Abstract
Objective
To examine the safety and efficacy of concomitant chemoradiotherapy using a modified TPF regimen (docetaxel + cisplatin + 5-fluorouracil) in patients with advanced sinonasal squamous cell carcinoma (SNSCC).
Study Design
Retrospective study.
Setting
Tertiary center (university hospital).
Methods
Seven patients with previously untreated T3-T4 SNSCC were enrolled. They underwent radiotherapy once daily (total dose, 70 Gy) with 2 courses of concomitant 120-hour infusion of 5-fluorouracil (600 mg/m2/d), docetaxel (50 mg/m2, day 2), and cisplatin (60 mg/m2, day 2)
Results
Grade 4 leukopenia, grade 4 neutropenia, and grade 3 lymphopenia were observed in 1, 3, and 4 patients, respectively. Grade 4 creatinine elevation was observed in 1 patient. However, other grade 3 or 4 adverse events were not common. Complete response was obtained in all patients. At 60 months there was 85.7% disease-free survival and 100% overall.
Conclusion
Concomitant chemoradiotherapy with a modified TPF regimen may be feasible and effective in patients with advanced SNSCC.
Squamous cell carcinoma of the nasal cavity and paranasal sinuses (sinonasal squamous cell carcinoma [SNSCC]) is rare when compared with other sites of the head and neck. 1 Early-stage SNSCC can be resected; however, surgical treatment of advanced-stage SNSCC demands considerable experience and advanced techniques because of the complex anatomy of the sinonasal area and proximity to the skull base and orbital cavity. Although multimodality treatments such as surgery, chemotherapy, and radiotherapy have been improved recently, prognoses of patients with SNSCC remain poor, with a 5-year survival rate of approximately 30% to 50%.2,3 Concomitant chemoradiotherapy (CCRT) has been recognized as the standard treatment for head and neck squamous cell carcinoma (HNSCC), and its efficacy has been proven in many clinical studies. 4 We assumed that the CCRT used for HNSCC did not improve the prognosis in patients with SNSCC because of the selected chemotherapy regimen. The standard regimen of CCRT for HNSCC comprises cisplatin (100 mg/m2 triweekly); however, prognoses of patients with advanced SNSCC who underwent CCRT with cisplatin were reported to be poor.
The TPF regimen (docetaxel + cisplatin + 5-fluorouracil) has been widely used as induction chemotherapy for patients with HNSCC, and its usefulness has been analyzed and reported elsewhere.5,6 However, CCRT with the TPF regimen is not extensively used because physicians and medical oncologists fear the possible toxicity based on the standard doses of individual agents. We used a modified TPF regimen with reduced doses of docetaxel, cisplatin, and 5-fluorouracil with concomitant radiotherapy to treat patients with SNSCC.
Here, we report the effectiveness and adverse events of CCRT with a modified TPF regimen to treat patients with advanced SNSCC and the promising outcomes in the patients who underwent this treatment.
Patients and Methods
In total, 41 patients with SNSCC were treated with curative intent between 2011 and 2019 at Iwate Medical University Hospital. All the patients had histopathologically proven squamous cell carcinoma originating from the nose or maxillary, ethmoid, sphenoid, or frontal sinuses. Of these 41 patients, the clinical courses of 7 patients who underwent CCRT with a modified TPF regimen were analyzed in this study. Criteria for the selection of these 7 patients were as follows: stage IVA or IVB, origin from nasal cavity or ethmoid/frontal sinus, origin from maxillary sinus but not applicable to RADPLAT (supradose selective intra-arterial cisplatin and concomitant radiation), and neither induction nor adjuvant chemotherapy. Most of the maxillary cancers (21 patients) were treated with RADPLAT. Three patients underwent surgery; 3, CCRT with cisplatin; 1, CRT with docetaxel; and 3, CCRT with modified TPF with induction or adjuvant chemotherapy. Three patients with poor performance status underwent radiotherapy alone. The location and extent of tumors were determined with contrast-enhanced computed tomography, magnetic resonance imaging, and 18-fluoro-2-deoxyglucose positron emission tomography. Tumors were staged per the most recent version of the TNM Classification of Malignant Tumors (eighth edition), published by the Union for International Cancer Control. 7 This study was approved by the Institutional Review Board of Iwate Medical University (MH2020-061).
This study was a retrospective chart review. All procedures followed were in accordance with the ethical standards of the responsible committee on human experimentation and with the Helsinki Declaration of 1975 as revised in 2013. 8 Written informed consent was obtained from all the patients.
The modified TPF regimen used to treat patients with advanced SNSCC comprised intravenous administration of 5-fluorouracil (600 mg/m2) on days 1 to 5 and a bolus injection of docetaxel (50 mg/m2) and cisplatin (60 mg/m2) on day 2. 9 These dosages were based on the results of our phase I study that assessed the dose-limiting toxicity of this protocol (data not shown). Briefly, 8 patients were enrolled in this study, and dose-limiting toxicities were observed in 2 patients: one had grade 4 pneumonitis and the other, grade 4 renal failure. Similar results and recommended doses were obtained in another phase I study. 10 Intensity-modulated radiotherapy, mainly used as the concomitant radiotherapy, was administered once daily up to a total dose of approximately 70 Gy. We planned to administer 2 courses of the modified TPF regimen during the first and fifth weeks of radiotherapy. Adverse events were assessed via the Common Terminology Criteria for Adverse Events (version 4.0; US National Cancer Institute). The Kaplan-Meier method was used to determine the survival rates of the patients. The log-rank test was used to assess significant differences.
Results
Patients’ ages ranged from 50 to 65 years (median, 61 years), and the follow-up period ranged from 18 to 96 months (median, 38 months). Locations of tumors were as follows: 3 patients, nasal cavity; 3, ethmoid sinus; 1, maxillary sinus. Clinical classification of tumors was performed according to the TNM classification: 3 patients had T4aN0M0 and 1 each had T3N2cM0, T4aN2bM0, T4bN0M0, and T4bN1M0. Patient characteristics are summarized in Table 1 . One patient with maxillary cancer was not eligible for RADPLAT because of multiple cervical lymph node metastases.
Characteristic Features of the Patients.
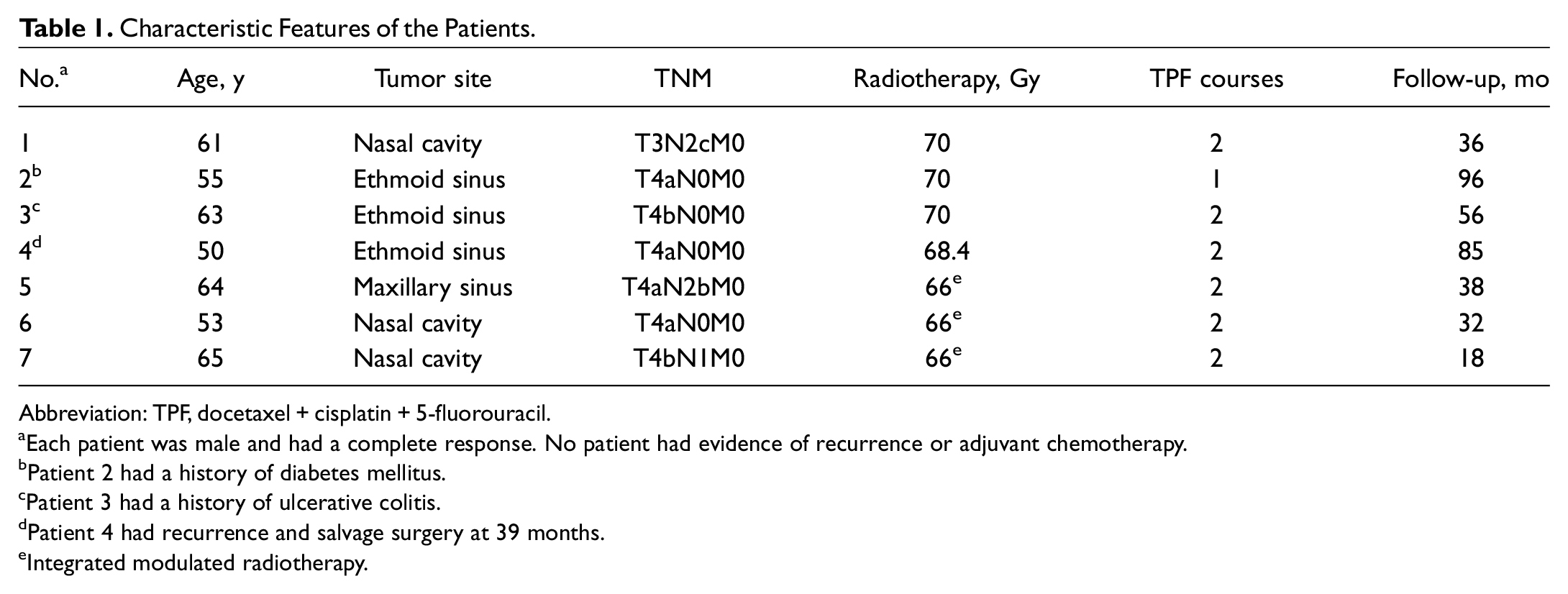
Abbreviation: TPF, docetaxel + cisplatin + 5-fluorouracil.
Each patient was male and had a complete response. No patient had evidence of recurrence or adjuvant chemotherapy.
Patient 2 had a history of diabetes mellitus.
Patient 3 had a history of ulcerative colitis.
Patient 4 had recurrence and salvage surgery at 39 months.
Integrated modulated radiotherapy.
The radiotherapy dosage ranged from 66 to 70 Gy (average, 68.1 Gy). Three patients received intensity-modulated radiotherapy, and 6 received 2 courses of the modified TPF regimen with concomitant radiotherapy. One patient (case 2) exhibited grade 4 creatinine elevation after the administration of the first TPF course; hence, the second course was canceled.
Adverse events in the patients who underwent CCRT with the modified TPF regimen are presented in Table 2 . Grade 4 neutropenia was observed in 3 patients (42.9%), and grade 3 lymphopenia was observed in 4 (57.1%). Grade 4 serum creatinine elevation was observed in 1 patient; therefore, the second course of TPF was canceled, as mentioned earlier. Grade 4 mucositis was observed in 1 patient (14.3%); however, he recovered at the time of discharge. All patients developed mucositis (any grade), and grade 3 or 4 bone marrow suppression was common and frequently occurred in patients with SNSCC who underwent CCRT with the modified TPF regimen. Visual disturbance was observed in some cases as a late complication of the radiation therapy, but no neurologic deficits were observed.
Adverse Events.
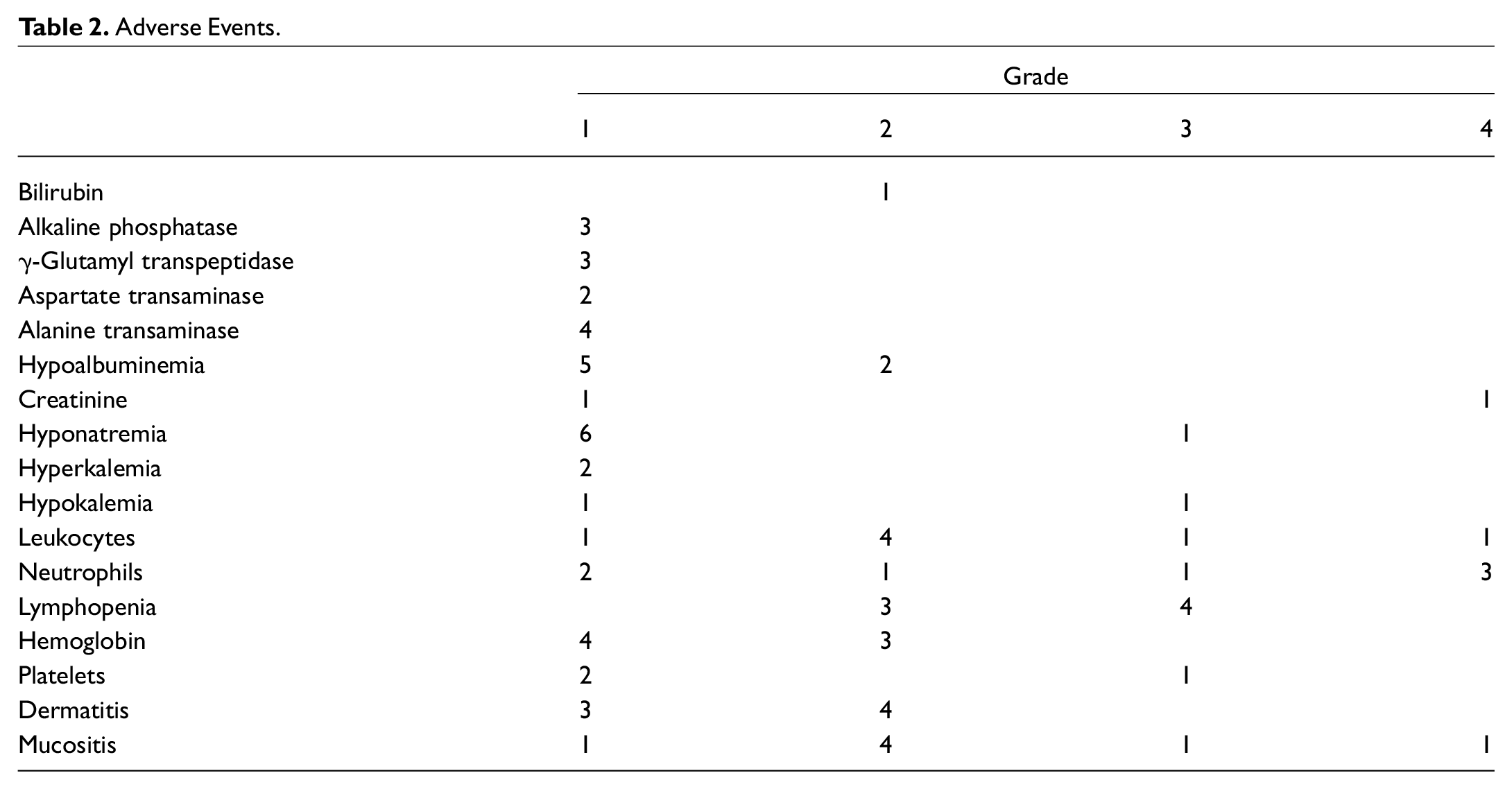
The typical result is shown in Figure 1 . The tumor is located in the right ethmoid sinus (panels A and C), invading into the orbital cavity and skull base up to the brain, but disappeared posttreatment (panels B and D). Complete response was obtained in 7 (100%) patients ( Table 1 ). Local recurrence was observed in 1 patient, who underwent successful salvage surgery after 39 months of CCRT. Survival rates of the patients were calculated by the Kaplan-Meier method. At 5 years after CCRT, the overall and disease-free survival rates were 100% and 85.7%, respectively ( Figure 2 ). All patients were alive without disease at the time of manuscript preparation.
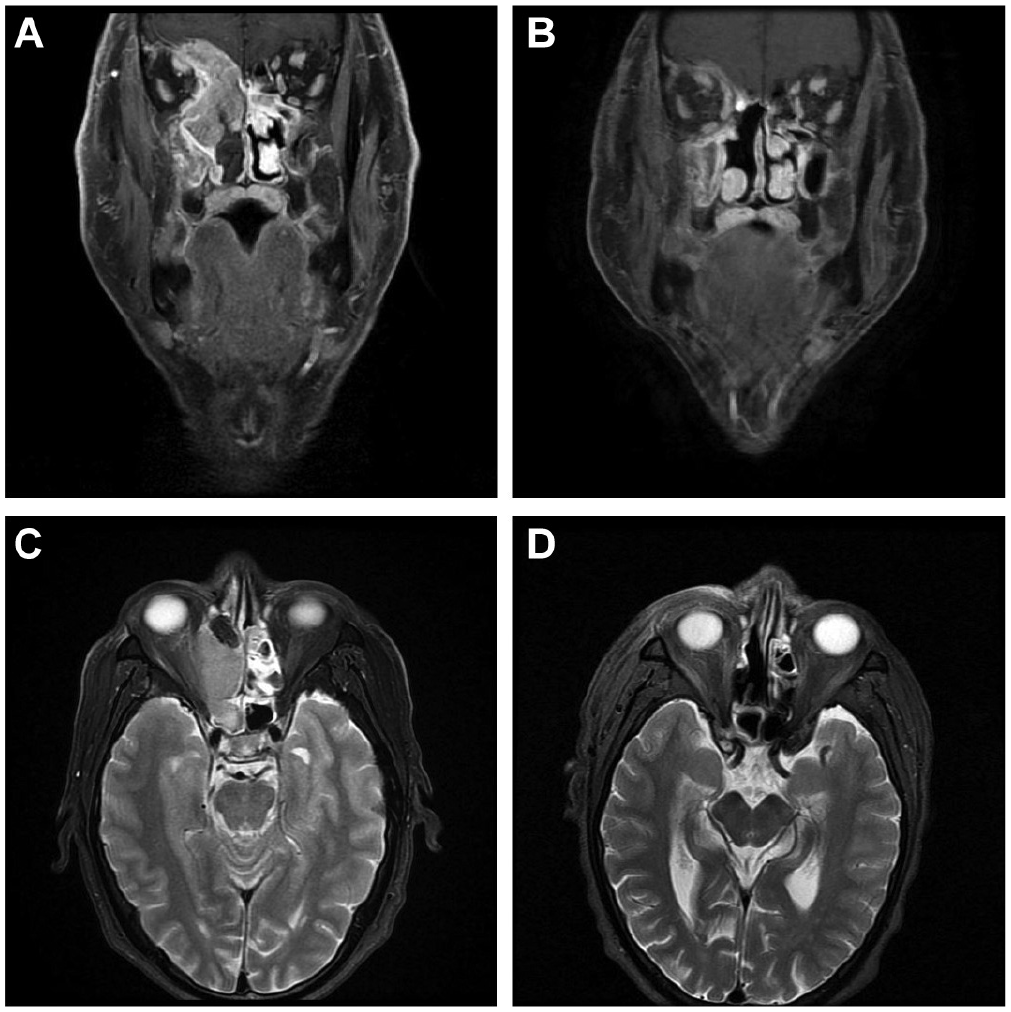
Typical result of modified TPF with CCRT (case 3). (A, C) Pre- and (B, D) posttreatment (3 months after CCRT) T2-weighted magnetic resonance imaging scans. CCRT, concomitant chemoradiotherapy; TPF, docetaxel + cisplatin + 5-fluorouracil.
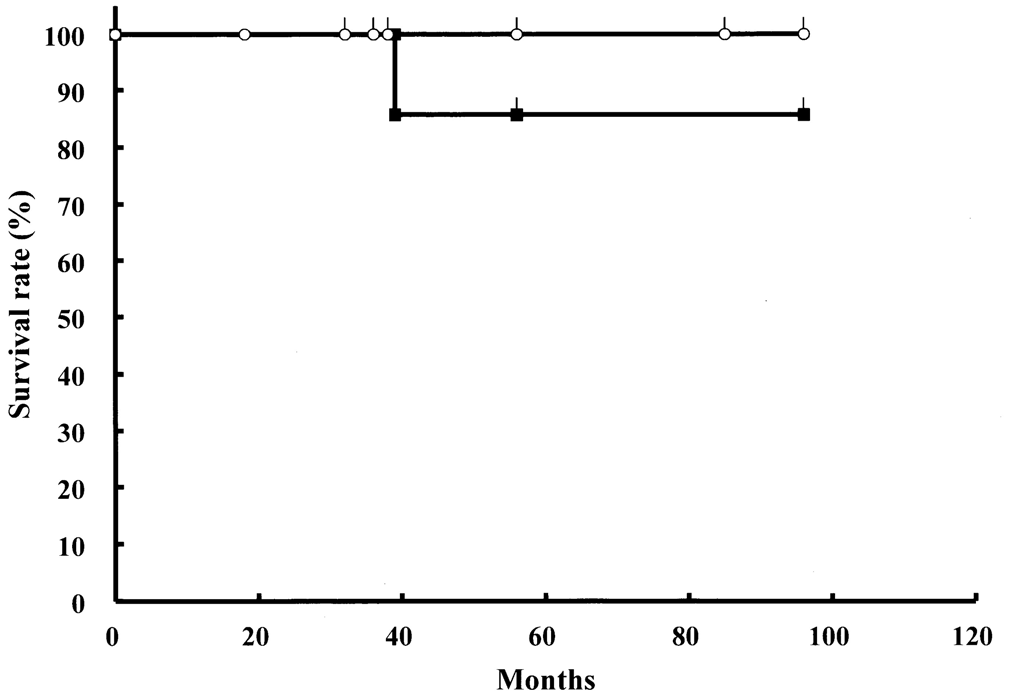
Survival curves of the patientswith sinonasal squamous cell carcinoma. Closed squares indicate disease-free survival and open circles, overall survival. At 5 years after concomitant chemoradiotherapy, the calculated rates were 85.7% for disease-free survival and 100% overall.
Discussion
The standard treatment for SNSCC has been controversial and unestablished because of the rarity of SNSCC. Although surgical resection and postoperative radiotherapy are performed for resectable tumors, treatment of advanced and unresectable SNSCC is difficult because of its proximity to the skull base and orbital cavity.
In this study, we used a modified TPF regimen as a concomitant chemotherapy of CCRT for patients with SNSCC. Although grade 4 adverse events such as neutropenia, serum creatinine elevation, and mucositis were observed in some cases, all patients completed the full course of radiotherapy, and 85.7% received scheduled courses of TPF ( Table 1 ). Although 1 patient underwent salvage surgery, the complete response rate and 5-year overall survival rate were both 100%. These results indicate that CCRT with a modified TPF regimen is effective and safe for patients with advanced SNHCC.
The common treatment for HNSCC is CCRT, in which platinum-based chemotherapy has been found to be an effective radiosensitizer. In general, patients receive CCRT with cisplatin (100 mg/m2) on days 1, 22, and 43 of the radiation therapy. However, few studies have been conducted on CCRT for SNSCC, and clinical data are largely limited to retrospective analyses.11,12 Kang et al retrospectively analyzed the data of patients with advanced maxillary cancer and reported that patients who underwent surgery, radiotherapy, and/or chemotherapy had better progression-free survival and overall survival than those who underwent CCRT. 13 Conversely, Kim et al indicated no difference in survival rates between patients who underwent surgery and postoperative radiotherapy and those who received CCRT—including locoregional recurrence–free, distant metastasis–free, disease-specific, and overall survival. 14
There has been a consensus regarding multidrug therapy for several types of cancer, including leukemia, lymphoma, colon cancer, and lung cancer, considering action and resistance to drugs. One of the crucial reasons for failure of cancer treatment is the development of drug resistance by cancer cells; hence, multidrug therapy is a promising approach to prevent cancer cell resistance.15,16 However, there is little comprehension of TPF for HNSCC.
Maeda et al reported that docetaxel enhances the cytotoxicity of cisplatin in gastric cancer cells by modifying intracellular platinum metabolism. 17 Although the cell type in gastric cancer is different from that in HNSCC, intracellular cisplatin concentration could be the key factor for the effectiveness of the TPF regimen because the standard chemotherapeutic agent that is first recommended for HNSCC is cisplatin. Hence, the order of drug administration is the key point for the effectiveness—that is, docetaxel first and then cisplatin. Saiki et al stated that the HNSCC cell line with acquired cisplatin resistance exhibited hypersensitivity to taxane. 18 This phenomenon could explain the effectiveness of the TPF regimen in patients with HNSCC who acquired cisplatin resistance after receiving cisplatin chemotherapy and exhibited a response to the TPF-based chemotherapy.
CCRT for advanced HNSCC usually comprises triweekly cisplatin, and its effectiveness and tolerance have been recognized so far. In addition, the TPF regimen has been used for advanced HNSCC as induction chemotherapy, and its usefulness and effectiveness have been reported.5,6 To improve the locoregional control of CCRT and outcome of the patients, Katori et al 10 developed CCRT with a modified TPF regimen. In the phase I trial, although CCRT with TPF was well tolerated and efficacious in patients with locally advanced HNSCC, it was associated with a high incidence of severe mucositis (79%; grades 3 and 4), 19 which prevented this protocol from being accepted as the standard therapy for advanced HNSCC. Therefore, we determined the drug doses based on the results of our phase I study to assess the dose-limiting toxicity of this protocol (data not shown), which are unexpectedly the same as those reported by Tsukuda et al. 19 Fortunately, the irradiation area in SNSCC comprises the nasal cavity, paranasal sinuses, and a limited region of the oral and pharyngeal mucosa. In fact, grade 3 or 4 mucositis occurred in 2 (28.6%) of our patients. Our results indicate that CCRT with the modified TPF regimen seems to be tolerable and shows promising outcomes in patients with SNSCC. The effectiveness and tolerability of CCRT with the modified TPF regimen in patients with squamous cell carcinoma of the external auditory canal have also been reported.9,20
The TPF regimen has been widely used in head and neck cancer as induction chemotherapy,5,6 and some reports have analyzed its efficacy in SNSCC.21,22 According to our results, patients who underwent CCRT using the modified TPF regimen had good treatment outcomes, as did patients in the previous studies of TPF induction chemotherapy followed by CRT. Adverse events in patients who underwent CCRT with modified TPF were not more severe than those in patients who underwent TPF induction chemotherapy followed by CRT. However, our results suggest that CCRT with the modified TPF regimen had benefits for the patients in terms of shorter treatment period and lower cost than TPF induction chemotherapy followed by CRT.
A limitation of this study was the study design. Because SNSCC is rare, a prospective study is difficult or impossible to conduct. Moreover, a standard treatment has not been established in most hospitals and institutions, and various treatments are used to treat patients with advanced SNSCC. Therefore, an organized retrospective study is required to evaluate treatment results. Stronger evidence is required to reveal the efficacy of CCRT with a modified TPF regimen for SNSCC treatment. A multi-institutional prospective study is warranted to clarify this concept, if possible.
