Abstract
Diabetic foot ulcers and infections are common complications of diabetic foot disease. Additionally, these complications are a common cause of morbidity and impose a substantial burden to the patient and society. It is imperative to understand the major contributing factors, namely, diabetic neuropathy, peripheral arterial disease, and immune system dysfunction in order to guide treatment. Management of diabetic foot disease begins with a detailed history and thorough physical examination. This examination should focus on the manifestations of diabetic neuropathy and peripheral arterial disease, and, in particular, any evidence of diabetic foot ulcers or infection. Prevention strategies should include a multi-disciplinary approach centered on patient education.
Introduction
Diabetes mellitus (DM) is a serious systemic disease with an increasing incidence in the United States and worldwide. More than 30 million people in the United States are affected by DM, and much like a malignant disease, multisystem organ involvement is frequent. 1 Lower extremity manifestations are frequently associated with substantial morbidity and mortality. Diabetic foot disease arises from chronic pathologic processes such as neuropathy, peripheral artery disease (PAD), biomechanical problems, and impaired wound healing. Although patients with diabetic foot disease have high rates of premature mortality, they have been shown to fear major amputation more than death. 54
One of the most common problems in the care of the diabetic patient is the diabetic foot ulcers (DFU), with studies reporting an average annual incidence of 2.2%. 2 Even with appropriate care, DFUs can ultimately lead to serious complications such as infection, amputation, and even death. Infections occur in up to 58% of patients presenting with a new foot ulcer. 36 A European study group found that up to 5% of diabetic patients with a DFU required a major amputation in 1 year. 37 Five-year mortality rates are as high as 45% for neuropathic ulcers and 55% for ischemic ulcers. 32 These rates have been shown to be similar or worse than many common types of cancers, including prostate, breast, and colon 21,22,48 (Figure 1).

Chart illustrating the relative 5-year survival of patients with newly diagnosed diabetic foot ulcers in comparison to common cancers. As seen in the chart, the survival in patients with diabetic foot ulcers is worse than many common cancers.
In addition to the morbidity of diabetic foot disease, there are major socioeconomic implications associated with this disease. Hospital admissions involving DFU can average more than $100 000 per admission if amputations or revascularization is required. 43 One study reported that the hospital cost for managing a complicated heel ulcer with PAD worldwide was estimated to range from $188 000.00 in the United States to $3060.00 in Tanzania. 13 Given the profound impact of diabetic foot disease on patients and society, there has been a growing body of literature surrounding this topic. This review focuses on the pathophysiology, evaluation, treatment, and prevention of diabetic foot ulcers and infections.
Pathophysiology
The etiology of diabetic foot disease is multifactorial, and includes complications of diabetic neuropathy, vasculopathy, immunopathy, and poor glycemic control. Diabetic neuropathy results in sensory, motor, and autonomic nerve dysfunction and is the most common cause of diabetic lower extremity ulcers. With proper screening, approximately 75% of diabetic patients undergoing foot and ankle surgery will be found to have neuropathy. 44 Because of an inability to determine injury or trauma, peripheral neuropathy is mostly associated with high rates of skin breakdown and neuropathic fractures. The inciting trauma could be caused simply by ill-fitting shoes or minor sprains and strains. The risk of developing a first DFU has been shown to be 7 times higher in those with moderate or severe sensory loss compared to patients with preservation of sensation. 57 Without protective sensation, a neuropathic patient lacks the physical symptoms that would normally cue healthy individuals to examine or rest their feet, thereby increasing the extent of skin damage before presenting for treatment. Autonomic neuropathy also contributes to ulcer formation as it affects both physiologic secretions and the arteriovenous systems leading to dry, flaking, and fragile skin. This increases the risk for fissuring and skin breakdown, creating potential sites of infection. Motor neuropathy can lead to structural changes to the foot. These changes are in part due to muscular imbalance and weakness caused by intrinsic atrophy, frequently manifesting as claw toes, hammertoes, prominent metatarsals, and other deformities. These deformities change pressure patterns on the foot making certain areas more susceptible to trauma or ulceration (Figure 2).
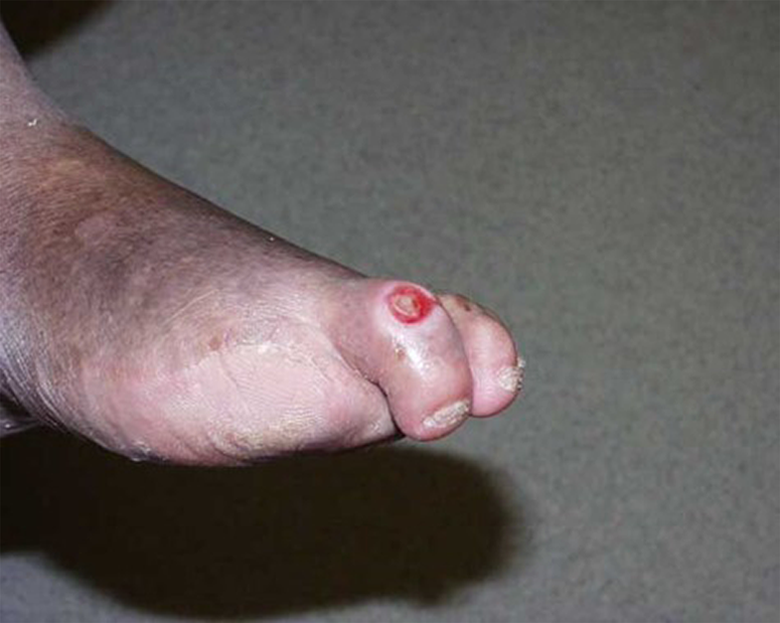
Neurogenic claw toes in a patient with previous toe amputations. The deformity resulted in dorsal toe ulceration secondary to trauma from shoe wear.
Peripheral arterial disease (PAD) is commonly seen in combination with neuropathy, in the diabetic population, and can contribute to foot complications. Approximately 50% of patients with diabetic foot disease have some degree of PAD. 55 Compared to diabetic patients with Charcot neuropathy, patients with DFUs are significantly more likely to have PAD, critical limb ischemia, and more often require revascularization. 53 Endothelial damage and vessel sclerosis of both large and small vessels leads to decreased peripheral perfusion. This places patients at an increased risk for ulceration and leads to impaired wound-healing and infection-fighting abilities. Moreover, patients with diabetes have an impaired ability to mount an inflammatory response to infection (immunopathy). Impaired neutrophil function, chemotaxis, phagocytosis, as well as a decreased t-cell response have been found in patients with diabetes compared to those without. 39 The root etiology for this dysfunction appears to be hyperglycemia, which impairs host defenses at a cellular level, affecting leukocytes, macrophages, and other cell types. 6 PAD and immunopathy do not directly cause ulceration; however, these patient factors can increase the risk of diabetic foot complications in those with diabetic neuropathy.
Evaluation
Clinical Examination
Proper management of diabetic foot problems begins with a thorough evaluation of the patient. A detailed history should be obtained, including the duration of diabetes, insulin dependence, existing comorbidities, operative history, family history, social history (tobacco or alcohol abuse, support network), and current medications. Symptoms of claudication and neuropathy are of particular importance and should be elicited in the history.
Physical examination should include an assessment of protective sensation with a 5.07 Semmes-Weinstein monofilament, vibratory perception with a 128-Hz tuning fork, and ankle reflexes. 44 The skin should be examined for signs of autonomic neuropathy, such as dry skin with cracks or fissures (Figure 3). Signs of motor neuropathy can be seen as muscular imbalances that frequently cause claw toes, hammer toes, or prominent metatarsal heads. Swelling is common; it should be quantified using either a tape measure or water volume displacement measurements. The standing alignment of the foot and ankle should be noted. The evaluation of gait may disclose abnormal pressure patterns, instability, or tightness of the gastrocnemius-soleus complex. Vascular assessment involves palpation of the popliteal, posterior tibial, and dorsalis pedis pulses. Secondary signs of peripheral artery disease include coolness of the foot, decreased or absent hair growth, shiny skin, or dependent rubor.

Left foot photograph of a patient with autonomic neuropathy and a plantar diabetic foot ulcer. The dry skin is prone to cracking, fissuring, and ulceration as illustrated beneath the first metatarsal head.
Vascular Tests
Patients with diabetes are at risk for developing both macrovascular and microvascular disease. Macrovascular disease associated with diabetes typically involves the deep femoral artery and the infrapopliteal trifurcation. 55 Microvascular disease typically presents as diabetic retinopathy, diabetic nephropathy, or diabetic neuropathy. Noninvasive vascular studies can assess flow, velocity, and waveforms (ie, triphasic, biphasic, or monophasic), and abnormal test results are indicative of macrovascular disease. The most commonly performed vascular studies include calculation of the ankle-brachial index (ABI), toe-brachial index (TBI), and absolute toe pressures (Figure 4). An ABI <0.91 and/or a TBI <0.7 indicates the presence of PAD. 53,55 The ABI can be falsely elevated in patients with calcinosis of the media of arterial walls, which results in reduced arterial compliance. For this reason, a toe brachial index (TBI) should complement the ABI. The TBI is less likely to be falsely elevated because the digital vessels are not affected by calcifications to the same degree as the proximal vasculature. The inclusion of the TBI has been shown to improve the ability to diagnose PAD. 55 Although the absence of peripheral pulses is associated with a 4.9 times higher likelihood of PAD, the presence of pulses does not exclude PAD. 55 Transcutaneous oxygen pressures (TcPO2) can provide additional information about wound healing potential and can identify changes in the microvascular circulation. Any evidence of ischemia should warrant referral to a specialist experienced in endovascular therapy and angiography. Depending on the location of the occlusion, this may include vascular surgeons, interventional radiologists, or interventional cardiologists. Revascularization can restore circulation at the macrovascular level; however, pathologic changes in the microvascular system will persist and may negatively impact wound healing.

Algorithm assessing the evaluation of peripheral artery disease in patients with diabetes
Diabetic Foot Ulcers
Diabetic foot ulcers (DFUs) frequently occur in diabetic patients as a result of repetitive trauma or abnormal pressure patterns in an insensate foot. A recent meta-analysis of diabetic patients reported a nearly 2-fold increased risk of all-cause mortality in diabetic patients with a DFU compared to diabetic patients without ulceration (relative risk [RR] 1.89, 95% confidence interval [CI] 1.60, 2.23). 9 In the presence of an ulcer, a provider should question the patient regarding the onset, duration, and progression of the wound. Documentation and measurements should be recorded noting the location, size, depth, and wound margins, in order to monitor its course.
Classification
Classification of DFUs serves to improve communication amongst providers and stratifies severity. The most commonly used classifications are the Wagner and University of Texas Wound Classification Systems. 34 The Wagner system focuses on ulcer depth, the presence of abscesses, osteomyelitis, joint sepsis, and gangrene but fails to characterize the extent of ischemia or neuropathy (Table 1). The University of Texas Wound Classification System provides the additive benefit of documenting the presence of ischemia and has been shown to be a better predictor of outcomes 24,34 (Table 2).
Wagner Wound Classification. 37

University of Texas Wound Classification.

Treatment
The initial treatment of DFUs includes sharp debridement, offloading, and local wound care. Debridement converts a chronic wound into an acute wound by removing necrotic tissue, decreasing bacterial burden and biofilm, and reducing inflammatory cytokines. This promotes an environment for healthy granulation tissue formation (Figure 5A, B). This can often be accomplished without anesthesia because of the peripheral neuropathy. Sharp debridement (Figure 5A, B) converts this chronic wound into an acute wound, decreasing biofilm burden and reducing plantar pressure. Achieving a healthy bleeding ulcer base combined with proper offloading is essential for healing. A healthy wound environment can be maintained with topical therapies and appropriate dressings. There is a paucity of evidence supporting the use of any one particular dressing or topical wound product over another. 20 Modification of host factors is critical, including optimizing glycemic control, smoking cessation, improving limb vascularity, and optimizing nutrition. If less than 50% reduction in wound size occurs over the course of 4 weeks with good offloading and wound care, the chance of spontaneous healing is low. 42 At this point, reassessment for signs of infection, nutritional deficiency, or arterial insufficiency is paramount. If these factors have been addressed, the use of advanced skin substitutes or bioengineered alternative tissues should be considered to facilitate healing. Multiple types of advanced wound care treatments are available to include skin grafts, living skin equivalents, growth factors, placental and amniotic membranes, xenografts and cadaver skin. However, further studies are still needed to demonstrate the superiority of any specific modality.
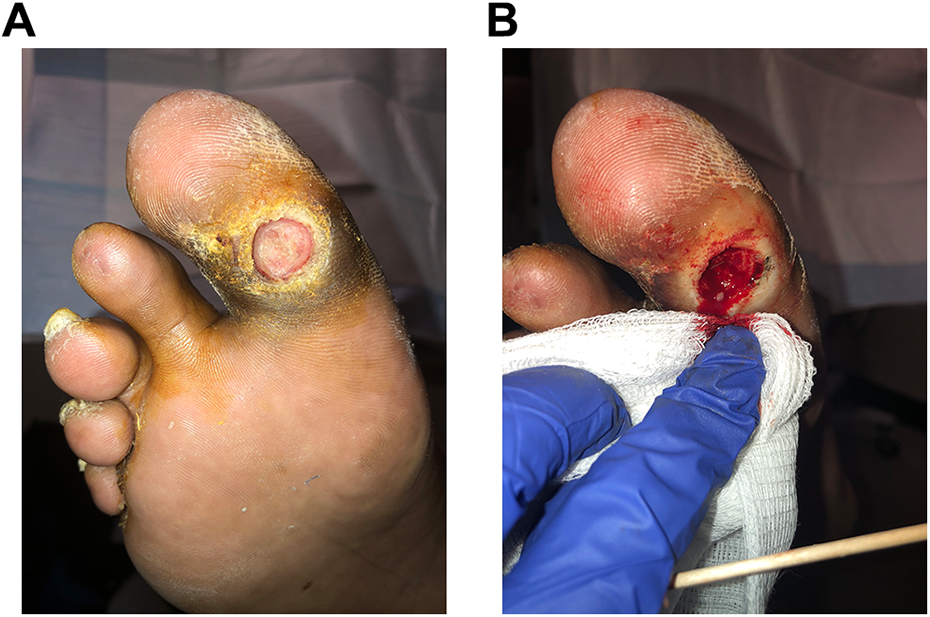
Illustration of (A) pre- and (B) postdebridement photos of a clinically uninfected right plantar hallux ulcer performed in the outpatient clinic. Sharp debridement with a No. 15 blade converts a chronic wound to an acute wound.
Offloading
One of the most important components of DFU treatment is offloading. Offloading can be accomplished through the use of footwear modification, braces, walkers, and total-contact casting. Total-contact casting or a nonremovable boot walker remains the gold standard for offloading. 26 A recent systematic review and meta-analysis demonstrated improved wound healing with the use of these orthoses over removable cast walkers, therapeutic shoes, and conventional therapy. 17 The superiority of nonremovable orthoses over removable ones is due to compliance with offloading, as patients wear their removable-cast walkers for only a total of 28% of their total daily activity. 5 Thus, physicians should choose an orthosis that does not rely on patient compliance, such as total-contact cast or nonremovable boot, to optimize wound healing. Further research is needed to determine the timing of removal of the offloading orthosis after ulcer healing as well as the efficacy of footwear modifications in preventing ulcers. Operative off-loading techniques for plantar forefoot ulcers include percutaneous and open Achilles tendon lengthening. Operative off-loading should be performed when the ankle is unable to dorsiflex to neutral or forefoot pressures are greater than 100 lb/in. 2 Achilles lengthening has been shown to assist in wound healing and decreases recurrence rates at midterm follow-up. 33
Advanced Healing Modalities
Recent attention has been directed toward advanced healing modalities, such as hyperbaric oxygen (HBO) and negative-pressure wound therapy. There is considerable debate on the efficacy of HBO, with a recent prospective, double-blind, randomized controlled trial failing to show any reduction in amputation rate with HBO and wound care when compared to wound care alone. In comparison, negative-pressure wound therapy has been shown to improve wound healing when compared to advanced moist wound therapy. 7,16,18,31 Current evidence does not support the use of antibiotic therapy in the management of noninfected ulcers (Figures 3 and Figure 5A).
Recurrence
Patient education plays a key role in preventing recurrence by increasing patient compliance through improvement in understanding. Despite these interventions, recurrence rates of ulcers remain high, with some studies reporting upwards of 40% recurrence. 10,11,35 The high recurrence rate is likely due to the fact that the underlying pathology (the peripheral neuropathy and PAD) are still present regardless of ulcer resolution. With chronic or recurrent ulcers, physicians should carefully monitor for infection and be aware of the risk factors for infection discussed below.
Diabetic Foot Infections
Diabetic foot infections are an increasingly common problem, as greater than 50% of DFUs will become infected. 36 These infections have a profoundly negative impact on patient self-reported quality of life. 38 The infection often begins with an invasion of foreign organisms into an area of skin breakdown. Following colonization, the microbes induce an inflammatory response leading to tissue destruction. Independent risk factors associated with diabetic foot infections include ulcers that probe to bone (odds ratio: 6.7), ulcers that have been present for >30 days (odds ratio: 4.7), recurrent ulcers (odds ratio: 2.4), wounds with traumatic etiology (odds ratio: 2.4), and the presence of PAD (odds ratio: 1.9). 25
Evaluation: Physical Examination
Management of diabetic foot infections begins with a proper history and physical examination supplemented by laboratory and imaging studies. Patients with diabetic foot infections may not manifest the typical signs and symptoms of infection (nausea, vomiting, anorexia, malaise, fever) because of an altered leukocytic immune response. 6 One of the earliest signs of infection in a patient with a DFU may be unexplained recent hyperglycemia. Patients may also report symptoms of the “diabetic flu” such as nausea, vomiting, anorexia, fevers, and chills, and these symptoms should prompt close inspection of the foot for ulcers or signs of a severe infection. 52 On close inspection, attention should focus on ulcer size, depth (probe to bone test), odor, margins, and drainage. An erythrocyte sedimentation rate >70 mm/h, ulcer >2 cm2, positive probe to bone test, and abnormal radiographic findings such as cortical disruption should increase suspicion for osteomyelitis. 12 The probe to bone test has been shown to be highly sensitive and specific for the presence of osteomyelitis. 23 Studies have reported positive predictive values ranging from 53% to 89% for diagnosis of osteomyelitis with a positive probe to bone test. 23 Perhaps more important, the inability to probe to bone is associated with a negative predictive value of 98%. The extremity should be elevated to determine whether the erythema stems from Charcot or infection. The lower extremity should be elevated with the patient supine for 5 to 10 minutes while observing for resolution of erythema, thus indicating a noninfectious etiology. 8 The diagnosis of infection can then be made clinically based on the presence of purulent drainage and/or the presence of at least 2 signs of inflammation (erythema, warmth, induration, swelling, tenderness, or pain). 51,52
Evaluation: Laboratory Testing
The addition of laboratory data is valuable to quantify the severity of the infection and response to treatment. These data should include a complete blood count, metabolic panel, erythrocyte sedimentation rate, and C-reactive protein. Albumin and prealbumin should be obtained to assess nutritional status. Glucose levels should be obtained, as loss of glycemic control is often one of the first signs of infection. Imaging modalities are of value to characterize the extent of soft tissue and bony involvement. Plain radiographs are useful in the initial workup of a diabetic foot infection. Particular attention should be paid to any radiographic abnormalities, such as cortical erosions, periosteal reactions, soft tissue gas, or radiopaque foreign bodies. Plain radiographs can be repeated to assess for signs of progression. Radiographs, however, are not very sensitive and thus should be combined with other imaging modalities to better characterize a suspected infection. 49 Magnetic resonance imaging (MRI) has been shown to have good sensitivity and specificity in the evaluation of osteomyelitis. 24 Nuclear medicine scans can be used when MRI is unavailable or if a patient is unable to undergo a MRI.
Evaluation: Microbiology
After identifying an infection, cultures are needed to direct antibiotic therapy. Deep tissue cultures should be obtained. These will more reliably detect the causative organism compared to superficial swabs. Especially in the face of osteomyelitis, superficial swabs do not correlate with organisms associated with underlying bone infection. 45 Although bone cultures are ideal, deep soft tissue cultures may be beneficial in identifying the offending organism in osteomyelitis. 18,34 After proper evaluation, infections should be classified using the IDSA system to assist in selecting the best treatment and to improve communication among providers.
Classification
The Infectious Disease Society of America (IDSA) classification system has been validated for grading the severity of infections 30,52 (Table 3). A mild infection is small in size and depth and is superficial. A moderate infection is deeper and more extensive, and a severe infection causes systemic or metabolic perturbations. 52 Increasing severity of infection is associated with higher rates of major amputation and longer hospital admissions. 51,52
Infectious Disease Society of American Diabetic Foot Wound Classification.

Treatment
The treatment of diabetic foot infections is dictated by the severity of the infection. Superficial infections should be cleaned and debrided of any necrotic tissue, and moist dressings can be applied with adequate offloading. Mild infections are usually treated with oral antibiotics in the outpatient setting. Selection of empiric antibiotics should be directed at aerobic gram-positive cocci and aerobic streptococci while considering recent patient antibiotic use and local susceptibilities. 29 The ideal duration of antibiotic therapy is controversial but should be continued until the resolution of infection. Most moderate and all severe infections, including those with evidence of necrotizing fasciitis, rapid progression of infection, or sepsis, warrant hospitalization and urgent consultation with an appropriate specialist to reduce the risk of amputation (Figure 6). Initial operative intervention consists of incision and drainage with thorough debridement of necrotic soft tissue and bone. Return trips to the operating theater are frequently necessary to obtain a healthy granulating wound bed (Figure 7). IV antibiotic therapy is often needed for severe infections with the duration dependent on the extent of the infection.

Photograph of a severe diabetic foot infection in a patient who presented with fever (38.6°C), leukocytosis (12 400 cells/mm3), nausea, and vomiting. There is a large plantar abscess with extensions toward the tarsal tunnel.

Photograph of the dorsal right foot demonstrates a granulating wound after incision and debridement of a dorsal diabetic foot infection.
Management of osteomyelitis is an important component in the treatment of diabetic foot infections and bone cultures are ideal to direct antibiotic therapy. Consultation with an infectious disease specialist assists with selection of antibiotics and treatment duration. A multicenter randomized study noted no difference between a 6- and a 12-week course of antibiotics on osteomyelitis remission rates at 1 year. 45 Considerable debate exists regarding the need for surgery in treating osteomyelitis, but our opinion is that surgery and antibiotic treatment produces a synergistic response. This is especially true in patients with exposed bone, patients with midfoot or hindfoot osteomyelitis, and those with deformity that could lead to skin breakdown. Nonoperative treatment of osteomyelitis, particularly in the forefoot, is a viable option for some patients. 19,41 The components of effective treatment of diabetic foot infections include prompt diagnosis, deep cultures, early empiric antibiotics followed by culture specific antibiotics, and local wound care vs operative debridement. Conservative surgery, that is, the avoidance of amputation, is successful in approximately 50% of patients. 3 Diabetic foot osteomyelitis involving the calcaneus is particularly difficult to treat, often requiring significant bone resection to achieve negative margins (Figures 8 and 9). These patients will often require lifelong bracing to accommodate for the lack of the Achilles mechanism. An understanding of risk factors and early involvement of multiple specialties is the key to successful outcomes and limb salvage. In a series of 229 patients hospitalized with diabetic foot infections, the diagnosis of osteomyelitis was associated with a 3.4 times increased odds of major amputation and significantly longer hospital stays than patients with soft tissue infections. 40

Predebridement clinical photo of a 73-year-old diabetic male with a necrotic right heel ulcer and osteomyelitis of the calcaneus.
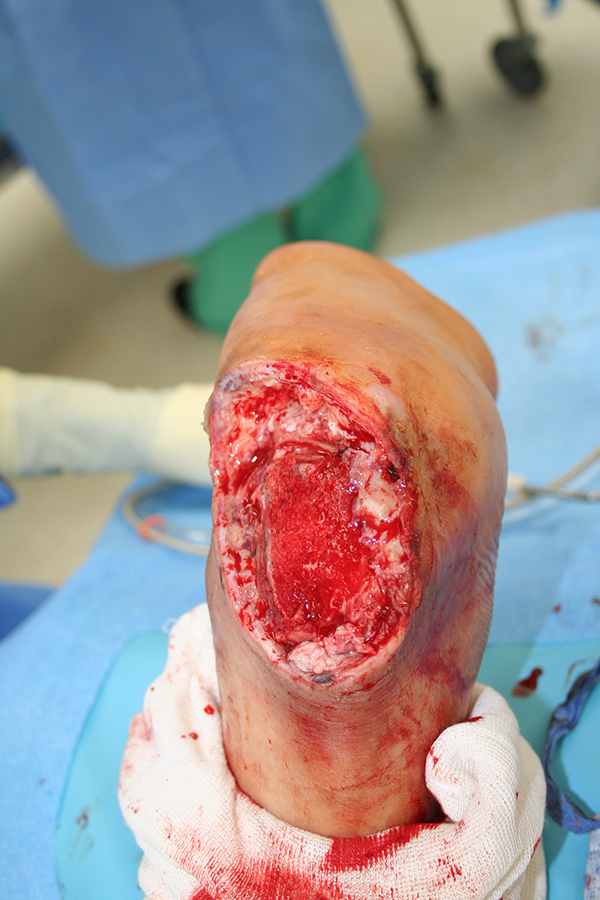
Clinical photo of same patient (as in Figure 8) after sharp excisional debridement of the soft tissues and resection of the calcaneal tuberosity to healthy bleeding bone.
Amputations
Even with proper management, infections can be difficult to control and patients should be counseled about the possibility of amputation. Amputation is the removal of a nonviable limb and should be considered in patients with uncontrolled infections or nonhealing wounds. Studies have shown that infection is an independent risk factor for minor amputation (odds ratio: 1.56, CI 1.05-2.30) and greater than 20% of moderate or severe DFI eventually lead to amputation. 46,51,52 Other studies have demonstrated that patients who developed foot infections had a 55.7 times greater risk of hospitalization and 154.5 times greater risk of amputation than those who did not develop infection. 25 There are many different levels of amputations including the forefoot, midfoot, Syme, below-knee, and above-knee amputations. The surgeon should consider patient-specific factors, such as current functional status and level of social support when deciding the appropriate level. In addition, the surgeon must assess the patient’s wound healing ability prior to proceeding with amputation with the ultimate goal of preserving as much length as possible in efforts to reduce energy expenditure. The morbidity and mortality after major amputation is substantial. After a median follow up of 109 weeks, 29.4% of patient died after transtibial amputation, and patients with end-stage renal disease had significantly higher mortality rates than patients not on dialysis (52% vs 24%, respectively). 48 One-third of patients developed a contralateral foot problem, and 10% of patients required a contralateral transtibial amputation. 48 Despite the morbidity associated with major amputation, select patients can significantly improve their quality of life after amputation. 49 In addition, an amputation may provide a better chance at recovery compared to multiple salvage attempts in a sick patient. Improvement in physical function after lower extremity amputation is related to achieving and maintaining ambulation.
Prevention
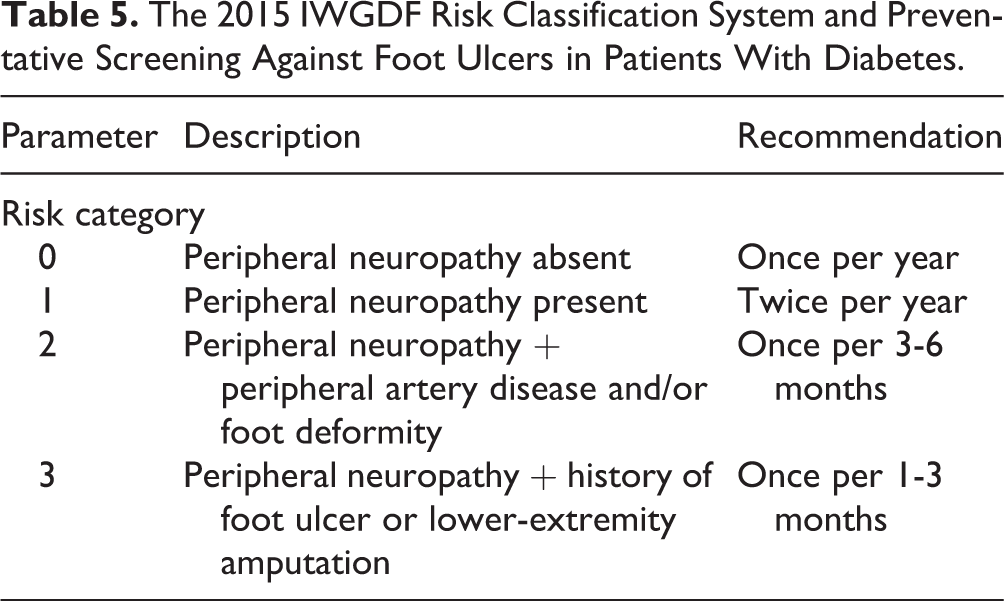
Given the morbidity and mortality associated with diabetic foot ulcers and infections, more attention should be focused on prevention. The International Working Group on the Diabetic Foot performed a systematic review of the current literature regarding the prevention of DFUs. They found high-quality evidence supporting the efficacy of consistent use of plantar pressure–relieving footwear on the prevention of plantar foot ulcer recurrence. However, there was a clear lack of evidence to support any intervention meant to prevent a patient’s first ulcer. 11,47 Data also suggest that elevated foot temperatures can predict early inflammation, and patients who self-monitored their foot temperature had a reduced recurrent ulcer rate. 4,27,28 A recent study compared patients with a history of DFU to a control group of patients with diabetic neuropathy, evaluating peak plantar pressure and plantar shear stress, and found that plantar shear stress was significantly higher in patients with a history of DFU whereas the average peak plantar pressure was not significantly different from that in the control group. 56 Consequently, reducing shear stress may be an important component of preventing recurrent DFUs. Despite an increase in focus on ulcer prevention, there still remains no evidence-based data providing clear recommendations for ulcer prevention. Efforts directed solely on patient education have failed to prove clinically relevant for reducing ulcers and amputations. 14 Given this paucity of data, large, multicenter studies are needed to define the means of preventing an initial ulcer. Until clear evidence is found, physicians should employ a multispecialty approach involving primary care doctors, endocrinologists, podiatrists, and orthotists based on the recommendations of the International Working Group on the Diabetic Foot 10,11 (Tables 4 and 5). The success of prevention depends on this multidisciplinary approach with focus on frequent foot inspections, basic wound care, offloading modalities, close glycemic monitoring, and patient education. 15,50
Recommendations From 2015 IWGDF Guidance on the Prevention of Foot Ulcers in At-Risk Patients With Diabetes.

IWGDF, International Working Group on the Diabetic Foot.
The 2015 IWGDF Risk Classification System and Preventative Screening Against Foot Ulcers in Patients With Diabetes.
Conclusion
Diabetes is a systemic disease with serious lower extremities manifestations including diabetic foot ulcers and diabetic foot infections that lead to substantial patient morbidity and mortality. The etiology of diabetic foot disease is multifactorial, and includes complications of diabetic neuropathy, vasculopathy, immunopathy, and poor glycemic control. Proper management of diabetic foot problems begins with a thorough clinical evaluation of the patient followed by early treatment with an emphasis on preventative strategies. The best preventative approach centers on, education, close follow-up, and clear communication between a multidisciplinary team composed of surgeons, hospitalists, endocrinologists, infectious disease specialists, and wound care experts. Further multicentered randomized controlled trials are needed to continue to drive treatment recommendations and prevention strategies.
Supplemental Material
Supplemental Material, FAO788864-ICMJE - The Evaluation and Treatment of Diabetic Foot Ulcers and Diabetic Foot Infections
Supplemental Material, FAO788864-ICMJE for The Evaluation and Treatment of Diabetic Foot Ulcers and Diabetic Foot Infections by Michael A. Del Core, Junho Ahn, Robert B. Lewis, Katherine M. Raspovic, Trapper A. J. Lalli, and Dane K. Wukich in Foot & Ankle Orthopaedics
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article. ICMJE forms for all authors are available online.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
