Abstract
Objective
This review aimed to systematically determine the optimal nasal saline regimen for different types of sinonasal diseases.
Data Sources
PubMed, Embase, SCOPUS, Cochrane Library, Web of Science, ClinicalTrials.gov. The last search was on December 6, 2021.
Review Methods
Study selection was done by 2 independent authors. Randomized controlled trials and meta-analyses were included. The effects of nasal saline treatment through various devices, saline tonicities, and buffer statuses were evaluated in patients with allergic and nonallergic rhinitis, acute and chronic rhinosinusitis (CRS), CRS with cystic fibrosis, and postoperative care, including septoplasty/turbinoplasty and endoscopic sinus surgery.
Results
Sixty-nine studies were included: 10 meta-analyses and 59 randomized controlled trials. For allergic rhinitis, large-volume devices (≥60 mL) were effective for treating adults, while low-volume devices (5-59 mL) were effective for children. Isotonic saline was preferred over hypertonic saline due to fewer adverse events. For acute rhinosinusitis, saline irrigation was beneficial in children, but it was an option for adults. Large-volume devices were more effective, especially in the common cold subgroup. For CRS, large-volume devices were effective for adults, but saline drop was the only regimen that had available data in children. Buffered isotonic saline was more tolerable than nonbuffered or hypertonic saline. The data for CRS with cystic fibrosis and nonallergic rhinitis were limited. For postoperative care, buffered isotonic saline delivered by large-volume devices was effective.
Conclusion
Nasal saline treatment is recommended for treating most sinonasal diseases. Optimal delivery methods for each condition should be considered to achieve therapeutic effects of saline treatment.
Intranasal saline treatment is effective, inexpensive, and safe. 1 It mechanically washes out mucus, secretory aggregation, and inflammatory cytokines from the nose. In addition, it promotes mucociliary clearance and helps reduce mucosal edema. Currently, it is recommended as an adjunct therapy for sinonasal diseases, such as rhinosinusitis, rhinitis, and upper respiratory tract infections. 2 It is unclear whether nasal saline treatment benefits all or only specific types of sinonasal diseases. There are several kinds of devices, such as spray, drop, syringe, pot, aerosol, and squeeze bottle. These devices deliver the saline solution into the nose and paranasal sinuses with different volumes and pressure. Different solutions (buffered or nonbuffered saline) and tonicities (hypertonic or isotonic) are used. These factors contribute to the difference in treatment outcomes.
There are limited systematic reviews that determine the optimal device and regimen of nasal saline treatment for different sinonasal diseases. This systematic review aimed to assess the therapeutic effects and safety of nasal saline treatment for sinonasal diseases and identify an optimal delivery method for each sinonasal disease.
Methods
A systematic search was conducted through databases for relevant publications: PubMed, Embase, SCOPUS, Cochrane Database Library, Web of Science, and ClinicalTrials.gov. The date of the last search was December 6, 2021. The search strategy employed combinations of the following Medical Subject Headings keywords: nose, nasal, irrigation, spray, inhalation, atomization, vaporization, saline, and sodium chloride. Randomized controlled trials (RCTs) and meta-analyses of systematic reviews were included. The search terms are provided in the Supplemental File (available online).
Study selection was performed independently by 2 authors using a web-based application for systematic review (covidence.org). Titles and abstracts were screened. Nonrelevant studies and duplicate studies were excluded. Full text of the screened articles was assessed for eligibility. Any disagreement on the study selection process was resolved by discussions until a final consensus was reached. Study selection was based on the following inclusion criteria: (1) RCT or meta-analysis conducted in humans, (2) patients with any sinonasal disease and any age, (3) intranasal saline treatment with any delivery method (tonicity, buffered, pH, temperature, volume, and device), (4) comparison between saline treatment and no-saline treatment or between 2 delivery methods, and (5) any clinical outcomes.
Studies were excluded per the following criteria: (1) systematic reviews without meta-analysis, (2) conference abstracts without complete data, (3) non-English articles, (4) delivery methods unavailable in the market, (5) medicated saline, and (6) comparison between saline and other medications. Data from studies with mixed populations were excluded when not reported separately. The meta-analyses were thoroughly cross-checked for the included studies, duplication, and analyses.
The device was categorized according to the volume of saline delivered into the nose: very low (<5 mL), low (5-59 mL), and large (≥60 mL). 3 Very low-volume devices included spray, drop, and aerosol; low-volume devices, syringe and jet flow; and large-volume devices, pot and squeeze bottles.
Sinonasal diseases were categorized into 3 groups:
Rhinitis: allergic rhinitis (AR) and nonallergic rhinitis (NAR)
Rhinosinusitis: acute rhinosinusitis (ARS), chronic rhinosinusitis (CRS), and chronic rhinosinusitis with cystic fibrosis (CRS-CF)
Postoperative care: after septoplasty/turbinoplasty and endoscopic sinus surgery (ESS)
The effects of saline treatment in each group were evaluated between saline and no-saline treatment, among different devices, between hypertonic and isotonic solution, and between buffered and nonbuffered saline.
Results
A total of 8090 publications were retrieved, of which 69 were included for review. The PRISMA flowchart (Preferred Reporting Items for Systematic Reviews and Meta-analyses) is displayed in Figure 1 . There were 10 meta-analyses and 59 RCTs (1 study by Ural et al 4 assessed 3 patient subgroups). Twenty-three studies assessed nasal saline treatment in patients with rhinitis (23 AR and 0 NAR). Thirty-five studies assessed rhinosinusitis (17 ARS, 17 CRS, and 1 CRS-CF). Thirteen studies assessed postoperative care (2 septoplasty/turbinoplasty and 11 ESS).
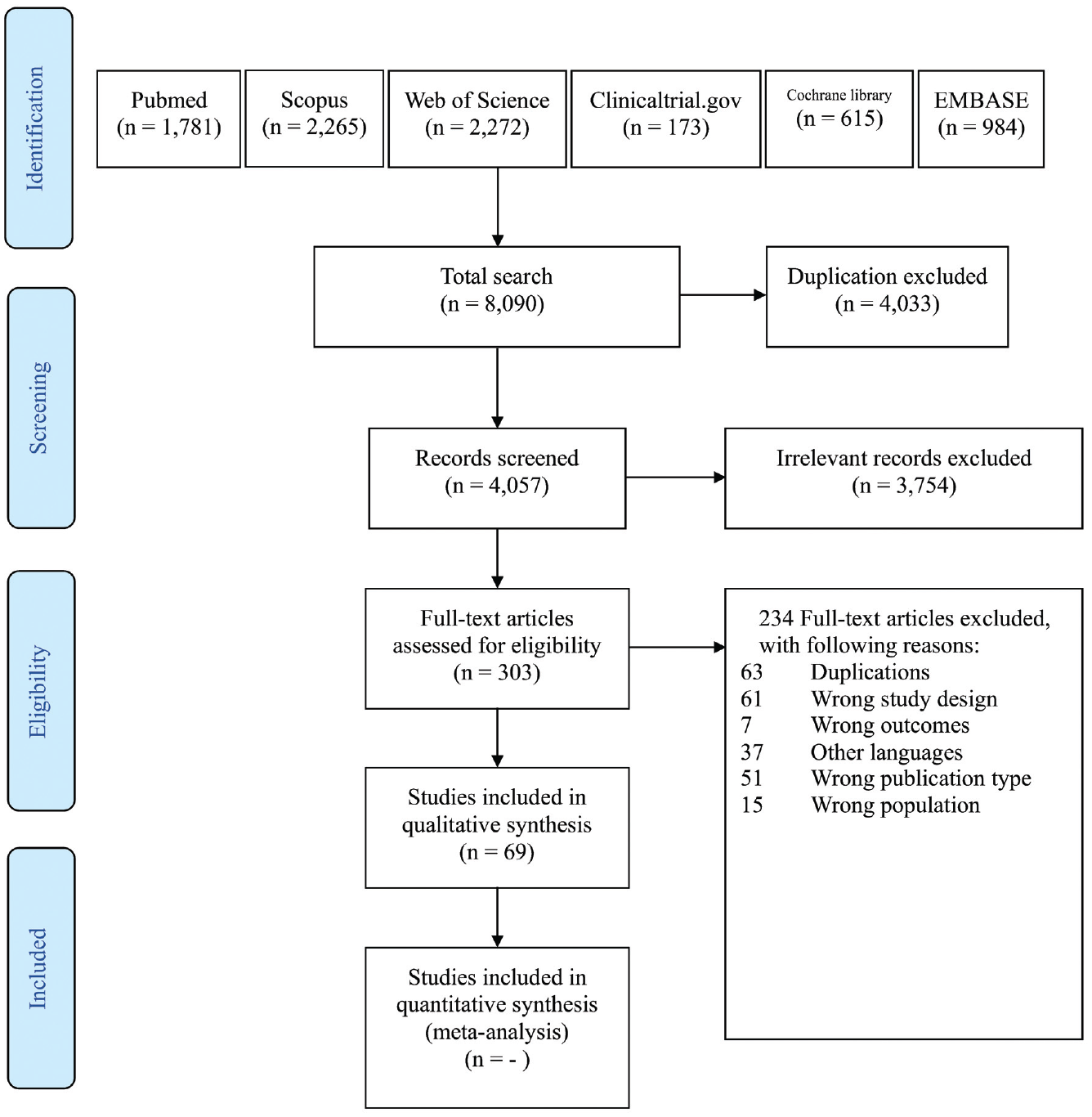
Diagram of study selections based on the 2009 PRISMA statement (Preferred Reporting Items for Systematic Reviews and Meta-analyses).
Rhinitis
Allergic Rhinitis
Of the 23 studies that assessed the effects of nasal saline treatment in patients with AR, 19 were RCTs and 4 were meta-analyses. Eleven RCTs assessed adult AR,4-14 and 8 RCTs assessed pediatric AR.15-22 Three meta-analyses pooled the data from adult and pediatric AR,1,3,23 and 1 meta-analysis examined data only from children. 24
In adult AR, 4 RCTs compared the effects of saline treatment with no-saline treatment.6,7,10,13 Duration of treatment ranged from 2 to 6 weeks. Three studies favored the saline treatment in symptom reduction.6,7,10 The benefits were demonstrated after 2 to 3 weeks and continued until 4 to 6 weeks. Patients in the saline treatment group used fewer antihistamines.7,10,13 The meta-analyses showed that saline treatment was superior in symptom improvements.3,23 Adverse effects were not different from control.
In pediatric AR, 6 RCTs compared the effects of saline treatment with no-saline treatment.15,17,19-22 All RCTs reported benefits of nasal saline treatment over control. Decreases in symptom score and antihistamine usage favored the nasal saline treatment at 4 weeks. 19 The addition of saline spray to nasal steroid showed beneficial effects, such as reduced dosage of intranasal steroid spray, at 8 to 12 weeks.17,22 In children with asthma, quality of life (QoL) improvement was shown at 12 weeks. 15 The meta-analyses showed decreases in symptoms3,23 and antihistamine usage. 24 However, the disease-specific health-related QoL was not affected. 3 Temporary otalgia and epistaxis were noted. 15
Devices
In adult patients, 1 RCT showed that nasal saline treatment with a squeeze bottle (240 mL) was better than a syringe (20 mL) in reducing symptoms. 12 There were no adverse effects in either device. 12 Spray was effective when compared with baseline.9,10 Yet, there were no comparisons between spray and other devices.
No study directly compared the effects among different devices in children. Many devices (spray, atomizer device, or large-volume syringe) provided beneficial effects. Minor adverse events, such as otalgia, ear fullness, and epistaxis, occurred in 30% of the patients who used a large-volume device 15 but none in very low and low-volume devices.17,19-21
Tonicity
In adult AR, 5 RCTs compared hypertonic saline (range, 1.8%-3%) and isotonic saline.4,5,9,11,14 Symptom reduction and QoL improvement favored the hypertonic solution at the duration of 1 to 8 weeks.5,9,11 Three meta-analyses with a mixed population of adults and children reported greater benefits of hypertonic saline.1,3,23 Isotonic saline shortened the saccharin transit time (STT), but hypertonic saline did not. 4 In 1 study, 2 of 40 patients (5%) noted nasal discomfort (light pain sensation) from the 3% hypertonic saline. 14
In pediatric AR, 3 RCTs compared the effects between hypertonic and isotonic saline.16,18,19 The meta-analysis, which included these RCTs, favored the hypertonic saline over the isotonic saline. The antihistamine usage was not different between the tonicities. Adverse effects were reported without statistical differences. 24
Buffer
One RCT assessed the effects of buffered saline in adult AR among 3 groups: mild alkalinity (pH 7.2-7.4), high alkalinity (pH8.2-8.4), and nonbuffered saline. 8 There were no differences among the groups in nasal symptoms, mucociliary clearance time, and nasal patency, although the mild alkaline buffered saline was preferred by the patients.
In children, there was no study that compared the buffered and nonbuffered saline. Buffered and nonbuffered saline showed benefits over the no-saline group.15,17,19-21
Summary
Nasal saline treatment decreased symptoms of AR. The duration of treatment was at least 2 weeks in adult patients and 4 weeks in pediatric patients. There was a slight chance of local nasal irritation in pediatric patients. A large-volume device (≥60 mL) was more effective and recommended in adult patients, while a very low- to low-volume device (<60 mL) was recommended in children. Hypertonic saline treatment was more effective in adults and children with AR. However, adverse events were reported in a small number of patients. Therefore, isotonic saline should be used first. Buffered and nonbuffered saline can be used in adults and children. A summary of the studies in AR is shown in Table 1 .
Studies of Patients With Allergic Rhinitis. a
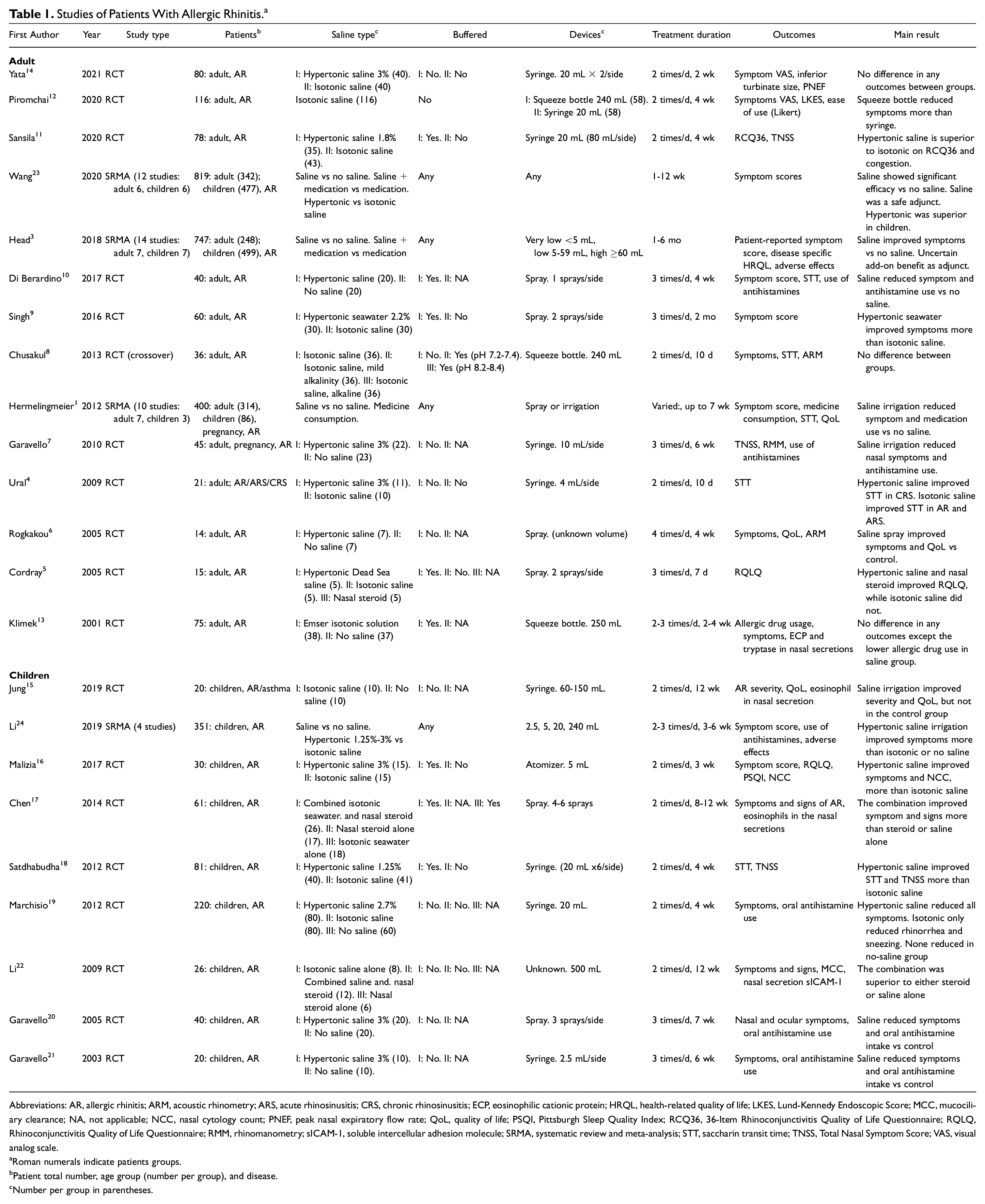
Abbreviations: AR, allergic rhinitis; ARM, acoustic rhinometry; ARS, acute rhinosinusitis; CRS, chronic rhinosinusitis; ECP, eosinophilic cationic protein; HRQL, health-related quality of life; LKES, Lund-Kennedy Endoscopic Score; MCC, mucociliary clearance; NA, not applicable; NCC, nasal cytology count; PNEF, peak nasal expiratory flow rate; QoL, quality of life; PSQI, Pittsburgh Sleep Quality Index; RCQ36, 36-Item Rhinoconjunctivitis Quality of Life Questionnaire; RQLQ, Rhinoconjunctivitis Quality of Life Questionnaire; RMM, rhinomanometry; sICAM-1, soluble intercellular adhesion molecule; SRMA, systematic review and meta-analysis; STT, saccharin transit time; TNSS, Total Nasal Symptom Score; VAS, visual analog scale.
Roman numerals indicate patients groups.
Patient total number, age group (number per group), and disease.
Number per group in parentheses.
Nonallergic rhinitis
There was no study evaluating saline treatment in NAR. Tomooka et al investigated a mixed population of NAR, AR, and CRS, but the data could not be extracted separately. 25
Rhinosinusitis
Acute Rhinosinusitis
There were 17 studies that assessed the effects of nasal saline treatment in ARS. Ten RCTs4,26-34 and 1 meta-analysis 35 investigated the effects in adults. Five RCTs36-40 and 1 meta-analysis were conducted in children. 41
In adult patients, 5 RCTs assessed nasal saline vs no-saline treatment.26,27,30,31,33 Duration of treatment ranged from 1 to 4 weeks. There were no differences in symptom reduction,26,30,31,33 QoL improvement,30,31,33 and STT. 27 However, a post hoc analysis of an RCT showed that the common cold subgroup benefited from saline irrigation on symptom reduction and QoL. 31 The duration of illness after saline treatment showed mixed results. One RCT 26 and 1 meta-analysis 35 reported that the nasal saline treatment did not shorten the duration of illness. In contrast, 1 RCT cited decreases in the duration of illness and usage of medications. 30 Minor adverse effects were indicated, such as dry nose and pain/irritation. 26
In children, 4 RCTs assessed nasal saline vs no-saline treatment.36-39 There were no differences between the groups at the duration of 2 days. 36 Symptom reductions (secretion and nasal obstruction) favored the saline at a longer duration (5 days to 3 weeks).37-39 A meta-analysis showed benefits on nasal symptom reduction. 41 Minor adverse effects were noted, such as nosebleed and burning sensation. 37
Device
Three RCTs compared the effects of saline treatment among different devices in adult patients.28,29,32 The saline treatment, either nasal irrigation with a 250-mL hanging bag (large volume, no pressure) or atomized nasal douche at 7 to 8 mL (low volume, high diffusion, pressure), reduced symptoms and improved nasal patency.28,29 One RCT assessed the overall effectiveness of a syringe (10 mL) with and without a silicone tip applicator. The patient-reported outcomes showed that the syringe with a silicone tip was more effective. 32
Two RCTs compared the effects of saline treatment among different devices in pediatric patients.37,40 One study compared a low-volume syringe (20 mL) with a large-volume squeeze bottle (240 mL). The effects favored the squeeze bottle on reducing nasal obstruction and rhinorrhea. 40 The other study compared a very low-volume spray (3 mL) with a low-volume jet flow (9 mL). There were no differences in the benefits and adverse events between the devices. 37
Tonicity
Four RCTs compared the effects of saline treatment among different tonicities in adult patients.4,26,27,34 There were no differences between the hypertonic and isotonic saline for nasal symptom score26,34 and days to resolution. 26 The STT was markedly improved in the isotonic saline group in 1 study 4 but not different in another. 27 Nasal irritation was more frequent in the hypertonic saline group. 26
In children, 1 study compared 2.3% hypertonic saline with isotonic nasal saline drops for 5 days. There was no difference in symptom score reduction and no report of adverse effects in this study. 39
Buffer
There was no comparative study between the buffered and nonbuffered saline. Both solutions were shown to be effective when compared with the baseline data.
Summary
The effects of nasal saline treatment for ARS had mixed results. It should be considered an option for adults but recommended for children with a duration >5 days. Devices with high diffusion, regardless of volume or pressure, were favored for adult patients, while large-volume with positive pressure devices were recommended for children. Isotonic saline is suggested for adults and children due to its low adverse event rate. Characteristics of the studies are displayed in Table 2 .
Studies of Patients With Acute Rhinosinusitis. a
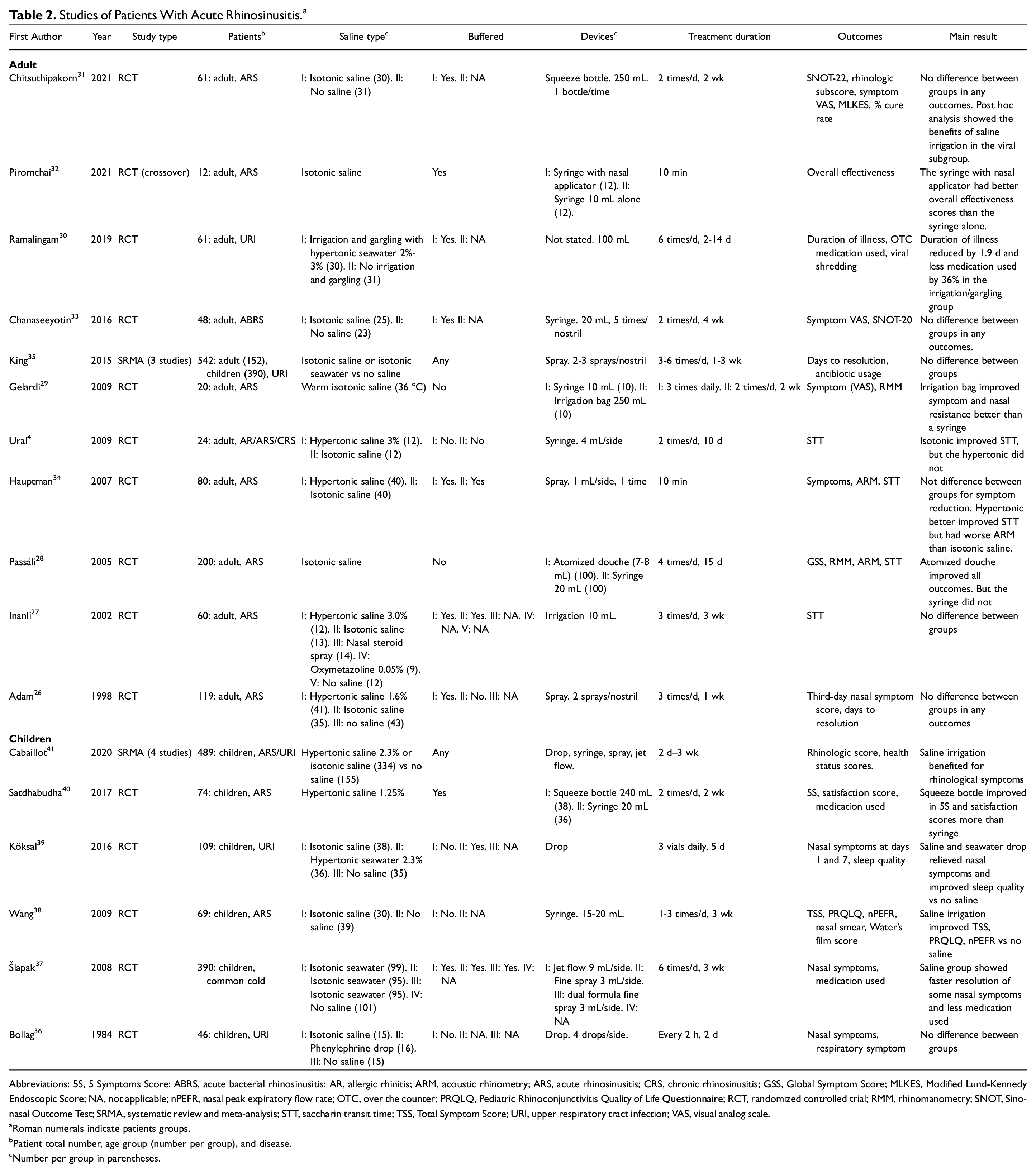
Abbreviations: 5S, 5 Symptoms Score; ABRS, acute bacterial rhinosinusitis; AR, allergic rhinitis; ARM, acoustic rhinometry; ARS, acute rhinosinusitis; CRS, chronic rhinosinusitis; GSS, Global Symptom Score; MLKES, Modified Lund-Kennedy Endoscopic Score; NA, not applicable; nPEFR, nasal peak expiratory flow rate; OTC, over the counter; PRQLQ, Pediatric Rhinoconjunctivitis Quality of Life Questionnaire; RCT, randomized controlled trial; RMM, rhinomanometry; SNOT, Sino-nasal Outcome Test; SRMA, systematic review and meta-analysis; STT, saccharin transit time; TSS, Total Symptom Score; URI, upper respiratory tract infection; VAS, visual analog scale.
Roman numerals indicate patients groups.
Patient total number, age group (number per group), and disease.
Number per group in parentheses.
Chronic Rhinosinusitis
There were 17 studies that assessed the effects of nasal saline treatment. Sixteen articles (13 RCTs and 3 meta-analyses) assessed adult CRS or mixed adults and children with CRS.4,42-57 One RCT was conducted in children. 58
Four RCTs compared the effects of nasal saline with no-saline treatment.42,43,46,52 The benefits of 2-week nasal saline treatment were not shown. 43 One RCT showed significant symptom improvement in the saline irrigation group over no saline at 4 weeks. 52 Taccariello et al found that saline douche and saline spray for 8 weeks improved the QoL and nasal endoscopic appearance. 42 One RCT 46 and 2 meta-analyses47,50 did not provide additional data. Adverse effects were indicated in the nasal saline group, including irritation, burning, tearing, and nosebleeds, 50 with a prevalence of 0% to 23%.43,59
No study assessed the effects of saline irrigation vs no saline in children. There was 1 RCT by Shoseyov et al that assessed the effects among different tonicities in children. 58
Device
Two RCTs assessed the effects of different devices.42,43 One study demonstrated that the nasal douche (60 mL) improved endoscopic appearances but not QoL, whereas the very low-volume spray had the opposite effects. 42 The other study compared 2 large-volume devices (bulb syringe vs irrigation pot) and showed no difference in the 31-item Rhinosinusitis Outcome Measure. 43
In children, there was no direct comparison of devices. One study showed that nasal drops relieved nasal symptoms with acceptable tolerability. 58
Tonicity
Nine studies (8 RCTs4,44,46,48,49,54,56,57 and 1 meta-analysis 55 ) assessed different tonicities of nasal saline treatment in adult CRS. The data from 1 RCT could not be used. 46 Hypertonic saline was more effective than isotonic saline in improving symptoms (nasal congestion48,49,54,56,57 and nasal discharge),48,49,54,56,57 the Lund-Mackay computed tomography score, 56 and STT. 4 In contrast, 1 RCT reported similar effectiveness between hypertonic and isotonic saline. 44
The risks of adverse events in the hypertonic saline group was significantly higher than the isotonic saline. Most events were irritation and burning sensation. 55
One RCT assessed hypertonic saline (3.5%) vs isotonic saline (0.9%) drops in children. 58 Both tonicities showed significant improvements in postnasal drip, cough, and radiologic score from the baseline. Hypertonic saline significantly reduced cough and radiologic score better than isotonic saline but not the postnasal discharge score. Adverse effects in the pediatric group, such as itching and burning sensation, were indicated in the high tonicity group in the first 3 to 4 days. 58 Two meta-analyses did not provide additional data.47,50
Buffer
One RCT compared buffered saline with nonbuffered saline. 45 Buffered saline improved symptoms and the Rhinoconjunctivitis Quality of Life Questionnaire scores. No adverse event was reported from the study.
No study compared buffered and nonbuffered saline in children.
Summary
Nasal saline treatment for at least 4 weeks is recommended for adults. The saline should be buffered and delivered in a large-volume device. Due to its higher adverse events, hypertonic saline is suggested only after isotonic saline has failed to improve the symptoms. Nasal drop can be used in pediatric CRS, preferably with isotonic saline. A summary of the studies in patients with CRS is displayed in Table 3 .
Studies of Patients With Chronic Rhinosinusitis. a
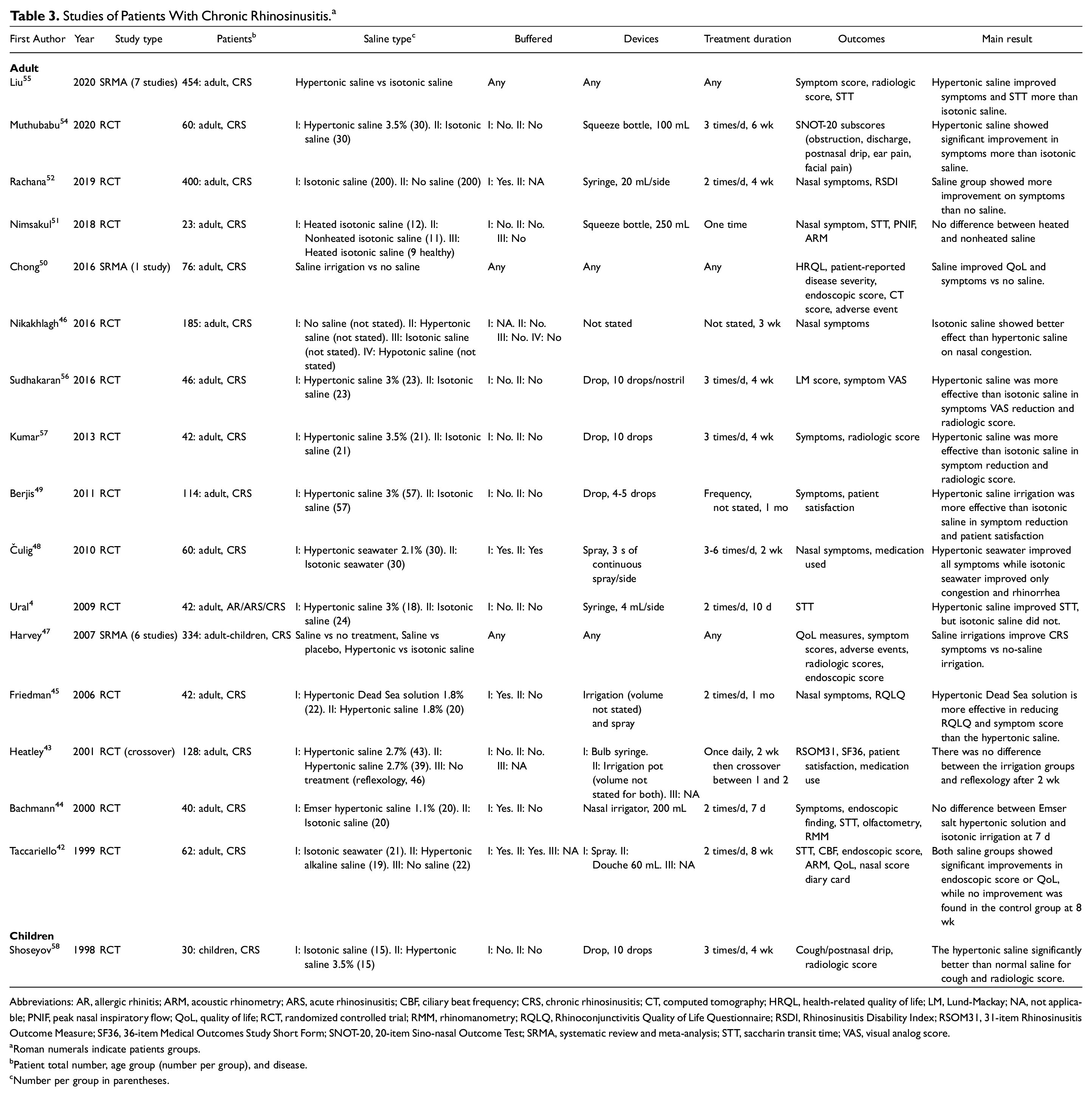
Abbreviations: AR, allergic rhinitis; ARM, acoustic rhinometry; ARS, acute rhinosinusitis; CBF, ciliary beat frequency; CRS, chronic rhinosinusitis; CT, computed tomography; HRQL, health-related quality of life; LM, Lund-Mackay; NA, not applicable; PNIF, peak nasal inspiratory flow; QoL, quality of life; RCT, randomized controlled trial; RMM, rhinomanometry; RQLQ, Rhinoconjunctivitis Quality of Life Questionnaire; RSDI, Rhinosinusitis Disability Index; RSOM31, 31-item Rhinosinusitis Outcome Measure; SF36, 36-item Medical Outcomes Study Short Form; SNOT-20, 20-item Sino-nasal Outcome Test; SRMA, systematic review and meta-analysis; STT, saccharin transit time; VAS, visual analog score.
Roman numerals indicate patients groups.
Patient total number, age group (number per group), and disease.
Number per group in parentheses.
Chronic Rhinosinusitis With Cystic Fibrosis
No study evaluated the effects of saline vs no saline in CRS-CF. Just 1 study by Mainz et al was identified. 60 It compared hypertonic saline (6.0%) with isotonic saline via a randomized crossover study design.
Device
The study utilized an atomizer delivering a very low volume (1 mL per nostril) of saline once daily for 28 days. Benefits were shown when compared with the baseline. 60 Other devices were not assessed.
Tonicity
Both tonicities showed improvements from baseline in 20-item Sino-nasal Outcome Test (SNOT-20) score and nasal symptoms. The improvements were not different between the tonicities. Minor adverse events were noted in both tonicities. 60
Buffer
There was no comparison between buffered and nonbuffered saline treatment. Nonbuffered saline demonstrated improvements from the baseline. 60
Summary
Limited data showed that saline treatment could be used for symptom control and QoL improvement. The saline may be hypertonic or isotonic and delivered by a very low–volume device (atomizer). Only data of nonbuffered saline were available.
Postoperative Care
After Septoplasty/Turbinoplasty Surgery
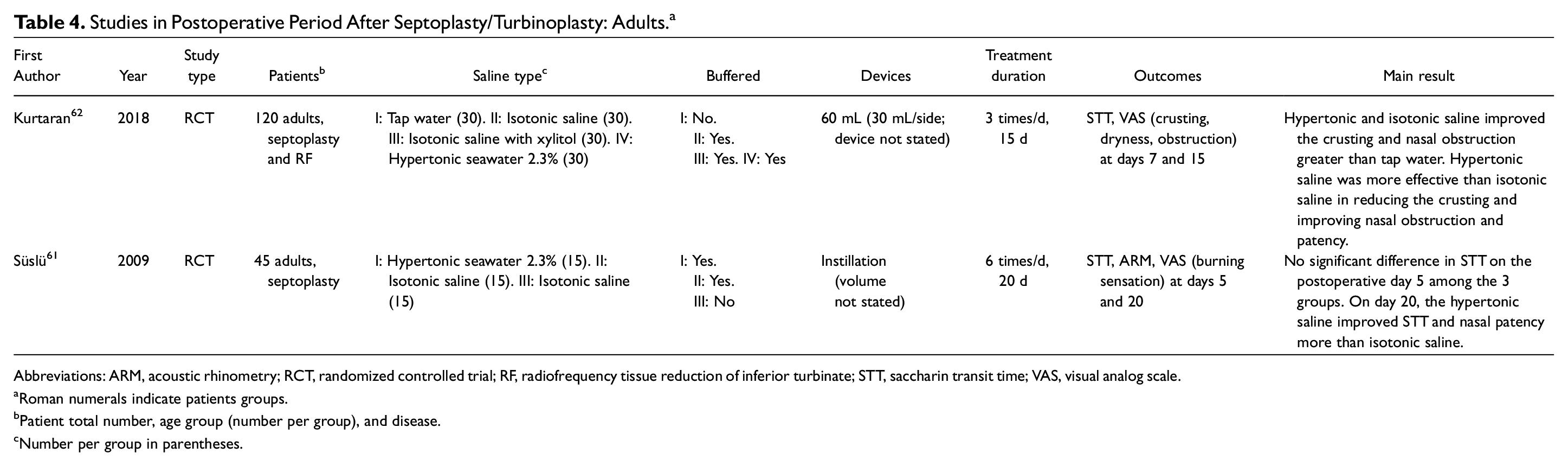
Two RCTs evaluated the effects of saline treatment after septoplasty/turbinoplasty.61,62 None of these studies used the no-saline treatment as control. Saline irrigation decreased the crusting and improved nasal obstruction, which was assessed by visual analog scale 62 and anterior rhinomanometry. 61 Benefits were shown during postoperative days 7 to 15. 62 The STT showed mixed results.61,62 Minor adverse effects were noted. There was no eligible study in pediatric patients.
Device
None of these studies compared different devices. Kurtaran et al studied the effects of a large-volume device (60 mL). 62 The improvements in crusting, nasal obstruction, and STT were reported on postoperative day 15. Süslü et al did not state the volume of nasal saline. 61
Tonicity
Two RCTs compared the effects of 2 tonicities.61,62 Hypertonic and isotonic saline improved the crusting and nasal obstruction greater than tap water. 62 Hypertonic saline was more effective than isotonic saline in improving the crusting, nasal obstruction, and nasal patency.61,62 However, hypertonic saline caused more adverse effects, such as burning sensation. 61
Buffer
Süslü et al compared buffered with nonbuffered isotonic saline. 61 Nasal patency improvement was not different between the groups. Yet, nonbuffered saline (pH 5.5) caused more burning sensation than buffered saline (pH 7.4). The authors believed that the burning sensation might be caused by the acidity of the solution. 61
Summary
With minor adverse events reported, nasal saline irrigation may be used after septoplasty/turbinoplasty. The large-volume saline is suggested to reduce crusting. Although hypertonic saline showed greater beneficial effects than isotonic saline, it was more likely to cause undesirable effects. Therefore, isotonic saline is recommended. The effects of buffered and nonbuffered saline were comparable, although nonbuffered saline caused more irritation. The buffered saline is recommended due to its better tolerability. Characteristics of the studies are displayed in Table 4 .
Studies in Postoperative Period After Septoplasty/Turbinoplasty: Adults. a
Abbreviations: ARM, acoustic rhinometry; RCT, randomized controlled trial; RF, radiofrequency tissue reduction of inferior turbinate; STT, saccharin transit time; VAS, visual analog scale.
Roman numerals indicate patients groups.
Patient total number, age group (number per group), and disease.
Number per group in parentheses.
After Endoscopic Sinus Surgery
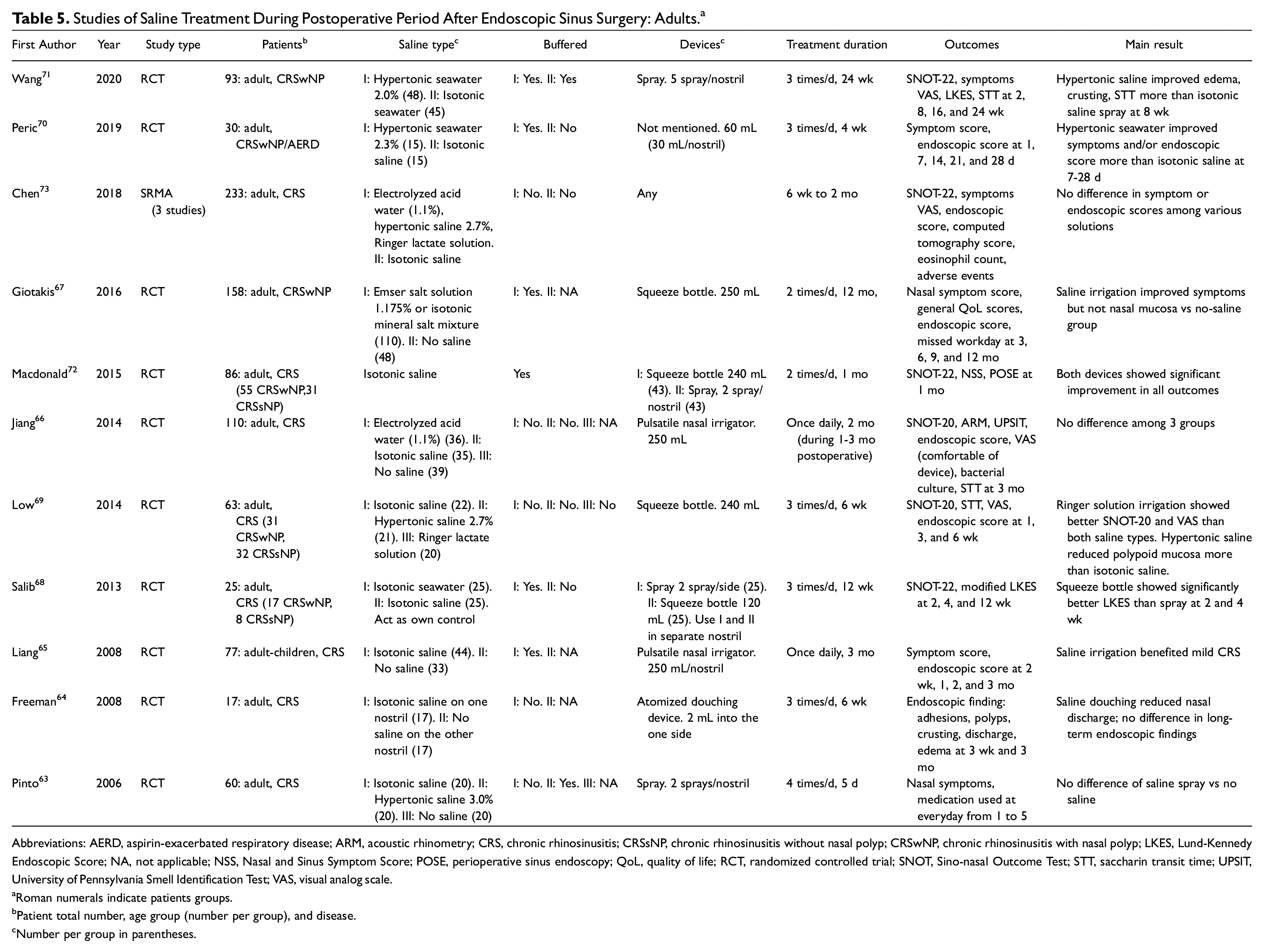
There were 10 RCTs and 1 meta-analysis that assessed post-ESS saline treatment.63-73 All RCTs assessed adult populations, of which 5 evaluated nasal saline vs no-saline treatment.63-67 The study duration ranged from 5 days to 12 months.
One RCT evaluated the outcomes at the first 5 postoperative days. The saline spray did not show benefit on nasal symptoms. 63 At 3 weeks, Freeman et al demonstrated the benefits of atomized douching on nasal discharge reduction. 64 At 3 months, 4 RCTs reported mixed results.64-67 One RCT cited no benefits on adhesion, polyps, crusting, mucosal edema, and nasal discharge improvements. 64 Another study indicated no benefit of saline treatment at 3 months on the SNOT-20, minimal cross-sectional area, smell test, STT, and endoscopic scores. 66 In contrast, 2 RCTs reported beneficial effects of saline treatment on nasal symptoms,65,67 nasal endoscopy, 65 and QoL. 67 Clinical outcomes were significantly improved after 3 months and up to 12 months. 67 When the benefits were assessed by severity of CRS, Liang et al found that the benefits were shown in the mild CRS subgroup with computed tomography scores ≤12. 65
There was no study in the pediatric population.
Device
Two RCTs compared the effects of saline treatment between 2 devices.68,72 Salib et al randomly assigned nasal saline treatment via 2 puffs of saline spray into 1 nostril and a squeeze bottle (120 mL) into the other nostril in the same patient. The large-volume irrigation showed better endoscopic findings at 2 and 4 weeks. Nasal endoscopic findings were not different at 3 months. The benefits assessed by the patient’s perception favored the large volume throughout the follow-up period. 68 The other RCT evaluated the feasibility of a multicenter trial and recruited a small number of patients. 72
Five other studies that utilized large-volume devices (240 mL) for saline treatment showed improvements from the baseline.65-67,69,70 Very low-volume devices used in 2 studies did not show beneficial results.63,64
Tonicity
Five RCTs compared hypertonic with isotonic saline.63,66,69-71 There were no differences between hypertonic and isotonic saline in improving symptoms,63,69,71 SNOT-20 scores,66,69,71 endoscopic scores, 66 minimal cross-sectional area, 66 smell threshold, 66 and STT.66,69,71 However, hypertonic saline demonstrated more benefits than isotonic saline during the first 6 weeks,69,71 but these differences did not persist in the longer follow-up period. 71 One meta-analysis compared different solutions with isotonic saline and reported no differences in symptoms or endoscopic scores. 73
More patients receiving hypertonic saline refused or discontinued the treatments than isotonic saline. 66 The adverse events, as reported in the hypertonic saline group, were irritation, 66 increased mucous secretion, and pain. 63
Buffer
Three studies compared the effectiveness between the buffered and nonbuffered saline.63,68,70 All were inconclusive due to discrepancies of other factors between the experimental arms. Pinto et al 63 and Perić et al 70 compared buffered hypertonic with nonbuffered isotonic saline. Salib et al compared buffered isotonic saline spray with nonbuffered isotonic saline irrigation using a bottle. 68 Although the differences between buffered and nonbuffered saline were inconclusive, both solutions revealed beneficial effects as compared with baseline.
Summary
The benefits at a short postoperative period were not demonstrated. Saline treatment is recommended when the duration of treatment is >3 weeks and up to 3 months after ESS. A large-volume device is preferred. Hypertonic and isotonic saline showed benefits on subjective and objective outcomes. Due to the potential of increasing pain and irritations caused by hypertonic saline, isotonic saline is recommended for the postoperative period after ESS. Buffered and nonbuffered saline can be used. Characteristics of the studies are displayed in Table 5 .
Studies of Saline Treatment During Postoperative Period After Endoscopic Sinus Surgery: Adults. a
Abbreviations: AERD, aspirin-exacerbated respiratory disease; ARM, acoustic rhinometry; CRS, chronic rhinosinusitis; CRSsNP, chronic rhinosinusitis without nasal polyp; CRSwNP, chronic rhinosinusitis with nasal polyp; LKES, Lund-Kennedy Endoscopic Score; NA, not applicable; NSS, Nasal and Sinus Symptom Score; POSE, perioperative sinus endoscopy; QoL, quality of life; RCT, randomized controlled trial; SNOT, Sino-nasal Outcome Test; STT, saccharin transit time; UPSIT, University of Pennsylvania Smell Identification Test; VAS, visual analog scale.
Roman numerals indicate patients groups.
Patient total number, age group (number per group), and disease.
Number per group in parentheses.
Discussion
Intranasal saline treatment may not be a one-size-fits-all for the different sinonasal diseases. In patients with AR, the saline treatment showed benefits on symptom reduction after 2 to 4 weeks. A large-volume device was recommended for adult AR, but it could cause adverse events in some children. Therefore, a low-volume device was recommended for children. Isotonic saline was suggested due to its effectiveness and tolerability. There were no differences between the buffered and nonbuffered saline. Therefore, either could be chosen.
In adult patients with ARS, the benefits of saline treatment were not revealed in most RCTs. However, symptom and QoL improvements were demonstrated in the common cold subgroup through large-volume saline irrigation. In addition, Ramalingam et al showed a reduction in duration of the common cold. 30 Due to this conflict in high-level evidence, the saline treatment was suggested as an option in adult ARS. The improvements in nasal obstruction and rhinorrhea were demonstrated in children after 5 days of saline treatment and up to 3 weeks. These findings were supported by the majority of RCTs and 1 meta-analysis. Thus, it was recommended for pediatric ARS, and isotonic saline delivered via a large-volume device was suggested.
Improvements in symptoms, QoL, and nasal endoscopic findings were demonstrated in adults with CRS after 4 weeks of nasal saline treatment. Therefore, it was recommended for CRS and should be administered for at least 4 weeks. Buffered isotonic saline delivered by a large-volume device was suggested. The evidence in children was limited to nasal saline drops.
The nasal saline treatment enhances mucociliary function, which benefits patients with CRS-CF. Just saline atomizer was recommended because it was the only device used in 1 study. Nasal saline irrigation after septoplasty/turbinoplasty and ESS reduced crusting and nasal obstruction. Isotonic saline delivered by a large-volume device effectively cleared out the clotted blood, mucous, and debris in the postoperative cavity. Beneficial effects after ESS were demonstrated up to 3 months, and then mixed results were reported. Effects of saline for healing and re-epithelization should be clearly seen during the first 3 months.
This review identified knowledge gaps and research opportunities. The beneficial effects of saline treatment are still unclear in adults with NAR, ARS, and CRS-CF. There is insufficient evidence of saline for pediatric CRS, CRS-CF, and postoperative care. Optimal devices for saline treatment for children with CRS require further studies.
This review had limitations. The recommendations suffered from heterogeneity of delivery methods and timing of evaluations. For instance, the outcomes of nasal saline spray was assessed at 5 days after ESS, while other studies evaluated the larger-volume device after 2 weeks or later. In addition, there were other confounding factors, such as the endotype of CRS that might affect a later stage of postoperative healing.
This review showed different levels of evidence among conditions. AR had very strong evidence supporting the saline treatment, whereas others conditions had relatively much weaker evidence. This review did not intend to discourage the saline usage in those without firmly supportive data. It rather informed how much the evidence currently existed and which delivery methods provided the best possible outcomes based on the available data. Future research is likely to provide or change the answers to a certain topic and/or change the recommendations. Physicians can always use their discrete decision to use the saline treatment or to wait until more evidence becomes available.
Conclusion
Evidence supported the use of the nasal saline treatment for AR, ARS, CRS, CRS-CF, and postoperative patients, but there were no data for NAR. For AR, large-volume devices were effective for treating adults, but low-volume devices were effective for children. Isotonic saline had fewer adverse events than hypertonic saline. For ARS, the evidence supported the use of saline irrigation in children, but there was weak evidence for adults. Large-volume devices were more effective, especially in the common cold subgroup. For CRS, large-volume devices were effective for adults, but saline drop was the only available data in children. Buffered isotonic saline was more tolerable than nonbuffered or hypertonic saline. There were limited data for CRS-CF. For postoperative care, buffered isotonic saline delivered by large-volume devices was effective.
Supplemental Material
sj-docx-1-opn-10.1177_2473974X221105277 – Supplemental material for Optimal Device and Regimen of Nasal Saline Treatment for Sinonasal Diseases: Systematic Review
Supplemental material, sj-docx-1-opn-10.1177_2473974X221105277 for Optimal Device and Regimen of Nasal Saline Treatment for Sinonasal Diseases: Systematic Review by Wirach Chitsuthipakorn, Dichapong Kanjanawasee, Minh P. Hoang, Kachorn Seresirikachorn and Kornkiat Snidvongs in OTO Open: The Official Open Access Journal of the American Academy of Otolaryngology-Head and Neck Surgery Foundation
Footnotes
Author Contributions
Disclosures
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
