Abstract
Novel methods in nuclear magnetic resonance (NMR) spectroscopy have recently been developed to investigate the binding properties of intermolecular complexes endowed with biomedical functions. Among these methods is the saturation transfer difference (STD), which enables the mapping of specific binding motifs of functional ligands. STD can efficiently uncover the specific and preferential binding sites of these ligands in their intermolecular complexes. This is particularly useful in the case of glycosaminoglycans (GAGs), a group of sulfated polysaccharides that play pivotal roles in various biological and pathological processes. The activity of GAGs is ultimately mediated through molecular interactions with key functional proteins, namely, GAG-binding proteins (GBPs). The quality of the GAG-GBP interactions depends on sulfation patterns, oligosaccharide length, and the composing monosaccharides of GAGs. Through STD NMR, information about the atoms of the GAG ligands involved in the complexes is provided. Here we highlight the latest achievements of the literature using STD NMR on GAG oligosaccharide-GBP complexes. Interestingly, most of the GBPs studied so far by STD NMR belong to one of the three major classes: coagulation factors, growth factors, or chemokine/cytokines. Unveiling the structural requirements of GAG ligands in bindings with their protein partners is a crucial step to understand the biochemical and medical actions of GAGs. This process is also a requirement in GAG-based drug discovery and development.
Introduction
In the past two decades, a variety of analytical techniques have been developed for explicit characterization of the interactions between proteins and their ligands. Among these are mass spectrometry, 1 surface plasmon resonance spectroscopy, 2 microarray, 3 and nuclear magnetic resonance (NMR) spectroscopy. 4 NMR has emerged as a powerful technique capable of uncovering a great amount of details regarding protein-ligand complexes in solution. This analytical technique can study a large variety of proteins because of the assessment of an impressive range of binding affinities, going from 10−9 to 10−3 molar. 4
With respect to the diversity of NMR experiments that can be conducted to investigate properties of intermolecular complexes in solution, three individual goals can be defined. The first would be the understanding of the specific amino acid residues involved in the protein-ligand interactions. The second would be the mapping of atoms of the binding ligand(s) involved in the interactions. The third would be the bound conformations of the ligand necessary for the proper and best interactions and how these conformations change from the free (unbound) status to the bound status. Regardless of the goal wanted in research, NMR spectroscopy can surely provide methods capable of overviewing all of these three aspects of the protein-ligand complexes. The first goal can be addressed by chemical shift perturbation (CSP), the second can be addressed by saturation transfer difference (STD), and the third can be addressed by the transferred nuclear Overhauser effect (trNOE).5,6
STD
The assessment of the ligand properties necessary for the specific interactions with their protein partners is crucial in any science concerned with the functionality of these proteins, including those of medical interest. As pointed above, the NMR STD method can properly address the questions related to how the ligands bind to their protein partners during intermolecular complex formation. As presented by Mayer and Meyer,
7
the NMR spectrum of the saturated state (on-resonance) in interaction with its protein partner is compared with the NMR spectrum of the unsaturated state (off-resonance) of the unbound ligands in solution (
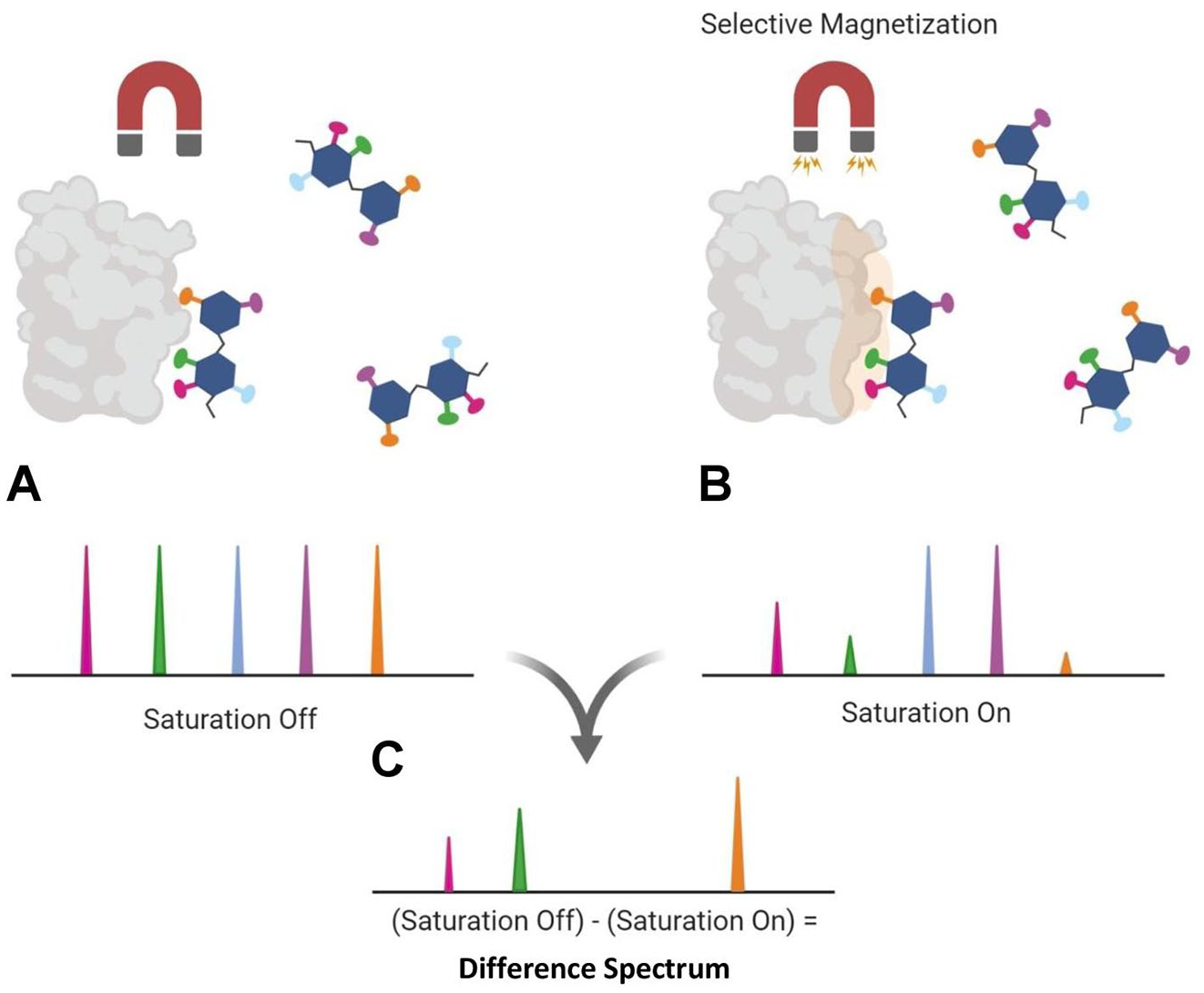
The overall saturation transfer difference (STD) nuclear magnetic resonance (NMR) mechanism is illustrated. (
Through STD NMR, dissociation constants of the complexes (KD) can be determined. 8 This experiment is typically performed with a sample prepared with ~5- to 100-fold molar excess of ligand in comparison with the protein concentration in solvent. This ensures that all observed signals are strictly from the ligand under investigation. Therefore, it is important to perform an on-resonance control experiment of the ligand being observed in solution without the protein. This ensures that no signals from the ligand are being observed from the selective pulse designed for the protein. In case signals from the protein are still present from the selective magnetization, these unwanted signals from the protein without the bound ligand can be filtered out from the STD spectra. This method is called saturation transfer double-difference (STDD).9,10 Although primarily conducted as a one-dimensional (1D) NMR experiment, STD can also be applied in two-dimensional (2D) NMR experiments. An example are the studies involving NMR in the heteronuclear single-quantum coherence (HSQC) spectrum 11 in events in which signal overlap of the complexed ligand is too difficult for interpretation through the usual 1D techniques. 12
The application of STD is subject to several experimental conditions, besides the concentration requirements as described above. These conditions must be carefully considered during the experiment so that accurate and well-resolved information can be acquired from the STD NMR technique. Among them is molecular weight (MW). STD signals can be limited by the MW of the molecule with which the ligand is interacting. Medium-sized or small macromolecule (<15 kDa) will not be subject to the intense 1H- 1 H cross polarization seen for larger complexes.13,14 The magnetization source leading to spin diffusion is the selective saturation applied to the protein in the on-resonance spectrum. As described above, this magnetization is intended to only excite the protein involved in the complex. Thankfully, in the case of carbohydrates, the frequently irradiated regions of the proteins do not overlap with the carbohydrate signals. 15 Because many binding proteins show their binding sites composed of lysine and arginine residues, the methylene protons of these amino acids can be chosen as targets for specific magnetization. 16 The length of this magnetic excitation is also crucial to the mechanism of STD. The excitement must be long enough to allow proper magnetization of the ligand from the initially radiated protein in the bound state. To assess this phenomenon, signal intensity buildup curves should be performed often to determine the optimal length of saturation time. 17 The preparation of the samples to be analyzed by STD NMR is also routinely performed using buffered solutions in which the salt concentrations can mimic the physiological conditions. 18 Temperature may also play a key role in the quality of the STD-derived information. Although frequently performed at room temperature (25 °C), the use of lower temperatures can be explored to increase resolution.15,19
STD was originally developed for use in screening of potential pharmaceutical agents from libraries of small MW compounds. Currently, STD NMR has been applied in a more diverse set of investigations. Recent examples are studies in which multiple ligands exist for interactions with functional biomacromolecules. More specifically, STD has been applied in the research of nucleic acids 20 and peptides 21 and, in a later stage, optimized for carbohydrate studies. 22 Recent analytical advancements have also greatly increased the types of binding targets subjected to scientific investigations. Whole cells, 23 viruses, 24 and G protein–coupled receptors (GPCRs) 25 are among these recent new binding targets successfully studied nowadays through STD NMR. Conclusions from these experiments are viable for therapeutic applications. The use of multiple ligands in solution allows for the further mapping of the specific binding epitopes of structurally similar ligands under the same screening. This information is exceptionally relevant in investigations of structurally complex polysaccharides that participate in a wide range of biomedical functions. Example of these polysaccharides endowed of numerous biomedical actions are the glycosaminoglycans (GAGs). GAGs are vastly exploited in the market and research as both pharmaceutical and nutraceutical ingredients.
GAGs and GAG-Binding Proteins
GAGs are a structurally diverse group of complex linear polysaccharides that are often highly sulfated and of high MW.5,26 GAGs are expressed as heterogeneous chains composed of disaccharide building blocks. These building blocks are in turn composed of alternating units of one hexosamine (N-acetylglucosamine [GlcNAc] or N-acetylgalactosamine [GalNAc]) and one hexuronic acid (glucuronic acid [GlcA] or iduronic acid [IdoA]) or a galactose (Gal) unit.5,26 Depending on the composition, GAGs fall within one of the four major specific families: heparin (Hp)/heparan sulfate (HS), hyaluronic acid (HA), chondroitin sulfate (CS)/dermatan sulfate (DS), and keratan sulfate (KS). All of these can vary greatly in terms of N- and O-sulfation patterns, except HA, which is naturally not sulfated.26,27 The structures of these four groups are depicted in
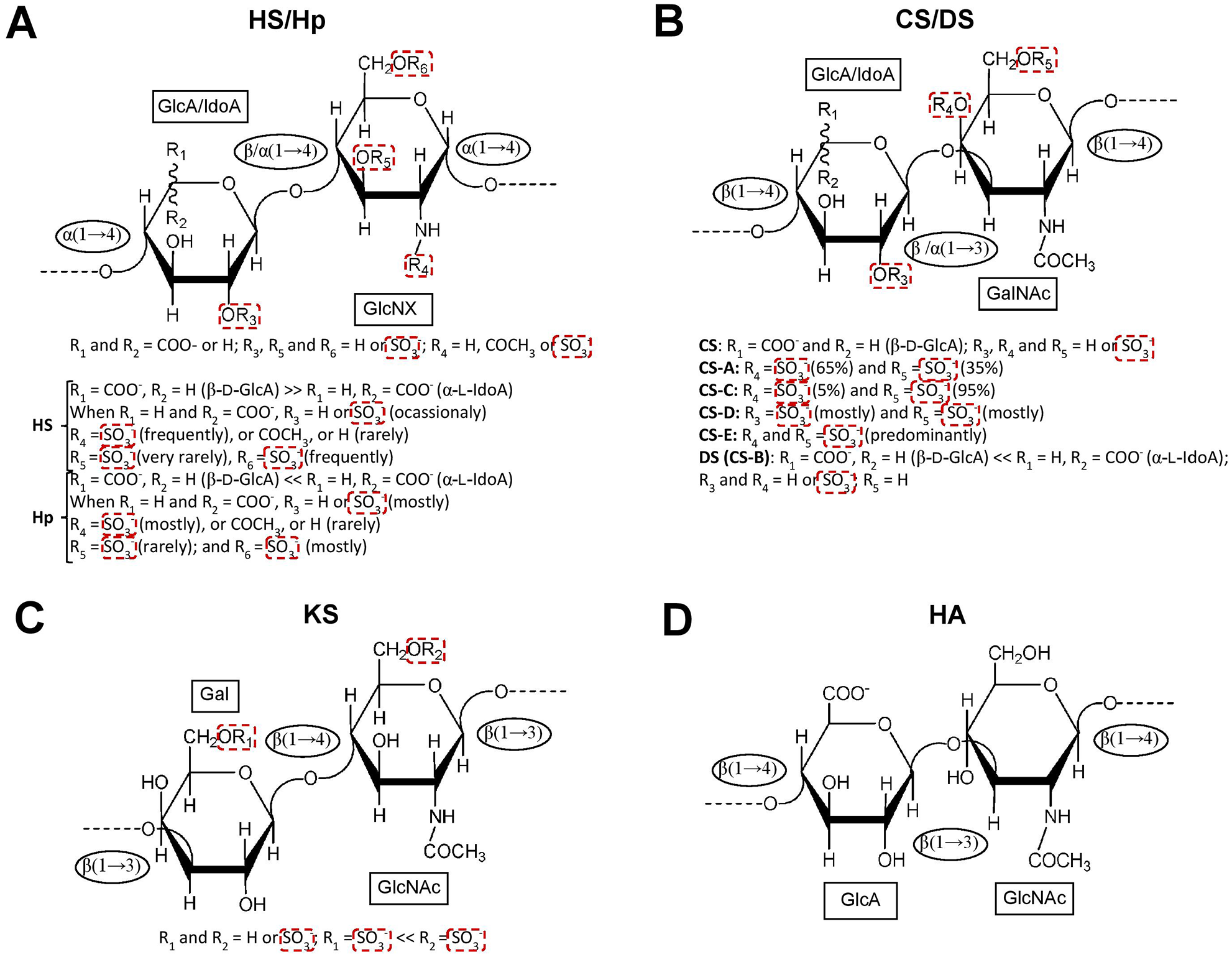
The structural representation of the four major glycosaminoglycan (GAG) families is provided. (
GAGs are highly evolutionarily conserved across animalia as a key component of the extracellular matrix (ECM). These molecules act as binders and regulators of a variety of key proteins involved in numerous physiological and pathological events.28–33 Because GAGs show high degrees of structural diversity, they can interact and control multiple proteins. These proteins are colloquially named GAG-binding proteins (GBPs). This structural complexity depends on the microheterogeneity of the oligosaccharide sequences seen in their polymeric chains. Structural specificity is a key determinant for protein interactions. 32 Hence, the discovery of binding motifs or functional epitopes in GAG molecules is a priority.
The GBPs are key players in the coagulation system,34,35 inflammatory response, 36 tumorigenesis, 37 tissue development, repair and fibrosis, 38 neurologic functions, 39 and virus infections.40,41 Although hundreds of GBPs have been identified 42 and are currently available for biomedical and physicochemical studies, those who have been investigated by STD NMR lately are in fact the minority. Interestingly, over the past decade, STD NMR studies of GAGs have been primarily focused on three major groups of GBPs: coagulation factors, fibroblast growth factors (FGFs), and cytokines/chemokines. A few other reports may exist but can still be generalized as exceptions.43,44 In all three of these GBP groups, the binding appears to be reliant on variable features within GAGs. These are chain length, specific sulfation pattern and planar 3D arrangement of the sulfate groups, and the monosaccharide composition.
Because the GAG structures are often expressed heterogeneously in nature, a common theme within the STD studies of the three GBPs is the exploitation of synthetically produced GAG-related defined oligosaccharides. The production of structurally defined oligosaccharides in terms of size and sulfation patterns assist the structure-activity relationship to define the proper or best GAG ligand or motif responsible for GBP interaction and regulation. This is usually accomplished through the use of oligosaccharides with lengths of four (tetrasaccharide), six (hexasaccharide), or eight (octasaccharide) units. These ligands enable more robust conclusions regarding the binding contributions of the individual monosaccharides and their sulfation patterns. Identification of the individual units within longer oligosaccharides becomes much more difficult primarily because of the chemical shift overlap (internal monosaccharides with nearly identical physicochemical properties). On the other hand, smaller oligosaccharides are not always the functional epitopes, although key conclusions can be reached using these short fragments. 45 Below, we highlight the main findings obtained so far by STD NMR studies of GAGs in interactions with the three major GBPs.
Coagulation Factors
Antithrombin
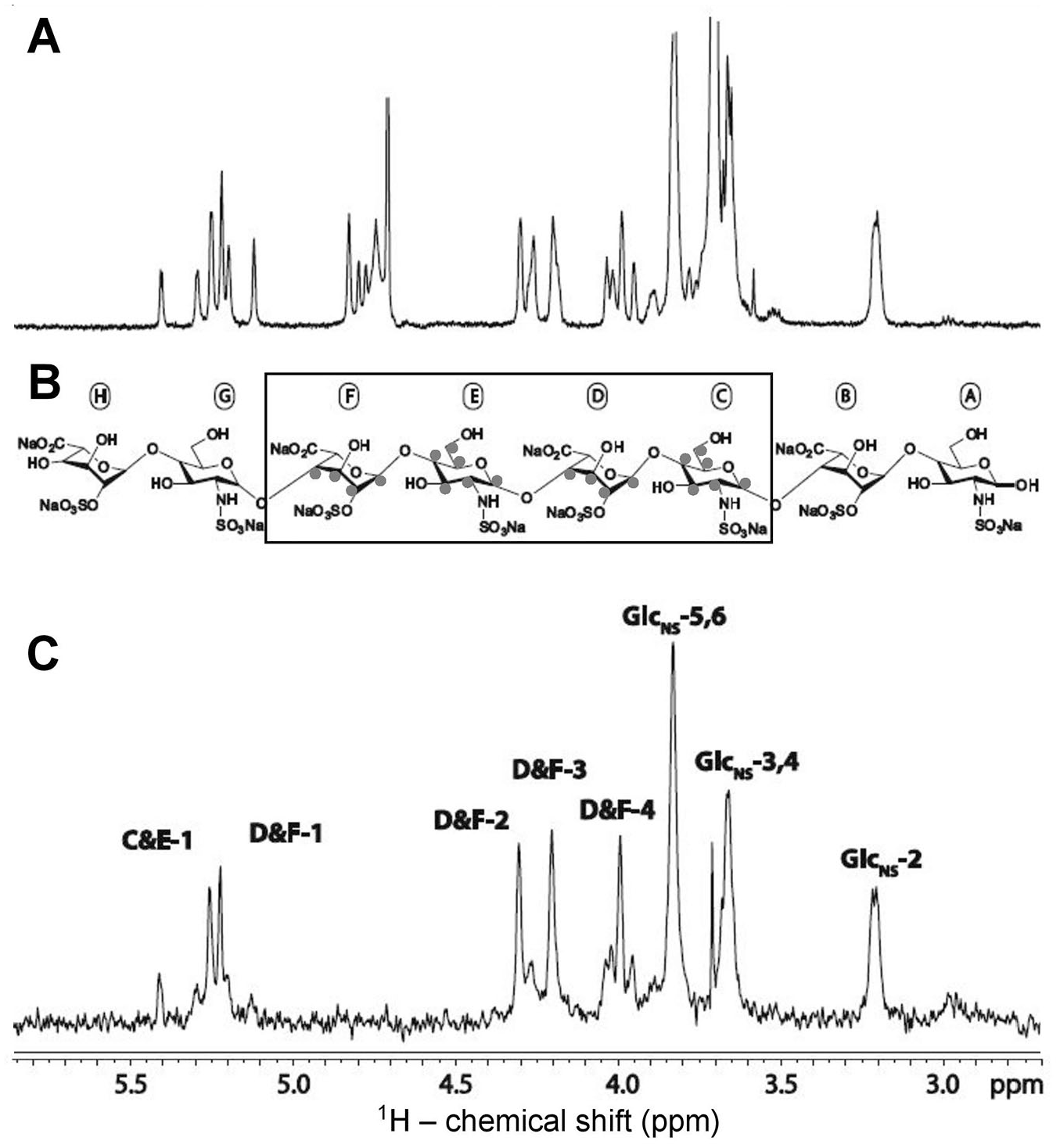
The most commonly exploited medical application of GAGs is the anticoagulant activity that is exhibited by Hp (
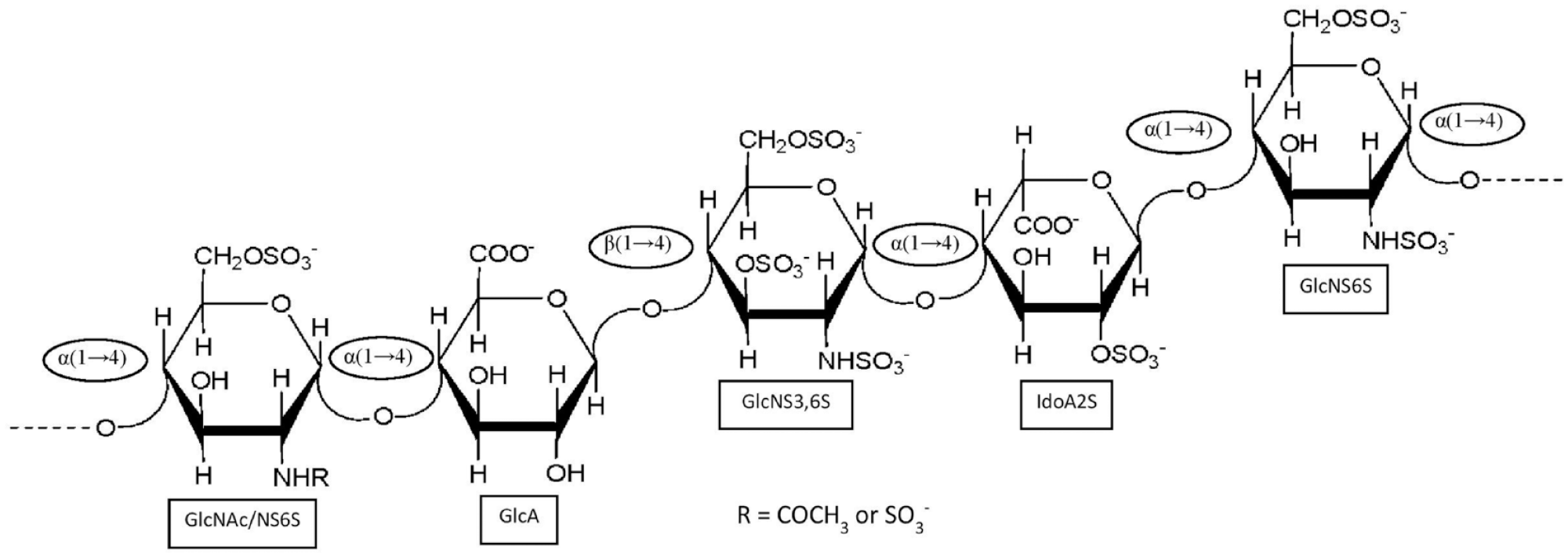
The antithrombin-binding heparin sequence is composed of the following pentasaccharide structure: [→4)-D-GlcNAc/GlcNS6S-α-(1→4)-D-GlcA-β-(1→4)-D-GlcNS3S6S-α-(1→4)-L-IdoA2S-α-(1→4)-D-GlcNS6S-α-(1→].
Stancanelli et al.
47
sought to further elucidate the importance of the pentasaccharide motif (
The hexasaccharides used in saturation transfer difference (STD) studies in the interaction with antithrombin (AT) are pictured. Both oligomers contain the AT-binding heparin pentasaccharide but with slight variation on the sulfation pattern as well as an addition of the glucuronic acid present at the reducing end. The hexasaccharide at (
In further work, Guerrini et al. 48 identified an increase in binding magnitude with the addition of sulfation on the 3-O-position of the GlcNS3S6S unit in the pentasaccharide sequence. 48 This work has validated the contribution of the rare and functional 3-O-sulfation site of the pentasaccharide for the proper interaction with AT. Viskov et al. 49 performed STD of a decasaccharide consisting of two contiguous pentasaccharide moieties as described earlier. The STD spectrum has revealed signals across the entire decasaccharide. However, the most intense signals were observed in the nonreducing end pentasaccharide sequence, suggesting some preference for one specific side of the decasaccharide. 49
FGFs
FGF2 and FGF10
FGFs are another family of GBPs that has been widely investigated using STD NMR technology. FGFs are key signaling proteins involved in multiple biological processes. Examples of these events are regeneration of both hard and soft tissues across multiple organ systems.50–52 These regulatory functions of FGFs have been exploited for therapeutic applications toward certain diseases such as Parkinson’s disease and diabetes. 53 The 23 currently known FGFs exhibit a conserved amino acid sequence. 53 A few of them display the lack of binding to GAGs, particularly to Hp and CS. 54 Because this relationship is so extensive, STD is a powerful tool to elucidate whether the structure-activity relationship between GAGs and this GBP family is a conserved mechanism or not.
The work of Yu et al. 16 provided excellent information about the FGF interactions. In their report, the authors compared two members of the FGF family, FGF2 and FGF10, in intermolecular complexes with a small library of three synthetic Hp octasaccharides. These Hp octasaccharides differ solely on sulfation patterns. 16 Regarding the Hp oligosaccharide structures, it is crucial to point out that each disaccharide unit is disulfated. However, only octasaccharide 2 contains a disulfated monosaccharide unit, GlcNS6S. Both octasaccharides 1 and 3 are sulfated on the C2 position of IdoA but with distinct sulfations at the hexosamine. Octasaccharide 1 bears the GlcNAc6S unit, whereas octasaccharide 3 bears the GlcNS unit. 16
FGF2 and FGF10 show markedly different patterns in the STD approach. In FGF2 interactions, all three octasaccharides display a transfer of saturation, indicating binding between the protein and ligands. However, only octasaccharide 3 displayed discrete intensities that are indicative of protons crucial to a specific binding interface. To further highlight the specificity of interaction with octasaccharide 3 as compared with the other two Hp oligosaccharides, all three fragments were immersed in the same solution for the STD study. A similar STD on-resonance spectrum of octasaccharide 3 alone was observed. This suggests selectivity of octasaccharide 3 for interaction with FGF2 as compared with the other two octasaccharides. This experiment has proved a preferential affinity toward the Hp fragment containing the GlcNS unit. When viewing the proton STD signals involved in this specific event, it is apparent that only the innermost disaccharide units within the oligosaccharide are involved in the binding. This is supported by the expected lack of signals from both reducing and nonreducing ends of the oligosaccharide chain. The STD results indicate that the Hp ligand binds to the FGF2 preferably by its internal middle region. Taking all data together,
54
it becomes apparent that the GAG binding to FGF2 not only requires a specific spatial arrangement of sulfation present but is also selective toward a residue during the binding (
Nuclear magnetic resonance assignments of the saturation transfer difference (STD) spectrum obtained from the complex of a heparin (Hp) octasaccharide and fibroblast growth factor 2 (FGF2). (
Conclusions yielded from STD NMR studies of FGF10 and the same Hp oligosaccharides proved a much more promiscuous binding event in comparison with the FGF2 study. Of the three used Hp fragments, both octasaccharides 2 and 3 displayed preferential NMR signals alone and of similar intensities when co-incubated in solution. Octasaccharide 2 displayed signals from both monosaccharide units; however, STD signals were mostly pronounced from the GlcNS6S units. This was expected because of the presence of sulfation at only this unit. The resulting STD spectra of octasaccharide 3 with FGF10 displayed similar signals observed in the STD study with FGF2. In both Hp octasaccharides, the signals observed are localized to the innermost residues of their chains. Negligible intensity of reducing and nonreducing ends was seen. This set of results has pointed again toward the fact that GAG oligosaccharides prefer to interact with the two forms of FGFs through the middle of their oligosaccharide chain. 16
Cytokines and Chemokines
Another application of STD in studies of GAGs is to understand their regulatory mechanisms of action over chemokines and cytokines. Regulation of these signaling proteins, mediated by GAG interactions, is crucial for the prevention and mediation of serious autoimmune disorders and inflammatory issues. In comparison with coagulation and fibroblast growth factors, STD-derived information related to chemokines and cytokines has been much more heavily explored lately. This is likely because of the functional and structural diversity of these GBPs themselves. This structural diversity has assisted the information generation about the GAG-binding aspects of these proteins. Motifs found in GAG oligosaccharides are required to form the functional GAG-chemokine/cytokine complexes, and thus, STD NMR studies are key to reveal these findings. The GAG binding is a requirement step for the functions of chemokines/cytokines. Upon interaction, GAG will induce chemokine/cytokine oligomerization. The oligomerized chemokines/cytokines are consequently activated and hold to the surface of vessels in a much tighter fashion so that they can exert their signaling roles to the migration of cells.
CXCL12α
Laguri et al.
55
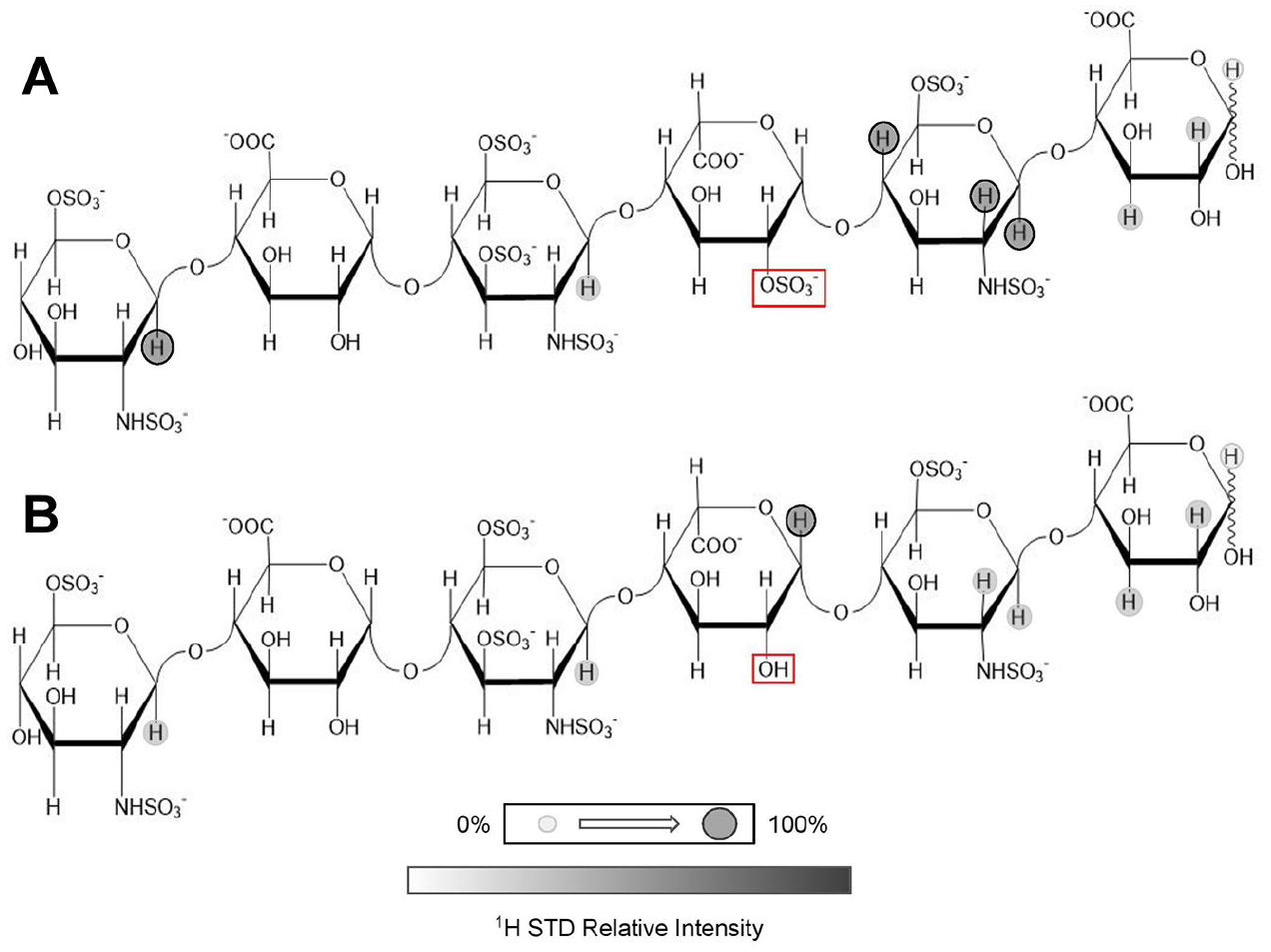
used a synthetic 13C-labeled HS (
STD NMR studies of the CXCL12α-HS oligosaccharide complex provided interesting conclusions regarding the GAG moieties involved in binding of CXCL12α.
Figure 6
illustrates the major findings from the GAG ligand viewpoint. Basically, during the analysis, the resultant NMR spectra displayed a loss of signals from residues not present in the innermost units likely because of the nature of the binding being again focused on the center of the oligosaccharide chain.
55
This occurred in a similar fashion to the results obtained by Yu et al.
54
with FGFs (
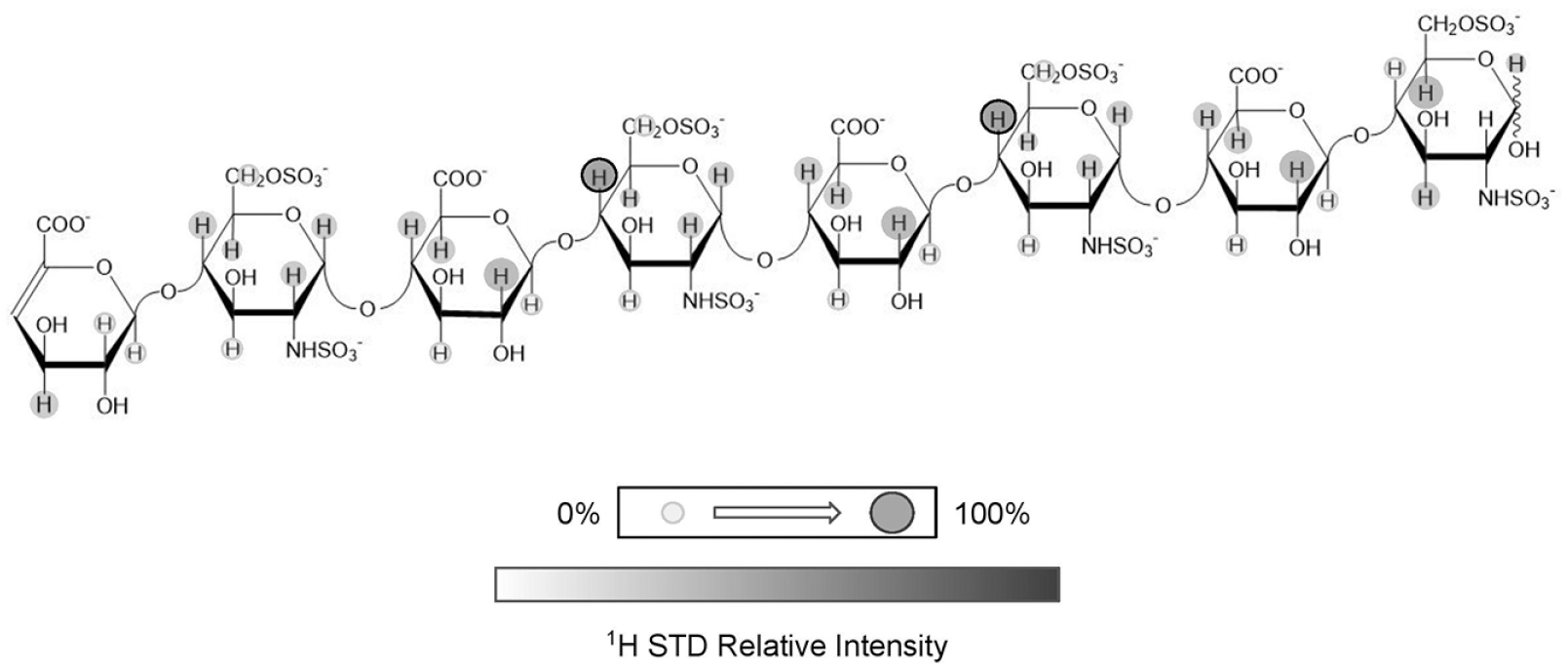
The heparan sulfate (HS) octasaccharide containing the GlcNS6S motif used in the saturation transfer difference (STD) study in the interaction with CXCL12 is displayed. STD intensities at appropriate protons are shown qualitatively through the gray markers present. The intensities are derived from the STD spectra of the excess HS octasaccharide in interaction with CXCL12α in solution after selective magnetization of the chemokine. Adapted with permission from ref. 55, 2011 American Chemical Society.
Midkine
Using CS oligosaccharides, Solera et al.
59
applied STD to investigate their binding with the cytokine Midkine (MK).
59
MK is a GBP that has great binding affinity to Hp (
In their STD studies, Solera et al.
59
employed two synthetic CS tetrasaccharides (
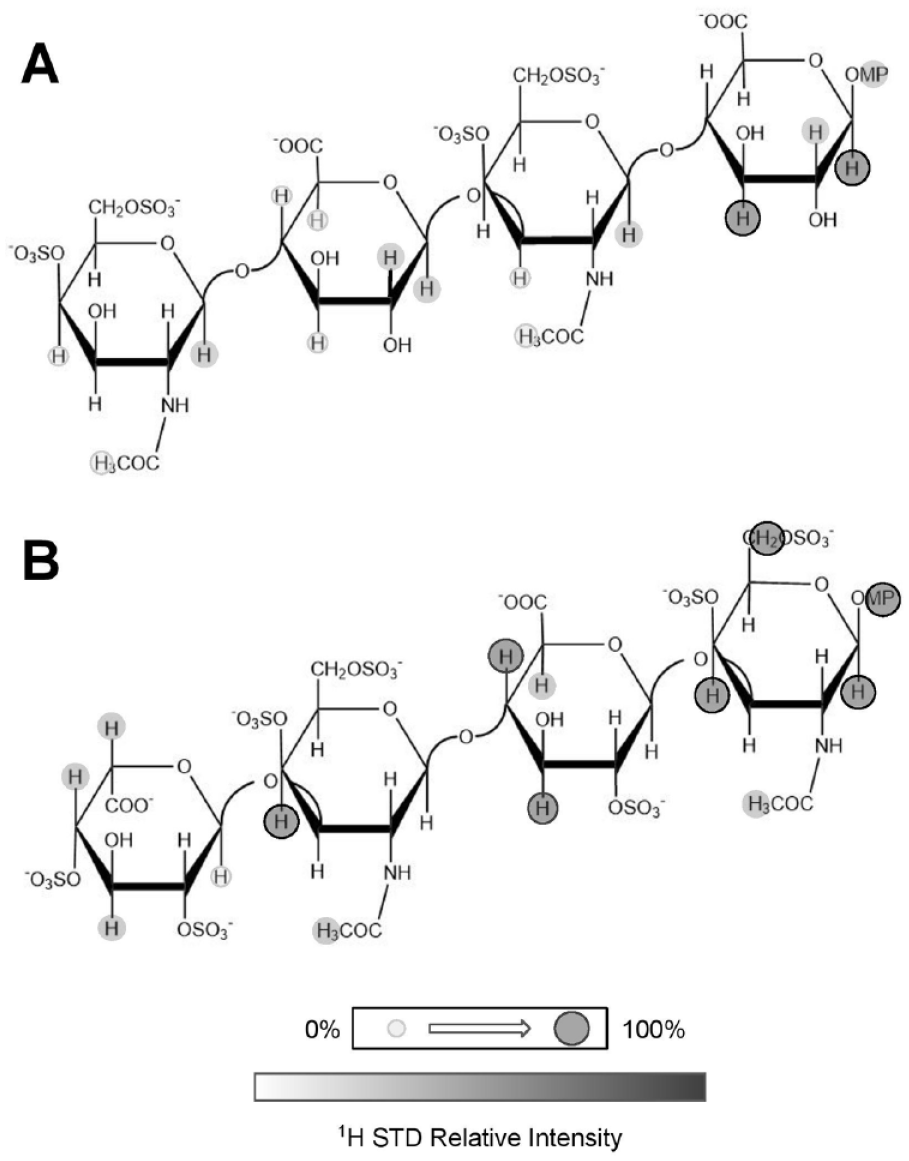
Structures of chondroitin sulfate (CS) tetrasaccharides used in the interaction with midkine. The CS tetrasaccharide at (
From Figure 7 , significant STD effects are noted on both planes of the CS tetrasaccharides. This apparent double interaction is due to the binding of both domains within MK, which displays a hinge action required for proper function. 64 Regarding the structural GAG binding mechanism of MK, two independent domains fold and interact with one GAG, as seen through NMR studies of MK in interactions with Hp 65 and through simulations of molecular dynamics. 59 As opposed to the previous results from the STD analysis of GAGs in the interactions with FGFs 16 and the CXCL12α, 55 for which the binding predominates in the middle of the GAG oligosaccharide chain, the binding of GAG oligosaccharides with MK prefers the reducing ends. 59 This comparative analysis exemplifies the specificity of interactions regarding the GBPs and GAGs.
CCL5
CCL5, colloquially known as regulated on the activation of normal T-cell expressed and secreted (RANTES), is, like most chemokines, actively involved in cell migration during the events of inflammation. CCL5 presents binding properties with GPCRs and ECM GAGs, where the interactions with GAGs are crucial for its biological activity in vivo.
66
Regulation and expression of CCL5 have become a topic of discussion recently because of its functions in multiple disease states such as liver fibrosis,
67
atherosclerosis,
68
and progression of pancreatic cancer.
69
Disruption of cellular binding that leads to these issues through GAG competition is currently under exploration as a possible route to mitigate CCL5’s proinflammatory activity. This has been observed in the work of Singh et al.,
70
in which Hp (
In the work of Deshauer et al., 71 two CS hexasaccharides extracted from natural sources and with different sulfation patterns at the C4 and C6 positions ( Fig. 2B ) were studied in interactions with CCL5. Using a fully 4-sulfated CS hexasaccharide, the STD method has proved to be a powerful tool to determine which protons from both GlcA and GalNAc units, and not only mostly from the sulfate unit, participate in the binding interface with CCL5. In addition to the standard STD experiment in which the GBP is first magnetized, the inverse of the STD experiment was performed in which the ligand itself is selectively set up to be magnetized in the experiment. Through this case of inverse STD, isotopically labeled CCL5 in molar excess concentration in solution was investigated through the 1H- 15 N HSQC spectrum (2D HSQC STD NMR). The amino acids mapped were in concordance with the NMR observations based on CSP used to diagnose the GAG binding site on CCL5 in the same work. The technology of inverse STD and CSP can be applied to any GBP in interaction studies with any GAG in which amino acid residues of the GBP have already been properly assigned.
Interleukin-10
Interleukin-10 (IL10) is a tightly regulated pleiotropic cytokine that functions in both pro- and anti-inflammatory processes. IL10 facilitates the migration of cells, where its primary function is a downstream effector in response to pathogens.
72
Multiple disease states are associated with IL10 when this cytokine is dysregulated. Among these diseases are autoimmune disorders such as inflammatory bowel disease,
73
viral pathogenesis,
74
and tumorigenesis.
75
As for all GBPs, it is important to distinguish how GAGs can play a role in the binding of IL10. Künze et al.
18
employed a suite of various GAGs in binding studies with IL10, including CS, DS, HA, and Hp (
Künze et al. 18 successfully carried out standard 1D STD and pseudo-2D STD experiments of their GAG ligands in complex with IL10, providing particularly robust conclusions about the binding interface. The 1D STD experiments indicated that most of the protons necessary for interaction are nearby sulfate groups. This conclusion is supported by the removal of sulfate groups and a consequent higher dissociation constant demonstrated by the reduction of the STD-related signal intensity when a comparative approach is made between a trisulfated CS disaccharide versus two different disulfated CS disaccharides. The CS disulfated disaccharides showed comparable signal intensity to each other but were markedly less intense than the trisulfated counterpart. The use of the HSQC experiment in this case has allowed more concise conclusions about the binding protons, especially when there is significant signal overlap in the spectrum. 18
As previously mentioned, GAGs are biologically synthesized as long-chain polysaccharides. Hence, it is crucial to understand how chain length can affect the strength of the consequent interactions with GBPs. Interestingly, it has been observed that binding affinity is directly affected by the length of the GAG chain.33,76 To explore this concept, Künze et al.
19
capitalized the usage of various Hp (
Leukocyte Common Antigen-Related
Leukocyte common antigen-related (LAR) is a receptor-like protein-tyrosine-phosphatase involved in numerous biological processes. 77 Some of these events are controlled by interactions with ECM GAGs. Binding to CS proteoglycans (CSPGs) is an example in which CSPGs function to inhibit axon growth and elongation. 78 This is in stark contrast to HS proteoglycans, which invoke an opposite response in which interactions with LAR, axon guidance, and proliferation are promoted.79,80 Because of this complex and antagonistic relationship between LAR and GAGs, it is important to determine the structural differences that mediate binding and differentiation of downstream effects.
Using the known binding epitope of the Hp pentasaccharide sequence of high affinity for AT (
Closing Remarks
STD is a very powerful NMR technique used to investigate at the atomic level the contacts of a ligand, such as GAG oligosaccharides, in interactions with proteins. Through the experimental results revisited here, the potential of STD has been proven very useful to distinguish between ligands in a mixture of GAG oligosaccharides and to understand the minimal structural features, such as sulfation patterns/contents, monosaccharide composition, chain length, and local binding preferences. All of these features are necessary and work altogether for proper complex formation with the GBPs. Among all structural biology–related studies that are recently being generated in this current glycosaminoglycanomics era, STD seems to play a crucial role in deciphering among the best binding moieties of GAG ligands required for the proper interaction with GBPs. Interestingly, the three major GBP groups mostly studied so far by STD are the coagulation factors, FGFs, and chemokines/cytokines. The STD NMR studies using these GBPs have shown that a diverse and distinguished set of GAG structures is required for adequate interactions. This observation strongly supports the fact that GAG-GBP complexes are highly specific. Understanding the structures of GAG epitopes necessary for GBP interactions is a requirement in drug discovery and development in which both biological macromolecules (GAGs and GBPs) must act cooperatively.
Footnotes
Acknowledgements
We are grateful to Christine Diedrich (SLAS Technology editorial manager) and Joe Olechno (SLAS Technology associate editor) as well as Drs. Parastoo Azadi and Christian Heiss (guest editors of the special issue “Carbohydrate Structure Analysis: Methods and Applications” in SLAS Technology) for their kind invitation to have one article of our group in their special issue.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received financial support for the research, authorship, and/or publication of this article: Financial support is received from the University of Mississippi and the American Association of Colleges of Pharmacy 2019 New Investigator Award. Copyrights and licenses were properly obtained for all figures reused or modified from the literature.
