Abstract

Keywords
Systemic lupus erythematosus (SLE) is a common autoimmune condition targeting various organ systems throughout the body. SLE is frequently undiagnosed or misdiagnosed due to its wide range of symptoms. Hypertrophic pachymeningitis (HP), defined by inflammation of the dura mater, is a rare clinical condition that can manifest with a variety of symptoms, such as headache and multiple cranial neuropathies. HP may be idiopathic or associated with a concurrent disease, typically an autoimmune disorder. Correlation between HP and SLE is rare, with few reports in the literature. 1 Review of the literature identified a single report from Japan of SLE associated with HP, manifesting as profound hearing loss. 2 We present a patient with SLE-related HP who experienced sudden unilateral sensorineural hearing loss (SNHL) with associated vertigo and an unexpectedly favorable response to treatment.
Case Report
A 56-year-old African American woman with SLE presented to an academic tertiary referral center with several weeks of headache, progressive right hearing loss, dizziness, and ipsilateral tinnitus. Her medical history included thrombotic thrombocytopenic purpura and pulmonary embolism. She followed a drug regimen, including hydroxychloroquine and warfarin within therapeutic range. Magnetic resonance imaging (MRI) showed dural enhancement, and she was admitted and medicated with intravenous vancomycin, ceftriaxone, and acyclovir for presumed meningitis. Lumbar puncture findings were negative, and the otolaryngology division was consulted.
Review of history confirmed the progressive right hearing loss over several weeks. She had developed severe rotary vertigo about a week prior to admission, and this had improved. At the time of examination, she noted mild dizziness exacerbated by rapid head acceleration in any direction. Examination revealed that she was alert and oriented. Otoscopy results were normal bilaterally. There was mild dizziness with left-beating nystagmus in left lateral gaze, accentuated after the head thrust maneuver. Audiometry demonstrated profound right SNHL; the left ear was normal. Word recognition was 0% on the right, and the Stenger test result was negative.
A targeted high-resolution MRI scan demonstrated extensive dural enhancement along the right petrous portion of the temporal bone and internal auditory canal ( Figure 1A ). At this time, the association of aseptic dural enhancement with the patient’s existing SLE led to the diagnosis of HP involving the right internal auditory meatus (IAM) as the cause of the patient’s hearing loss. In reference to previous treatment in the literature,1-5 antibiotics were discontinued, and the patient started a course of corticosteroids, receiving 40 mg of oral prednisone per day for 14 days.
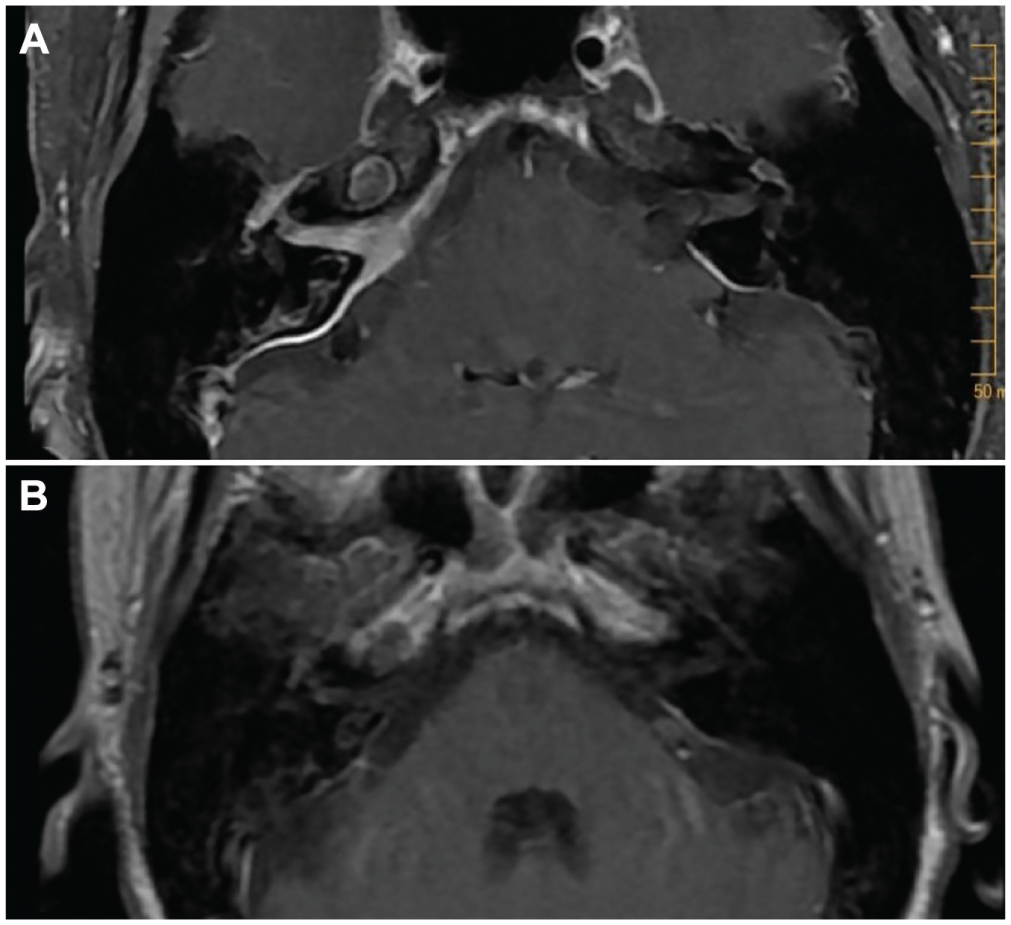
Contrasting the T1-FLAIR gadolinium-enhanced MRI scans taken before and 1 year after treatment with prednisone. (A) The acute MRI scans show extensive enhancement and thickening of dura on the posterior surface of the right temporal bone extending into the IAM. The left temporal bone has similar mild enhancement. The right geniculate ganglion and facial nerve enhanced but were clinically uninvolved. (B) At 1 year after treatment, there is marked reduction in dural inflammation. FLAIR, fluid-attenuated inversion recovery; IAM, internal auditory meatus; MRI, magnetic resonance imaging.
The patient experienced dramatic improvement in hearing after completing the course of oral corticosteroids, showing a near complete regaining of right ear function. The follow-up audiogram showed essentially normal thresholds with 100% word recognition. One-year posttreatment MRI demonstrated near total resolution of dural inflammation ( Figure 1B ).
Discussion
Dural inflammation due to HP can be localized to various areas of the cranial vault or skull base and result in a variety of central nervous system symptoms imitating various pathologies, such as meningitis. Additionally, various cranial neuropathies occur. Our patient presented with headache and cochleovestibular nerve involvement causing vertigo and SNHL. In HP diagnosis, gadolinium-enhanced T1-weighted MRI showed the presence of dural inflammation; in the case of our patient, the dura of the IAM was particularly affected. This inflammatory dural response could affect any cranial nerve. Interestingly, the geniculate ganglion and tympanic segment of the right facial nerve enhanced intensely but were clinically uninvolved. Preexisting SLE is a documented cause of HP, and HP has been associated with SNHL in previous cases.
Our patient demonstrated a nearly complete resolution of hearing after a course of corticosteroids. Given the poor prognostic indicators of profound thresholds, vertigo, and extensive IAM involvement demonstrated by MRI findings, the hearing recovery was unexpected. One might speculate that the mechanism of hearing loss involved reversible compromise of the arterial supply to the inner ear. Treatment with corticosteroids decreased the dural inflammation on MRI and resulted in complete restoration of hearing, further suggesting that the cause of the hearing loss was inflammatory in nature.
Conclusion
For patients with a known autoimmune disorder who present with profound hearing loss, HP should be included in the differential, as treatment may lead to complete resolution of cranial nerve deficit.
Author Contributions
Disclosures
Footnotes
No sponsorships or competing interests have been disclosed for this article.
