Abstract
Objective
Patients with coronavirus disease 2019 (COVID-19) may present with smell/taste dysfunctions in addition to the most frequent symptoms (fever, cough, and shortness of breath) or as the first symptom or even the only symptom. There is paucity of documentation of prevalence and characteristics of smell/taste dysfunction in COVID-19 in sub-Saharan Africa. The aim of this study was to determine the prevalence of smell/taste symptoms in our setting to institute local evidence-based practice.
Study Design
Cross-sectional study.
Setting
COVID-19 treatment centers in Uganda.
Methods
Patients hospitalized for COVID-19 at 3 treatment sites from November 2020 to March 2021 were recruited. Following written informed consent, their demographics, comorbidities, and smell/taste symptoms data were collected using a questionnaire.
Results
Of 614 patients recruited, 409 (63.8%) had mild symptoms and 232 (36.2%) had moderate to severe symptoms; 64.3% were male, and the mean age was 48.6 ± 15.51 years. In total, 23.1% were health responders and 12.2% had contact with a positive case. Smell and taste impairment was seen in 425 (66.3%) patients, second to cough (71.6%). Smell and taste impairment was seen in 162 (38.1%) as the first symptom, in 128 (30%) as the only symptom, and significantly more in those with mild COVID-19 symptoms (P < .001).
Conclusion
COVID-19 manifests with various symptoms, including impairment of smell and taste. This study shows that smell and taste impairment is common and can be the first and only symptom in less severe COVID-19 infections. Therefore, inclusion in the Ministry of Health guidelines is strongly recommended.
Infection with coronavirus disease 2019 (COVID-19), caused by severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) virus, is a pandemic that started in early December 2019 in Wuhan, China. 1 As of December 2021, over 269 million people have been affected globally, with 5.3 million deaths. The hardest hit country in Africa is South Africa, followed by Tunisia. Uganda ranks 12th in Africa and is the second most-hit country after Kenya in East Africa. 2 The daily update from Uganda Ministry of Health as of December 10, 2021, showed that Uganda had 127,796 cases with 3263 deaths. 3
The presentation of COVID-19 varies from mild to severe forms, with the largest number of patients being asymptomatic. As of mid-September 2020, more severe forms were seen in patients aged 60 years and older and those with comorbidities such as cardiovascular diseases and diabetes mellitus. 4 The commonest symptoms of COVID-19 that were reported both globally and locally were fever, cough, dyspnea, headache, and myalgias.4-6 These symptoms are adequately screened for in the current National Guidelines for Management of COVID-19 from the Ministry of Health of Uganda in addition to history of recent travel out of the country or contact with a COVID-19–positive case. 7 However, atypical symptoms like dysfunction of the senses of smell and taste were reported in several studies as presentations of COVID-19.8-11 Unfortunately, patients who had loss of smell and loss of taste were not stipulated as high-risk patients in the Ugandan National Guidelines for COVID-19 triage protocol. These patients would unknowingly be vectors of the disease in the community, as they would otherwise be “asymptomatic.”
Prevalence of loss of smell or taste ranged from of 5% to 98%, with a paucity of reports from sub-Saharan Africa. 8 Studies from the Western world and Asia reported that some patients with COVID-19 had loss of smell and taste as the only symptom and others as the first symptom of COVID-19.9,10,12,13 Most patients in 1 study done in Italy had complete loss of sense of smell and taste, with more than half having it before diagnosis of COVID-19, and 13% had it as their first symptom. 9 Many suggested that a sudden severe new-onset loss of smell or taste should be suspected as COVID-19 until proven otherwise. 10 In fact, a study in France to identify COVID-19 showed the specificity of loss of smell and taste as 90.3% and the positive predictive value as 78.5%. 14 A household study tracing index COVID-19 cases with household members found that loss of smell/taste had the highest positive predictive value (83%) among household contacts. 15 We therefore sought to describe the smell and taste symptoms of patients with COVID-19 in Uganda.
Methods
This cross-sectional study was prospectively carried out in 3 largest sites designated by the Ministry of Health (MoH) of Uganda as COVID-19 treatment centers: Mulago National Specialist Hospital and Namboole Treatment Centre in Kampala and Entebbe Regional Referral Hospital in Entebbe. At the time of the study, these centers admitted every person who had a laboratory confirmation of COVID-19 by polymerase chain reaction (PCR) testing, even when asymptomatic (eg, the contacts of symptomatic COVID-19–positive patients who likewise tested positive for COVID-19 were admitted as required by the MoH guidelines). Similarly, some asymptomatic patients in the triage protocol (eg, cross-country truck drivers) had mandatory COVID-19 testing because of recent travel out of the country. Other individuals opted to have COVID-19 testing while in the hospital for other reasons. These were quarantined when found positive for COVID-19. The testing was done by both private and public laboratories with a central reporting system to the MoH. The study was carried out in accordance with the principles of Declaration of Helsinki. Ethical approval was obtained from the Mulago Clinical Research Ethics Committee MHREC 1955 (decision no. 05/22), as well as approval from the Ministry of Health (scientific research no. 2020-04-29T15_22_34). Written informed consent was obtained from the patients.
The study was done between November 2020 and March 2021, during the first epidemic peak in Uganda, before different variants (Delta and Omicron) and before any vaccinations were available locally. An interviewer-administered questionnaire (for details, see Supplemental Data S1_crf in the online version of the article) was answered by all documented PCR-tested COVID-19–positive patients admitted on the general wards and high-dependency units (HDUs) who consented to the study. The HDUs had facilities for nasal prong or mask administration of piped oxygen and more health care workers compared to the general wards. If the patient’s condition deteriorated, they would then be stepped up to the intensive care unit (ICU), where intubation and mechanical ventilation facilities were available. In this study, those with mental illness or critically ill requiring intubation and intensive care were excluded. Variables collected in the questionnaire included age, sex, comorbidities, risks factors for COVID-19 (ie, health care responders or close contact with a confirmed COVID-19–positive patient), symptoms (ie, fever, cough, dyspnea, fatigue or muscle pain, diarrhea or nausea). Outcome variables were the specific information about presences of smell/taste symptoms (isolated or combined), including its timing of presentation. The study patients were later grouped as group A (those with mild symptoms, admitted to the general ward) and B (moderate to severe symptoms, admitted to the HDU) for comparison of patient characteristics between the groups. The MoH national guidelines classifies mild cases as asymptomatic cases or those with uncomplicated upper respiratory tract viral infection. Moderate cases present with pneumonia without the need for oxygen, while severe cases require hospital-level interventions such as oxygen therapy. 3
Statistical Analysis
The questions were coded and the data entered using the EpiData version 3.1 (EpiData Software). The data were exported to Statistical Package for Social Scientists (SPSS) version 23 (SPSS, Inc), where data analysis was done. Participant characteristics were expressed as categorical and/or continuous variables. Continuous variables were expressed as means and standard deviations, while categorical data were expressed as frequencies with their respective proportions. The main outcome of this study was prevalence of smell and taste symptoms, which was presented as frequencies and proportions. We compared the different patient characteristics between the groups that had mild symptoms and those that had moderate to severe symptoms as a secondary objective.
Results
Overall, a total of 614 COVID-19–positive patients were described; 412 (64.3%) were male, and the mean age was 48.6 ± 15.51 years. Specific risks included being a health responder (23.1%) and being in contact with a confirmed case (12.2%). Comorbidities were present in 54% and included hypertension (29.1%), diabetes (20.6%), respiratory disease (2.8%), and sinusitis and allergy (1.1%). The common symptoms that were manifested were cough, followed by smell and taste impairment and then fever. Smell and taste impairment was seen in 425 (66.3%). It was the first symptom in 162 (38.1%) and the only symptom in 128 (30%). Symptom duration was 10.8 ± 11.8 days. Baseline characteristics are shown in Table 1 .
Baseline Characteristics of Patients With COVID-19 Infection.

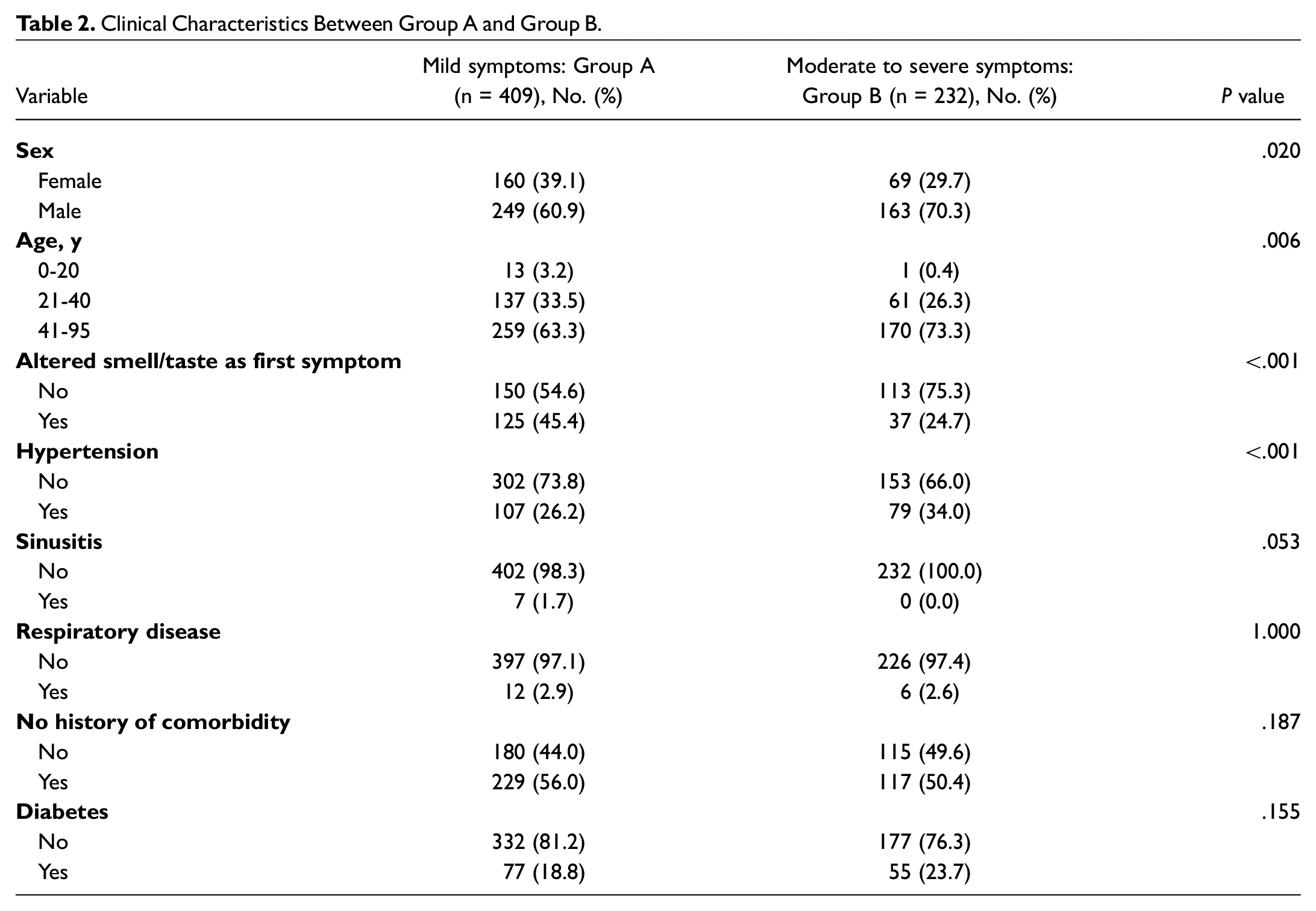
With comparison between group A (those with mild symptoms) and group B (those with moderate to severe symptoms) demonstrated in Table 2 , significant differences were found with more males being affected, the largest proportions being in those aged above 40 years, and having hypertension. The smell/taste alteration as the first symptom was more in group A at 45.5% compared with group B at 24.7% (P < .001). There was no significant difference with sinusitis/respiratory disease, diabetes, or history of comorbidity.
Clinical Characteristics Between Group A and Group B.
Discussion
Our study shows a high prevalence of smell and taste alteration in COVID-19 patients at 66.3% and mostly in those with mild disease. It was the second most common symptom, with 38% having it as their first symptom and 30% having it as their only symptom. It should be noted that every patient who tested positive was quarantined due to institutional policy at that time.
Tong et al, 16 in their systematic review and meta-analysis, found the prevalence of smell and taste impairment in COVID-19 was 53% and 44%, respectively. Saniasiaya et al 17 also found a pooled prevalence of 48% of taste disorders in their meta-analysis. Although our prevalence of smell and taste impairment is higher than their global trend, there was no representation from Africa for regional comparison of our study findings. Variations of presentation are noted among geographical regions. Lechien et al 12 showed a prevalence of 85.6% among 417 COVID-19 patients in a multicenter study done in Europe. In the United States, a prevalence of 73% was reported. 18 In sharp contrast, low prevalence was found in Asia, with frequencies of 30% in Korea, 14.8% in India, 8.9% in Japan, and 5.6% in China.19-22 The contrast between Europe and the Americas and Asia was thought to be due to ethnic differences between the number of angiotensin-converting enzyme (ACE) receptors in the olfactory epithelium of whites and Asians. 23 As there is scarce documentation involving the black race, our study presents an opportunity to have data from Africa that may help elucidate differences in racial and ethnic symptoms on presentation.
Qualitative symptoms of smell (phantosmia and parosmia) or taste (phantogeusia and parageusia) have also been shown to be prevalent especially in the wake of long COVID-19. 24 Parosmia was prevalent in 40% of those with previous COVID-19, 6 months after a negative COVID-19 PCR test. 24 Dysgeusia had a pooled prevalence of 41.3%. 17 Regrettably, we did not assess these symptoms in our study, a feature we hope to include in further studies. In addition, objective assessment of the smell and taste dysfunctions yielded higher outcomes than subjective tests.17,25 At the time of our study, performing objective chemosensory testing was perceived to increase risk of viral transmission to the investigators, and therefore it was felt prudent to evaluate chemosensory deficits using subjective assessments. However, objective methods will be assessed in future studies to note any differences between these methods. Paderno et al 26 reported resolution of smell and taste disorders in 87% and 82%, respectively, by 30 days. Risk factors for later resolution included presence of nasal congestion, grade of symptoms, and female sex. Kattar et al 27 suggested olfactory retraining therapy for those with unresolved olfactory symptoms for improvement of their chemosensory deficits. We focused more on the magnitude of the smell/taste disorders as a baseline for future studies including recovery rates and interventions to improve quality of life after COVID-19.
Several studies agree with ours concerning smell and taste alterations manifesting more in mild COVID-19.12,13,28-31 Patients who tested positive for COVID-19 but did not have smell dysfunction were 5 times more likely to be hospitalized, 7 times more likely to be intubated, and 7 times more likely to die than those who had smell dysfunction in 1 meta-analysis. 28 This was proposed for risk stratification. Paderno et al 29 studied 508 patients and found a prevalence of smell disorders in 93.1% in the quarantined patients vs 73.4% in the hospitalized patients. Bianco et al 30 in Italy compared 50 COVID-19–positive patients (hospitalized and nonhospitalized). Nonhospitalized patients had more mild symptoms like altered smell, sore throat, and rhinorrhea while hospitalized patients had more common symptoms like dyspnea, fatigue, fever, and cough. A significant difference was seen between the prevalence of smell alteration in nonhospitalized patients at 81.8% compared with 28.5% in the hospitalized patients. Yan et al 31 reported that hospitalized patients were 10 times less likely to report smell alterations. Izquierdo-Domínguez et al 13 reported smell and taste dysfunction more in younger (<60 years old) and nonhospitalized patients. Lechien et al 12 also reported a similar trend in those with mild to moderate COVID-19.
Our COVID-19–positive patients had smell and taste loss as their first symptom in 38%. This is higher than most studies. Dell’Era et al 9 noted a 9% prevalence, although 54% generally had loss of smell before being diagnosed. In a multicenter study by Izquierdo-Domínguez et al, 13 18% to 19% patients reported loss of smell and/or taste as the first symptom of COVID-19. Parderno et al 26 reported 14% had olfactory dysfunction as their first symptom, with 16% reporting gustatory disorders as their first symptom. As regards smell and taste alteration as the only symptom, our study still has higher proportions than Cho et al, 10 who found that 4 of 39 of their COVID-19 patients had smell impairment as their only symptom.
As smell and taste loss has a high positive predictive value of COVID-19, we join several otolaryngology societies in sounding the alarm concerning sudden loss of smell and taste as a symptom for COVID-19.14,15 With our study findings, we therefore recommend inclusion of loss of smell and taste as a COVID-19 defining symptom that should be part of triage. As this study was done prospectively, we were able to collect most variables for our study. However, due to the subjective nature of the questionnaire, there may have been recall bias in terms of timing of the smell and taste symptoms. Moreover, for safety concerns, we did not carry out any endoscopic examinations of the nose or any objective smell or taste tests. Using our findings should therefore be done with caution. Our questionnaire also did not inquire about any past problems with smell or taste to rule out any preexisting conditions that could be potential confounders of this study. This would have enriched our study findings. Likewise, we made no inquiry of qualitative symptoms of smell at inception of the study. This presents an avenue for future studies that would include qualitative symptoms and longer follow-up, especially in the wake of long COVID-19. Although this study prioritized the prevalence of the sensory dysfunctions as a symptom for triage due to its potential contribution to prevent further spread of the COVID-19, a future study evaluating the rates of recovery of sense of smell and taste in our setting would inform local ENT practice to improve quality of life. With the ever-changing trends of the COVID-19 presentation, as witnessed by the recent drop in the prevalence of smell and taste dysfunctions during the Omicron outbreak, it is still important to document the findings in the first wave in case any future patterns resurface.
Conclusion
The proportion of patients with smell/taste symptoms due to COVID-19 during the first wave in our country is high. A significant number presented with smell and taste loss as the first or only symptom. We strongly recommend the listing of smell/taste dysfunction as a high-risk symptom for COVID-19 infection screening. Patients are encouraged to go for PCR testing and isolate according to MoH guidelines, to curb further spread of COVID-19.
