Abstract
The fabrication of engineered vascularized tissues and organs requiring sustained, controlled perfusion has been facilitated by the development of several pump systems. Currently, researchers in the field of tissue engineering require the use of pump systems that are in general large, expensive, and generically designed. Overall, these pumps often fail to meet the unique demands of perfusing clinically useful tissue constructs. Here, we describe a pumping platform that overcomes these limitations and enables scalable perfusion of large, three-dimensional hydrogels. We demonstrate the ability to perfuse multiple separate channels inside hydrogel slabs using a preprogrammed schedule that dictates pumping speed and time. The use of this pump system to perfuse channels in large-scale engineered tissue scaffolds sustained cell viability over several weeks.
Introduction
To effectively engineer artificial tissue for research or clinical usage, culture platforms must mimic the properties of native tissue in order to achieve cellular behavior analogous to that of the in vivo phenotype. Three-dimensional (3D) tissue-engineered microenvironments have allowed researchers to more accurately model native tissue and study cellular interactions that are not typically observed in two-dimensional (2D) culture systems.1,2 For example, in 3D tissue in vivo, almost all cells are surrounded by other cells and reside in a complex extracellular environment; because 2D cell cultures are much simpler, they sometimes provide misleading and nonpredictive data for in vivo or 3D responses. 3 Platforms that enable perfusion using a scaffold that supports user-controlled flow are also appealing for 3D studies, as they afford the ability to control presentation of soluble factors in either a spatial or temporal dimension, mimicking diffusion profiles documented in large tissues and organs. 4 However, the function of these perfusion systems is dependent on a reliable pumping mechanism, without which experiments involving live cell assays on a chip, shear flow studies, and bioreactors with thick tissue cannot be maintained for extended culture periods.5–7
For long-term cell culture within 3D scaffolds, the pumping system must be able to perfuse the scaffold in a watertight chamber without interruption, run in an incubator without undergoing corrosion, and reliably deliver fluids according to user-defined programs. However, most pump perfusion systems fail to meet at least one of these criteria.8–10 Perfusion systems typically require expensive pumps, and few can be utilized in a humidified incubator.11,12 The commercially available peristaltic pumps used in most studies are generally large in size (20–30 cm) and can weigh up to 5 kg. 12 These systems are typically kept outside of the incubator to prevent corrosion due to high humidity and heat in the incubator space. At some pumping speeds, a pump sitting outside the incubator may result in the fluid being delivered at a temperature cooler than 37 °C, as well as improper gas balance in gas-permeable tubing. The ability to keep a pump inside the incubator would enable appropriate temperature and O2-to-CO2 balance in the fluid delivered to a tissue-engineered scaffold. Moreover, commercial units using a single motor lack the flexibility to pump multiple independent fluid conduits at different speeds and time intervals simultaneously in an automated, user-programmed manner.13–15 This flexibility in programming a perfusion system accommodates the changing physiological demands of different cell types within a tissue structure as it matures or ages.
In this work, we have addressed a current gap in tissue engineering platforms through the development of a customizable, inexpensive, and lightweight perfusion pump system. Using this platform, we show the ability to sustain cell viability by continuously perfusing large hydrogel scaffolds embedded with red fluorescent protein (RFP)-expressing fibroblasts, run multiple motors at various speeds, and schedule the motors to run at various speeds. With the system we have developed, we are able to increase the complexity of perfusion experiments that may be designed with a single pump unit.
Materials and Methods
Cell Culture
RFP-expressing fibroblasts were purchased from Angio-Proteomie (Boston, MA) and maintained in a humidified 5% CO2-containing atmosphere (37 °C). Culture medium consisted of DMEM supplemented with 10% v/v fetal bovine serum (FBS), 50 U/mL penicillin, and 50 µg/mL streptomycin, all procured from Life Technologies (Carlsbad, CA).
AutoCAD Design and 3D Printing
AutoCAD software (Autodesk Inc., San Rafael, CA) was used to make all 3D designs, including a housing component for the Arduino board and six EasyDriver chips, and a fluid-pump attachment (pumphead, Fig. 1A ) for each stepper motor (StepperOnline, Nanjing City, China). Detailed instructions for assembling this system and code can be found at instructables.com (https://www.instructables.com/id/Custom-Peristaltic-Pumps-x6/), and STL files and print settings can be found at thingiverse.com (https://www.thingiverse.com/thing:2832314). The components were printed using a Dimension SST 768 3D printer (Statasys, Eden Prairie, MN) with acrylonitrile butadiene styrene (ABS; Stratasys). To form the rollers on the pumphead, Teflon tubing was cut to length, placed between a vertical pair of pumphead holes, and secured with a Philips machine screw and hex nut.

(
Arduino Programming and Speed Calibration
Code for individually controlling the speed, direction, and on/off status of each of the six stepper motors was written in the Arduino program version 1.6.5. To determine a relationship between the arbitrary speeds in the Arduino code and actual flow rates, we performed a flow calibration as follows. Tygon lab tubing (1/16 in. ID × 1/8 in. OD) (Cole Palmer, Vernon Hills, IL) was cut to various lengths (98–105 mm) and placed around a pumphead on a stepper motor ( Suppl. Fig. S1 ). By varying the length of tubing placed between the bracket and pumphead, we were able to vary the tension on the tubing and thus the occlusion. This allowed us to control the volume dispensed in each pulse, and thus the flow rate for a given motor rotation rate. One side (the inlet) was placed in a 1 L beaker of water. The flow rate through the tubing at various motor speeds was measured with a Sensirion SLI-1000 flow meter (Staefa ZH, Switzerland). Each motor and tubing pair was run at 1, 10, 100, 175, 250, and 500 arbitrary units (AU; controlled by Arduino code) and the volume delivered was recorded. The relation between motor speed (AU) and flow rate was determined by a line of best fit. Ultimately, a tubing length of 102 mm was used due to the 1:1 relationship between AU and microliters per minute. After determining the relationship between motor speed and fluid flow rate, an additional experiment was performed to verify the ability to deliver a specified fluid volume. In this experiment, six motors were run at speeds of 1, 10, 100, 175, 250, and 500 AU for 10 min each, and the total water dispensed for each motor at a given speed was weighed on a microbalance and compared with the expected volume.
Polydimethylsiloxane Molds for Perfusion Chambers
Polydimethylsiloxane (PDMS; Sylgard 184, Dow Corning, Midland, MI) was mixed in a 1:10 ratio, poured into a small paraffin mold with two 18-gauge needles placed through the sides to serve as large channels, and placed in an oven at 60 °C for 2 h. PDMS blocks were then removed from the paraffin, and needles were withdrawn and replaced with 1/16 in. silicon tubing (Cole Palmer). A square was then cut out of the center of the PDMS block and discarded, leaving a four-walled PDMS mold to be filled with a hydrogel. The mold was placed on a thin slab of uncrosslinked PDMS and placed in the oven for crosslinking.
Hydrogel Device Fabrication
Gelatin powder (Sigma Aldrich, St. Louis, MO) was dissolved in complete cell media to achieve either 5% or 10% w/v concentration and sonicated at 37° C for 1 h to thoroughly mix. Microbial transglutaminase (mTG; MooGloo, Eliot, ME) was dissolved in PBS to achieve 10% w/v concentration. All fluids were sterile filtered through a 0.2 µm filter (Thermo Fisher, Waltham, MA). Trypsinized RFP fibroblasts were then added to 8 mL of gelatin and 2 mL of mTG and thoroughly mixed. The mixture of gelatin and mTG was then poured into the PDMS mold over the silicon tubing (1/16 in.) and incubated at 37 °C. After the hydrogel solidified, the silicon tubing pieces were withdrawn until the end of the tubing was located just within the edge of the hydrogel. The tubing pieces (ports) were then connected to the pump tubing, and the PDMS mold containing the hydrogel was placed into a custom-built perfusion chamber. All steps were performed using sterile technique.
Cell Viability in Hydrogels
To assess long-term cell viability in 3D gelatin hydrogels supported by the perfusion platform, cell-laden hydrogels were continuously perfused for 21 days, while control cell-laden gels (with identical channels) were not perfused. On the 21st day of culture, the hydrogels were perfused with 5% v/v Calcein-AM and Sytox Blue for 1 h (Thermo Fisher). RFP signal from fibroblasts and intracellular Calcein from all cells were imaged on a Zeiss 710 confocal microscope (Zeiss Microscopy, Munich, Germany). Cell viability was assessed using ImageJ (National Institutes of Health, Bethesda, MD). No-flow controls were performed and imaged 48 h after seeding and stained with Calcein-AM for cell viability.
Results
Customizable 3D-Printed Parts and Motor Brackets
The six-sided pumphead was able to minimize the pulsatile effect of turning the system to pump fluids and reduce backflow into the fluid lines. The tubing collars were able to slide into the laser-cut motor brackets and sustain a desired tension on the tubing in order to deliver a consistent amount of fluid on all motors.
Customization and Programming of the Perfusion System
Peristaltic pumps are used to deliver fluid by alternating compression and relaxation of a tube around a pumphead. As the pumphead rotates, a seal is created between the roller and the tubing. This seal passes fluid forward, while a vacuum is created on the back end that draws more fluid forward. The ability to control the speed at which fluid is delivered is essential when operating pumps for perfusing engineered tissue. We have demonstrated that our Arduino code can be used to control six stepper motors, each at individual speeds, and allows for schedules that turn on and off motors and adjust speeds at designated times (
Perfusion System Setup and Calibration
We sought to develop a system in which using a USB cable, users can upload code that controls the speed at which the motors turn, the amount of torque the individual motors output, the ability to increase or decrease volume rates, and the ability to turn individual motors on or off at specified times. To find the relationship between AU speed values in the code, flow rate, and volume dispensed, we connected the inlet tubing to a water source and delivered water through a flow sensor. Using the data for six motors run at motor speeds ranging from 1 to 500 AU, a linear fit was applied to derive a conversion from motor speed to average fluid flow rate ( Fig. 2 ). A tubing length of 102 mm was selected for the final design because of the 1:1 relationship between AU and flow rate (in µL/min) with this setup. Beyond a motor speed of 500 AU, the relationship between AU and fluid flow rate became nonlinear and eventually saturated. Using the linear fit for motor speeds from 1 to 500 AU, the resulting minimum and maximum flow rates using 1/16 in. ID tubing were 1 and 500 µL/min, respectively ( Table 1 ). The range of possible flow rates can be adjusted to suit the specific needs of different applications by using tubing with a larger or smaller inner diameter ( Suppl. Table S1 ). When used to perfuse channels in our hydrogels, we chose to use 1/16 in. ID tubing because the channels created in our hydrogels had a diameter of 1/16 in. After determining the relationship between motor speed and fluid flow rate, the accuracy of fluid volume delivery was verified by weighing water output after running each motor for 10 min at a given speed ( Suppl.Fig. S3 ).
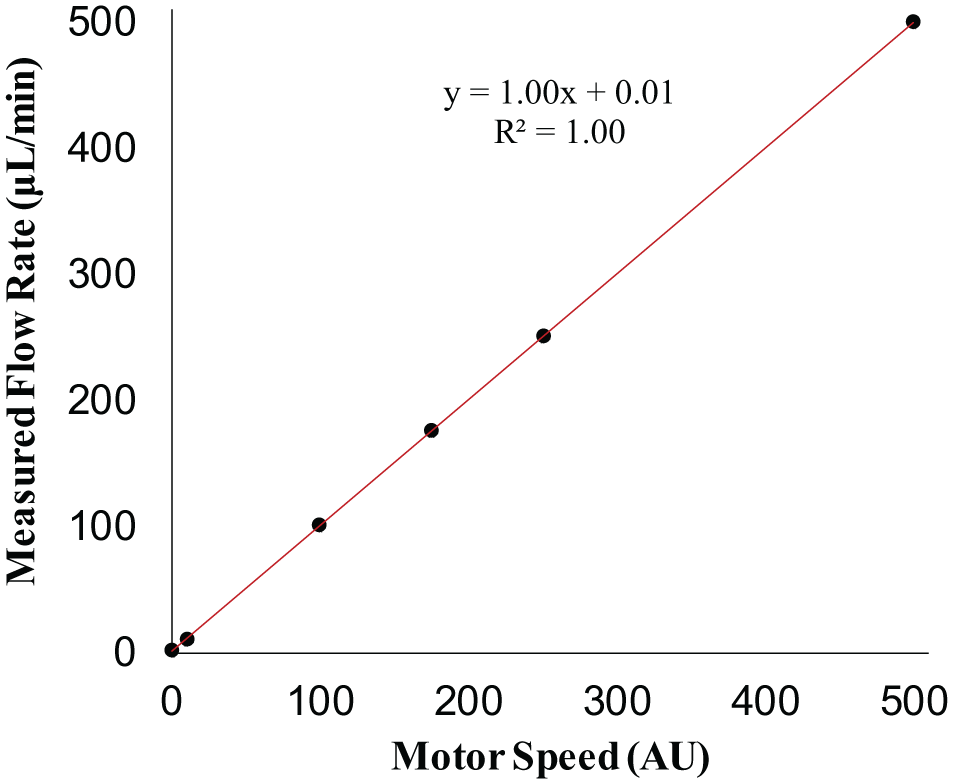
The actual fluid flow rate measured by a Senserion flow meter (µL/min) is linearly dependent on motor speed (AU) for calibrated motor–tubing pairs. Data represent mean ± standard error for six individual motors.
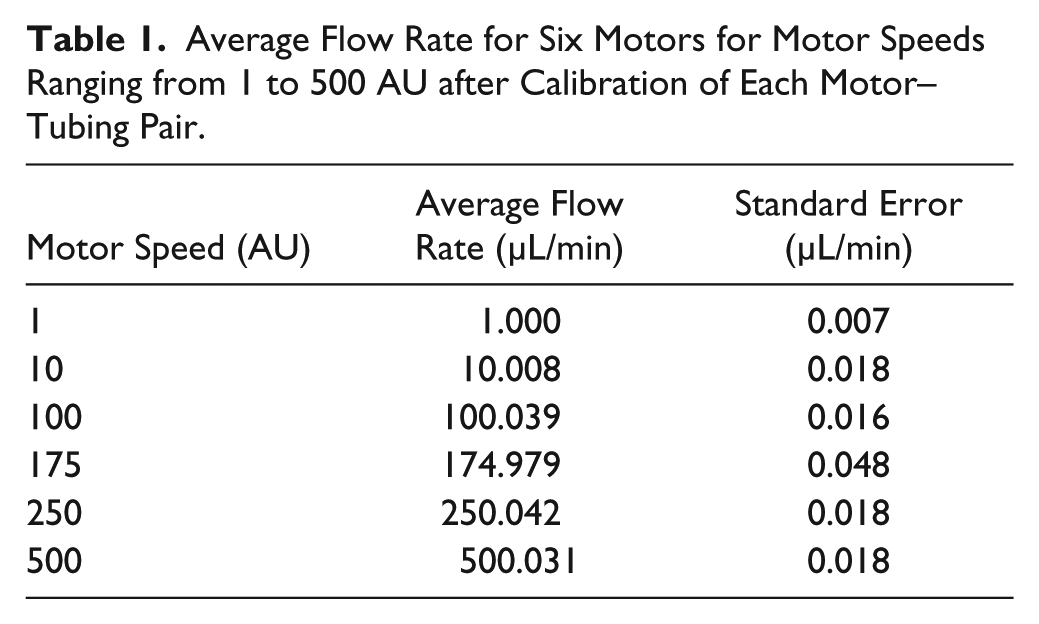
Average Flow Rate for Six Motors for Motor Speeds Ranging from 1 to 500 AU after Calibration of Each Motor–Tubing Pair.
Long-Term Cell Survival and Small-Molecule Diffusion in a Hydrogel
RFP-expressing fibroblasts were embedded in 3D hydrogel scaffolds with two parallel large channels for perfusion (schematic in Fig. 3A ). Cell-laden hydrogels were incubated in a media bath and perfused continuously by the pump system for 21 days ( Fig. 3B ). Long-term cell survival was assessed using confocal imaging of Calcein-AM delivered to RFP fibroblasts after 21 days of incubation ( Fig. 4 ). Figure 4A shows RFP-expressing fibroblasts stained with Calcein-AM located approximately 3 mm deep inside the gel. Cells survived and proliferated within and across the surface of the hydrogel over the 21-day period as shown by a z-stack projection at 3 mm ( Fig. 4B , D and Suppl. Fig. S4 ). Cells were imaged for RFP using a confocal microscope on days 1, 4, 7, and 21. RFP was used to provide a total cell count for cell proliferation over the course of a 21-day period. Calcein-AM and Sytox Blue were used to assess cell viability ([Calcein-AM count]/[Calcein-AM + Sytox Blue count]) on days 1 and 21. On day 1, 91.6% of the embedded cells were living. On day 21, 96.5% of the embedded cells were alive in the perfused hydrogel ( Fig. 4D ). Static culture of the cell-laden hydrogel resulted in nearly all cells dying ( Fig. 4C ).

(
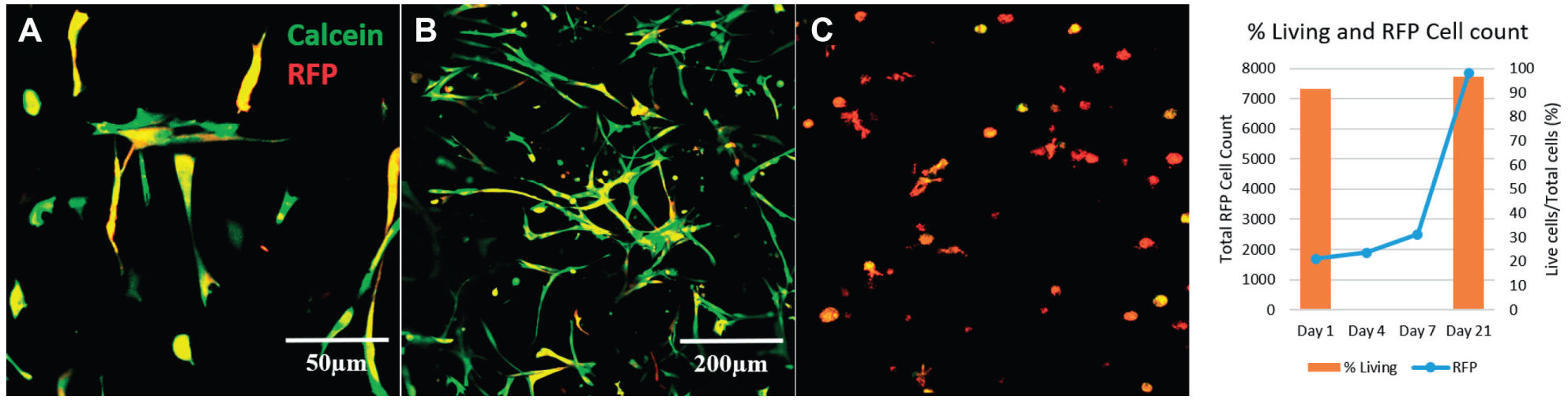
Images of gelatin hydrogel with RFP-expressing fibroblasts (red) stained with Calcein (green). Calcein staining of RFP-expressing fibroblasts shows cell viability on day 21 for cells embedded near the surface of the hydrogel (
Discussion
In the field of tissue engineering, the development of 3D scaffolds that mimic native tissue is important to furthering our understanding of the structure–function relationships in cellular interactions in basic science research, as well as to translational research in which tissues are engineered for clinical use.16–18 Although tissue engineering has progressed from 2D to 3D systems in recent years,12,19,20 several limitations remain in the toolset available to tissue engineering researchers. These limitations include the cost and size of specialized equipment (e.g., commercial peristaltic pumps and syringe pumps), compatibility of perfusion systems with incubator use, and customizability and flexibility in the temporal and spatial delivery of fluids (e.g., media and morphogens). To address these limitations in currently available microfluidic systems for tissue engineering applications, we have developed a customizable, inexpensive, and portable pump system.
Commercially available pumps, such as the Harvard Apparatus Standard Infusion/Withdraw PHD ULTRA syringe pump, Thermo Fisher FH100M Multichannel Pump System, and Ismatec IPC Low-Speed Digital Peristaltic Pump, have been used successfully in tissue engineering applications.12,21,22 However, there are drawbacks to each of these models. The PHD ULTRA syringe pump is a highly accurate system (0.25% of the desired rate of delivery) with a maximum flow rate of 216 mL/min (140 mL syringe) and minimum flow rate of 1.56 pL/min (0.5 mL syringe). This pump system can be scheduled to perfuse a specific amount and reversed to pull media through a system and back into the syringe. However, this device is 4.5 kg and large, can run two syringes at only one rate, and costs several thousand dollars. The FH100M multichannel pump system (Cole Palmer) is programmable, can support up to 12 tubes, has a lower pulsation because of the nature of the rollers, has a valveless diaphragm, and is reversible with a maximum rate of 760 mL/min and minimum rate of 2 µL/min. 22 However, it is expensive ($2000–$4000) and large, and it can run only one speed for all 12 channels. The Ismatic IPC low-speed digital peristaltic pump (Cole Palmer) is accurate (~1% of desired rate of delivery), can be scheduled, and can support up to 24 tubes. However, it weighs 4.5 kg, has large dimensions, can only run all 24 channels at one speed, and is expensive ($5000–$7000). Recently, alternatives to these large pump systems have become available, such as the MP 2 (Elemental Scientific, Omaha, NE) and nano 3 (Elemental Scientific) peristaltic pump systems. These pumps have the advantage of a more compact size, a cheaper price, and being equally as accurate as the models previously mentioned. The MP 2 allows for multiple tubing attachments to a single pumphead to run at one programmable rate, and the nano 3 pumps consist of three independent pumpheads that can be programmed to run at three independent speeds. However, six of the MP 2 systems or two of the nano 3 systems would be required to match our system in terms of the number of individually controlled channels. These commercial systems are compared with our system in Supplemental Table 2 .
In contrast, our pump system is composed of a compact, 3D-printed housing unit; stepper motors; EasyDriver chips; a power supply; and an Arduino board that weighs 0.42 kg and costs approximately $122 per unit. Additional common electrical supplies used in building our pump system (e.g., wiring, solder, and serial connectors) cost approximately $200, but these stock supplies can be used for building at least four units. While this system requires more effort to assemble than an off-the-shelf pump, the cost savings are significant and a single unit can be scheduled to deliver fluid to up to 12 separate hydrogels at six speeds (2 hydrogels per motor) or deliver fluid to multiple ports in fewer hydrogels. The Arduino board enables control over all six motors. While the Arduino and motor driver electronics are located outside of the incubator, the motors and associated pump hardware are placed in the incubator, resulting in a smaller footprint within the controlled incubator environment compared with placing a commercial system in the incubator. In our lab, a single pump unit consisting of six stepper motors placed inside the incubator routinely runs without interruption or failure for months (the time frame of the desired experiment). The code used for this system allows the user to run specific sets of motors for a specified period of time and then shut off, run motors at several speeds for a set length of time, or run motor or speed programs for a specified number of loops. This ability to control the number of motors, the time they run, the rate at which they deliver fluid, the number of cycles, and the sequence of variable cycles enables the user to set up complex perfusion experiments that run reliably for extended periods of time (on the order of months). This is a useful tool when studying the dynamics of cells that require highly coordinated delivery or the absence of specific environmental cues.
In the current work, we have described the construction of the pump system as well as the customizable programming that enables perfusion of 3D scaffolds with precise schedules and fluid flow rates ( Suppl. Video S1 ). Each stepper motor is calibrated to determine the relationship between the program input values to flow rates ( Table 1 ) and the corresponding actual volume dispensed ( Suppl. Table S3 ). Our calibration experiments showed that our system can consistently deliver fluids at a minimum rate of ~1 µL/min (±0.0067) and a maximum rate of ~500 µL/min (±0.018) ( Suppl. Table S3 ). However, the maximum and minimum rates increase and decrease with a change in the size of tubing ( Suppl. Table S1 ). This range of flow rates is useful for tissue-engineered scaffolds with various cell types that may need high rates of fluid flow, such as human umbilical vein endothelial cells (HUVECs), 23 or cell types that do not require high flow but need constant delivery of media, such as mesenchymal stem cells (MSCs). 24
We demonstrated the utility of the pump system by perfusing 3D gelatin scaffolds containing cells. In experiments with RFP-expressing fibroblasts embedded in these hydrogels, cell viability within the hydrogel was maintained for 21 days as evidenced by live cell staining (Calcein) and spreading of fibroblasts ( Fig. 4 ). The perfusion of large hydrogels and Calcein experiments demonstrate that our perfusion system is an effective means of sustaining cell viability for extended periods of time.
Overall, these results demonstrate that our pump system has a high level of customization compared with most systems on the market and do-it-yourself perfusion systems. It can be used for automated and accurate control of fluid delivery to maintain cell viability through continuous perfusion.
Supplemental Material
Supplemental_Material_for_Perfusion_System_by_O_Grady_et_al – Supplemental material for A Customizable, Low-Cost Perfusion System for Sustaining Tissue Constructs
Supplemental material, Supplemental_Material_for_Perfusion_System_by_O_Grady_et_al for A Customizable, Low-Cost Perfusion System for Sustaining Tissue Constructs by Brian J. O’Grady, Jason X. Wang, Shannon L. Faley, Daniel A. Balikov, Ethan S. Lippmann and Leon M. Bellan in SLAS Technology
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was funded and supported by NSF BMAT 1506717 (L.M.B.) and a NARSAD Young Investigator Award from the Brain and Behavior Research Foundation (E.L.).
Supplemental material is available online with this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
