Abstract
Purpose:
The purpose of this article is to review and analyze reported cases of ocular venous air embolism (OVAE) to develop a reliable clinical definition of OVAE and effective prevention strategies.
Methods:
We reviewed all reports of suspected air embolism during vitrectomy published in PubMed since the introduction of pars plana vitrectomy, and 5 cases found elsewhere and separately reported concurrent with this review.
Results:
OVAE is a precipitous drop in end-tidal CO2, a choroidal detachment, or a choroidal wound, followed by signs of impending or actual cardiovascular collapse, during vitrectomy air infusion. In each case meeting the above clinical definition, entrained air was found whenever it was sought (8/8, 100%), either by antemortem imaging or postmortem forensic investigations. Most OVAE cases were fatal (9/13, 69%), with 8 of 9 deaths (89%) occurring the day of surgery.
Conclusions:
OVEA is a rare but usually fatal complication of air infusion into the eye during vitrectomy. Although received with skepticism when first reported (2005), OVAE may be the most lethal type of surgical air embolization because of its high entrainment pressure and proximity to the heart. Because the effective response time to avoid a fatal OVAE outcome can be less than 1 minute, use of preventive measures is critical—most notably a “time out” before air infusion to confirm infusion cannula positioning, and immediate cessation of air infusion if choroidal detachment is detected.
Keywords
Introduction
From the advent of (pars plana) vitrectomy in 1971 until the introduction of cannulated vitrectomy in 2002, surgical entry to the vitreous cavity was accomplished directly through sclerotomies, after incision and displacement of the overlying conjunctiva. 1,2 Infusion lines with 20-gauge cannula flanges were sutured directly to the sclera to enable 3-port, closed vitrectomy.
Cannulated vitrectomy introduced a new entry system in which 3 cylindrical metal cannulae, riding on insertion trocars, are placed transconjunctivally through the eye wall into the vitreous cavity. An infusion line is then inserted into 1 trocar-placed, unsutured cannula, while the remaining cannulae receive fiber-optic and vitrectomy probes.
The advantages of cannulated sclerotomies over plain sclerotomies include: transconjunctival entry; fluid management via valved cannulae during instrument exchanges; rapid, sutureless wound closure upon removal of the cannulae; and quick, relatively painless recovery postoperatively. In sum, these advantages have almost eliminated direct scleral entry in vitrectomy.
However, cannulated vitrectomy also eliminated suturing of infusion cannulae to the scleral wall. And a rare, often fatal complication, ocular venous air embolism (OVAE), more easily associated with unsecured infusion cannulae, has now been reported. 3 -5
Any vitrectomy that uses air infusion may result in OVAE if insidious slippage of an infusion cannula during vitrectomy causes suprachoroidal infusion of pressurized air. Resultant tearing of the vortex veins can allow silent entrainment of suprachoroidal air into the systemic venous circulation at a rate that is usually fatal unless promptly detected (Figure 1). 5
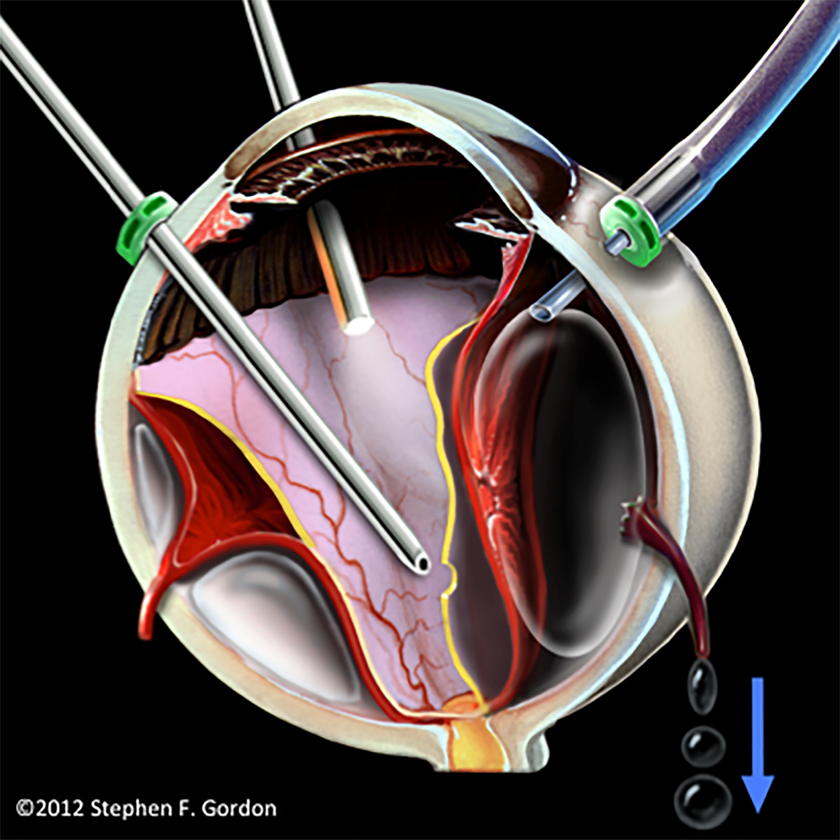
Concept illustration of pressurized air from a slipping, unsecured cannula causing accidental choroidal detachment, torn vortex vein ampullae, and air entrainment through vortex veins. (Reprinted with permission from the British Journal of Ophthalmology. 5 )
OVAE can also result from vitreous cavity air accessing intact vortex veins through large choroidal wounds, such as from trauma or choroidal melanoma resection. 6,7 Although these cases involve a particularly high risk for developing OVAE, they are rare relative to all cases of vitrectomy air infusion, and the informed vitrectomy team can take special precautions during any necessary air infusion.
As in other surgical fields affected by venous air embolism (VAE), responses to the iatrogenic OVAE complication have often been skepticism, denial, or reluctance to report secondary to reputational and medicolegal concerns. 8,9 Consequently, 13 years after the initial report of OVAE, 3 most vitreoretinal surgeons appear to remain unaware of, and thus susceptible to, the OVAE complication.
The evidence for OVAE, however, has become overwhelming, demanding widespread dissemination and establishment of reliable preventive measures. After a systematic analysis of all reported cases and experimental evidence, we recommend precautions that could substantially reduce or even eliminate the risk of fatal OVAE during vitrectomy.
Materials and Methods
The authors began investigations as a result of direct personal knowledge of a suspected case of fatal OVAE for which a consensus opinion as to the cause of death could not be achieved. After experimentally confirming suprachoroidal air infusion and consequent OVAE (in banked eyes) as a possibly fatal complication of vitrectomy, 5 we reviewed articles found in PubMed from 1970 to 2018 related to pars plana vitrectomy and air embolism, articles referenced in these PubMed articles, and 5 OVAE cases discovered elsewhere and concomitantly reported in a separate article along with this review. 10
We also reviewed the anesthesia literature on VAE in all types of surgery, including the historical literature before the development of modern databases. Additionally, in January 2017 we surveyed 30 vitreoretinal physicians in the United States, randomly selected from the American Society of Retina Specialists (ASRS) membership directory, as to whether they were aware of the potential OVAE complication. This survey was conducted via email with phone follow-up as needed.
Results
A total of 13 cases of OVAE were reviewed and are presented in table form (Table 1). A narrative summary of each case is then presented in Supplemental Report 1. A reliable clinical definition of OVAE derived from this review and important lessons for prevention of OVAE are described in the following discussion section.
OVAE Review.
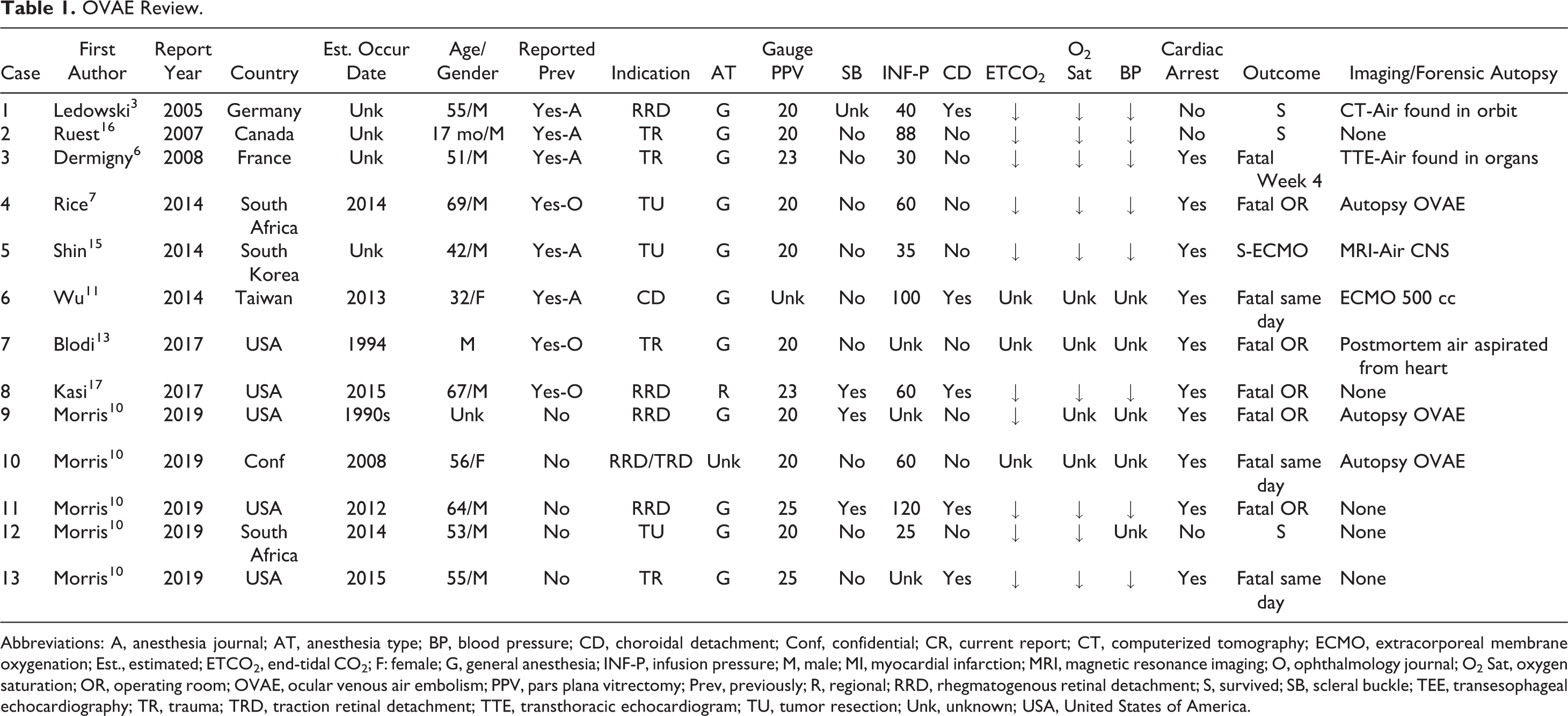
Abbreviations: A, anesthesia journal; AT, anesthesia type; BP, blood pressure; CD, choroidal detachment; Conf, confidential; CR, current report; CT, computerized tomography; ECMO, extracorporeal membrane oxygenation; Est., estimated; ETCO2, end-tidal CO2; F: female; G, general anesthesia; INF-P, infusion pressure; M, male; MI, myocardial infarction; MRI, magnetic resonance imaging; O, ophthalmology journal; O2 Sat, oxygen saturation; OR, operating room; OVAE, ocular venous air embolism; PPV, pars plana vitrectomy; Prev, previously; R, regional; RRD, rhegmatogenous retinal detachment; S, survived; SB, scleral buckle; TEE, transesophageal echocardiography; TR, trauma; TRD, traction retinal detachment; TTE, transthoracic echocardiogram; TU, tumor resection; Unk, unknown; USA, United States of America.
Survey of Awareness
In our January 2017 randomized survey (see Methods) of ASRS vitreoretinal specialists in the United States, only 6 of 30 respondents (20%) were aware of the OVAE complication.
Discussion
OVAE Definition
We define OVAE as a precipitous drop in end-tidal CO2 (ETCO2), a choroidal detachment, or a choroidal wound, followed by signs of impending or actual cardiovascular collapse during vitrectomy air infusion. This clinical definition encompasses all known cases of suspected air embolization occurring during vitrectomy air infusion.
In support of this definition, we note that entrained air was found each time it was sought in OVAE cases (8/8) so defined: in the heart, during intraoperative imaging (Case 6)
11
; in the heart during postmortem radiography (Case 4, Figure 2)
7
;
in the heart during 3 forensic autopsies (Cases 4, 9, and 10)
7
and in an in vivo experiment
12
; by aspiration of air (from the heart) postmortem (Case 7)
13
; in the venous system, by cannulation of the common femoral artery and vein, enabling extracorporeal membrane oxygenation (ECMO, Figure 3, Case 6)
11
; in the orbit behind the eye, by postoperative imaging (Case 1)
3
; and finally, with a patent foramen ovale present,
14
paradoxical embolization into the left main coronary artery and the brain (Cases 3 and 5)
6,15
was documented postoperatively on OVAE patients who survived the embolic event.
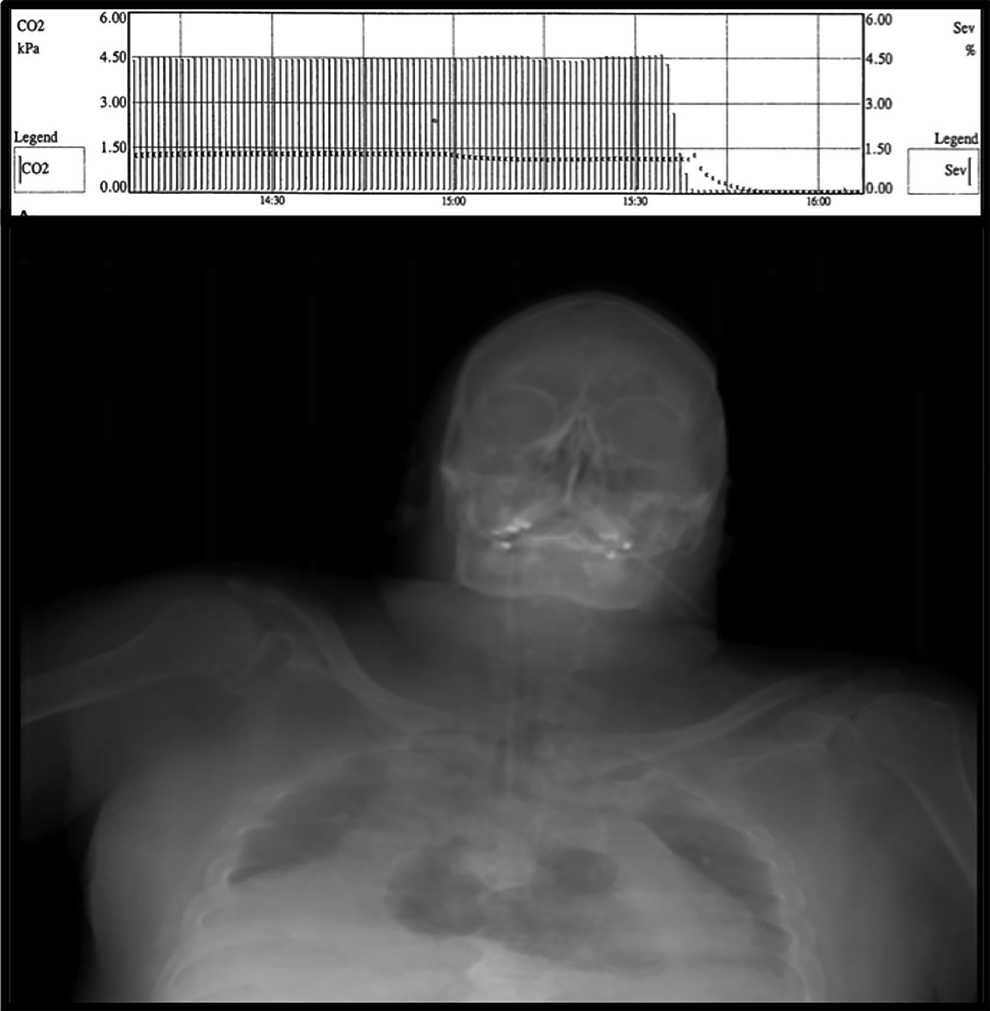
Postmortem roentgenogram revealing extensive air in the heart of a patient who suffered ocular venous air embolism during choroidal melanoma resection (Case 4). A precipitous drop in end-tidal CO2 is seen in the graph above. (Reprinted with permission from Retinal Cases and Brief Reports. 7 )

Extracorporeal membrane oxygenation showing blood frothy with entrained air exiting the body (arrow), and normal blood (above) being returned. More than 500 cc of air was removed from the circulating blood. (Case 6) (Reprinted with permission from the Journal of Anesthesia. 11 )
In the minority of OVAE patients who survive, this clinical definition (supported by the 100% confirmation rate noted above) recognizes that OVAE actually occurred. In mortality conferences for those who don’t survive, it justifies acceptance of clinical OVAE so defined as the probable cause of death, shifting the burden of proof from OVAE proponents to those who contend otherwise.
If a case meets the stringent criteria established by these circumstances, this clinical definition allows future authors to abandon the OVAE event descriptions of “possible, presumed, or suspected.” 3,5,15 -17 Finally, this definition encourages vitreoretinal surgeons to recognize and report even OVAE cases that are effectively stopped at an early stage, improving our knowledge base for this life-threatening complication.
Chronology of the OVAE Complication
The first 3 published cases of OVAE (Cases 1, 2, and 3) were reported in the anesthesia literature in 2005, 2007, and 2008. 3,6,16 By chance, Scottish ophthalmologist L.T. Lim found these reports and brought them to the ophthalmic literature in 2010. 4 However, Lim’s conclusion that OVAE had indeed occurred was challenged by retina specialists, 8 and a subsequent article (in German) by Gamulescu et al received little notice. 18
In 2012, we learned of the first case of suspected OVAE in which a patient died in the operating room, apparently caused by accidental suprachoroidal air infusion (Figure 1). When opinions regarding the cause of death were inconclusive, we investigated the possibility of OVAE by performing vitrectomy in banked, enucleated eyes, intentionally creating choroidal detachment by pulling the air infusion cannula slightly outward, simulating cannula slippage. When these eyes were then partially submerged in a water bath, air loudly bubbled out only after Serrefine clamps on the vortex vein stumps were removed. 5 These were the first experimental investigations of OVAE, providing striking visual evidence for its occurrence (Figure 4 and Supplemental Video).
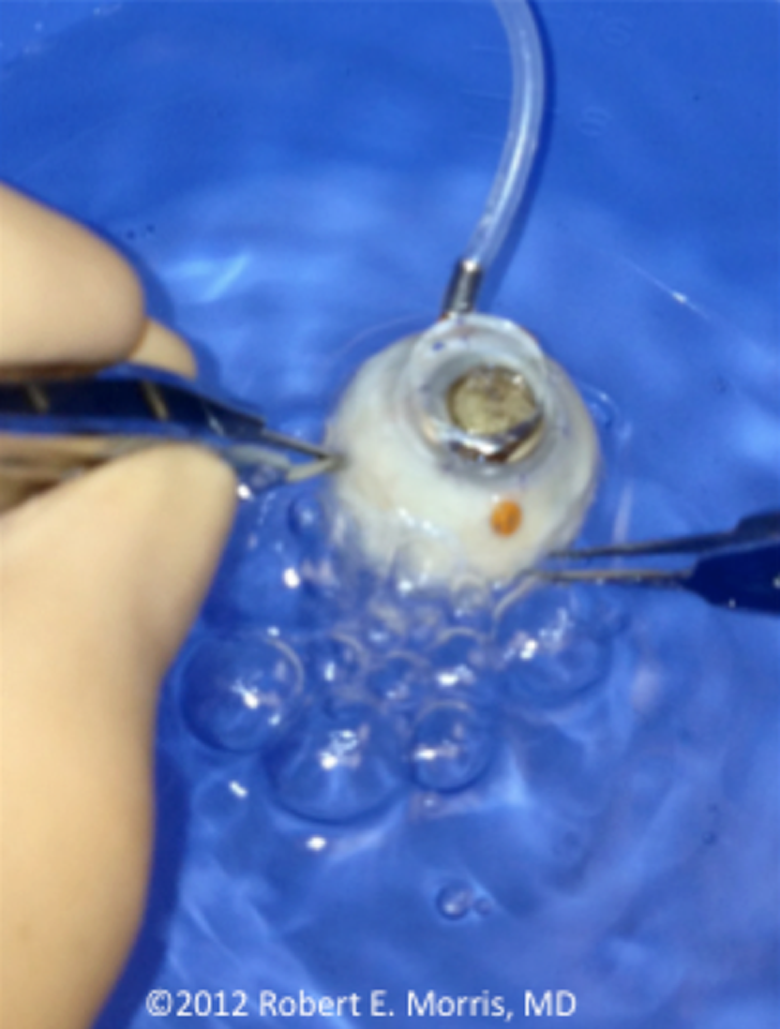
Image from a video of a banked eye in which air loudly bubbles from torn vortex veins after intentional suprachoroidal air infusion caused choroidal detachment (see Supplemental Video). (Image courtesy of Retina Specialists of Alabama, LLC.)
After the 2013 publication of our experiments, 3 additional clinical OVAE reports were published in 2014 from South Africa, Taiwan, and Korea. 7,11,15 In these reports, 2 patients died. A third patient nearly died after cardiac arrest, defibrillation, and 87 minutes of unsuccessful cardiopulmonary resuscitation (CPR). However, emergency extracorporeal life support (ECLS—a portable, rapidly deployable form of cardiopulmonary bypass) 19 was established during CPR through cannulation of the common femoral artery and vein, and normal cardiac rhythm returned within 6 minutes of its employment (Case 5). 15
In a 2016 experiment, Gayer and colleagues produced fatal, autopsy-proven OVAE in a porcine model. 12 The animal died within 7 minutes of beginning air infusion. Very importantly, both the Morris in vitro and the Gayer in vivo experiments concluded that fatal air entrainment could occur in less than 1 minute during vitrectomy air infusion. 5,12 This rate varies with infusion pressure, 20 which may also determine the number of torn vortex veins entraining air. (Clinical corroboration is seen in Cases 8 and 11, in which a precipitous drop in ETCO2 occurred within 1 minute of increasing infusion pressure).
In a 2017 letter to the editor commenting on the Gayer experiment, Blodi reported a (1994) fatal complication of vitrectomy which, aided by the Gayer et al article, he now recognized as OVAE. He had learned of this case in the course of litigation review 2 decades earlier (details provided by personal communications). 13
Also in 2017, Kasi et al reported an intraoperatively fatal OVAE case occurring during a 2015 combined vitrectomy/scleral buckle repair of rhegmatogenous retinal detachment—the first-ever formal case report of OVAE from the United States, 17 4 previous cases having gone unreported. Finally, concurrent with this review, Morris, Boyd et al report herein 5 previously unknown OVAE cases that occurred between 1990 and 2015. 10
OVAE Prevention and Treatment
Initial reports of life-threatening air embolization through the eye were shocking and were questioned because of the “small diameter of choroidal veins.” 8 But the vortex veins can be relatively large (Figure 5) with thin walls that can expand substantially under pressure. And because of pressurized infusion and proximity to the heart, OVAE may be the most lethal of all forms of accidental surgical VAE (Figure 6). 20,21 Air entrainment was so rapid and severe that 9 of 13 OVAE patients (69%) died despite intensive resuscitative efforts, and 8 of the 9 deaths (89%) occurred the day of vitrectomy.
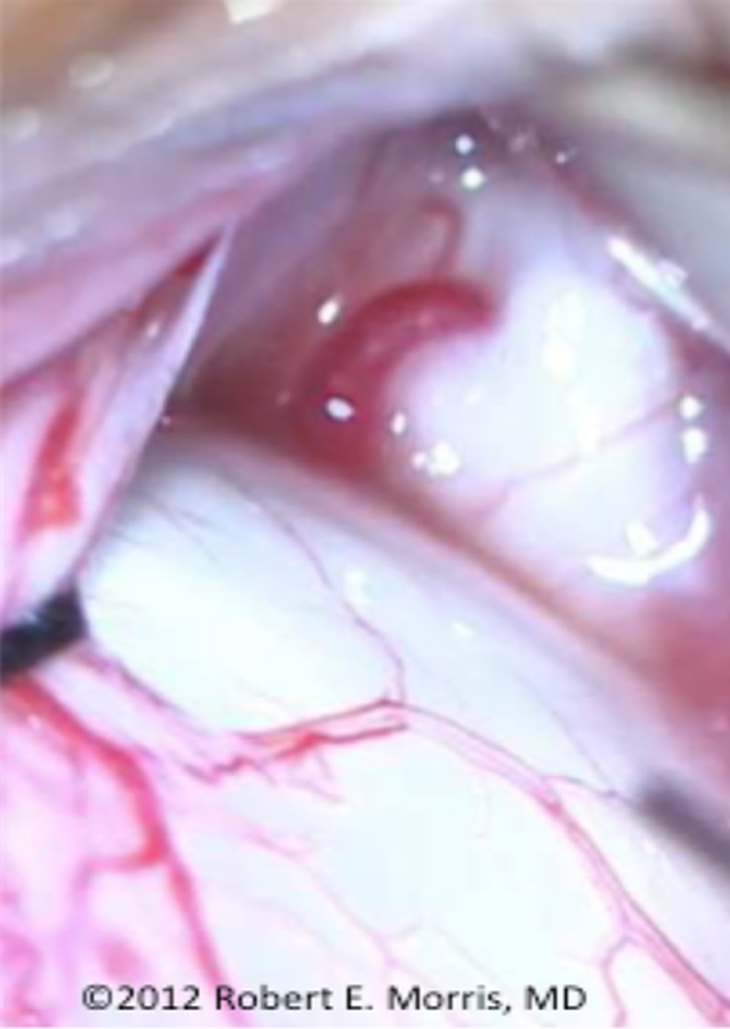
Human vortex vein exiting the posterior sclera, conjunctiva retracted intraoperatively. Note the large size of the perfused vortex vein relative to adjacent episcleral vessels. (Reprinted with permission from the British Journal of Ophthalmology. 5 )
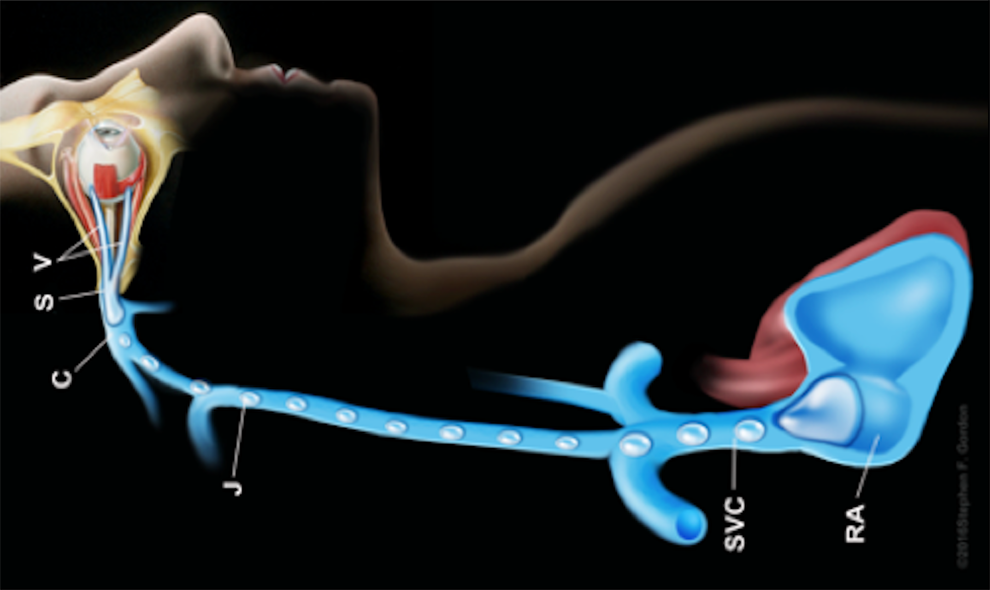
Artist Stephen Gordon’s depiction of noncontinuous but potentially fatal air emboli (infusion pressures typically ≤ 60 Hg) leaving the eye through vortex veins and eventually entering the heart through the systemic venous circulation. CS indicates cavernous sinus; J, jugular vein; RA, right atrium; RV, right ventricle; SVC, superior vena cava; V, vortex vein. Some vortex veins drain into the superior orbital (ophthalmic) vein and then into the cavernous sinus. Other vortex veins drain into the inferior orbital (ophthalmic) vein and then into the pterygoid plexus. (Reprinted with permission from the artist Stephen F. Gordon ©2018.)
The prospect of such rapid entrainment requires immediate OVAE detection and cessation of air infusion as the most important steps to avoid a fatal outcome. In fact, routinely employing prevention strategies is the optimal form of OVAE management. 20
However, dating back to the first report in 2005, only 1 of 6 known OVAE cases (Case 4) had been directly reported in the ophthalmic literature before 2017. 7 Consequently, most United States vitreoretinal surgeons we polled in January 2017 remained unaware of OVAE and the need to initiate an adequate prevention strategy. Better dissemination of information regarding the OVAE risk is of obvious importance and was the impetus for this review.
During general anesthesia, the ETCO2 parameter is recognized as the earliest reliable, routinely monitored clinical indicator of life-threatening VAE. 12,19 It immediately reflects reduced blood flow to the lungs caused by air lock in the right heart during OVAE. But a precipitous ETCO2 decrease is not as easily noticed during regional block anesthesia, commonly used in vitreoretinal surgery, since expired air is not completely captured (Supplemental Report 2).
Because of difficulties in detecting OVAE with impending cardiovascular collapse quickly enough to prevent a fatal outcome, the vitreoretinal surgeon should immediately recognize accidental suprachoroidal infusion/choroidal detachment and stop air infusion before OVAE can even commence. 5,12 The opposite reaction—increasing air infusion pressure, presumably to oppose suspected suprachoroidal hemorrhage—was pursued in 3 of the 4 cases (6, 8, and 11) in which choroidal detachment developed intraoperatively, and it was invariably fatal. 11,17
OVAE due to suprachoroidal air infusion can be prevented by a (vitrectomy team) “Time Out” immediately before air infusion is initiated, to confirm the infusion cannula tip remains normally positioned in the vitreous cavity. Here is a typical conversation, involving 23 total words:
Infusion confirmed.
Air infusion on.
Air infusion on at 30.
Air approaching (watching air descend in the line).
Air in the eye (watching air immediately bubbling into the vitreous cavity).
Understand, air in the eye.
To avoid iatrogenic injury to the eye, it has always been the vitreoretinal surgeon’s responsibility to monitor the infusion cannula position throughout a vitrectomy operation. We now know that this same duty ensures the patient’s survival during air infusion. Especially when the eye is moved about in combined vitrectomy/scleral buckling procedures (Case 8, 9, 11, and possibly Case 1), secure suturing or close monitoring of the air infusion cannula is essential to prevent OVAE. 3,5,17
In contrast to the insidious nature of cannula slippage, the particularly high OVAE risk with large choroidal wounds (from lacerating injury, melanoma resection, or intentional choroidotomy) can be anticipated, planned for, and prevented. 6,7,22 Ideally air infusion should be completely avoided in such cases, but if air infusion is anticipated in the presence of a large choroidal wound, general anesthesia with close ETCO2 monitoring and a precordial Doppler monitor are advisable. 23,24
When OVAE prevention fails, cardiac arrest is likely (10 of 13 cases, 77%). But only 1 of the 10 OVAE arrests was successfully resuscitated with standard CPR, and that patient died within 4 weeks of embolic complications. 6 Thus, even as CPR is begun, the majority of OVAE patients could reasonably be considered for emergency extracorporeal life support via cannulation of the femoral artery and vein. 19 The sole survivor of OVAE cardiac arrest received ECLS after 87 minutes of failed CPR (Case 5). 15
If a patient survives the immediate OVAE event, pulmonary inflammation, platelet aggregation, and increased microvascular permeability can still lead postoperatively to (fatal) acute respiratory distress syndrome (ARDS, Supplemental Report 2). 20 The 55-year-old patient in Case 13 apparently died from ARDS. Close monitoring of each patient should continue postoperatively, with expert pulmonary and cardiovascular expertise available, ideally including emergency ECLS. 25,26
If an OVAE death occurs, consultation with a forensic pathologist should be quickly sought to perform postmortem imaging; to perform a prompt forensic autopsy specifically to detect and document air embolization; and to rule out other causes of death (Supplemental Report 3). 7,27
Conclusion
Since 2005, OVAE has occurred in at least 8 countries on 5 continents. 3,6,7,11,13,15 -17 Armed with knowledge accumulated over this period, during which 9 known patients have died from OVAE, vitreoretinal surgeons, anesthesia providers, and their hosting institutions can adopt measures to safeguard future patients from the OVAE risk. To maintain sensitivity of vitrectomy teams to this rare but usually fatal complication, a downloadable OVAE poster (Figure 7) suitable for an annual period of display in vitrectomy operating suites is included in the supplemental materials (Supplemental Poster).
Ocular venous air embolism poster. (Reprinted with permission from the artist Stephen F. Gordon ©2018.)
Supplemental Material
Supplemental_Report_1 - Ocular Venous Air Embolism (OVAE): A Review
Supplemental_Report_1 for Ocular Venous Air Embolism (OVAE): A Review by Robert E. Morris, Gwendolyn L. Boyd, Mathew R. Sapp, Matthew H. Oltmanns, Ferenc Kuhn, and Maurice S. Albin in Journal of VitreoRetinal Diseases
Supplemental Material
Supplemental_Report_2 - Ocular Venous Air Embolism (OVAE): A Review
Supplemental_Report_2 for Ocular Venous Air Embolism (OVAE): A Review by Robert E. Morris, Gwendolyn L. Boyd, Mathew R. Sapp, Matthew H. Oltmanns, Ferenc Kuhn, and Maurice S. Albin in Journal of VitreoRetinal Diseases
Supplemental Material
Supplemental_Report_3 - Ocular Venous Air Embolism (OVAE): A Review
Supplemental_Report_3 for Ocular Venous Air Embolism (OVAE): A Review by Robert E. Morris, Gwendolyn L. Boyd, Mathew R. Sapp, Matthew H. Oltmanns, Ferenc Kuhn, and Maurice S. Albin in Journal of VitreoRetinal Diseases
Footnotes
Acknowledgments
The authors thank the UAB Callahan Eye Hospital and the Alabama Eye Bank for providing facilities and tissue that enabled the first experimental confirmation of suprachoroidal air infusion as a source of life-threatening OVAE, 5 and for their encouragement of our continuing OVAE investigations.
The authors gratefully acknowledge Ms Jessica Haynes, Ms Stephanie Hill, Mrs Christina Sullivan, Mr DeWayne Conn, and Ms Alanna Weems for their important research and clerical assistance. They also thank Mr Stephen Gordon for his masterful illustrations of the OVAE complication; Christopher Blodi, MD, for many valuable suggestions upon review; Niels Hauff, MD, for his description of ETCO2 monitoring and ARDS in the supplemental material, and his review of the article; Linda Liebenberg, MMedPath (Foren), for her description of optimal postmortem autopsy considerations in the supplemental materials; Stephanie Casterline, CRNA, for providing the information in Case 13 as an educational, life-saving memorial to her brother; Silvio Litovsky, MD, for expert pathology consultation; and C. Diane Scharper for graceful grammar and style revisions.
This report is dedicated to the memory of Maurice S. Albin, MD, who first elucidated the threat of air embolization in modern surgery; Robert Machemer, MD, the inventor of pars plans vitrectomy and revered preceptor to the first author; and to the memory of those individuals, known and unknown, who have died from OVAE.
Authors’ Note
None of the authors were surgeons in any of the cases included in this article.
Ethical Approval
This study was approved by the UAB Institutional Review Board for Human Use and is in accordance with the Declaration of Helsinki.
Statement of Informed Consent
No informed consent was necessary because of the retrospective nature of the study, and because all data collection and analysis were performed without patient identifiers.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was partially funded by the Helen Keller Foundation for Research and Education, through a grant from the Pete Hanna Charitable Foundation.
Supplemental Material
Supplemental material is available online with this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
