Abstract
Background:
Total ankle replacement (TAR) is an established treatment for end-stage ankle arthritis, offering pain relief and improved functionality. The Scandinavian Total Ankle Replacement (STAR) was first used in the United States in 1998 and although unilateral STAR has been extensively studied, there is limited documentation on the outcomes of simultaneous bilateral STAR procedures. This study aimed to evaluate the survivability and patient-reported outcomes of simultaneous bilateral STAR, focusing on implant survival rates and improvements in quality of life, function, and pain relief.
Methods:
A retrospective cohort study was conducted on all patients managed with primary TAR performed simultaneously with bilateral STAR prostheses at our institution from July 1998 to February 2008. Demographics and perioperative characteristics were collected. Poisson regression was used for analyzing implant survivability, and generalized linear mixed models were applied to assess patient-reported outcomes, including the visual analog scale (VAS), Short Musculoskeletal Function Assessment (SMFA), 36-Item Short Form Health Survey (SF-36), and American Orthopaedic Foot & Ankle Society (AOFAS) hindfoot, at multiple follow-up time points.
Results:
Twenty-one patients were included for a total of 42 implants. The average length of follow up was 4.9 ± 2.7 years. The implant failure rate at 5 years was 12.0% (95% CI 4.5%-32.0%). The last recorded failure occurred at 8 years postimplant, with an implant failure rate of 19.2% (95% CI 17.2%-51.1%). Statistically significant improvements were observed in all patient-reported outcome measures from baseline to the 5-year follow-up (P < .05) except SMFA bother because of high variability and small sample size.
Conclusion:
At midterm follow-up, patients who underwent bilateral simultaneous TAR with the STAR prosthesis experienced a significant and durable improvement in patient-reported outcome scores across multiple functional indices. Failure rates were within range to those reported in previous studies evaluating unilateral STAR, with an estimated average survivorship of 80.8% at 8 years.
Level of Evidence:
Level IV, case series.
Keywords
Introduction
Total ankle replacement (TAR) has become a viable alternative to ankle arthrodesis for severe ankle arthritis, offering pain relief, enhanced joint function, and the preservation of range of motion. The Scandinavian Total Ankle Replacement (STAR) was first implanted in the United States in 1998 as a compassionate use device before obtaining FDA approval in 2009. 18 Studies of its use demonstrated promising results in terms of outcomes and survivorship in unilateral recipients. However, despite being a widely studied TAR implant, there is still a scarcity of data on the outcomes of simultaneous bilateral procedures, underscoring the need for further investigation.
Patients with ankle osteoarthritis experience a significant decline in quality of life, comparable with that seen in severely disabling conditions such as end-stage kidney disease, congestive heart failure, and end-stage hip arthritis. 12 Although the survival of TAR implant systems is crucial to patient outcomes and well documented in the literature,17,21 long-term patient-reported data on quality of life and functional improvements remain notably lacking.11,19,22
TAR candidates who present with ankle arthritis of both ankles may require bilateral TAR procedures. In this study, "bilateral" TAR will subsequently refer to patients who underwent TAR on both ankles using STAR prostheses during the same surgical session, rather than undergoing two separate unilateral procedures. Studies have explored the biomechanical benefits and patient satisfaction following unilateral TAR, but these findings are not directly transferrable to bilateral cases, where the dynamics of recovery and adaptation may differ notably. 8 Moreover, data on implant survivability and the trajectory of patient-reported outcome (PRO) measures in bilateral TAR recipients are limited,1,4,9 and even more so for those specifically evaluating the STAR implant, 14 providing insufficient evidence to assess comprehensively the effectiveness of this approach.
Given these gaps, this study aimed to elucidate midterm clinical outcomes of patients who underwent bilateral STAR procedures. By evaluating implant survival rates and PROs of pain, function, and quality of life over a period of up to 8 years, this study seeks to contribute valuable insights to the existing body of literature, offering a more robust basis for clinical decision making in the treatment of bilateral end-stage ankle arthritis.
Methods
Study Design and Population
A retrospective cohort study design was employed to assess all patients who received simultaneous bilateral STAR implants. To identify eligible patients, an internal database of all patients undergoing TAR at our single tertiary care center was reviewed. All patients who underwent a primary simultaneous bilateral TAR with usage of the STAR prosthesis from July 1998 through February 2008 were included in this study. Bilateral STAR TAR was indicated in patients with bilateral advanced or end-stage arthritis that were severely symptomatic and unresponsive to conservative treatment, provided there were no contraindications to surgery. Patients undergoing unilateral, staged, or revision STAR TAR were excluded. All surgeries were conducted at a single institution by fellowship-trained foot and ankle surgeons with substantial expertise in performing TAR. The institutional review board approved the study, and all patients provided informed consent for the use of their data in research.
Data Collection
Demographic and clinical data were exported from the REDCap database and included gender, race, age, diabetes status, body mass index (BMI), smoking status, American Society of Anesthesiologists (ASA) score, and surgery duration.
PRO measures were gathered preoperatively and postoperatively using standardized assessments frequently used to evaluate functional outcomes in patients undergoing TAR. These PROs included the visual analog scale (VAS) pain score, Short Musculoskeletal Function Assessment (SMFA) function and bother questionnaires, the 36-Item Short Form Health Survey (SF-36), and the American Orthopaedic Foot & Ankle Society (AOFAS) summary, pain, and function subscales. PRO scores of the eligible patients were exported from the REDCap database for analysis. Follow-up time points were compiled into 1-year buckets, in which the patient’s closest observation to each 1-year follow-up time point was included. Follow-up after 7 years was excluded because of limited sample size available at these later time points.
Additionally, implant failure data were exported for analysis. Failure was defined as need for revision surgery including implant removal or exchange, arthrodesis, or amputation.
Statistical Analysis
Descriptive baseline characteristics were summarized on the patient level (at first implant). Continuous variables are displayed as mean ± standard deviation (SD) for normally distributed variables, or median with 25th and 75th percentiles (Q1-Q3) and range for skewed variables. Categorical variables are presented as frequency and percentage of nonmissing values.
Poisson regression modeling was performed with implant failure as the endpoint. The log-transformed follow-up time (last follow-up or death) was treated as an offset parameter. A random intercept was included in the model to account for patients having 2 observations (one for each side). This method provides robust incidence rate estimates despite incomplete or variable follow-up intervals. The estimated incidence rate of implant failure at 5 years and the last recorded time to event (8 years) are reported with 95% CIs.
Comparative analyses between the preoperative and follow-up PROs were depicted using generalized linear mixed effects models. Models included the respective preoperative value and year as the primary predictors. A random intercept was included in the model to account for each patient having 2 sides. The least squares mean (LS-mean) at each time point was compared to baseline. P values were adjusted for multiple testing using the Dunnett post hoc method.
Analyses were conducted using SAS 9.4 (SAS Institute Inc, Cary, NC), and statistical significance was assessed at α = .05.
Results
Patient Characteristics
The study included 21 patients who received bilateral STAR implants, totaling 42 implants. Patients were primarily female (n=13, 61.9%) and all were Caucasian. The median (Q1-Q3) age at surgery was 63.5 years (48.5-66.0). The median (Q1-Q3) BMI was 24.8 (23.0-31.2), the majority of patients had never smoked (n=12; 57.1%), did not have a diagnosis of diabetes at the time of surgery (n=18; 85.7%), and were ASA 3 (n=14; 66.7%). Preoperative diagnoses included post-traumatic (n=9; 42.9%), rheumatoid (n=8; 38.1%), and osteoarthritis (n=4; 19.0%). Complete baseline characteristics are detailed in Table 1.
Demographics and Clinical Characteristics (N=21).

Abbreviations: ASA, American Society of Anesthesiologists; BMI, body mass index.
Implant Survivorship
Of the 42 implants, 5 had a failure (11.9%). The average number of years of follow-up (defined as time from implant to last date of contact, implant failure, or death) was 4.88 ± 2.73 years (range, 0-10 years). The 5-year survivorship was 88.0% with an implant failure incidence rate of 12.0% (95% CI 4.5%-32.0%). The last event occurred at 8 years postimplant. At this time, the estimated incidence rate of implant failure based on Poisson regression modeling was 19.2% (95% CI 17.2%-51.1%).
Patient-Reported Outcomes
There was overall a significant improvement in PRO scores after bilateral STAR procedures. Table 2 summarizes baseline and follow-up values for the VAS, SMFA, SF-36, and AOFAS measures. Specific results of PRO improvements are discussed in further detailed below.
Patient-Reported Outcome Measures (Baseline Through 7-Year Follow-up).
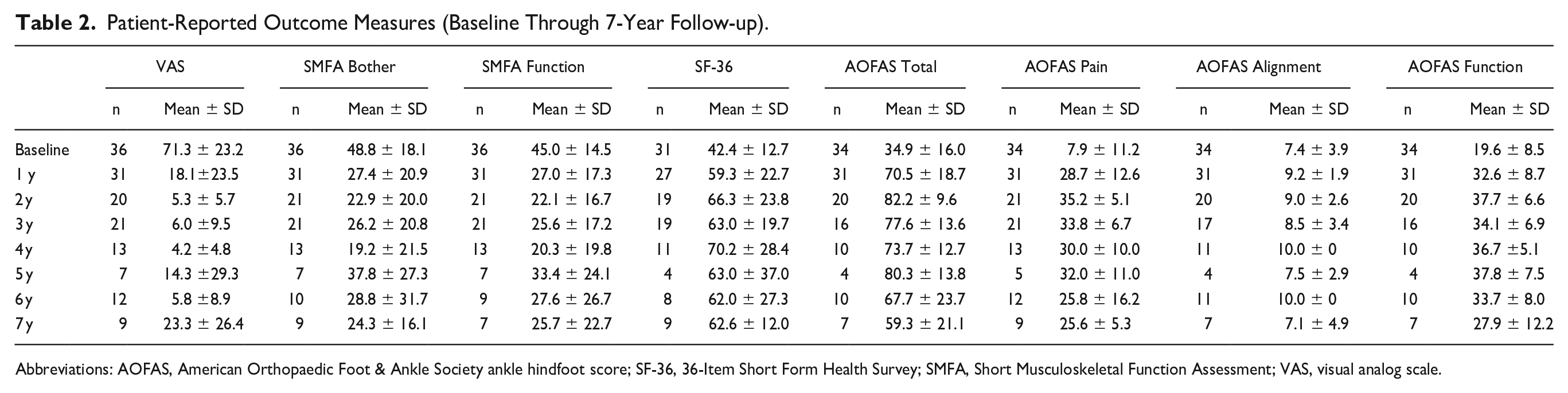
Abbreviations: AOFAS, American Orthopaedic Foot & Ankle Society ankle hindfoot score; SF-36, 36-Item Short Form Health Survey; SMFA, Short Musculoskeletal Function Assessment; VAS, visual analog scale.
VAS
Preoperative VAS scores were initially high (71.3 ± 23.2), indicating significant pain levels. Following the implantation of the STAR prosthesis, VAS scores markedly decreased and remained consistently lower throughout the follow-up period (Figure 1). Every follow-up time point demonstrated significant reductions from baseline, with all adjusted P values <.001, indicating substantial and sustained pain relief.

Box plots showing VAS scores over time, with a significant decrease in pain levels post-implantation and sustained improvements across all follow-up years. VAS, visual analog scale.
SMFA indices
Preoperative SMFA bother and function scores were relatively high, indicating considerable bother and functional impairment because of ankle issues. Postimplantation, both scores decreased and remained stable (Figure 2). All follow-up time points for the SMFA function index were significantly lower than baseline, whereas for the SMFA bother index, all follow-up time points except for year 5 were significantly lower than baseline (all P <.05).

Box plots of SMFA bother (left) and function (right) indices over time, illustrating notable decreases in bother and improvements in functional outcomes postimplantation, with scores remaining lower than baseline across most follow-up years. SMFA, Short Musculoskeletal Function Assessment.
SF-36
Preoperative SF-36 scores were relatively low, indicating poor general health. Following STAR implantation, these scores improved and remained stable (Figure 3). Except for year 7, all follow-up time points were significantly higher than the baseline (all P < .05), highlighting enhanced general health and well-being among the patients.

Box plots of SF-36 scores over time, depicting an increase in general health postimplantation with sustained improvements compared with baseline. SF-36, 36-Item Short Form Health Survey.
AOFAS hindfoot scores
Preoperative AOFAS hindfoot scores, including hindfoot pain and function subscales, were relatively low, indicating poor foot and ankle health and significant pain. Postbilateral STAR implantation, these scores improved and remained stable, with all follow-up time points significantly higher than baseline (all P <0.05) except for hindfoot function at year seven (Figure 4).

Box plots of AOFAS Hindfoot score over time (top), and subscales showing pain relief (bottom left), and improvement in function (bottom right) post bilateral STAR implantation, with scores significantly higher (all P <.05) than baseline across the follow-up period, except for function at year 7. AOFAS, American Orthopaedic Foot & Ankle Society ankle hindfoot score.
Discussion
This study provides valuable insights into the outcomes of bilateral STAR TAR in patients with end-stage ankle arthritis of both ankles. Our findings demonstrate sustained implant survivability, and significant improvements in PROs and overall quality of life.
In general, the STAR implant system has shown variable survivorship rates across studies. Five-year survival rates range from 85.9% to 93.3%, whereas 10-year rates vary from 71.1% to 80.3%.15,23,24 Long-term follow-up reveals declining survivorship, with single-center survivorship reports of 45.6% at 14 years and 63.3% at 15 years.7,16
There are few studies in the literature that examine bilateral TAR, with ongoing debate regarding the effectiveness of sequential vs simultaneous TAR for treating end-stage ankle arthritis. Barg et al 3 evaluated 26 patients who received bilateral simultaneous HINTEGRA total ankle system TAR and observed a secondary revision rate of 11.5% for 52 ankles, with notable improvements in VAS pain and SF-36 scores. Their study reported survival rates of 91% at 5 years and 78% at 8 years. 3 In comparison, our analysis revealed a 5-year implant survivorship of 88.0% and an 8-year survivorship of 80.8%. These comparable survival rates suggest that simultaneous bilateral STAR procedures offer similar durability and effectiveness in pain relief and our significant reductions in VAS scores align well with the pain improvements documented in their study.
Another study by Barg et al. compared bilateral sequential TAR to unilateral TAR, finding that although unilateral TAR had better immediate postoperative outcomes, results were equivalent at 1- and 2-year follow-ups. 4 This suggests that bilateral STAR can achieve similar long-term outcomes to unilateral TAR after the initial recovery period.
Similarly, Desai et al 9 examined the quality of life in 53 patients with sequential bilateral TAR compared to 106 with unilateral TAR, with various implant types pooled together in analysis. Their results found equivalent improvements in PROs and component survival rates despite worse preoperative health status in the bilateral group. The postoperative SF-36 PCS scores showed similar improvement from baseline in both patients with unilateral and bilateral TAR. Metal component revision was indicated in 6% of the unilateral group and 8% of the bilateral group, with a mean implant survival time of 10.9 years in the bilateral cohort and 9.2 years in the unilateral group. In our study of simultaneous bilateral STAR implants, we observed significant improvements in SF-36 scores, indicating enhanced general health and well-being postimplantation. Our findings align with the Desai et al results, as we also noted equivalent improvements in PROs despite the complexities associated with bilateral procedures. 9 Additionally, our study reported a comparable implant survival rate, with 80.8% survivorship at 8 years, further supporting the effectiveness of bilateral STAR procedures.
Fletcher et al 10 evaluated 50 patients (100 ankles) undergoing either simultaneous or sequential bilateral TAR with various implant types. The results showed that both groups had significant improvements in PROs, with no significant differences in complication rates or reoperations between the groups. 10 The present study's focus on the STAR system offers a more specific perspective, and our similarly high patient satisfaction rates align with these broader findings, reinforcing the viability of bilateral STAR TAR. Unlike fixed-bearing designs, the STAR prosthesis uses a mobile-bearing polyethylene component, which may influence wear patterns and revision rates.
Perhaps most relevant is the first report of a series of patients undergoing bilateral simultaneous TAR by Karantana et al. in 2010. 14 This study included five patients, all of whom received the STAR system implants. Postoperatively, these patients experienced no complications and reported high satisfaction levels as measured by AOFAS scores. However, the study did not provide data on implant survivorship. In our larger study, we similarly observed high patient satisfaction and significant improvements in functional outcomes, including AOFAS scores, which further support the viability of simultaneous bilateral TAR using the STAR system for patients with bilateral end-stage ankle arthritis. Additionally, our study contributes valuable data on implant survivorship, enhancing the existing literature on the efficacy and durability of bilateral STAR procedures.
It is important to note that while unilateral TAR has been extensively studied, bilateral TAR presents unique challenges and considerations. The dynamics of recovery and adaptation in bilateral cases differ notably from unilateral cases, as patients must manage rehabilitation on both sides simultaneously. Despite these challenges, our study indicates that bilateral STAR provides comparable benefits to unilateral procedures in terms of pain relief, function, and quality of life. In contrast to unilateral arthroplasties, where discrepancies in limb function can pose challenges, a bilateral approach appears to mitigate some of these concerns, as it could potentially facilitate a more balanced and effective rehabilitation phase because of decreased pain and the relative symmetry of the newly replaced joints. When comparing to bilateral knee or hip replacements, which have well-documented outcomes,2,5,13 bilateral TAR with STAR implants shows a similarly positive impact on PROs. Studies on bilateral knee replacements have demonstrated that although the initial recovery may be more challenging compared to unilateral procedures, the long-term outcomes are generally favorable.6,13 This parallel suggests that bilateral STAR can achieve similar success, provided the patients are carefully selected and managed.
This study has several limitations. It is recognized that subsequent modifications to implant design and surgical technique may have been made since this original cohort underwent bilateral TAR with STAR implants. The sample size is relatively small, and the follow-up duration varies among patients. It is possible that earlier cases in the cohort were subject to higher failure rates because of the learning curve associated with TAR procedures. However, no correlation between failures and early-phase surgeries was observed in this study, even though this phenomenon has been reported in other early STAR cohorts. 20 Given this is a single-center study, the results may not be generalizable to all patient populations. A further limitation of our study is that we did not systematically record minor complications such as wound healing problems or implant-associated fractures. Additionally, the retrospective nature of the study introduces potential biases related to data collection and reporting. Because this is a historical cohort, we used legacy PRO instruments (VAS, SMFA, SF-36, AOFAS). Current practice often employs PROMIS measures, which may provide additional precision in assessing foot and ankle function. Future, prospective, studies with larger cohorts, validates PROs, and longer follow-up periods are needed to validate our findings and further explore the long-term outcomes of bilateral STAR. 20 Furthermore, bilateral TAR should be evaluated in a gait laboratory to compare its effects with both unilateral TAR and cases of ankle arthritis without joint replacement. There is currently a lack of data regarding how ankle joint mechanics, both before and after TAR, impact the ankle joint and the forces experienced by the hip and knee.
In conclusion, bilateral STAR appears to have been a viable and effective option for patients with bilateral end-stage ankle arthritis, providing significant pain relief, functional improvements, and enhanced quality of life. The survivorship rates are comparable to those reported for unilateral STAR, suggesting that bilateral procedures do not compromise implant longevity. These findings contribute valuable information to the existing body of literature and support the continued use and refinement of this treatment modality in appropriate patient populations.
Supplemental Material
sj-pdf-1-fao-10.1177_24730114251329349 – Supplemental material for Outcomes of Bilateral Scandinavian Total Ankle Replacement: A Detailed Analysis of Implant Survivability and Patient-Reported Outcomes
Supplemental material, sj-pdf-1-fao-10.1177_24730114251329349 for Outcomes of Bilateral Scandinavian Total Ankle Replacement: A Detailed Analysis of Implant Survivability and Patient-Reported Outcomes by Alexandra Hunter Aitchison, Albert T. Anastasio, Kevin A. Wu, Emily Poehlein, Cynthia Green, Samuel B. Adams, James K. DeOrio, Mark E. Easley and James A. Nunley in Foot & Ankle Orthopaedics
Footnotes
Ethical Approval
Ethical approval for this study was obtained from the Duke Health Institutional Review Board (Pro00107443)
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Samuel B. Adams, MD, reports general disclosures of royalties from DJO/Enovis and Conventus, and consulting fees from DJO/Enovis, Conventus, Stryker, and Artelon. James K. DeOrio, MD, reports general disclosures of royalties from Exactech, Merete, and Stryker; consulting fees from Additive, Crossroads, Exactech, and Mirus; and research support from Exactech and Treace Medical. Mark E. Easley, MD, reports general disclosures of royalties from Exactech and Treace Medical; consulting fees from Exactech, Paragon 28, and Treace Medical; and research support from Exactech, Paragon 28, and Treace Medical. James A. Nunley, MD, reports general disclosures of royalties from Exactech; consulting fees from Exactech, Vilex, and Mirus; and research support from Trimed and Treace Medical; and stock or stock options with Bristol-Myers Squibb. Disclosure forms for all authors are available online.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
