant vision at 2 years compared with eyes with smaller CST fluctuations. 3 In analyses of RTV in anti-VEGF–treated eyes with age-related macular degeneration, high RTV was associated with poor VA outcomes at 2 years.2,15 Similarly, anti-VEGF–treated eyes with DME demonstrated a direct correlation between RTV and visual outcomes at 12 months. 5
By contrast, previous reports on the 0.19 mg FAc implant reinforce its utility in controlling RTV and, therefore, patient outcomes. In 3 smaller studies, the 0.19 mg FAc implant reduced RTV in both retrospective and prospective analyses.4,9,16 In the present study, PALADIN demonstrated on a larger scale that the 0.19 mg FAc implant is associated with significant reductions in RTV, with an increased percentage of eyes with dry retinas at 36 months vs baseline (≤300 µm; Figure 2) and significant RTV reductions before vs after FAc implant across all 3 analysis measures (Figure 3). This reduced RTV post FAc implant comes even though most patients received anti-VEGF and/or steroid therapies in the 36 months before receiving FAc. 9 However, eyes in Q1 and Q2 had the least amount of RTV and the largest improvements in functional outcomes (ie, BCVA gains and yearly treatment burden) vs baseline, while eyes in Q3 and Q4 had the greatest amount of RTV and the worst visual outcomes (Figure 4).
Because RTV is an umbrella term used to describe the extent of edema control over time, no single measurement accurately describes the full RTV trajectory; however, in conjunction with RTA and RTSD, these parameters together give a more accurate and detailed depiction of RTV before and after FAc. Also, while CST-AUC evaluates the whole disease course, RTA and RTSD evaluate the disease course’s post-pharmacologic effect. Previous analyses of the PALADIN data found that while the pharmacologic effect of FAc is seen as early as day 7, it reaches a steady state of disease control around 6 months after FAc.4,9 Therefore, the first 6 months of pharmacologic effect of FAc were excluded, allowing RTV to be evaluated based on the ability of FAc to establish and maintain long-term disease control rather than the initial CST improvement. In line with this information, the present data show that, numerically, analyzing RTV via RTA or RTSD correlates better with long-term visual outcomes (eg, last-observed BCVA in the present study) than CST-AUC (Figure 5). When considering baseline visual acuity as a covariate to account for a ceiling effect in BCVA, the multivariate regression leads to further improved correlations (Table 1).
While RTA, RTSD, and CST-AUC did correlate with the last-observed BCVA, the unadjusted R2 values were somewhat low overall. Because approximately one third of study eyes had a baseline BCVA of 20/40 or better, it is likely that the poor correlations of RTV measures with the last-observed BCVA were due to a BCVA ceiling effect. Variability between investigators’ assessments is an additional possibility, as a limitation of the PALADIN study is that no images were collected and all data were entered and quality checked by the principal investigator and study site. When using a multivariate linear regression with baseline BCVA as a covariate, the R2 values improved from approximately −0.13 to −0.44.
Another point of consideration is the influence of supplementary anti-VEGF therapy on RTV. A previous report demonstrated that the mean number of yearly supplemental treatments decreased from 3.5 before FAc to 1.7 after FAc, with approximately 25% of eyes requiring no supplemental therapy through 36 months after FAc. 12 In the eyes that received supplemental therapy, most were given aflibercept; these eyes did not experience additional improvement from baseline to 36 months. 12
Evidence continues to mount regarding the importance of RTV in understanding the efficacy and durability of treatments in DME. Improving patient outcomes in DME requires careful consideration of which treatments are used and in what order. Because of the demonstrated link between high RTV and poor visual outcomes, reducing RTV at treatment onset could prevent or minimize the development of RTV-related damage. Although short-acting treatments such as anti-VEGF injections are the current standard of care for DME, real-world studies have demonstrated that their efficacy is linked to high injection frequency, often monthly. 17 This high injection frequency is associated with fluctuations in drug availability at the retina between treatments, which can lead to inconsistent disease control, greater RTV, 16 increased mechanical stress on the retina, and ultimately, irreversible vision loss. 15 By contrast, the 0.19 mg FAc implant is the only therapy that continuously treats DME for up to 36 months.
Previous analyses of the PALADIN study established that the 0.19 mg FAc implant provides effective DME treatment of visual, anatomic, and treatment burden outcomes.6,12 The present analysis, however, expands on earlier findings and demonstrates that the 0.19 mg FAc implant significantly reduces RTV, thereby improving both BCVA and treatment burden, 2 outcomes that are vital for patients with DME.
Footnotes
Authors’ Note
Presented in part at the American Society of Retina Specialists Annual Meeting 2022, New York City, New York, USA, July 13–16, 2022; EURETINA 2022 Congress, Hamburg, Germany, September 1–4, 2022; Club Jules Gonin Meeting 2022, Dubrovnik, Croatia, September 7–10, 2022; Retina Society 2022, Pasadena, California, November 2–5, 2022.
Ethical Approval
This study was approved by the Institutional Review Boards (IRBs) of the following institutions: Sterling (the study’s central IRB; approval 5020), Johns Hopkins Medical Center (approval IRB00068317), Tufts Medical Center (approval 11980), University of California Davis (approval 798838-2), Mayo Clinic (approval 15-0028269), University of Illinois (approval 2016-0018), University of Utah (approval 0087306), Wayne State University (approval 060915M1F), University of Maryland (approval HP-00068708), and Washington University (approval 201602044). This study adhered to the guidelines of the Declaration of Helsinki.
Statement of Informed Consent
Written informed consent was obtained by all participants in the study.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Dr. Sheth received financial support from 4D Molecular Therapeutics, Allergan, Ashvattha Therapeutics, Chengdu Kanghong, Eyepoint, Genentech, Gyroscope, Ionis, Iveric Bio, NGM Biopharmaceuticals, Novartis, Opthea, Outlook, Oxurion, Recens Medical, Regeneron, Regenxbio, Roche, SalutarisMD, and SamChungDang, and Santen; is a consultant to Alimera Sciences, Apellis, Eyepoint, Genentech, Graybug, Iveric Bio, Novartis, Regeneron, and Vial; and is a lecturer for Alimera Sciences, Apellis, and Genentech. Dr. Singer is a consultant to Aerie, Alimera Sciences, Allegro, Allergan Eyepoint, Genentech Kodiak, Novartis, Regeneron, and Santen; is a lecturer for Aerie, Alimera, Sciences, Allegro, DRCR, Genentech, Icon, Ionis, Kalvista, Kodiak, Novartis, Opthea, Optos, Regeneron, Santen, Senju, and Sydnexis; provides expert testimony for Allergan, Genentech, Mallinckrodt, Regeneron, and Spark; and owns equity in Aviceda, Inflammasome, Novartis, and Nanoscope. Dr. MacCumber is a consultant to Alimera Sciences, Bausch + Lomb, Genentech, Novartis, and Regeneron. Dr. Cutino is an employee of Alimera Sciences and owns equity in Alimera Sciences. Dr. Kasper is an employee of Alimera Sciences. Dr. Coughlin is an employee of Alimera Sciences. Dr. Riemann received financial support from AGTC, Alcon, Alimera Sciences, Allergan, Arepio, Chengdu Kanghong, Clearside, Genentech/Roche, Gyroscope, Janssen/Johnson & Johnson, Lineage (Formerly BioTime), Lowry-MacTel Registry, Neurotech, Nightstar/Biogen, NotalVision, Novartis, Ophthotec/Iveric, Regeneron, RegenexBio, and Spark; is a consultant to Alcon, Alimera, Alimera Deutschland GmBH, Allergan, Animal Eye Institute, Bausch + Lomb/Valeant, BMC/Eyetube, DORC, Genentech/Roche, Gore, Gyroscope, Haag Streit AG, Haag Streit Surgical, Haag Streit USA, HumanOptics AG, Iamc2, iVeena, Janssen/Johnson & Johnson, Kaleidoscope Engineering, Lineage (Formerly BioTime), MedOne, Neuracle, NotalVision, Orbit BioMedical, RegenexBio, Reliance Industries, SalutarisMD, Samsara, TrueVision, Vortex Surgical, and Zeiss; is a lecturer for Alcon, Alimera Sciences, Alimera Deutschland GmBH, Allergan, Bausch + Lomb/Valeant, CSTLII, Novartis, Regeneron, Reliance Industries, SalutarisMD, and TrueVision; owns equity in CVP (CEI Vision Partners)/ECP (Eye Care Partners), Digital Surgery Systems, ForwardVue Pharma, iVeena, TrueVision, and Vortex Surgical; receives royalties from MedOne, TrueVision, and Vortex Surgical; owns intellectual property with Haag Streit USA, Iamc2, Janssen/Johnson & Johnson, Kaleidoscope Engineering, MedOne, Reliance Industries; and has leadership roles with Aniridia Foundation International (Medical and Scientific Advisory Council), Chruman Research (Owner/Cofounder), Clovernook Center for the Blind and Visually Impaired (Board of Trustees), CVP (CEI Vision Partners)/ECP (Eye Care Partners) (Owner/Cofounder), Digital Surgery Systems (Owner), ForwardVue Pharma (Board of Directors), Iamc2 (Cofounder), Macor Industries (Owner), Northmark Pharmacy (Owner), and VEO (Owner/Cofounder).
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Alimera Sciences, Inc, Alpharetta, GA. The sponsor or funding organization participated in the design of the study, data management, data analysis, interpretation of the data, preparation, and review of the manuscript.
Collaborators: The PALADIN Study Group
Alimera Sciences, Inc, as the responsible party for the PALADIN study, attests that the PALADIN Study Group can be included as an author and that the below list of collaborators is accurate.
PALADIN Principal Investigators
Ahmed Abdelsalam, MD
Akbar Shakoor, MD
Ala Moshiri, MD
Andrew Barkmeier, MD
Asheesh Tewari, MD
Carl Baker, MD
Daniel Kiernan, MD
Daniel Rosberger, MD
Daniel Roth, MD
David Eichenbaum, MD
Deepika Malik, MD
Dennis Marcus, MD
Doug Dehning, MD
Elias Reichel, MD
Elisha Tilton, MD
Evelyn Fu, MD
Gregg Kokame, MD
Hani Salehi-Had, MD
Harit Bhatt, MD
Jared Nielsen, MD
Jay Prensky, MD
Jennifer Lim, MD
Jon Adleberg, MD
Joseph Beck, MD
Joseph Gunn, MD
Joseph Podhorzer, MD
Kent Small, MD
Lakshmana Kooragayala, MD
Lars Freisberg, MD
Lisa Schocket, MD
Maria Berrocal, MD
Martin Worrall, MD
Michael Cassell, MD
Michael Singer, MD
Michael Tsipursky, MD
Nancy Holekamp, MD
Pamela Weber, MD
Pauline Merrill, MD
Peter Campochiaro, MD
Pravin Dugel, MD
Rahul Khurana, MD
Rajendra Apte, MD
Rajiv Rathod, MD
Randy Katz, MD
Richard Chace, MD
Robert Kwun, MD
Ruben Grigorian, MD
Sam Mansour, MD
Saradha Chexal, MD
Steven Madreperla, MD
Victor Gonzalez, MD
William Aldred, MD
