Abstract
Komodo dragon attacks are rare occurrences, especially in the United States. Attacks are believed to be highly infectious and venomous, leading to demise of its prey. We present a case of a 43-year-old female zookeeper attacked by an adult dragon leading to tendon and neurovascular injuries.
Introduction
The Komodo dragon (Varanus komodoensis) is a carnivorous varanid lizard that is native to the islands of Indonesia. 3 They are known for being the world’s largest lizard, reaching a mass of up to 90 kg and a length of 3 meters. 5 Because of their large size, these lizards feed on prey that equal or exceed their own mass, including water buffalo, pigs, and deer. 3,5 The mechanism with which the dragon is capable of killing such large prey is an area of interest and debate. In addition to direct physical trauma, it has been hypothesized that pathogenic bacteria and/or venom, from the bite, can contribute to the ultimate demise of its prey. 3
Although there are reports of multiple attacks on humans at Komodo National Park, Indonesia, reports of attacks in the United States are rare occurrences. 14 However, Komodo dragons are popular exhibits in zoos throughout the United States, which pose a risk to the zookeepers that care for them. In addition, nonnative monitor lizards have become established in Florida, after being brought to the United States and released into the wild, thereby increasing the likelihood of human interactions. 18 There are currently only 2 published case reports of Komodo dragon bites in the United States. The first of a zookeeper sustaining a bite to the left small finger by a juvenile dragon that subsequently led to a mallet finger injury. 4 The second incident was also of a zookeeper bitten on the right hand, by a juvenile dragon, sustaining minor lacerations that healed without complications. 7 We present a case of a zookeeper that was attacked by an adult Komodo dragon (Figure 1) who sustained deep lacerations in her left upper and right lower extremities, leading to tendon and neurovascular injuries.

Adult captive Komodo dragon.
Case Presentation
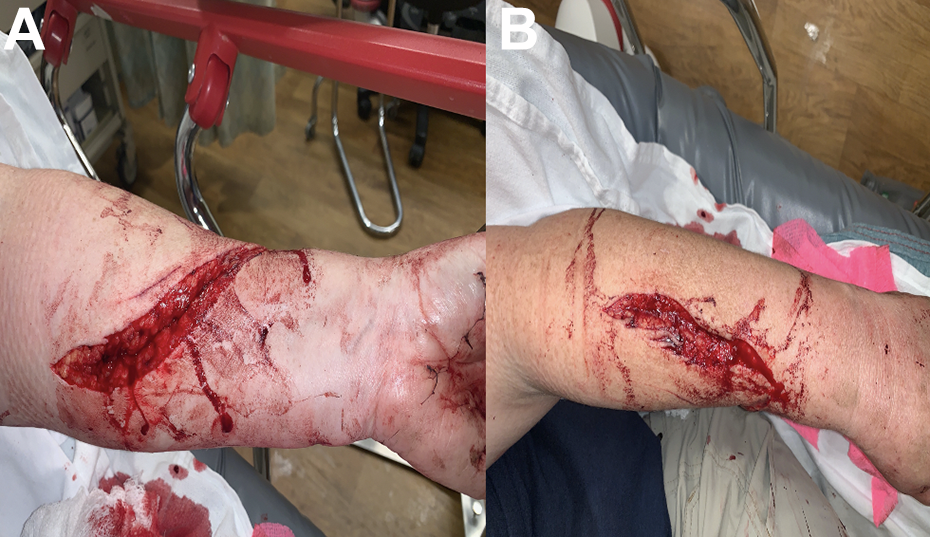
A 43-year-old, left-hand-dominant female zookeeper was attacked by an adult Komodo dragon at the local zoo during feeding. She sustained traumatic lacerations to her left forearm (Figure 2) and right lower leg (Figure 3). Hemostasis was adequately achieved with manual pressure and she was subsequently transported to the emergency department. The orthopedic surgery team was consulted secondary to suspected tendon and neurovascular involvement of her right lower extremity. On physical examination, her vital signs were within normal limits and the Glasgow Coma Scale score was 15. The patient’s left forearm and right lower extremity lacerations had some mild active venous bleeding but no apparent arterial bleeding requiring a tourniquet. The patient had an approximately 15-cm laceration of the volar left forearm and 10-cm laceration over the dorsal left forearm (Figure 2). The right lower extremity sustained multiple traumatic lacerations over the posteromedial and the anterolateral leg (Figure 3). The patient was neurovascularly intact to the left upper extremity and there were no apparent injuries to the flexor or extensor tendons in the forearm or hand. On neurologic examination of the right lower extremity, she exhibited no motor function to the extensor hallucis longus, tibialis anterior, and extensor digitorum longus. She had no sensation to light touch in the deep peroneal nerve distribution but normal sensation in all other distributions. We were unable to palpate a dorsalis pedis pulse; however, she had a 2+ posterior tibial pulse and good perfusion to the foot distally. Shortly after arrival to the emergency department, she was given intravenous piperacillin-tazobactam and 1 dose of levofloxacin per infectious disease team.
(A) Left dorsal forearm traumatic laceration and (B) volar forearm traumatic laceration with no apparent tendon or neurovascular involvement.

(A) Right lower extremity showing several traumatic lacerations over the anterolateral and posteromedial leg. (B) Traumatic lacerations starting over posteromedial leg and continuing posteriorly.
The patient consented and was taken emergently to the operating room for irrigation and debridement (I&D) of her right lower and left upper extremities with placement of negative pressure wound therapy. The volar and dorsal wounds to the left forearm were inspected and found not to violate the fascia. Sharp excisional wound debridement was performed followed by irrigation with sterile saline and placement of negative pressure wound vac. Inspection of the right lower extremity wounds revealed the tibialis anterior, extensor hallucis longus, and extensor digitorum longus, anterior tibial artery, and deep peroneal nerve had been traumatically lacerated. The wounds were thoroughly debrided followed by adequate irrigation with sterile saline (Figures 4 and 5). The anterior tibial artery was found to be thrombosed and the proximal and distal ends were subsequently ligated. The proximal stump of the deep peroneal nerve was traumatically lacerated and we were unable to identify the distal stump. Therefore, a traction neurectomy of the proximal stump was performed. The lacerated anterior compartment tendons were tagged with PDS suture for later repair. Antibiotic beads consisting of vancomycin and tobramycin were made and placed in the anterior compartment wounds followed by placement of a negative pressure wound vac. The infectious disease team was consulted for antibiotic recommendations and the patient was placed on piperacillin-tazobactam for 48 hours postoperatively then transitioned to oral amoxicillin-clavulanate for 10 days. Deep vein thrombosis prophylaxis was initially held for the first 48 hours secondary to the theoretical risk of the dragon’s venom to lead to a coagulopathy.

Left forearm showing volar (left) and dorsal (right) traumatic wounds following initial irrigation and debridement.
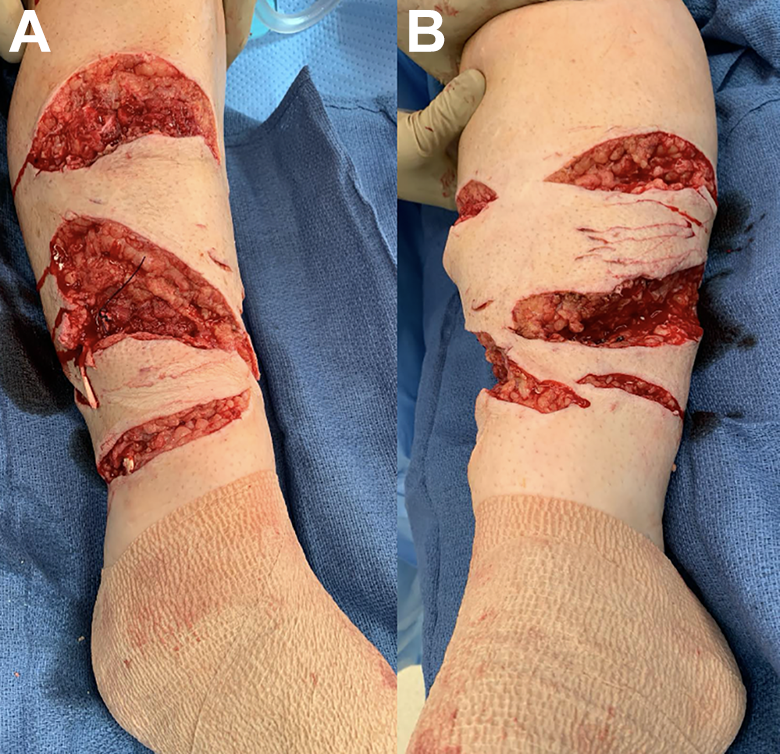
(A) Right lower extremity showing anterolateral traumatic wounds following initial irrigation and debridement (I&D). (B) Posteromedial traumatic wound following initial I&D.
The patient was taken back to the operating room 48 hours later for repeat I&D, tendon repair, and closure of wounds. The left forearm wounds were closed primarily after repeat I&D. The tibialis anterior, extensor hallucis longus, and extensor digitorum longus tendons were repaired with 2-0 FiberWire using a 4-strand Strickland technique. Following tendon repair and closure, there was a 2 × 3-cm and 7 × 8-cm area over the anterolateral leg with exposed viable muscle that could not be closed (Figure 6). Negative pressure wound vac devices were placed and the foot immobilized in maximum dorsiflexion with a short leg splint. The patient exhibited no signs of coagulopathy and was subsequently started on prophylactic enoxaparin sodium (Lovenox) in the hospital and transitioned to daily aspirin on discharge for deep vein thrombosis prophylaxis. Patient subsequently underwent split-thickness skin grafting by the plastic surgery team on hospital day 5 and then discharged home instructed to be nonweightbearing in a maximum-dorsiflexion splint. Patient ultimately healed her wounds and skin graft sites without any issues (Figure 7). She transitioned out of the splint at the 6-week follow-up appointment and was unable to actively dorsiflex her ankle. An ankle-foot orthosis was made in order to assist with ambulation and to prevent equinus contracture. She was then referred to a foot and ankle colleague and a magnetic resonance image was obtained that revealed torn tibialis anterior, extensor hallucis longus, and extensor digitorum longus tendons with retracted muscle indicating a failed extensor tendon repair. Nonoperative and operative options were discussed with the patient, and she elected to undergo posterior tibial tendon transfer to the lateral cuneiform and gastrocnemius recession approximately 3 months after the initial injury. She was placed in short leg cast for 6 weeks postoperatively and then transitioned to a boot and began formal physical therapy. At the 3-month follow-up appointment, she was doing well with active ankle dorsiflexion and good passive range of motion at the ankle (Figure 8). The boot was discontinued and subsequently transitioned to an ankle-foot orthosis so she could go back to work.

(A and B) Anterolateral right lower extremity showing the 7 cm proximal to distal and 8 cm anterior to posterior wound that was unable to be closed following second-stage I&D with closure.

Anterolateral and posteromedial right lower extremity wounds healed at 6 months from initial injury.

Right lower extremity 6 months from initial injury and 3 months postoperation from posterior tibial tendon transfer revealing active dorsiflexion past neutral.
Discussion
Fortunately, Komodo dragon attacks on humans in the United States are rare occurrences. However, because of the rarity of this injury, there is a paucity of evidence in the literature to help guide treatment. The mechanism with which the dragon kills its prey is a topic of debate. One potential mechanism is known as the “bacteria as venom model.” 1,8 A study of wild and captive Komodo dragons revealed 57 species of bacteria isolated from their saliva, with the most common isolates including Escherichia coli, Staphylococcus sp, and Streptococcus sp. 4,12 A study by Montgomery et al 12 revealed that mice injected with Komodo dragon nonfiltered saliva had a higher mortality than those injected with filtered saliva. In addition, Pasteurella multocida was the only bacteria isolated from dying mice injected with Komodo dragon saliva, which is theorized as a contributing factor to the death of its prey. 12 However, Goldstein et al 9 studied the saliva from 16 captive Komodo dragons and found, as with other carnivores, the captive Komodo oral flora is reflective of the gut and skin flora of their recent meals and environment. Therefore, it is unlikely that toxic bacteria reliably induce sepsis, in prey, to the extent that this would become an evolutionarily successful mechanism for V komodoensis to rely on for prey capture. 8,9
A second potential mechanism, discussed in the literature, for which Komodo dragons kill their prey, is through envenomation. 8 Fry et al 8 described venom glands within V komodoensis that contain proteins that may potentially lead to anticoagulative changes (PLA2 toxins) and hypotensive changes (CRISP, kallikrein, and natriuretic toxins) within its prey. In addition, Koludarov et al 10 reported on the fibrinogenolytic activity of varanoid lizard venoms, which could lead to decreased clot formation and increased bleeding times. 6 The V komodoensis is hypothesized to use its serrated teeth in a “grip and rip” mechanism to create deep wounds which allow entry of its venom. 8 Although not a V komodoensis, there is report of a monitor lizard, V bengalensis, bite leading to hemolysis, coagulopathy, rhabdomyolysis, and acute kidney injury that resulted in cardiac arrest. 15 However, this report has been challenged by White et al 19 regarding the identity of the animal responsible for the bite. In addition, Weinstein et al points out that the consensus definition of a venom is “a complex substance produced in a specialized gland and delivered by an associated specialized apparatus that is deleterious to other organisms in a given dosage and is actively used in the subjugation and/or digestion of prey and/or in defense.” 16,17 There is currently no clear objective supporting evidence of a venom function for V komodoensis. 16,17 However, the fact that V komodoensis possess proteins that could potentially lead to anticoagulant and/or hypotensive effects should be considered during evaluation and management.
The most agreed on mechanism with which V komodoensis kills its prey is through soft tissue injury, massive blood loss, and subsequent hypovolemic shock. 16 Although bite forces in V komodoensis are relatively weak, they possess strong postcranial musculature that provides the ability to resist forces generated in pulling on a prey item or a prey item pulling back. 8,13 As a result, they are able to kill large prey by using relatively weak bite forces that are amplified by sharp teeth and postcranial input resulting in significant soft tissue injuries. 8,11,20
Initial management of patients following a Komodo dragon attack should begin with ATLS protocol as with all patients presenting following a traumatic event. After confirmation of an airway, adequate breathing/ventilation and hemodynamic stability, antibiotic prophylaxis, and tetanus status should be addressed. Plain film imaging should also be obtained to ensure no underlying fracture and/or foreign body.
For patients with minor wounds, local irrigation and loose closure with prophylactic antibiotics is recommended. The preferred antibiotic agent for prevention of infection due to animal bite wounds is amoxicillin-clavulanate. 2 For patients unable to take this regimen, an agent with activity against P multocida and an agent with anaerobic activity (eg, doxycycline and clindamycin) should be chosen. 2 In addition, it may be beneficial to observe patients for a short time to ensure no signs of envenomation such as hypotension or hemodynamic instability. Although, there is currently no evidence to suggest clinically important envenomation in humans following varanid lizard bites, it is still certainly a possibility. 4,7,19
Patients presenting with more substantial wounds may require operative irrigation and debridement in a staged fashion. Antibiotic prophylaxis in this setting may require intravenous regimens for empiric prophylaxis. Recommended intravenous regimens include piperacillin-tazobactam or ceftriaxone plus an agent with anaerobic activity such as metronidazole or clindamycin. 2 We recommend continuing intravenous antibiotics for 48 hours post I&D followed by 7-10 days of oral amoxicillin-clavulanate. Because of the significant soft tissue injury and wound contamination in this patient, we chose to perform a multistaged irrigation and debridement. Deep vein thrombosis prophylaxis was held during the first 48 hours to ensure no potential anticoagulant effects from the “venom.” We did not encounter any abnormal bleeding, hypotensive episodes, or concerns of sepsis during this patient’s hospital stay. However, the clinician should be wary of a patient presenting with coagulopathy and hypovolemic shock because of the potential for significant soft tissue injury and envenomation from a Komodo dragon bite.
Supplemental Material
Supplemental Material, sj-pdf-1-fao-10.1177_24730114211015623 - Beware of the Dragon: A Case Report of a Komodo Dragon Attack
Supplemental Material, sj-pdf-1-fao-10.1177_24730114211015623 for Beware of the Dragon: A Case Report of a Komodo Dragon Attack by Brandon S. Boyd, Felipe Colon, Jesse F. Doty and Kristopher C. Sanders in Foot & Ankle Orthopaedics
Footnotes
Ethics Approval
Ethical approval was not sought for the present study because a case report does not require IRB review and approval.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article. ICMJE forms for all authors are available online.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
