Abstract
Rhamnogalacturonan II (RG-II) is a structurally complex pectic polysaccharide that exists as a borate ester cross-linked dimer in the cell walls of all vascular plants. The glycosyl sequence of RG-II is largely conserved, but there is evidence that galacturonic acid (GalA) methyl etherification and glucuronic acid (GlcA) methyl esterification vary in the A sidechain across plant species. Methyl esterification of the galacturonan backbone has also been reported but not confirmed. Here we describe a new procedure, utilizing aq. sodium borodeuteride (NaBD4)-reduced RG-II, to identify the methyl esterification status of backbone GalAs. Our data suggest that up to two different GalAs are esterified in the RG-II backbone. We also adapted a procedure based on methanolysis and NaBD4 reduction to identify 3-, 4-, and 3,4-O-methyl GalA in RG-II. These data, together with matrix-assisted laser desorption/ionization–time-of-flight mass spectrometry (MALDI-TOF) MS analysis of sidechain A generated from selected RG-IIs and their NaBD4-reduced counterparts, suggest that methyl etherification of the β-linked GalA and methyl esterification of the GlcA are widespread. Nevertheless, the extent of these modifications varies between plant species. Our analysis of the sidechain B glycoforms in RG-II from different dicots and nonpoalean monocots suggests that this sidechain has a minimum structure of an O-acetylated hexasaccharide (Ara-[MeFuc]-Gal-AceA-Rha-Api-). To complement these studies, we provide further evidence showing that dimer formation and stability in vitro is cation and borate dependent. Taken together, our data further refine the primary sequence and sequence variation of RG-II and provide additional insight into dimer stability and factors controlling dimer self-assembly.
Introduction
Rhamnogalacturonan II (RG-II) is a pectic polysaccharide that exists as a borate cross-linked dimer in the primary cell walls of all vascular plants.1,2 Current models of the primary cell wall predict that RG-II is attached to a homogalacturonan, which itself may be linked to rhamnogalacturonan I. 3 These three pectic polysaccharides are believed to form a macromolecular network that interacts with cellulose and hemicellulose and thus has a fundamental role in defining many of the wall’s biomechanical properties.4–7 RG-II is likely to have a central part in controlling these properties, since the inability to cross-link RG-II typically results in swollen walls that have reduced wall tensile strength.8–10 Moreover, numerous studies have shown that modifying RG-II structure decreases its cross-linking and reduces plant fitness.11–16
RG-II is one of the most structurally complex polysaccharides yet identified in nature. It is composed of 11 different sugars linked together by at least 20 different glycosyl linkages ( Fig. 1A ). The RG-II backbone is composed of (1→4)-linked α-D-galacturonic acid (GalA) residues. 17 This backbone is substituted with four different oligosaccharide sidechains (A, B, C, and D) and two distinct α-L-arabinofuranose (Araf) substituents (E and F). 18 Sidechains C and D are structurally conserved disaccharides. Sidechain B may, depending on the plant, contain between six and nine glycosyl residues, whereas sidechain A is an octasaccharide. The RG-II dimer contains two RG-II molecules linked to one another by a borate diester between the apiosyl residue of each A sidechain. ( Fig. 1B ). 19 This dimer self-assembles in vitro when the monomer is reacted with boric acid in the presence of a divalent cation.20,21 However, the mechanism of cross-linking in planta and the cellular location where this reaction occurs has not been determined. 22
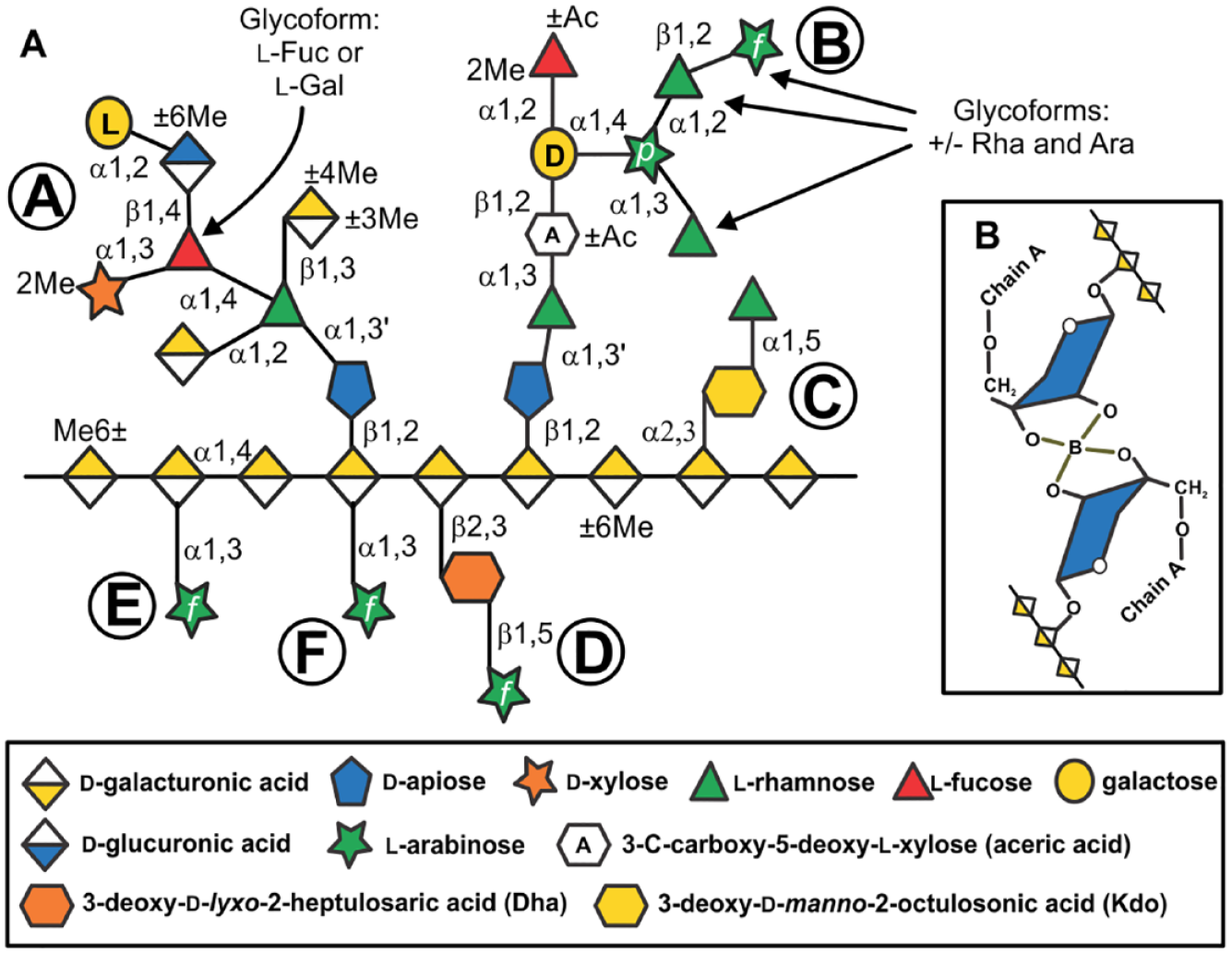
The primary structure of RG-II. (
The primary sequences of the A and B sidechains are not identical in RG-II isolated from different plant species and tissues.23–27 For example, sidechain A may contain 3,4-linked
The structural features of RG-II that are critical for dimer self-assembly must be determined, if we are to understand the physiochemical role of this unique pectic polysaccharide in wall assembly and function. Our approach toward this goal is to identify structural features that are highly conserved in RG-II from diverse plants. This led us to develop new methods to identify O-methylated GalA and methyl-esterified GlcA in sidechain A, as well as a new procedure to identify and locate methyl-esterified backbone GalAs. We complement these studies by describing some of our recent results on the effects of different cations as well as temperature on dimer formation and stability in vitro.
Materials and Methods
Plant Material
We obtained actively growing leaves from American sweet flag (Acorus americanus, Acorales), agave (Agave americana, Asparagales), banana (Musa acuminata, Zingiberales), common screwpine (Pandanus utilis, Pandanales), false bird-of-paradise (Heliconia wagneriana, Zingiberales), horsetail (Equisetum hyemale, Equisetales), natal lily (Clivia miniata, Asparagales), southern swamp lily (Crinum americanum, Asparagales), spiderwort (Tradescantia virginiana, Commelinales), and white bird-of-paradise (Strelitzia alba, Zingiberales) from the Plant Biology Greenhouse at the University of Georgia (Athens, GA). We purchased shoots of asparagus (Asparagus officinalis, Asparagales), celery petioles (Apium graveolens, Apiales), mango fruit (Mangifera indica, Sapindales), papaya fruit (Carica papaya, Brassicales), and starfruit (Averrhoa carambola, Oxalidales) from a local grocery store. Mistletoe (Viscum album, Santalales) was purchased from Allo Botanicals (allobotanicals.com).
We purchased Wolffiella lingulata (ID8041), Wolffiella repanda (ID9122), Wolffia arrhiza (ID7193), Wolffia borealis (ID9147), Wolffia globosa (ID9299), and Lemna tenera (ID9020) from the Rutgers duckweed stock cooperative (Rutgers University, NJ). These duckweeds were grown on Schenk and Hildebrandt basal salts (1.6 g/L; Sigma-Aldrich, St. Louis, MO), pH 5.8, containing sucrose (5 g/L; Sigma-Aldrich), morpholinoethanesulfonic acid (500 mg/L; Sigma-Aldrich), and agar (8 g/L; Sigma-Aldrich). Plants were kept in a controlled environmental growth cabinet (Adaptis A1000, Conviron, Winnipeg, Canada) at 19 and 15 °C with a 14 h light and 10 h dark cycle, respectively, with a light intensity of 120 µmol quanta/m2/s and 70% relative humidity. 30
We grew suspension-cultured ‘Paul’s Scarlet’ rose cells (Rosa sp.) 31 under constant dim illumination (~20 µmol quanta/m2/s) on an orbital shaker at 23 °C in MX1 medium containing glycerol (16 mL/L; Sigma-Aldrich) and boric acid (50 µM; Sigma-Aldrich). Suspension-cultured tobacco BY2 cells were grown in the absence of light on an orbital shaker at 22 °C in Murashige and Skoog medium (Sigma-Aldrich) containing sucrose (3%). 32 Suspension-cultured red kidney bean (Phaeseolus vulgaris) cells were grown in the absence of light as described. 33 Arabidopsis thalaiana (Col-0 and mur1-1) seeds were purchased from the Arabidopsis Biological Resource Center (abrc.osu.edu; Ohio State University, Columbus, OH). Seeds were germinated and grown on potting soil (Farfard 3B, Sun Gro Horticulture, Agawam, MA) at 19 and 15 °C with a 14 h light and 10 h dark cycle, respectively, with a light intensity of 120 µmol quanta m2/s in a controlled environmental growth chamber (Conviron). Tobacco seeds (Nicotiana tabacum K326) were germinated and grown on potting soil at 23 and 19 °C with a 14 h light (intensity of 120 µmol quanta m2/s) and 10 h dark cycle in a controlled environmental growth chamber (Conviron).
Preparation of Cell Walls
We prepared cell walls as their alcohol insoluble residues (AIRs). 34 Starch was removed by treating suspensions of the AIRs (0.5–1.0 g) in 50 mM sodium acetate (Sigma-Aldrich), pH 5.2, for 24 h at 45 °C with the glucoamylase Spirizyme (60 μL/g; Novozymes A/S, Bagsvaerd, Denmark) and the α-amylase Liquozyme (300 μL/g; Novozymes A/S). The de-starched AIR was collected by filtration through nylon mesh (40 μm pore size) and washed with deionized water.
Solubilization of RG-II
We used two different protocols, depending on the source of the plant material, to obtain materials enriched in RG-II:
Endopolygalacturonase (EPG) treatment of AIR: The de-starched AIR (Arabidopsis leaf, banana, celery, common screwpine, horsetail, mango, mistletoe, papaya, starfruit, and suspension cultured bean, rose, and tobacco cells) was suspended in 50 mM sodium acetate, pH 5.2, and treated for 24 h at 30 °C with EPG (M2, 5 units/g; Megazyme, Bray, Ireland). The EPG-treated AIR was collected by filtration through nylon mesh (40 μm pore size) and retreated with the EPG. The soluble materials released by EPG were combined, dialyzed (3500 molecular weight cutoff) against deionized water, and freeze-dried.
EPG treatment of the ammonium oxalate-soluble material: The de-starched AIR (agave, American sweet flag, asparagus, duckweeds, false bird-of-paradise, natal lily, southern swamp lily, spiderwort, tobacco leaf, and white bird-of-paradise) was extracted for 16 h at room temperature with 50 mM ammonium oxalate (pH 5.5, 100 mL/g; Sigma-Aldrich). The soluble materials were dialyzed (3500 molecular weight cutoff) against deionized water and freeze-dried. Solutions of the ammonium oxalate-soluble materials in 50 mM sodium acetate, pH 5.2, were treated for 24 h at 30 °C with EPG (5 units/g; Megazyme M2). The EPG-treated materials were dialyzed (3500 molecular weight cutoff) against deionized water and freeze-dried.
Isolation of RG-II
We isolated RG-II using size-exclusion chromatography (SEC). To this end, solutions of the EPG- and oxalate/EPG-generated materials (5–10 mg) in 50 mM ammonium formate, pH 5 (500 µL), were filtered (0.45 μm spin filters) and then fractionated on a Superdex-75 10/300 GL column (GE Healthcare Bio-Sciences AB, Uppsala, Sweden). We eluted the column at 0.5 mL/min with 50 mM ammonium formate (Sigma-Aldrich), pH 5. 20 SEC was performed with a Dionex UltiMate 3000 pump (Thermo Fisher Scientific, Waltham, MA) and a Shodex RI-101 refractive index detector (Showa Denko America Inc., New York, NY) under the control of a Dionex Chromeleon 6 workstation (Thermo Fisher Scientific). Fractions containing RG-II were collected manually and repeatedly freeze-dried to remove ammonium formate.
We isolated red wine RG-II from Yellow Tail (Yenda, Australia) Cabernet Sauvignon as described. 28 This RG-II exists as its borate cross-linked dimer. To generate the monomer, we treated the dimer for 1 h at room temperature with 0.1 M HCl to hydrolyze the borate diester. 20 The acid was removed by dialysis and the solution freeze-dried.
Identification of O-Methyl Ethers of GalA
To generate the methyl-esterified methyl glycosides of GalA and GlcA, we methanolyzed RG-II (~1 mg) for 4 h at 80 °C with 1 M HCl in anhydrous methanol (250 µL; Supelco, Bellefonte, PA). The solution was concentrated to dryness under a flow of air at 40 °C and the residue was washed with isopropanol (3 × 250 µL; Sigma-Aldrich) and then with methanol (3 × 250 µL; Sigma-Aldrich). Methyl-esterified uronic acids were converted to their corresponding neutral glycoses by overnight treatment at 4 °C with NaBD4 (20 mg/mL in water, 250 µL; Sigma-Aldrich). Aqueous solutions of NaBH4 have a pH between 9.3 and 10, which minimizes the hydrolysis of the reductant but does not cause substantial hydrolysis of methyl esters. Any remaining NaBD4 was destroyed by dropwise addition of glacial acetic acid and then concentrating the solution to dryness under a flow of air. We removed borate as its methyl ester by adding methanol containing 10% glacial acetic acid (3 × 250 µL; Sigma-Aldrich) and then concentrating the solution to dryness under a flow of air. Finally, the residue was washed with methanol (3 × 250 µL; Sigma-Aldrich). A solution of the residue in water (500 µL) was desalted using an OnGuard-II H cartridge (1 mL; Thermo Fisher Scientific), which had been prewashed with 1 M HCl (5 mL) and water (15 mL) and then freeze-dried. The desalted material was hydrolyzed for 1.5 h at 120 °C with 2 M trifluoroacetic acid (TFA; Sigma-Aldrich), and the released neutral monosaccharides were analyzed as their alditol acetate derivatives by gas–liquid chromatography–electron impact–mass spectrometry (GLC-EI-MS). GLC-EI-MS was performed with a 7890A gas chromatograph and 5975C mass selective detector (Agilent Technologies, Santa Clara, CA) using a 30 m SP2330 capillary column (Supelco) as described. 35
We used high-performance anion-exchange chromatography with pulsed amperometric detection (HPAEC-PAD) and a Dionex ICS-3000 ion chromatography system (Thermo Fisher Scientific) to determine the efficiency of NaBD4 reduction of methyl-esterified GalA and GlcA. GalA and GlcA (1 mg each) were methanolyzed for 4 h at 80 °C with 1 M HCl in anhydrous methanol and then converted to their corresponding neutral glycoses by overnight treatment at 4 °C with NaBD4 (20 mg/mL in water, 250 µL). The NaBD4-reduced methyl glycosides were hydrolyzed for 1.5 h at 120 °C with 2 M TFA, and the released neutral and acidic monosaccharides were analyzed by HPAEC-PAD using a CarboPac PA1 column (Thermo Fisher Scientific) as described. 30 At least 90% of the methyl-esterified methyl glycosides of GalA and GlcA are converted to Gal and Glc, respectively. Thus, our procedure will provide a semiquantitative measure of the abundance of the methyl-etherified uronic acids.
Identification of Naturally Occurring Methyl-Esterified GlcA in Sidechain A and Methyl-Esterified GalA in the RG-II Backbone
We treated RG-II (2–4 mg) overnight at 4 °C with NaBD4 in water (400 µL, 20 mg/mL) to convert naturally occurring methyl-esterified uronic acids to their corresponding 2H-labeled neutral glycoses. The remaining NaBD4 was destroyed by the addition of glacial acetic acid. Borate was removed with methanol containing 10% glacial acetic acid (3 × 250 µL) and then with methanol (3 × 250 µL). The residue in water (500 µL) was desalted using an OnGuard-II H cartridge (1 mL) and freeze-dried. A portion of this material (~500 µg) was hydrolyzed for 1.5 h at 120 °C with 2 M TFA. We used GLC-EI-MS analysis of the alditol acetate derivatives to determine the neutral monosaccharide composition. We determined the neutral and acidic monosaccharide compositions by HPAEC-PAD using a CarboPac PA1 column as described. 30
Generation of RG‑II Backbone Fragments by Selective Acid Hydrolysis
To generate backbone fragments, we treated NaBD4-reduced RG-II and native RG-II (500 µg) for 16 h at 80 °C with 0.1 M TFA (250 µL). 17 The hydrolysates were then concentrated to dryness with a flow of air and the residues washed with isopropanol (1 × 250 µL) and with methanol (3 × 250 µL). The residues were dissolved in water (200 µL) and a portion (25 µL) was analyzed by HPAEC-PAD using a CarboPac PA1 column as described. 17 A second portion (1 µL) was analyzed by negative ion MALDI-TOF MS using a Bruker Microflex LRF mass spectrometer and workstation (Bruker Scientific, Billerica, MA). MALDI-TOF MS-MS spectra were obtained with an ABI Sciex 5800 MALDI-TOF-TOF mass spectrometer and workstation (AB Sciex, Ontario, Canada). Spectra were obtained using a MALDI target plate precoated with Nafion117 36 (Sigma-Aldrich) prior to the addition of 2,5-dihydroxybenzoic acid (DHB; Bruker Scientific; 1 μL, 10 mg/mL in aqueous (aq.) 50% [v/v] acetonitrile; Sigma-Aldrich) and the oligosaccharides (1 μL, ~1 mg/mL in water). The matrix and analyte were co-crystalized by concentration to dryness using a flow of warm air from a hair dryer. Spectra from at least 200 laser shots were summed to generate each mass spectrum.
Generation of Sidechains A and B by Selective Acid Hydrolysis
To release sidechain A, we treated RG-II (200–500 µg) for 1 h at 80 °C with 0.1 M TFA (250 µL). 24 Sidechain B was released by treating RG-II (200–500 µg) for 16 h at 40 °C with 0.1 M TFA (250 µL). 37 The lower temperature minimizes hydrolysis of the acid-labile Araf residue that terminates some B sidechains (see Fig. 1 ).24,26 The hydrolysates were concentrated to dryness with a flow of air and the residues washed with isopropanol (1 × 250 µL) and then with methanol (3 × 250 µL). The residues were dissolved in water (500 µL) and the acidic oligosaccharides isolated using Supelclean ENVI-Carb graphitized carbon cartridges (1 mL; Supelco). 38 We conditioned the cartridges with aq. 80% (v/v) acetonitrile containing 0.1% TFA (2 mL) and then with deionized water (5 mL). The hydrolysates were dissolved in water (500 µL) and applied to the cartridge, which was then washed with water (2 mL). Acidic oligosaccharides were eluted with aq. 50% acetonitrile containing 0.1% TFA (3 mL). The solutions were concentrated to dryness under a flow of air and the residues then dissolved in water (100–300 µL). We obtained MALDI-TOF mass spectra of sidechain B in the positive ion mode (DHB matrix) and sidechain A in the negative ion mode (Nafion/DHB matrix 36 ).
Determination of the Rate and Extent of RG-II Dimer Formation and Stability In Vitro
Dimer Formation
We reacted the wine RG-II monomer (200 µg) in NaOAc, pH 4 (200 µL), at room temperature with boric acid (1 mM) and Pb(NO2)2 (0.5 mM; Sigma-Aldrich). The proportions of monomer and dimer present after 0.25, 0.5, 1.0, 2.0, 4.0, and 6.0 h were then determined by SEC with a Superdex-75 10/300 GL column and refractive index (RI) detection. The reaction mixtures were injected onto the column precisely at the specified time to minimize the lag period between reaction time and detection of the RG-II. Borate cross-linking of RG-II occurs slowly at the pH of the ammonium formate eluent (pH 5). This assay was also used to determine the effects of different concentrations of boric acid and lead, KCl (Sigma-Aldrich), and selected divalent (CaCl2, NiCl2, ZnCl2, MgCl2, CuCl2, SrCl2, and BaCl2; all from Sigma-Aldrich) cations on the rate and extent of dimer formation.
Dimer Formation and Stability at Different Temperatures
We determined the effect of temperature on dimer formation by reacting the wine RG-II monomer (200 µg) in NaOAc, pH 4 (200 µL), at temperatures between 4 and 80 °C with boric acid (1 mM) and Pb(NO2)2 (0.5 mM; Sigma-Aldrich). The proportions of monomer and dimer present were then determined by SEC. We determined the stability of the wine dimer (200 µg) by keeping it for 1 h at 80 °C in NaOAc, pH 4 (200 µL). The proportions of monomer and dimer present were then determined by SEC. The effects of different concentrations of boric acid and Ca2+ and other divalent cations (NiCl2, ZnCl2, MgCl2, CuCl2, Pb(NO2)2, SrCl2, and BaCl2) on the abundance of the dimer after treatment for 1 h at 80 °C were also determined using this protocol.
Results
Identification of O-Methyl Ethers of GalA
Methyl etherification of the β-linked GalA in the A sidechain of RG-II has been shown to differ across plant species.24–27 However, facile methods to identify such GalAs are lacking. These modified uronic acids are not typically reported when the glycosyl residue composition of RG-II is determined by analysis of its trimethylsilyl methyl glycoside derivatives. 39 By contrast, neutral O-methylated glycoses including 2-O-methyl xylose (MeXyl) and 2-O-methyl fucose (MeFuc) present in RG-II are detected by GLC-EI-MS analysis of their alditol acetate derivatives. 40 Thus, we believed that this procedure could be used to detect O-methyl-etherified GalA residues provided that the hexuronic acids are converted to their corresponding neutral hexoses. Methyl-esterified uronic acids are reduced to their corresponding neutral glycoses by treatment with sodium/potassium borohydride in imidazole buffer,41,42 but the high concentrations of buffer complicate further analysis. This issue was overcome when Bhat and coworkers showed that NaBD4 in water converts methyl-esterified hexuronic acids to their corresponding 6,6′- 2 H-hexoses. 43 Aqueous solutions of NaBH4 are relatively stable 44 and have a pH between 9.3 and 10.5, which is likely sufficiently basic to minimize the hydrolysis of the reductant but does not cause rapid hydrolysis of methyl esters.
We first methanolyzed suspension-cultured tobacco and rose cell RG-II with hot methanolic HCl to generate a mixture of methyl glycosides and the methyl-esterified methyl glycosides of GalA and GlcA (
Fig. 2A
). This mixture was treated with aq. NaBD4 and then hydrolyzed with 2 M TFA. We determined if any of the released monosaccharides contained 2H by GLC-EI-MS analysis of their alditol acetate derivatives. We observed a fivefold increase in the area of the peak corresponding to Gal and a twofold increase in the area of the glucose peak (
Suppl. Fig. S1
). The EI mass spectrum of each peak was consistent with the presence of the 6,6′-
2
H-hexitol derivatives (
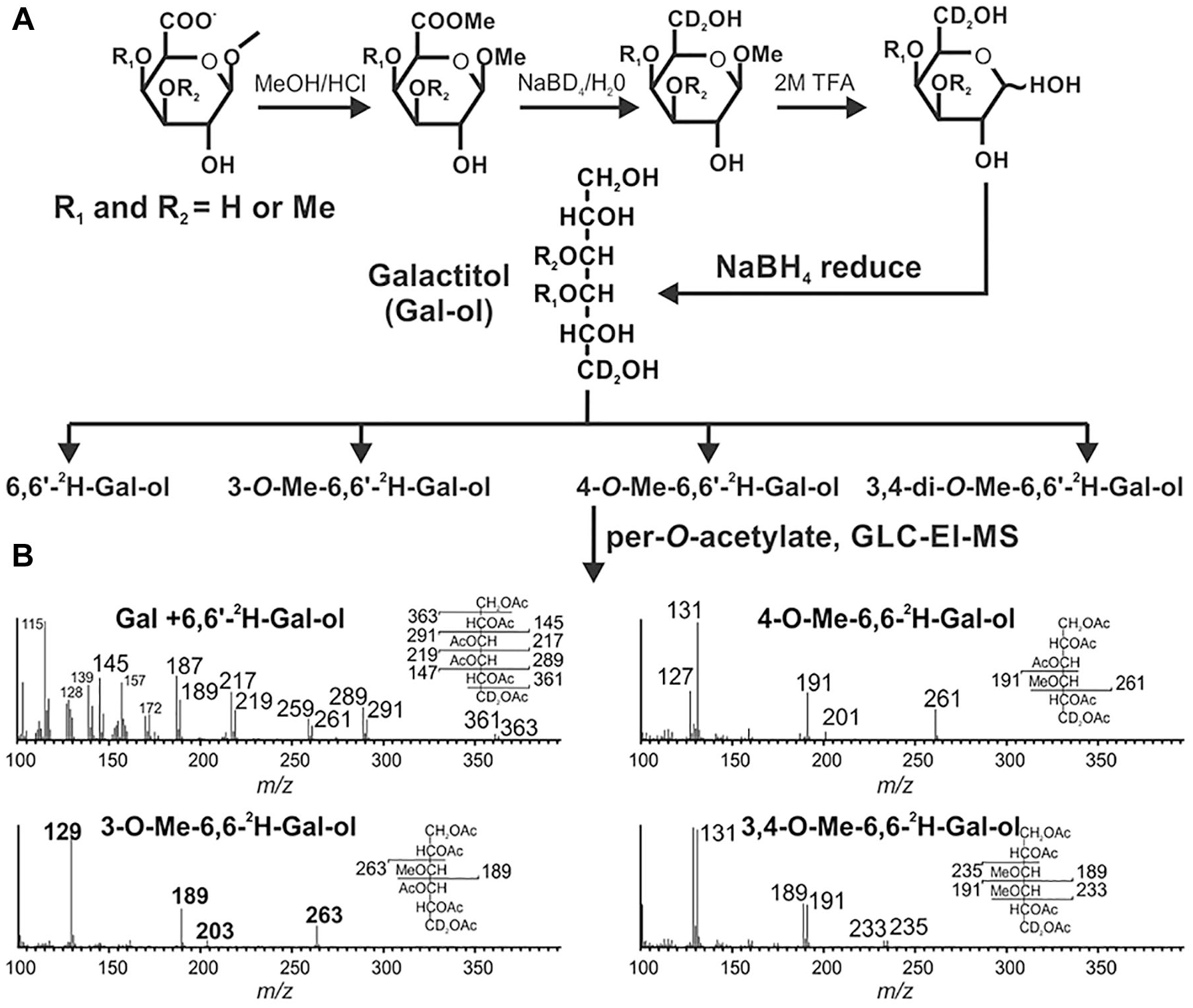
A procedure to identify methyl-etherified GalA residues in RG-II. (
Sidechain A Methyl Etherification and Methyl Esterification Differ in the RG-II from Different Plants
The presence of methyl-etherified β-linked GalA and methyl-esterified GlcA in sidechain A had been demonstrated in only a limited number of different plant species.25,27,30 We extend these data by determining the methylation status of sidechain A from the RG-II of several additional plants. Variable proportions of 3- and 4-mono-O-methyl and 3,4-di-O-methyl GalA but no 2-O-methyl GalA were detected ( Table 1 ). In several plants, most notably the members of the Lemnoideae, the GalA is substituted with between one and two methyl ethers. In other plants, including celery and mistletoe, there is less than one methyl ether per GalA, suggesting that only a small portion of the A sidechain GalA carries a methyl ether. There is no discernible correlation between the position and extent of GalA methylation and plant phylogeny, although 4-O-methyl GalA does predominate in the RG-II isolated from the limited number of monocots we analyzed ( Table 1 ).
Relative Proportions of Mono- and Di-O-Methyl GalA in the RG-II Isolated from the Cell Walls of Selected Plants.
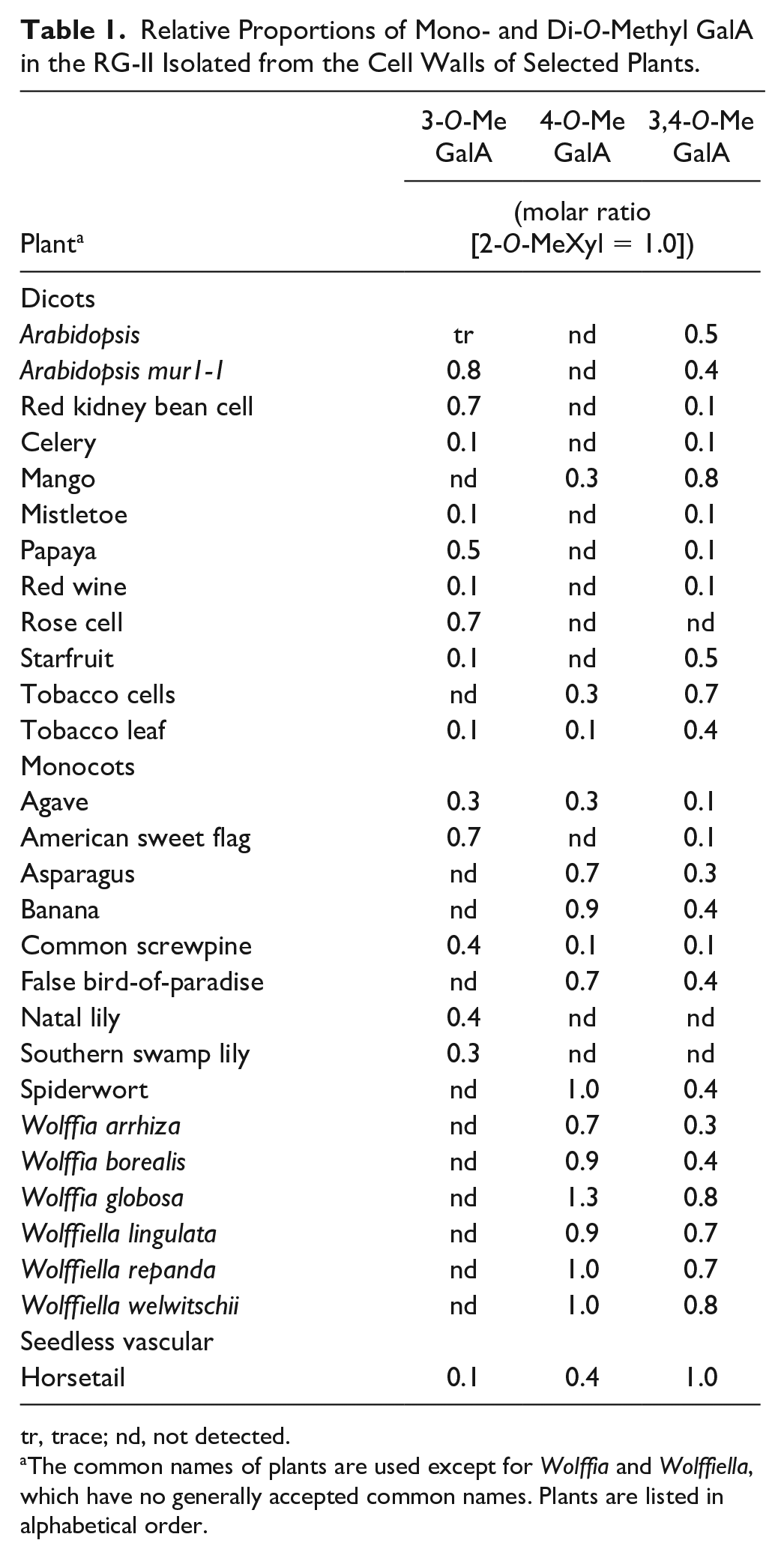
tr, trace; nd, not detected.
The common names of plants are used except for Wolffia and Wolffiella, which have no generally accepted common names. Plants are listed in alphabetical order.
We next developed a method using aq. NaBD4-reduced RG-II and MALDI-TOF MS that allowed us to distinguish methyl-esterified and methyl-etherified residues in sidechain A (see
Fig. 3
).The MALDI-TOF mass spectra of sidechain A from all plants contain ions that differ by 14 and 16 atomic mass units (amu;
Relative Abundance of the Methylated Glycoforms of Sidechain A Generated from Various RG-IIs before and after Reduction with NaBD4.
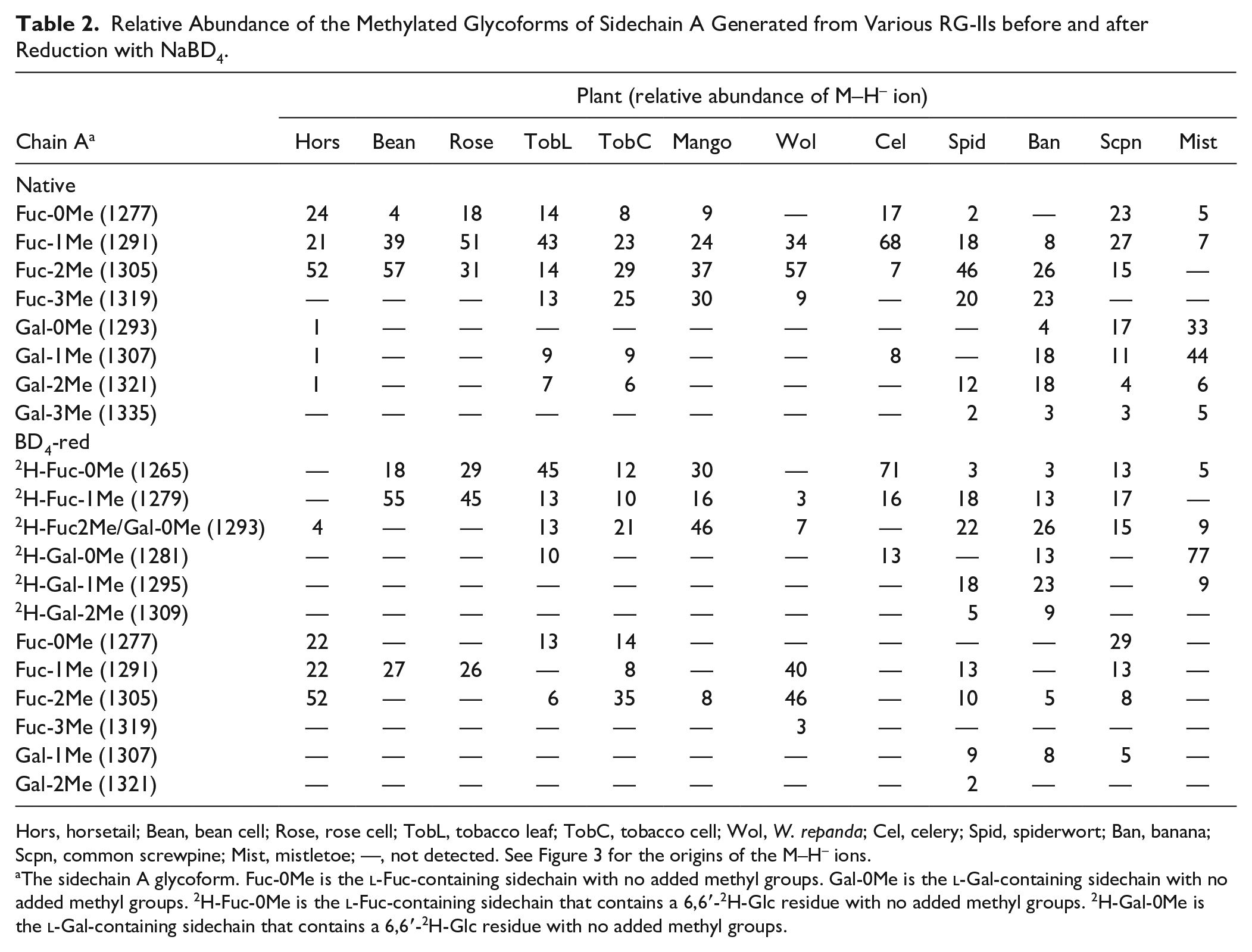
Hors, horsetail; Bean, bean cell; Rose, rose cell; TobL, tobacco leaf; TobC, tobacco cell; Wol, W. repanda; Cel, celery; Spid, spiderwort; Ban, banana; Scpn, common screwpine; Mist, mistletoe; —, not detected. See Figure 3 for the origins of the M–H– ions.
The sidechain A glycoform. Fuc-0Me is the
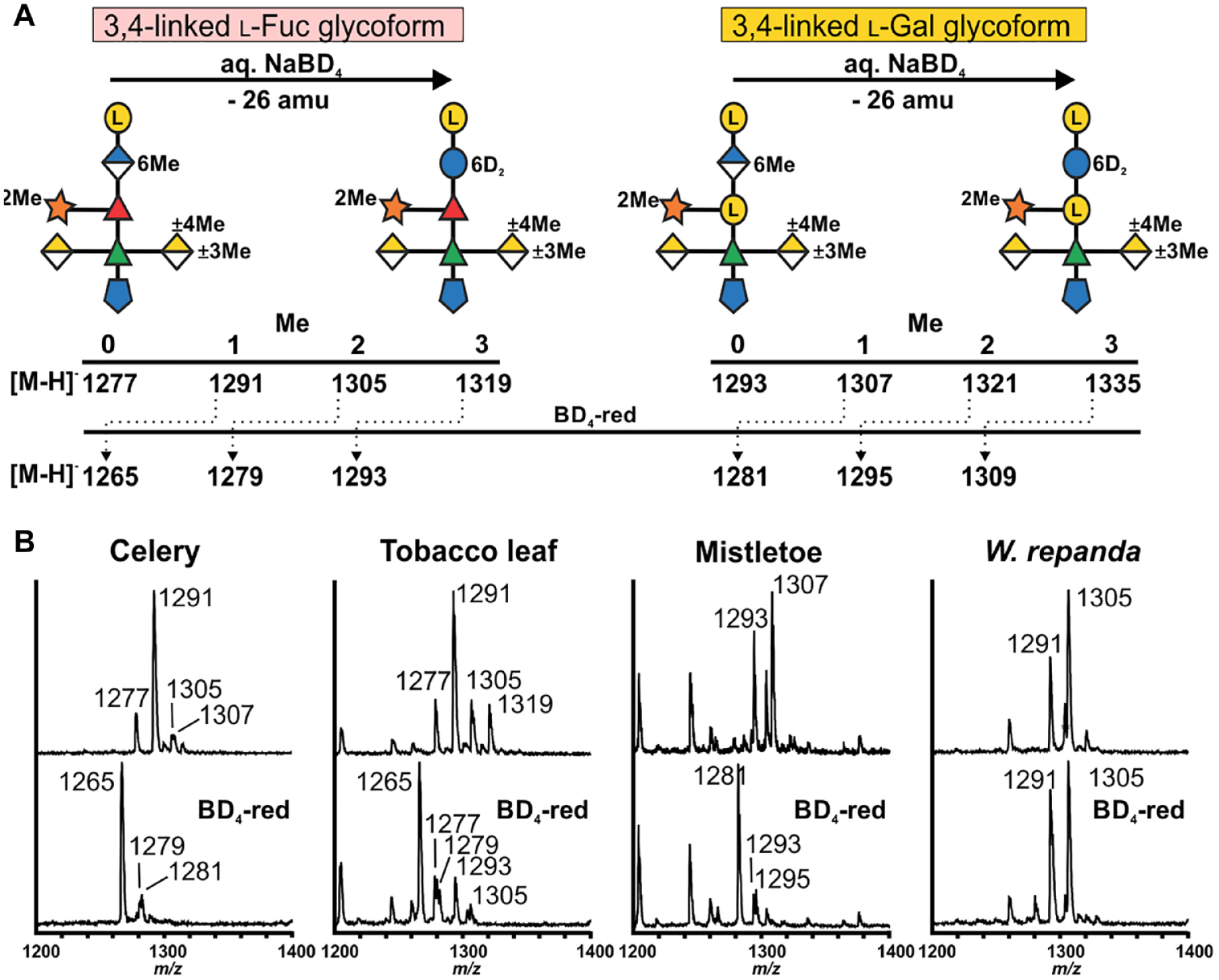
Differentiating methyl-etherified and methyl-esterified hexuronosyl residues in the A sidechain of RG-II. (
The MALDI-TOF mass spectra of sidechain A generated from the BD4-reduced RG-II still contain ion that differ by 14 and 16 amu (
Our data confirm that 3,4-linked Gal is far more abundant than 3,4-linked Fuc in the A sidechain of mistletoe RG-II. 27 The major ion we detected after NaBD4 treatment of mistletoe RG-II corresponds to a deuterated A sidechain with no methyl groups. The A sidechain lacking a methyl group (m/z 1293) generated from native mistletoe RG-II was not detected after NaBD4 reduction ( Table 2 ). This fragment is likely generated by de-esterification of sidechain A when the native RG-II is treated with TFA. We conclude that mistletoe and celery RG-II are similar since methyl-esterified GlcA accounts for the bulk of the methyl groups. Methyl-etherified GalAs are not abundant, a result consistent with the relatively low abundance of 3- and 4-O-Me GalA detected in mistletoe RG-II ( Table 1 ). Sidechain A from a different species of mistletoe (Viscum coloratum) has also been reported to contain little, if any, methyl-etherified GalA. 27 These authors also reported that the GlcA is not methyl-esterified, although the possibility could not be discounted that the GlcA was de-esterified during isolation of the RG-II.
The A sidechain from horsetail and Wolffiella contains zero, one, or two methyl groups irrespective of whether the RG-II had been treated with NaBD4. Sidechain A containing 6,6∙-2H-Glc accounted for less than 10% of the oligosaccharides. We suggest that most of the GlcA is not methyl-esterified and that the methyl groups that are present exist as O-methyl ethers. Indeed, methyl-etherified GalA is abundant in the RG-II from both these plants ( Table 1 ). These data are also consistent with our previous study of duckweed RG-II, which suggested that little, if any, of the A sidechain contains a methyl-esterified uronic acid. 30
A Hexasaccharide May Represent the Minimum Structure of Sidechain B
Numerous studies have shown that sidechain B exists as O-acetylated glycoforms that vary in the number of Ara and Rha residues.17,23–25,27,45 We partially characterized sidechain B generated from several different plants to determine if these glycoforms are correlated with the methylation status of sidechain A ( Table 2 ).
As expected, sidechain B exists as an O-acetylated oligosaccharide that varies in the number of Ara and Rha residues ( Table 3 ). Glycoforms containing between 5 and 9 glycoses were detected. The mono-O-acetylated species accounted for approximately 70% of sidechain B generated using our experimental conditions.
Relative Abundance of the B Sidechain Glycoforms in the RG-II Isolated from Selected Plants.
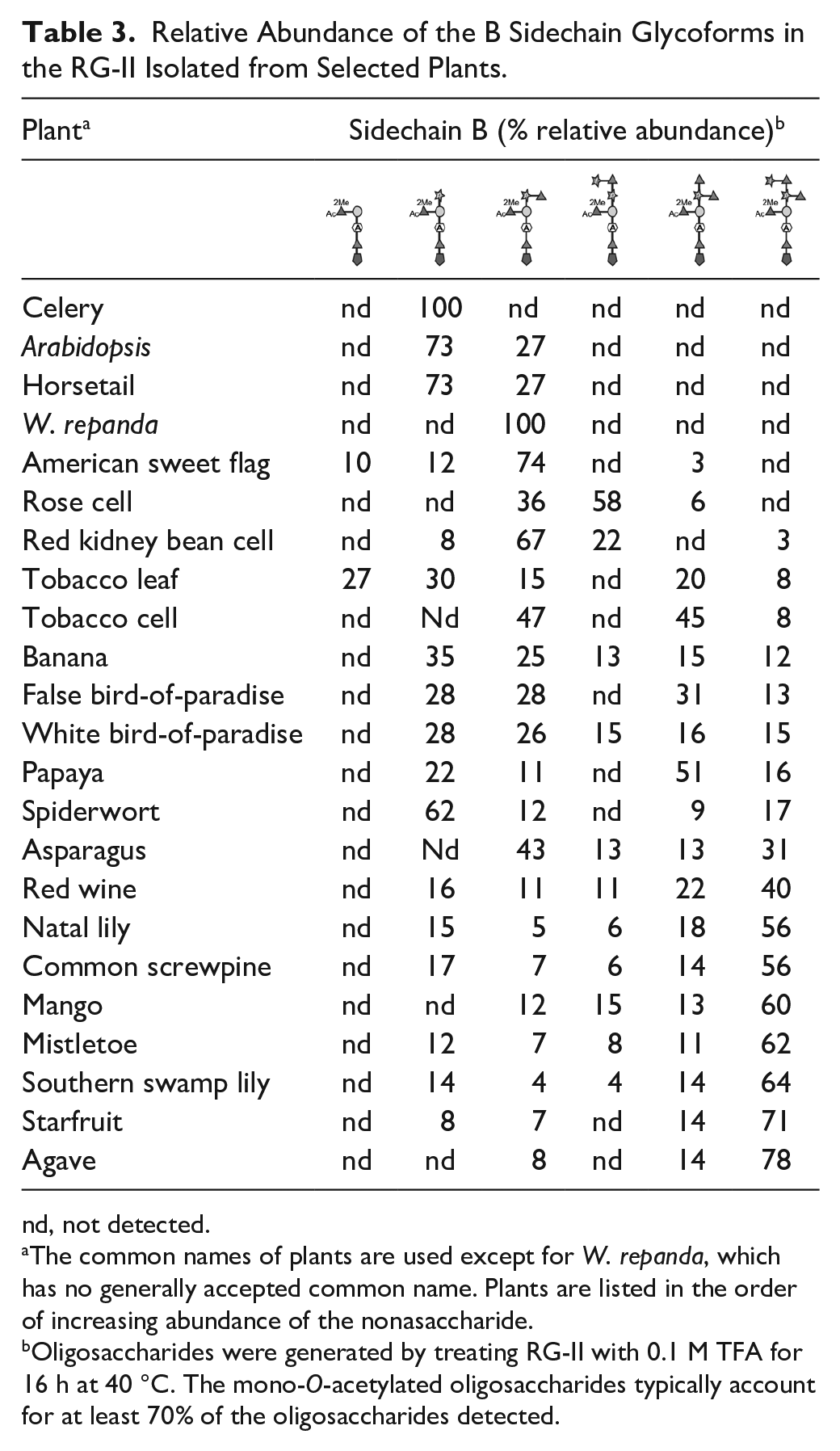
nd, not detected.
The common names of plants are used except for W. repanda, which has no generally accepted common name. Plants are listed in the order of increasing abundance of the nonasaccharide.
Oligosaccharides were generated by treating RG-II with 0.1 M TFA for 16 h at 40 °C. The mono-O-acetylated oligosaccharides typically account for at least 70% of the oligosaccharides detected.
The monoacetylated hexasaccharide (Ara-[MeFuc]-Gal-AceA-Rha-Api) was the only B sidechain glycoform generated from celery RG-II ( Table 3 ). Celery RG-II exists almost entirely as the borate cross-linked dimer ( Suppl. Fig. S3 ). Thus, the absence of the hepta-, octa-, and nonasaccharides does not affect the ability of RG-II to dimerize.
Small amounts of the monoacetylated pentasaccharide (MeFuc-Gal-AceA-Rha-Api) were generated from tobacco leaf and American sweet flag RG-II. However, this pentasaccharide accounts for <27% of the B sidechain glycoforms generated from these RG-IIs. Thus, we suggest that the hexasaccharide represents the minimum structure of sidechain B compatible with the function of RG-II.
The B sidechain hexasaccharide is, with the exception of celery RG-II, extended to a heptasaccharide by the addition of a single Rha residue. A second Rha is then added followed by an Ara residue to generate the nonasaccharide ( Table 3 ). The nonasaccharide is the largest B sidechain glycoform we detected. By contrast, Wu and coworkers have reported that the nonasaccharide is extended by an as yet unidentified hexose in the RG-II of sword-leaf dogbane (Apocynum venetum) 27 Further studies are required to determine if this glycoform exists in other plants.
We found no discernible correlation between the structures of sidechains A and B. For example, sidechain B is a hexasaccharide in celery and accounts for one-third of the B sidechain in banana RG-II. However, the 3,4-linked
The RG-II Backbone Contains at Least One Methyl-Esterified GalA
The results of previous studies28,29 suggest that the RG-II backbone contains one or more methyl-esterified GalA residues. However, methods to identify and locate such residues are lacking. We have shown that the methyl-esterified GlcA of sidechain A is converted to 6,6∙-2H-Glc by treating RG-II with aq. NaBD4. Thus, we anticipated that this treatment would also convert backbone methyl-esterified GalA to 6,6∙-2H-Gal.
We treated wine RG-II and wine RG-II that had been de-esterified with cold dilute NaOH with aq. NaBD4 (
Fig. 4A
). Deuterium-labeled galactonic acid (GalA-ol), which is formed by C-1 reduction of the reducing GalA at the end of the backbone, was generated from both RG-IIs (
Fig. 4B
). By contrast, NaBD4 reduction of esterified RG-II was the only treatment that resulted in a decrease in the abundance of GalA and GlcA and the appearance of the 6,6∙-2H-galactitol and 6,6∙-2H-glucitol derivatives (
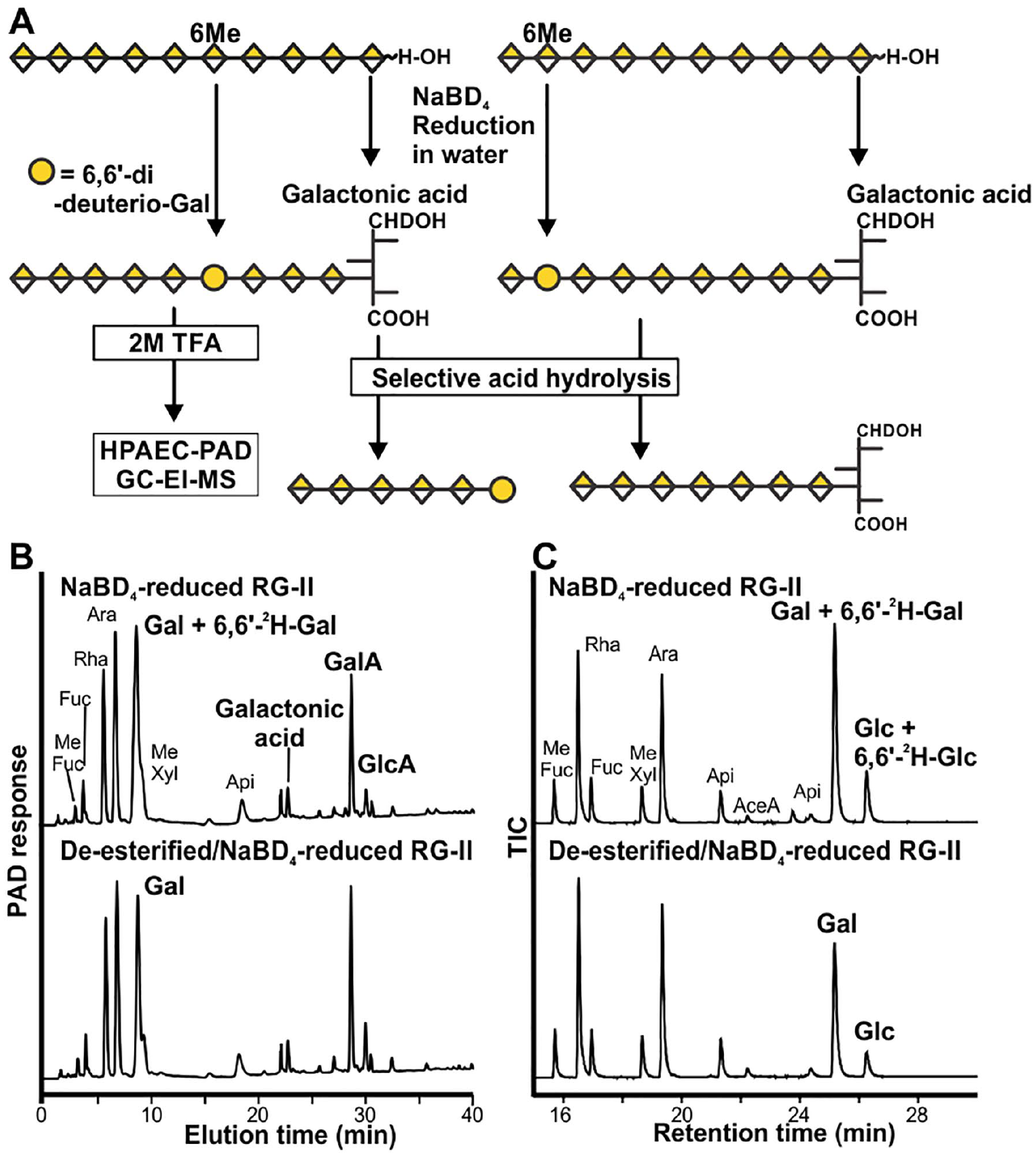
A protocol to convert the methyl-esterified backbone GalA residues to 6,6∙-2H-Gal. (
To determine the location of the methyl-esterified GalAs, we took advantage of the fact that backbone fragments can be generated by selective acid hydrolysis of RG-II. 17 The glycosidic bond of GalA is much more resistant to acid hydrolysis than the other glycosidic bonds. 46 Indeed, we detected no backbone fragments containing portions of any sidechain during MS analyses of the oligosaccharides formed by this treatment. The introduction of a Gal residue into the backbone by NaBD4 reduction generates a glycosidic bond (–4Gal–4GalA–) that is more susceptible to acid hydrolysis than the 4GalA-4GalA bond (see Fig. 4A ). We expected that selective acid hydrolysis would generate a diverse series of backbone fragments if this Gal is distributed randomly. A smaller number of fragments is anticipated if the Gal is not randomly distributed.
We treated NaBD4-reduced wine RG-II (BD4-RG-II) and its de-esterified and reduced counterpart (OH-RG-II) for 16 h at 80 °C with 0.1 M TFA and then analyzed the oligosaccharides generated by MALDI-TOF MS. The mass spectra of the fragments generated from OH-RG-II ( Fig. 5A ) and BD4-RG-II ( Fig. 5B ) differ substantially from one another. The BD4-RG-II spectra contain predominant ions at m/z 885 and 1061 corresponding to [M–H]– of GalA4-6,6∙-2H-Gal and GalA5-6,6∙-2H-Gal, respectively, as well as ions at m/z 1058, 1234, and 1410 that correspond to GalA5-GalAol, GalA6-GalAol, and GalA7-GalAol, respectively. The mass spectra of the OH-RG-II fragments ( Fig. 5A ) are dominated by ions at m/z 882, 1058, 1234, 1410, and 1586. The masses of the GalAn-GalAol fragments are 18 amu lower than expected for a series of oligosaccharides composed of GalA and galactonic acid. For example, GalA7-GalAol, which has a nominal mass of 1429, gives a predominant ion at m/z 1410 [M–18-H]–. The [M–H]– ion is far less abundant. Ions that are 18 amu lower than expected also dominate the mass spectrum of standard oligogalacturonides that had been treated with NaBD4 ( Suppl. Fig. S5A ). Oligogalacturonides that had not been treated with NaBD4 gave [M–H]– ions with the expected masses ( Suppl. Fig. S5B ). We suspect that oligogalacturonides terminated at their former reducing end with galactonic acid lactonize when concentrated to dryness in the presence of the Nafion/DHB matrix.
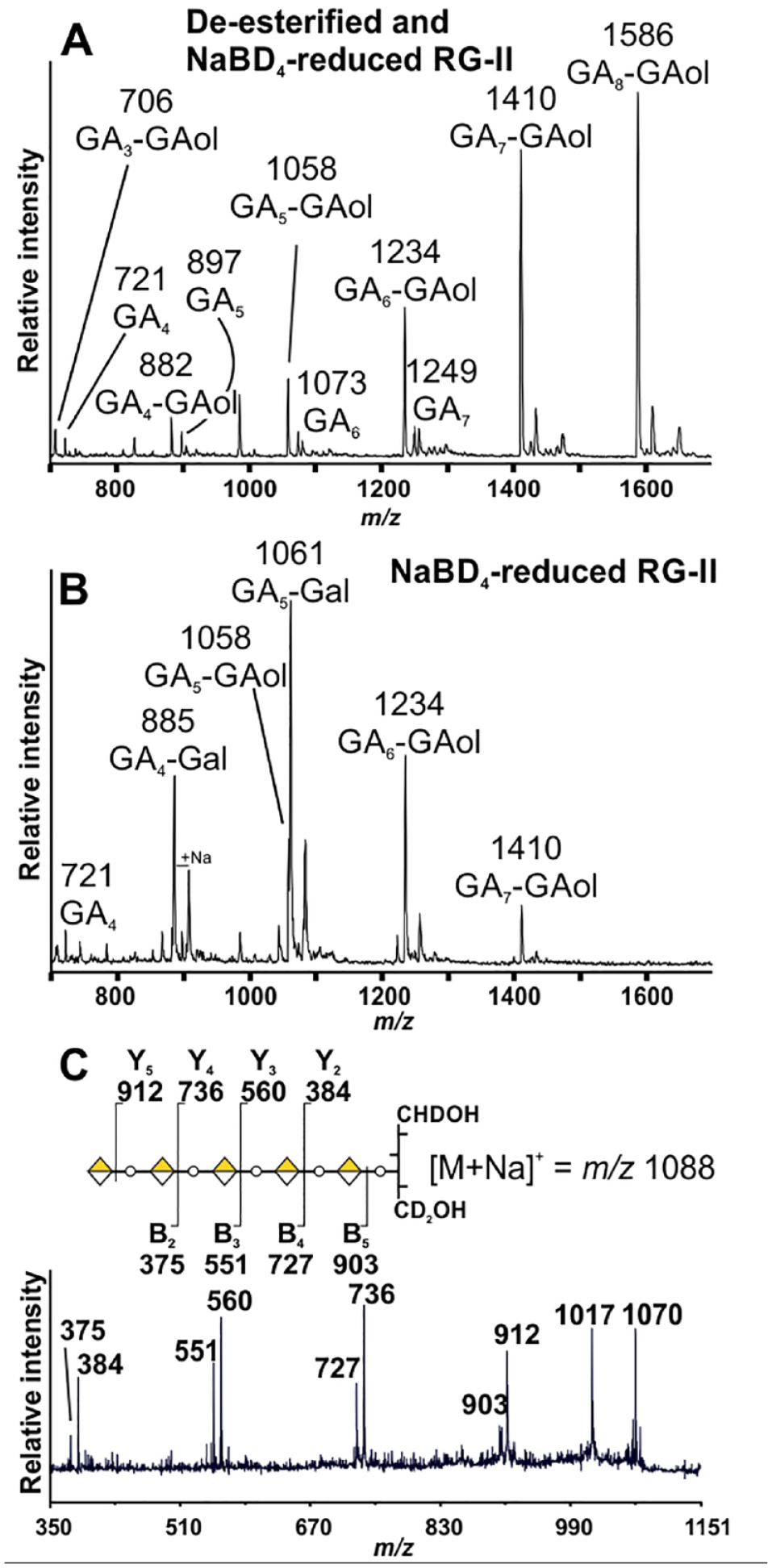
The backbone fragments generated by selective acid hydrolysis of NaBD4-reduced wine RG-II. (
To facilitate locating the Gal in the GalAn-6,6∙-2H-Gal fragments, we first converted the mixture of GalAn-6,6∙-2H-Gal fragments to their corresponding GalAn-1,6,6∙-2H-galactitols using NaBD4. The positive ion MS 2 spectra of GalA5-1,6,6∙-2H-galactitol contained a series of Y fragments ions 47 at m/z 384, 560, 736, and 912 and B fragments ions at m/z 375, 551, 727, and 903 ( Fig. 5C ), which are consistent with the sequence GalA-GalA-GalA-GalA-GalA-Gal. If this fragment was generated by hydrolysis of a Gal-GalA glycosidic bond and assuming that the RG-II backbone contains between 9 and 11 GalAs, then the methyl-esterified GalA is likely located between three and four residues from the reducing end of the backbone.
We next analyzed RG-II isolated from several different plants to determine if the number and location of the backbone methyl ester is conserved. The fragments generated from most of the RG-IIs have similar MALDI-TOF mass spectra ( Suppl. Figs. S6 and S7 ) and HPAEC-PAD oligosaccharide fingerprints ( Suppl. Figs. S8 and S9 ). The ion at m/z 1061, which corresponds to GalA5-6,6∙-2H-Gal, was predominant, which suggests that the methyl-esterified GalA is located between three and four residues from the reducing end of the backbone. A second ion corresponding to GalA7-GalAol (m/z 1410) varied considerably in its abundance. This oligosaccharide is particularly abundant in the fragments generated from celery, spiderwort, rose, and tobacco cell RG-II ( Suppl. Fig. S6 ). Such a fragment may be formed if a methyl-esterified GalA is located close to the nonreducing terminus. A methyl-esterified backbone GalA has been reported to be located between the E and F substituents, which are themselves close to the nonreducing terminus of wine RG-II. 18
The MALDI spectra of the backbone fragments generated from banana, horsetail, and Wolffiella RG-II were dominated by the ions at m/z 1234 (GalA6-GalAol) and 1410 (GalA7-GalAol). GalA5-6,6∙-2H-Gal was much less abundant ( Suppl. Fig. S7 ). Thus, the predominant site of GalA methyl esterification in these RG-IIs is likely located close to the nonreducing terminus.
The Role of Cations in Dimer Self-Assembly and Stability
In previous studies we showed that in vitro the RG-II monomer self-assembles into a dimer in the presence of boric acid and selected divalent cations, most notably Pb2+, Ba2+, and Sr2+.19,20 Here, we have further investigated the effects of Pb2+ on dimer formation and stability. We used acetate buffer in these experiments since adding aq. Pb2+ salts (>2 mM) to the phthalate buffer used previously resulted in the formation of a precipitate. No precipitate was discernible when we added the Pb2+ salts to 50 mM NaOAc, pH 4.
The rate of dimer formation is increased by increasing the Pb2+ concentration from 0.1 to 0.5 mM. However, dimer formation is inhibited at Pb2+concentrations above 0.5 mM ( Fig. 6A ). This inhibition is largely a consequence of slowing rather than preventing dimer formation ( Fig 6B ). Pb2+ inhibition is reversible as it suppressed by adding Ca2+ ( Fig. 6C ). Other cations are far less effective than Ca2+ ( Fig. 6D ). However, Ca2+ is itself far less effective than Pb2+ at increasing dimer formation at low concentrations of boric acid ( Fig. 6B,E ).
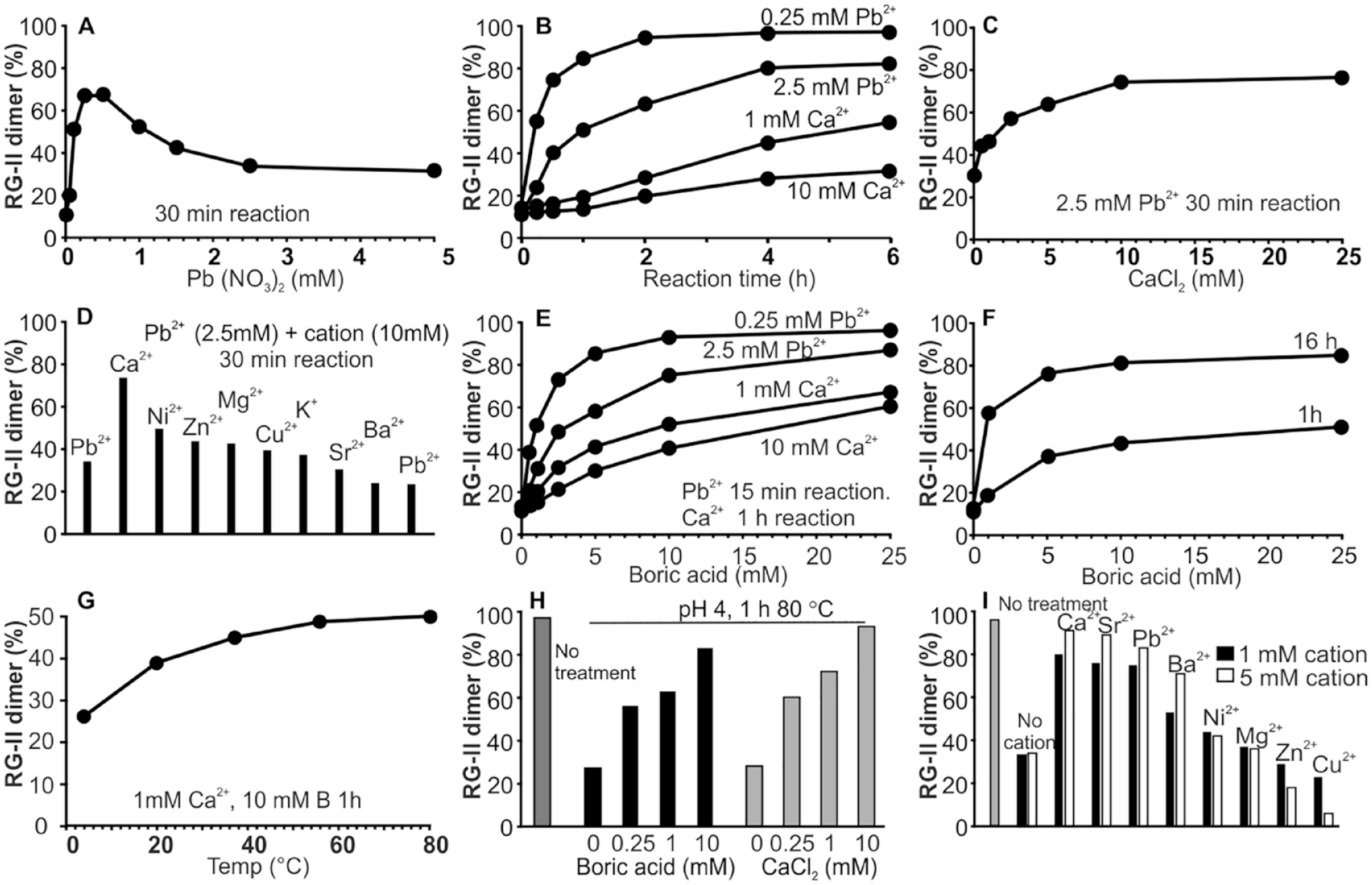
The effects of selected cations and temperature on RG-II dimer self-assembly in vitro. (
One or more of the GalA carboxylates present in RG-II likely form intramolecular electrostatic bonds with Pb2+ and Ca2+. 48 Polygalacturonates also form chelate complexes with Pb2+, and the resulting stabilized structure is likely to have a relatively high Ka value. 48 High concentrations of Ca2+ may prevent the formation of such a complex.
The Pb2+-dependent inhibition of dimer formation is also suppressed by increasing the boric acid concentration ( Fig. 6D ). This was unexpected, since the dimer forms slowly in the presence of boric acid alone ( Fig. 6F ). Determining the nature and geometry of Pb2+ binding to RG-II, as well as any cation- or boric acid-induced structural changes, will require solution conformational analyses and molecular modeling studies.
We have shown that dimer self-assembly occurs rapidly at room temperature. However, the temperature dependence of this reaction has not been investigated. We obtained a substantial increase in the amounts of dimer formed within 1 h when the reaction temperature was increased from 4 to 80 °C ( Fig. 6G ). This was not expected since substantial amounts of the monomer are formed when the dimer is kept for 1 h at pH 4 and 80 °C ( Fig. 6H ). This led us to suspect that at 80 °C the stability of the dimer is dependent on the presence of boric acid or Ca2+. Indeed, the dimer was stable at pH 4 and 80 °C when boric acid (0.25–10 mM) or Ca2+ (0.25–10 mM) was present. Comparable stabilizing effects were also obtained with Sr2+, Pb2+, and Ba2+. Other divalent cations (Ni2+, Mg2+, Zn2+, or Cu2+) were less effective ( Fig. 6I ). Together these data lead us to suggest that dimer stability is controlled to a large extent by the types of cations present and the concentrations of these cations and borate. Such data are also consistent with the long-term stability of the RG-II dimer in red wine, which typically contains micromolar concentrations of calcium and boric acid. 49
Discussion
There is an increasing awareness that nonglycosyl substituents including methyl ethers, methyl esters, and acetyl esters have important roles in controlling the functions, properties, and interactions of plant cell wall polysaccharides.50–52 In this study, we have developed new methods to study the methylation status of RG-II. Our data, together with the results of others,23–25,27,30 clearly show that even though the glycosyl sequence of RG-II is conserved there are notable differences in the primary structures of the A and B sidechains.
Most, if not all, plants form RG-II that contains sidechains A, B, C, and D and the F substituent ( Fig. 1A ). The E substituent may have a less widespread distribution. 18 The B sidechain shows the most structural variation as it differs most notably in the number of Ara and Rha residues added to a core hexasaccharide. Our data also provide evidence that methyl esterification of one or more backbone GalAs, methyl esterification of the A sidechain GlcA, and methyl etherification of the A sidechain β-GalA are near universal. However, the extent of esterification and etherification is not identical in each RG-II. These levels are likely controlled by the activities of the yet to be identified transferases that catalyze the addition of nonglycosyl substituents to RG-II. We do not know if the variability in the abundance of nonglycosyl substituents and the existence of sidechain glycoforms affect the biological functions of RG-II. RG-II isolated from healthy plants typically exists as a dimer, suggesting that natural structural variants are not compromised in their ability to form this complex. Nevertheless, it is conceivable that such structural variants exist to enable the in vivo rates of dimer formation to be tailored to the growth requirements of a plant.
Methyl etherification of GalA in sidechain A is a widespread modification, albeit one that differs in the relative abundance of each GalA methyl ether. Nothing is known about the biological roles of these methyl ethers or the biosynthetic routes that lead to their synthesis. O-Methylation of GalA is known to result in a small but discernible decrease in the pK of this hexuronic acid, 53 although it is unclear if this would affect the functions of RG-II. The β-GalA of sidechain A may be close in space to the apiose that is believed to participate in the borate cross-link. GalA forms a borate ester at O-3 and O-4. 52 Methylation at O-3 or O-4 will prevent the formation of this ester. Nevertheless, the nonstoichiometric methylation of GalA in many RG-IIs likely precludes this as a universal mechanism to control the site of borate esterification.
The B sidechain of RG-II isolated from several nonflowering plants contains 3-O-methyl Rha. 23 This methylated glycose is replaced by Rha in the RG-II of flowering plants, a change that has no discernible effect on the ability of the RG-II to form a dimer. Thus, the possibility cannot be discounted that O-methylation of GalA no longer serves a function even though the enzymes that catalyze O-methylation have not been eliminated during the evolution of vascular plants. Deciphering the role of O-methyl GalA in sidechain A will require the identification of the methyltransferases that catalyze the addition of this substituent, as well as the isolation of mutant plants that lack these enzymes.
Methyl esterification of GlcA is expected to have a substantial effect on the charge status of sidechain A, which may in turn affect the conformations that this sidechain can adopt. Altering the structure of sidechain A is known to decrease the ability of RG-II to form a dimer in vitro and in vivo.11,14,54 Nevertheless, the extent of GlcA methyl esterification varies in different plants. In celery, GlcA is methyl-esterified, whereas little esterified GlcA was detected in horsetail and Wolffiella RG-II, nor was it detected in one species of mistletoe (V. coloratum) RG-II. 27 The low abundance of this modification does not affect RG-II dimer self-assembly, since Wolffiella welwitschii RG-II readily forms a dimer in vitro. 30 However, the rates of dimer formation of Wolffiella RG-II and an RG-II that contains methyl-esterified GlcA have not been compared. New insight into the role of GlcA methyl esterification in RG-II function will require the identification and characterization of the methyltransferases that catalyze these modifications.
It is well established that 3,4-linked
Our study provides evidence that two of the backbone GalAs are methyl-esterified. We could not precisely locate these methyl esters using our current methods because the backbones of the RG-IIs studied are heterogeneous and typically contain between 9 and 11 GalAs. Nevertheless, one ester may be in the region of the reducing terminus and one toward the nonreducing terminus. A methyl-esterified backbone GalA located near the nonreducing terminus was also identified in a study of oligosaccharides generated by enzymic fragmentation of RG-II. 18
Adding methyl esters to specific GalAs within a homogalacturonan chain may provide structural cues that guide the glycosyltransferase involved in sidechain initiation during RG-II synthesis. Up to six glycosyltransferases—two apiosyltransferases, two arabinosyltransferases, one 3-deoxy-
Sidechain A contains two 6-deoxy hexoses (Rha and Fuc), a MeXyl, a methyl-esterified GlcA and a methyl-etherified GalA. The B sidechain contains at least one Rha, a MeFuc and up to two O-acetyl substituents. Natural variations in these substituents do not prevent self-assembly, since RG-II isolated from healthy plants exists as the dimer. Nevertheless, interactions between methyl- and acetyl-containing domains within each RG-II monomer may have a role in initiating dimer assembly or contribute to dimer stability.
Methyl esterification of a backbone GalA, irrespective of its position along the backbone, will limit its ability to interact with cations and may provide a mechanism to enable specific backbone–cation interactions. Cations are known to have a role in borate ester cross-linking in vitro20,21 and in vivo. 56 Our data suggest that divalent cations have a complex role in the self-assembly process and in maintaining dimer stability. Moreover, different sites within RG-II may have substantially different affinities for cations and thus have a role in maintaining the molecular conformations of RG-II required for self-assembly to occur.
We have provided evidence that the RG-II dimer is stable in the presence of borate or Ca2+, even at elevated temperatures. This may provide an explanation for why cell wall boron exists in a nonexchangeable form and why plants require a constant supply of borate for normal growth. 57 The RG-II dimer will be stable and its borate not available for reuse if the apoplastic Ca2+ concentration is at normal values (1–2 mM). 58 Progress in understanding the mechanisms involved in dimer self-assembly and stability is hindered by our current lack of knowledge of the solution conformations that RG-II can adopt and how these conformations interact with and are affected by borate and divalent cations.
The fact that RG-II self-assembles into a dimer makes it one of the few plant cell wall polysaccharides with a property that can be readily manipulated and investigated in vitro. Methods are now available to study if and how this process is affected by the natural structural diversity of RG-II, by modifying the RG-II structure with site-specific enzymes, 18 or by structural variants obtained from mutant plants with defects in one or more aspects of RG-II biosynthesis.11,14,54 The expansion of such studies may also shed light on whether the structural complexity of RG-II is required solely to maintain its interaction with borate and divalent cations or if it is required for other biological processes. Computational approaches to develop dynamic conformational models describing dimer self-assembly are required to unravel the molecular-level interactions that drive this process. However, such studies face considerable challenges due to the complex structure of RG-II and the diversity of its nonglycosyl substituents. Progress in this area will benefit greatly by identifying the minimum structural features of RG-II that are required for normal cross-linking to occur.
Supplemental Material
Supp-FigS1-S8_ONeill-Rev2 – Supplemental material for Locating Methyl-Etherified and Methyl-Esterified Uronic Acids in the Plant Cell Wall Pectic Polysaccharide Rhamnogalacturonan II
Supplemental material, Supp-FigS1-S8_ONeill-Rev2 for Locating Methyl-Etherified and Methyl-Esterified Uronic Acids in the Plant Cell Wall Pectic Polysaccharide Rhamnogalacturonan II by Malcolm A. O’Neill, Ian Black, Breeanna Urbanowicz, Vivek Bharadwaj, Mike Crowley, Sabina Koj and Maria J. Peña in SLAS Technology
Footnotes
Supplemental material is available online with this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The authors acknowledge the Division of Chemical Sciences, Geosciences, and Biosciences, Office of Basic Energy Sciences of the United States Department of Energy, through Grant DE-SC0008472 for funding structural studies of RG-II and Grant DE-SC0015662 for funding analytical instrumentation support at the Center for Plant and Microbial Complex Carbohydrates at the Complex Carbohydrate Research Center.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
