Abstract
Objective
Tracheostomies have been performed in patients with prolonged intubation due to COVID-19. Understanding outcomes in different populations is crucial to tackle future epidemics.
Study Design
Prospective cohort study.
Setting
Tertiary academic medical center in New York City.
Methods
A prospectively collected database of patients with COVID-19 undergoing open tracheostomy between March 2020 and April 2020 was reviewed. Primary endpoints were weaning from the ventilator and from sedation and time to decannulation.
Results
Sixty-six patients underwent tracheostomy. There were 42 males (64%) with an average age of 62 years (range, 23-91). Patients were intubated for a median time of 26 days prior to tracheostomy (interquartile range [IQR], 23-30). The median time to weaning from ventilatory support after tracheostomy was 18 days (IQR, 10-29). Of those sedated at the time of tracheostomy, the median time to discontinuation of sedation was 5 days (IQR, 3-9). Of patients who survived, 39 (69%) were decannulated. Of those decannulated before discharge (n = 39), the median time to decannulation was 36 days (IQR, 27-49) following tracheostomy. The median time from ventilator liberation to decannulation was 14 days (IQR, 8-22). Thirteen patients (20.0%) had minor bleeding requiring packing. Two patients (3%) had bleeding requiring neck exploration. The all-cause mortality rate was 10.6%. No patients died of procedural causes, and no surgeons acquired COVID-19.
Conclusion
Open tracheostomies were successfully and safely performed at our institution in the peak of the COVID-19 pandemic. The majority of patients were successfully weaned from the ventilator and sedation. Approximately 60% of patients were decannulated prior to hospital discharge.
In 2020, the novel coronavirus (SARS-CoV-2) caused >4 million deaths worldwide, with the United States having >600,000 deaths by June 2021.1,2 While many metropolitan and urban areas experienced a heavy case volume of coronavirus disease 2019 (COVID-19), New York City (NYC) was the early epicenter of the pandemic.3,4 With many patients experiencing acute respiratory distress syndrome and pneumonia, the rates of invasive mechanical ventilation and intubation were exceptionally high.
Due to the high volume of patients requiring mechanical ventilation in NYC, tracheostomy offered an opportunity to relieve resources and facilitate ventilator weaning, including personnel to manage ventilation and sedation, as well as equipment associated with endotracheal management. 5 Additionally, benefits of tracheostomy include improved comfort and reduced need of sedation, paralytic support, and subsequent intensive care unit (ICU)–level support, which reduced the risk of COVID-19 transmission among health care workers. 6 Therefore, percutaneous and open tracheostomies were common in many hospitals in NYC, and current data show that tracheostomy in patients with COVID-19 was safe and had high utility. Several series from other facilities in NYC demonstrated relatively consistent outcomes in several tracheostomy endpoints.5,7-9
In this study, we highlight the tracheostomy outcomes of the Department of Otolaryngology–Head and Neck Surgery at a major NYC hospital that experienced one of the highest censuses of COVID-19 cases in the United States. Notably, this hospital serves a highly diverse population with various ethnic and racial minorities. When compared with other populations within NYC and nationally, our population had a considerably higher rate of Hispanic and non-White patients. Additionally, the hospital represented in this study is located in a distinct socioeconomic neighborhood of Manhattan. 10 In light of the rise of new variants across the globe, ongoing review and analysis of tracheostomy outcomes in the COVID-19 setting are critical to better prepare for future variants and waves.
Methods
A prospectively established protocol for tracheostomy data within the Department of Otolaryngology–Head and Neck Surgery at a single institution was developed. Patients who had acute respiratory failure and COVID-19 confirmed by reverse transcriptase polymerase chain reaction and who underwent tracheostomy were included. Patients undergoing tracheostomy for other reasons during this period were excluded. The electronic medical record was queried for patients in the database (Epic Systems). Data were collected regarding dates of admission, ventilation initiation and termination, sedation initiation and termination, tracheostomy, and discharge and/or death, as well as information on comorbidities, anticoagulation, and tracheostomy-specific complications.
All tracheostomies conducted in this study involved an open approach. It was hospital protocol to perform tracheostomy after 20 days from admission. This policy was set in place at the onset of the pandemic primarily to protect the health care and surgical teams from viral spread based on the understanding of COVID-19 infectivity at the time. All procedures in this study were conducted at a time when this policy was in place. All procedures were conducted by otolaryngologists at a single institution, all of whom had sufficient personal protective equipment per hospital policy. Personal protective equipment included N95 masks, eye protection in the form of goggles or face shields, and protective gowns, and all procedures were performed in a room with outflow ventilation to prevent recirculation air. Among all procedures, tracheostomy was performed under apneic conditions to minimize viral dissemination into the surrounding air. Specifically, patients were all paralyzed for the procedure and placed in a mandatory vent setting. At the time of entering the airway and exchanging the endotracheal tube for the tracheostomy tube, the ventilator was paused to ensure that the patients did not cough or exhale into the room. Once the tube was placed with a heat-moisture exchanger, ventilation was resumed. All patients in this study received cuffed tracheostomy tubes (Shiley), and placement was conducted in varying environments, such as formal ICUs, operating rooms, and operating rooms converted into ICUs.
For analysis, the primary outcomes were duration of ventilation and duration of tracheostomy (time from procedure to decannulation). Secondary outcomes included duration of sedation and tracheostomy-specific complications. End of ventilatory and positive pressure support was defined as toleration of a tracheostomy collar for >24 hours. Sedation duration was defined as the time of initiation until the patient no longer required intravenous sedating drip medication for >24 hours. Major complications were procedural issues occurring during the tracheostomy or any complication requiring an unplanned return to the operating room. Minor complications included any bleeding requiring packing. We also noted rates of cuff leak, aspiration events, pneumothoraxes, mucous plugging, and wound cellulitis/infection of the tracheostomy site. These events were derived from clinical documentation, such as progress notes and nursing reports. Time to outcome events was estimated with the Kaplan-Meier method, and medians were compared via the log-rank test. Statistical significance was evaluated at the 0.05 alpha level. Analyses were performed in R Core Team (survival and tidyverse version 3.5.3). This study was approved by the Columbia University Irving Medical Center Institutional Review Board.
Results
Sixty-six patients between April and May 2020 underwent tracheostomy at a single institution. This was at the time when the original strain was the primary cause of infection. Table 1 illustrates the demographic variables and outcomes of this cohort. The majority of patients were male (n = 42, 64%). The average age of patients in this study was 62 years (SD, 13.7; interquartile range [IQR], 58-73). Many patients (49.5%) did not have a documented race. White patients represented approximately 20% of the cohort, and Black patients represented approximately 17%. Approximately 48% of our patients identified as Hispanic or Latino.
Demographic Characteristics and Outcomes for 66 Patients at a Single Hospital.

Abbreviation: IQR, interquartile range.
Within our cohort, patients were intubated for a median time of 26 days prior to tracheostomy (IQR, 23-30). Of the patients in our cohort, 77% (n = 18) were successfully weaned from ventilation. The median time to weaning from ventilatory support after tracheostomy was 18 days (IQR, 10-29). Of those sedated at the time of tracheostomy (n = 45, 68%), the median time to discontinuation of sedation was 5 days (IQR, 3-9).
Of the 59 patients who survived, 39 (66%) were decannulated. Of those decannulated (n = 39) before discharge, the median time to decannulation was 36 days following tracheostomy (IQR, 27-49). In Kaplan-Meier analysis ( Figure 1 ), which censored those who were discharged prior to decannulation and those who died, the median time to decannulation was 43 days (95% CI, 36-52 days). Additional analysis was conducted via Kaplan-Meier analysis to examine the time from tracheostomy to sedation weaning after censoring those who died ( Figure 2 ), in which the median time was 11 days (95% CI, 9-26). The median time from ventilator liberation to decannulation was 14 days (IQR, 7-18).
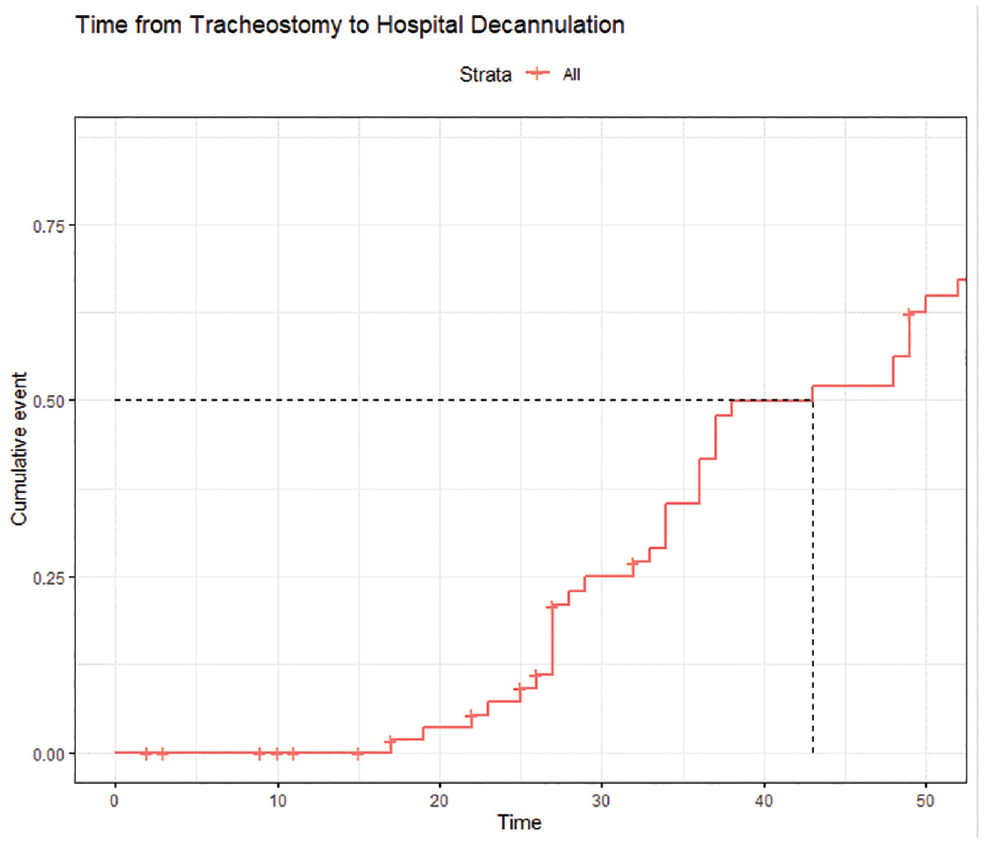
Time from tracheostomy to hospital decannulation. Patients who were discharged prior to decannulation or died were censored.
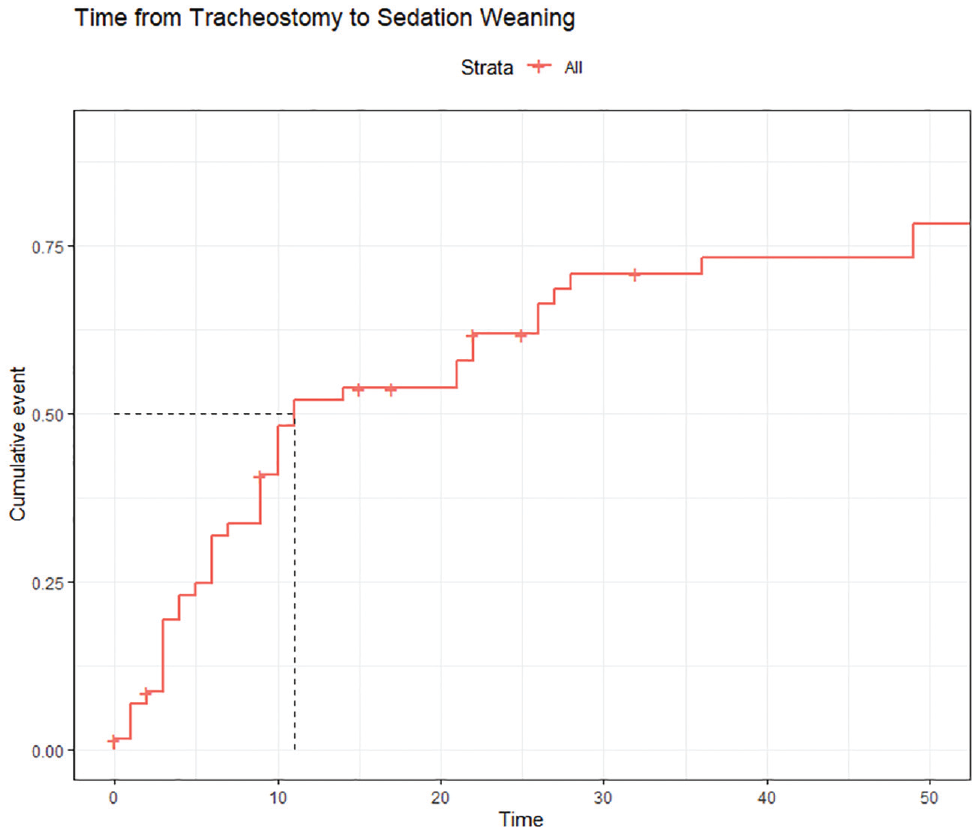
Time from tracheostomy to weaning from sedation. Patients who were weaned off sedation prior to tracheostomy were excluded, and those who died prior to sedation weaning were censored (removed from the analysis without achieving the outcome of interest).
Thirteen patients (20.0%) had minor bleeding requiring packing. For other minor complications, 8 (12%) patients experienced a cuff leak; 3 (5%), an aspiration event; 5 (8%), a pneumothorax; 4 (6%), a mucous plug; and 6 (9%), wound cellulitis/infection of the tracheostomy site. Two patients (3.0%) had bleeding requiring neck exploration. No other major complications were reported. The all-cause mortality rate was 10.6% (n = 7). No patients died of procedural causes. Additionally, no surgeons who completed the tracheostomies experienced COVID-19–related illness. All surgeons in this analysis had proper protective equipment.
Discussion
In this study, we analyze and report the outcomes of 66 patients undergoing tracheostomy at a major medical center in NYC. Notably, this center experienced a particular high burden in COVID-19 cases during the peak of the pandemic and treats an ethnically diverse population with a little less than half of patients identifying as Hispanic or Latino. This differs notably from other tracheostomy studies published during the pandemic; however, this did not dramatically affect practice patterns for tracheostomies in our cohort. By institutional protocol, we performed tracheostomies after 20 days. Systematic reviews of early-pandemic tracheostomies show similar utilization of late tracheostomy protocols, with approximately 70% being performed during the pandemic after 2 weeks. 11 In this cohort, most patients were in their sixth decade of life and exhibited a high body mass index. Finally, most patients were male in our cohort, which is similar to other published studies of tracheostomy outcomes.5,8,12-17 Table 2 summarizes some of the published data on tracheostomies in the United States, specifically regarding decannulation rates.
Select Published Studies of Tracheostomies for Patients With COVID-19 in the United States.
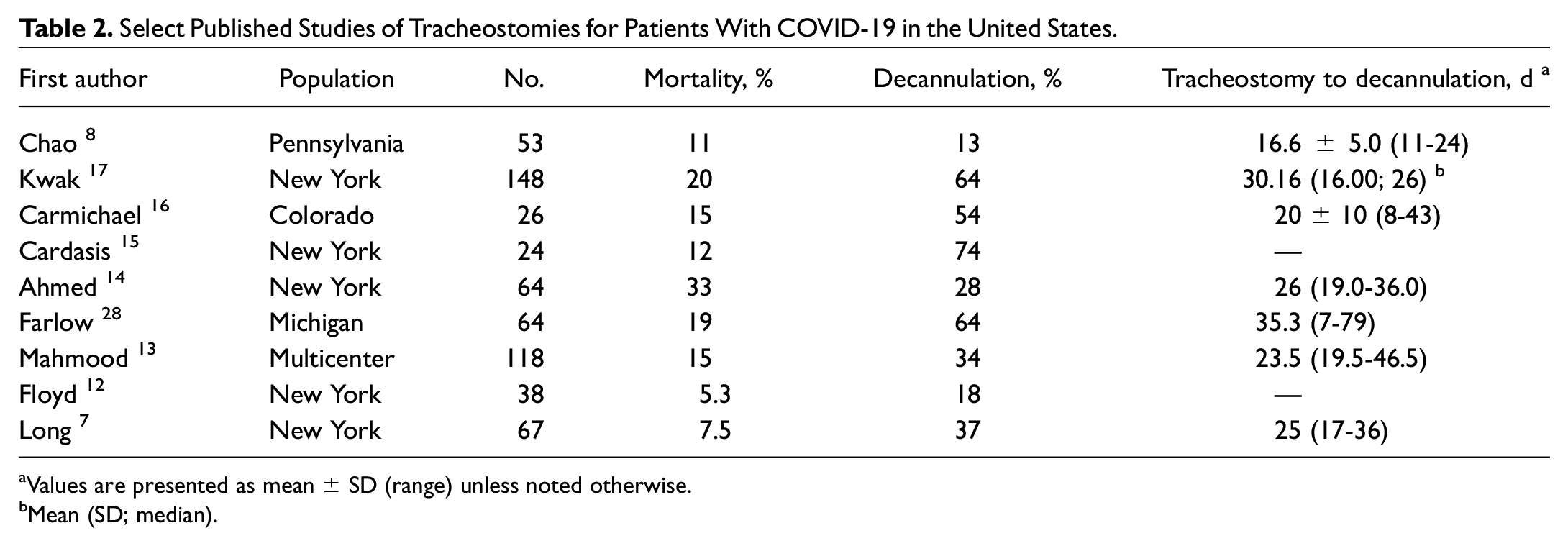
Values are presented as mean ± SD (range) unless noted otherwise.
Mean (SD; median).
Within our population, most patients were decannulated prior to discharge. Our decannulation rate (59%) is similar to that of other hospitals in the United States and showed improvement as compared with some institutions internationally.5-8,14,18-20 However, it is important to acknowledge that a considerable proportion of patients (40%) in our study were not decannulated prior to discharge or death. The median time to hospital decannulation was a little over a month after censoring those who were discharged or had died. This is similar to other series, including one at another major institution in NYC in which the time to decannulation was 32 days, although systematic analyses show an average time closer to 20 days.5,11 However, it is important to acknowledge that outcomes such as subglottic stenosis and other long-term events would be an important metric to examine when examining prolonged intubation and tracheostomy. 21 We will continue to follow the outcomes of this cohort, as understanding its long-term outcomes, especially with a post–20-day protocol, is important in establishing protocols to help combat future COVID-19 variants. Additionally, understanding the changes and impact of different tracheostomy protocols over the course of the pandemic is imperative. Our study focuses on the early pandemic, but later in the course of COVID-19 tracheostomy timing was less stringent, which represents a different patient cohort. A notably new avenue of research regarding post–COVID-19 survivorship includes quality of life and postintubation/intensive care syndromes that may afflict patients many months to years out from their initial presentation. Understanding these syndromes from an otolaryngologic perspective will be important in the years to come and should be scrutinized in addition to mortality rates. 22
The death rate in our cohort was about 10%, which is similar to other studies published in the United States ( Table 2 ). Additionally, several nonpandemic studies of tracheostomies in patients with acute respiratory distress syndrome exhibited mortality rates up to 40% to 50%.23,24 One large study of tracheostomies of patients with COVID-19 in Spain showed a mortality rate as high as 25%. Other studies in NYC demonstrated similar mortality rates to our cohort.5,7,14,17,19,20 Variations in mortality rate are secondary to a number of factors, such as inherent differences in patient characteristics as well as population-level variations in disease burden and facility resources.
No procedure-related complications were noted in our study. Yet, about 20% of patients experienced minor bleeds requiring simple packing, while 3% had bleeding requiring a return to the operating room. Our rate of bleeding is largely within the range of critically ill patients receiving tracheostomies in nonpandemic situations. 18 Finally, no surgeons completing the analyzed procedures experienced a personal COVID-19–related illness during the designated study period, indicating that overall tracheostomy in the setting of COVID-19 disease is safe for both the surgeon and the patient with proper precautions, as established in other major studies. 25 Our protocols for personal protection were similar to other national protocols established during the pandemic and appeared effective for surgeons performing these cases.26,27 However, additional studies should scrutinize other members of the care team as well as posttracheostomy care to provide a comprehensive understanding of risk of COVID-19 infection with tracheostomies.
This study has limitations, primarily in regard to the single-institution nature of the patient cohort. Additionally, it is difficult to account for the variations in the patient’s experience with COVID-19 illness when analyzing in aggregate. Also, while we notably present an ethnically diverse population, stratification analysis was difficult in our cohort, as a considerable number of patients had incomplete definitions of race/ethnicity on their admission forms and documentation.
Conclusion
We present an ethnically diverse population within a neighborhood of Manhattan that experienced a heavy burden of COVID-19 illness during the pandemic. We illustrate that tracheostomies were safe and providers did not contract COVID-19 from procedural exposure. Additionally, a majority of patients were able to be decannulated. Information regarding tracheostomy outcomes will be important in light of new waves and COVID-19 variants that are presenting across the globe. However, future studies following these patients is necessary to fully establish optimal protocols for these patients. Overall, we illustrate that tracheostomies in patients with COVID-19 are safe and should be utilized in light of a new wave.
Author Contributions
Disclosures
Footnotes
This article was presented at the 2021 AAO-HNSF Annual Meeting & OTO Experience; October 2-5, 2021; Los Angeles, California.
