Abstract
Natural triterpenes represent a group of pharmacologically active and structurally diverse organic compounds. The focus on these phytochemicals has been enormous in the past few years, worldwide. Asiatic acid (AA), a naturally occurring pentacyclic triterpenoid, is found mainly in the traditional medicinal herb Centella asiatica. Triterpenoid saponins, which are the primary constituents of C. asiatica, are commonly believed to be responsible for their extensive therapeutic actions. Published research work has described the molecular mechanisms underlying the various biological activities of AA and its derivatives, which vary for each chronic disease. However, a compilation of the various pharmacological properties of AA has not yet been done. Herein, we describe in detail the pharmacological properties of AA and its derivatives that inhibit multiple pathways of intracellular signaling molecules and transcription factors that are involved in the various stages of chronic diseases. Furthermore, the pharmacological activities of AA were compared with two natural compounds: curcumin and resveratrol. This review summarizes the research on AA and its derivatives and helps to provide future directions in the area of drug development.
Introduction
Centella asiatica, also known as Gotu kola, is an herbaceous, frost-tender perennial plant in the flowering plant family Apiaceae. Native to China, India, and other tropical countries, it has been used for centuries as a medicinal herb.1–3 In China, it is considered a miracle elixir of life. 4 C. asiatica is believed to be a medicine used by common folks, and its use has been described in ancient Chinese and Indian calligraphies. 5 C. asiatica accumulates large quantities of pentacyclic triterpenoid saponins, inclusively known as centelloids. These terpenoids include Asiatic acid (AA), asiaticoside, brahmic, brahmoside, brahminoside, centelloside, madecassoside, sceffoleoside, and thankuniside.6,7
Over the past few years, there has been a growing interest in natural triterpenoids, specifically focusing on the scientific aspects of extraction, isolation, structural analysis, and a wide spectrum of biological activities. AA is the most prominent constituent of the Centella saponins and is a naturally occurring pentacyclic triterpenoid. 4 This compound possesses a wide spectrum of biological activities, notably anticancer,8–17 anti-inflammatory and wound healing,18–21 antidiabetic,22–24 antioxidant and hepatoprotective,25–28 anti–hepatitis C virus (HCV), 29 and neuroprotective,30,31 among others. Chemically, AA (2,3,23-trihydroxy-urs-12-ene-28-oic-acid) is an aglycone form of asiaticoside and is easily formed by hydrolyzing the sugar moiety of the asiaticoside structure in acidic conditions. Its chemical formula is C30H48O5 and it has a molecular weight of 488.70 kD. The molecular structure of AA is given in Figure 1.
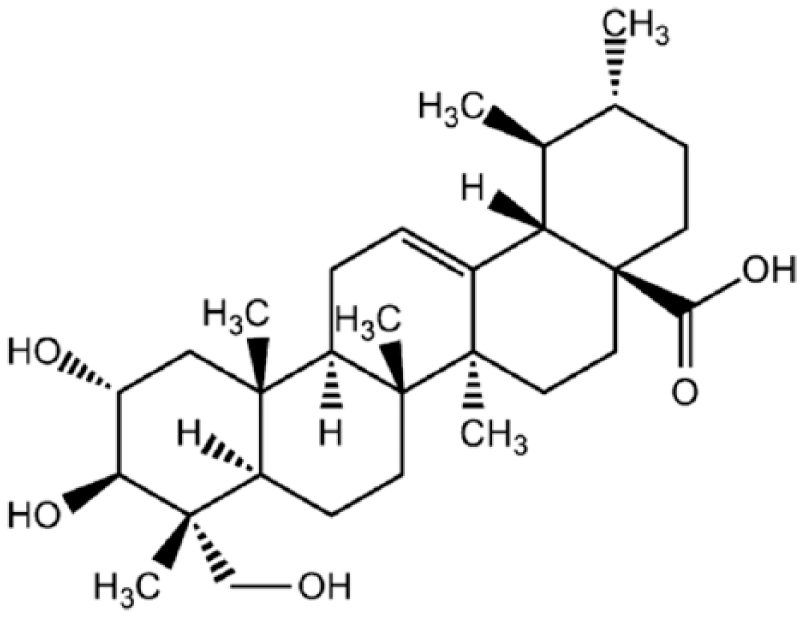
Structural formula of AA (C30H48O5), an aglycone form of asiaticoside. It is chemically known as 2,3,23-trihydroxy-urs-12-ene-28-oic-acid. AA is the most prominent of the Centella saponins and is one of the naturally occurring pentacyclic triterpenoids.
Potential Role of AA in the Prevention and Treatment of Various
AA, a naturally pentacyclic triterpenoid, seems to be promising due to its various pharmacological properties. Interestingly, some derivatives of AA were found to have strong activity in treating some diseases.32,33 Considering this perception, the present review highlights the established studies related to the pharmacological potential of AA and its derivatives against different diseases. The molecular mechanisms underlying the targets of AA and its derivatives that are liable for its therapeutic effect are also discussed.
Targeted Inhibition of Tumor Proliferation, Survival, and Metastasis by AA: Activity of AA in Cancer
AA has attracted great interest, and its anticancer effects have been studied in vitro and in vivo by many researchers in different types of cancer.8–17 The interactions of AA with molecular targets like nuclear factor erythroid-derived 2-like 2 (Nrf2), nuclear factor kappa B (NF-κB), protein kinase C (PKC), free radicals scavenging, and cell longevity pathway (CLP) that lead to anticancer, cytoprotective, chemosensitizer, and chemopreventive effects must be emphasized, as AA acts by inhibiting the proliferation of cancer cells and induces apoptosis. Many studies reported that AA could induce apoptotic cell death by modulating the protein expression of several apoptosis regulators, such as caspases, B-cell CLL/lymphoma 2 (BCL-2) family members, and glioblastoma multiforme (GBM) cells. BCL-2 and BCL-XL proteins were decreased by AA treatment. 9 Furthermore, AA induces endoplasmic reticulum stress (by increasing GRP78 and calpain, and decreasing calnexin and IRE1α expression), enhances free intracellular calcium, and damages cellular organization in GBM cells. 9 Disruption of the endoplasmic reticulum and variations in calcium homeostasis are early events in AA-induced death. AA inhibits the expression of NDR1/2 kinase and promotes the stability of the p21WAF1/CIP1 protein through attenuating the NDR1/2-dependent phosphorylation of p21WAF1/CIP1 in Hep G2 cells. 34 Lee et al. reported that AA induces apoptosis through increased intracellular Ca2+, which in turn enhances p53 expression in HepG2 cells. 35 Cho et al. also found that AA induces cell death by both apoptosis and necrosis, with Ca2+-mediated necrotic cell death predominating. 36 AA was found to significantly decrease interleukin-8 (IL-8) production in human colon cancer cells. 37 Park et al. also observed that AA-induced apoptosis may be mediated through generation of reactive oxygen species (ROS), alteration of the Bax/BCL-2 ratio, and activation of caspase-3, but it is p53 independent. 38 Tang et al. illustrated that the mitochondrial death apoptosis cascade plays a very important role in AA-induced cancer apoptosis. 39 Through the increase in mitochondrial membrane permeability and release of cytochrome C from mitochondria into cytosol, AA induces caspase-9 activity, which further stimulates caspase-3 and poly(ADP-ribose) polymerase cleavage, resulting in irreversible apoptotic death in tumor cells. Taken together, these works suggest that the mitochondrial death apoptosis cascade plays a key role in AA-induced cancer apoptosis.
AA significantly upregulates miR-1290, which sensitizes cells to AA-induced cytotoxicity and negatively regulates BCL-2 expression. 40 AA is able to regulate cell cycle progression in RPMI 8226 cells with the inhibition of signal transduction mediated by focal adhesion kinase (FAK). 41 AA may exert antitumorigenesis through inhibitory actions in NO and cyclooxygenase-2 (COX-2) signals. 42 Kavitha et al. also reported the strong antiangiogenic potential of AA and suggested its usefulness against malignant gliomas. 9
Activity against Breast Cancer
Hsu et al. conducted a study on the two human breast cancer cell lines: MCF-7 and MDA-MB-231. 43 They demonstrated that AA exhibited effective cell growth inhibition by inducing cancer cells to undergo S-G2/M phase arrest and apoptosis, due to increased p21/WAF1 levels. The study also illustrated that AA attenuated the Cdc2 function by accelerating the association of the p21/WAF1/Cdc2 complex and the level of inactivated phospho-Cdc2 and phospho-Cdc25C. The authors described in their study how exposure to AA treatment triggered the mitochondrial apoptotic pathway specified by changing the Bax/BCL-2 ratios, cytochrome C release, and caspase-9 activation, but it did not act on Fas/Fas ligand pathways and the activation of caspase-8. Simultaneously, in the same study it was found that mitogen-activated protein kinase (MAPK), extracellular signal-regulated kinase (ERK1/2), and p38, but not c-Jun NH2-terminal kinase (JNK), were critical mediators in AA-induced cell growth inhibition.
Kim et al. demonstrated that AA induced cell death in MCF-7 human breast cancer and AGS human gastric cancer cells, and upregulated expression of miR-1290 by 2.58- and 7.01-fold, respectively. 40 It was also found that miR-1290 promotes AA-induced apoptosis by decreasing the BCL-2 protein level in A549 non-small-cell lung carcinoma (NSCLC) cells. 40
Activity against Ovarian Cancer
Ren et al. investigated the effects of AA on the proliferation, apoptosis, and cell cycle progression of human ovarian cancer cells. 44 Hisamatsu et al. demonstrated that the PI3 kinase (PI3K)/Akt/mammalian target of the rapamycin (mTOR) pathway is commonly activated in ovarian cancer and has been suggested as a promising therapeutic target for this disease. 45 The effects of AA on the PI3K/Akt/mTOR pathway were also examined in ovarian cancer cells by the same authors.
Activity against Lung Cancer
MicroRNAs (miRNAs) are small RNAs, 19–25 nucleotides in length, that play an important role in growth, differentiation, and cell death. Various oncogenic miRNAs (oncomiRs) and tumor suppressor miRNAs have been recognized. 46 For example, miR-34a inhibits oncogenic cellular transformation by suppressing oncogenes, such as proto-oncogene c-Met, cyclin-dependent kinase 4 (CDK4), and BCL-2.47,48 The miR-21, which is a well-studied oncomiR, also targets many key tumor suppressor genes, such as phosphatase and tensin homolog (PTEN), programmed cell death 4 (PDCD4), and TP63. 49 Recently, miR-21 was implicated in the phenomenon of cancer cell addiction to key oncogenes.50–52 These findings strongly indicate that an examination of a miRNA-based cellular mechanism is important for the understanding of tumorigenesis and anticancer processes, and certain miRNAs may be critical targets for anticancer therapy. It is reported that treatments of human cells with C. asiatica extract altered the miRNA expression profiles; 53 this indicated that the effects of the extract on cells might involve miRNA-specific mechanisms. Kim et al. showed that AA modifies the expression profiles of specific miRNAs in A549 NSCLC cells using miRNA microarray. 40 They reported that miR-1290 is the most broadly upregulated miRNA, and that AA-induced anticancer activity is dependent on the expression of this miRNA, which promotes AA-induced apoptosis by decreasing the BCL-2 protein level in A549 cells. 40 In another study conducted by Wu et al., they demonstrated that AA significantly reduces lung cancer cell growth both in vitro and in vivo, that the associated apoptosis is mediated through mitochondrial damage. 54
Activity against Skin Cancer
AA can act as a natural chemoprotective agent against UVB-mediated injury in normal human dermal fibroblasts (NHDFs) through alteration in the expression of specific miRNAs. 53 AA induces apoptosis in SK-MEL-2 human melanoma cells, and it also induces apoptosis through ROS generation, alteration of the Bax/BCL-2 ratio, and caspase-3 activation in SK-MEL-2 human melanoma cells.
Park et al. suggested that role of AA may be a good candidature for the treatment for skin cancer. 38 They also reported that AA induces apoptosis in melanoma cells through increasing the levels of ROS. AA also showed a protective effect against OGD/R-injured PC12 cells, and the possible mechanisms were related to eliminating free radicals and inhibiting cell apoptosis. 55
Activity of AA in Liver Diseases
A number of studies have shown that AA can protect the liver from injury via mechanisms underlying antimitochondrial stress and the cellular antioxidant system. 56 Researchers have also reported that AA is capable of inhibiting collagen matrix production by hepatic stellate cells (HSCs) and keloid fibroblasts by blocking the autocrine effects of transforming growth factor β1 (TGF-β1) in vitro. Treatments with AA were able to induce hepatic Smad7, thereby blocking TGF-β/Smad signaling and fibrosis in a rat model of CCl4-induced liver fibrosis and in TGF-β1-activated HSCs. The in vitro study concluded that AA may be a novel therapeutic agent for liver fibrosis. Induction of hepatic Smad7 thereby reduces activation of TGF-β/Smad signaling, and may be a principal mechanism by which AA protects against chronic liver disease associated with fibrosis. 56 Thus, AA inhibits liver fibrosis by blocking the TGF-β/Smad pathway.
Wei et al. observed that intragastrical administration of AA protected the liver against ethanol-induced hepatotoxicity through the attenuation of oxidative stress. 57 Pakdeechote et al. indicated that 3 weeks of AA intake at 10 or 20 mg/kg/day improved metabolic abnormalities induced by a high-carbohydrate and high-fat diet, such as glucose intolerance, hypertension, and circulating oxidative and inflammatory stress in rats. 58 Lin et al. found that a high-fat diet decreased hepatic glutathione (GSH) content, lowered catalase (CAT) and glutathione peroxidase (GPx) activities, and increased cytokine release. 59 They further investigated the effects of AA at a dose of 10 or 20 mg/kg/day on lipid deposit and the protein expression of lipogenic enzymes and SREBPs in livers from mice treated with a high-fat diet. Their findings supported that AA could protect the liver via antilipogenic, antioxidative, and anti-inflammatory activities to improve diseases caused by high-fat diets. Duggina et al. reported that triterpene saponins may provide a protective and/or therapeutic alternative against immune-mediated liver diseases. 60
Activity of AA in Apoptosis
Apoptosis, or programmed cell death, is explained by numerous morphological and biochemical changes, including reduced cell volume, condensation of nuclear chromatin, DNA fragmentation, and an increased proportion of sub-G1-phase cells. 61 Initiation of apoptotic processes in cells induces activation of proapoptotic proteins, as well as inactivation of antiapoptotic proteins. 62 BCL-2 family proteins are involved in the regulation of apoptosis as either death antagonists or death agonists. 63 BCL-2 is a serious regulator of cell growth and antiapoptotic processes. Recently, AA was reported to induce apoptosis in several types of cancer cells, and to affect the level of expression of BCL-2 and BCL-XL. 63 However, the mechanisms underlying the AA-induced downregulation of BCL-2 remains largely unclear.
Activity of AA in Wound Healing
AA has been used for wound healing and research has been increasingly supportive of such claims.64–67 Wu et al. identified that AA is the major active ingredient responsible for the burn wound healing therapeutic effect. 64 Notably, AA is reported as the major worthy ingredient of a wound healing drug (commercial name Madecassol), and may also be able to accelerate nerve regeneration. 68 Studies show that both asiaticoside and madecassoside may help burn wound healing, possibly through the significant improvement of collagen I synthesis. 69 Somboonwong et al. demonstrated the wound healing effects of C. asiatica extracts for both incision and burn wounds. 67 Further, these extracts were analyzed by thin-layer chromatography and it was shown that the phytoconstituents, mainly β-sitosterol, AA, asiaticoside, and madecassocide, were respectively present in different solvent extracts. Somboonwong et al. claimed that AA in ethyl acetate extract is the most active ingredient for wound healing. 67
Bian et al. 70 reported that AA is responsible for keloid management, as it inhibited TGF-1-induced collagen and PAI-1 expression in keloid fibroblasts via peroxisome proliferator-activated receptor activation, and could be effective in the arsenal for combating keloid. AA can induce a steady gene expression response with its prevailing pharmacological effects in connective tissue disorders, such as wound healing and microangiopathy. AA, via its antiglycative activity, could protect against skin damage.71,72
Activity of AA in Inflammation
Inflammation is the response of body tissues to harmful stimuli, such as pathogens, damaged cells, or irritants. It is a protective response involving immune cells, blood vessels, and molecular mediators. 73 Inflammation, which is involved in injury and host defenses against infectious agents, is also responsible for the pathophysiology of many chronic diseases. A coordinated series of common effects or mechanisms of inflammation contribute to oxidative stress, tissue injury, angiogenesis, remodeling of the extracellular matrix, and fibrosis in various target tissues. Its progression involves many inflammatory mediators, controlled by cells of innate and adaptive immunity. In this field, there is a requirement for novel strategies to predict disease susceptibility, target and monitor therapies, and ultimately develop new approaches for the treatment and prevention of chronic diseases. 74
Various studies have been conducted to confirm AA’s effectiveness in inflammations. Huang et al. reported that AA possesses analgesic and anti-inflammatory effects. 75 They observed that the anti-inflammatory mechanism of AA may be associated with inducible nitric oxide synthase (iNOS) and connected with the increase in the activities of antioxidant enzymes (CAT, superoxide dismutase [SOD], and GPx). Yun et al. claimed that the anti-inflammatory properties of AA might be the result of the inhibition of iNOS, COX-2, IL-6, IL-1β, and tumor necrosis factor α (TNF-α) expressions through the downregulation of NF-κB activation via suppression of the IκB kinase (IKK) and MAPK (p38, ERK1/2, and JNK) phosphorylation in RAW 264.7 cells. 76 This study documented that ROS could activate signaling pathways, including MAPK and/or NF-κB in airway epithelium, which in turn elicits the production of oxidative and/or inflammatory-associated molecules, and facilitates the progression of respiratory diseases. 77 Thus, an agent with the capability to reduce ROS and prostaglandin E2 (PGE2) generations or suppress NADPH oxidase, COX-2, NF-κB, and MAPK expression may potentially protect airway epithelial cells to attenuate oxidative and inflammatory injury. Tsao and Yin demonstrated that AA, glycyrrhizic acid, and oleanolic acid protect human bronchial cells against oxidative and inflammatory injury via enhancing mitochondrial stability, decreasing ROS and PGE2 production, and suppressing protein expression of NAPDH oxidase, COX-2, NF-κB p65, and p-p38. 78 Pakdeechote et al. found that AA improves metabolic and hemodynamic abnormalities in rats with metabolic syndrome (MS) induced by a high-carbohydrate and high-fat diet by decreasing oxidative stress and inflammation, 58 whereas Chen et al. 79 investigated the anti-inflammatory effects of AA both in vivo and in vitro. Hongming et al. 80 reported that AA exhibited significant roles against lipopolysaccharide (LPS)/d-GalN-induced fulminant hepatic failure by inhibiting oxidative stress and inflammation. The proposed mechanism may be associated with the inhibition of MAPK and NF-κB activation. These studies show that AA induces anti-inflammatory actions via activation of antioxidant enzymes, and by suppression of IKK and MAPK (p38, ERK1/2, and JNK, and upregulation of Nrf2).
Activity of AA in Diabetes
Diabetes mellitus is a group of metabolic disorders in which blood sugar levels are affected; over a prolonged period, diabetes can cause many complications. 81 AA and its derivatives are promising agents in the prevention of diabetic complications. As AA has strong antioxidant activity, which can inhibit or slow the formation of advanced glycation end products, it is implicated in the pathogenesis of diabetic nephropathy, embryopathy, and neuropathy.22–24 Until now, very few clinical studies have been concerned with the application of AA in treating diabetes. However, due to the great therapeutic potential, this compound deserves special attention in this specific field. AA has been reported to lower blood glucose levels in type 1 diabetic rats at a dose of 25 mg/kg. 82 It preserves β-cell mass and mitigates hyperglycemia in streptozotocin (STZ)-induced diabetic rats. Thus, it was also investigated for its effect on the lipid profile and lipid-metabolizing enzymes in STZ-induced diabetic rats. 82 Ramachandran and Saravanan reported that AA improves the level of plasma insulin, thereby decreasing the glucose level and reversing the changes in the levels of the key carbohydrate metabolizing enzymes, and prevents lipid peroxidation while improving the antioxidant status in rats with STZ-induced diabetes. It also exerts an antihyperglycemic effect by enhancing glucose uptake in the skeletal muscles in insulin-deficient STZ diabetic rats via the PI3K-Akt signaling pathway. 83 The authors also illustrated the possible mechanism of action that AA may directly contribute to impairment in the PI3K/Akt signal pathway. Activation of PI3K and the serine-threonine PKB Akt are important steps in insulin action. Akt has been reported to be an arbitrator of the glucose uptake signaling pathway, which is controlled by insulin in both muscle and fat cells and hepatic gluconeogenesis. The PI3K/Akt pathway is activated downstream of the insulin receptor. 83 Hence, these findings provide a basis for the use of AA in the prevention of diabetes and related complications.
Activity of AA in Cardiovascular Diseases
Cardiovascular disease includes various coronary artery diseases, such as stroke, heart failure, hypertensive heart disease, angina and myocardial infarction, rheumatic heart disease, heart arrhythmia peripheral artery disease, and venous thrombosis. 84 Accumulating evidence indicates that AA has various cardiovascular activities. A wide range of potentially therapeutic cardiovascular effects of AA has been published. It is well established that L-NAME-induced hypertension produces not only cardiovascular alterations, but also inflammation and an increase in oxidative stress. Chronic inhibition of NOS with L-NAME induces pro-inflammatory phenotypic changes of the vascular wall, including an increase in the expression of intercellular adhesion molecule-1 (ICAM-1), vascular cell adhesion molecule-1 (VCAM-1), and inducible nitric oxide synthase (iNOS). 85 Bunbupha et al. reported that AA reduces blood pressure and oxidative stress markers of recovering eNOS and NADPH oxidase subunit p47phox expression, and thus improves NO bioavailability in L-NAME-induced hypertensive rats. 86 The authors also described that AA was able to reduce blood pressure and alleviate left ventricular (LV) hypertrophy, myocardial fibrosis, aortic collagen deposition, and thickening of the aortic wall in L-NAME-induced hypertensive rats. 86 A study confirmed that AA reduced blood pressure by decreasing renin-angiotensin over activity and sympathetic nerve over activity, and improving vascular function in rats with MS induced by a high-carbohydrate and high-fat diet. 87
AA plays an important role in the inhibition and treatment of cardiac hypertrophy.88,89 Cardiac hypertrophy is sometimes characterized by an increase in the size of the cardiac myocyte and protein synthesis, abnormal expression of fetal genes, and unwanted deposition of extracellular matrix. It can result in heart failure and ventricular arrhythmias. Ma et al. demonstrated an experimental verification for the function of AA in the treatment of cardiac hypertrophy and heart failure. 88 They demonstrated that IL-1β is a potent modulator of cardiac hypertrophy, both in vitro and in vivo, through activating NF-κB signaling. Xu et al. reported that AA inhibited cardiac hypertrophy in an IL-1β stimulation experiment in vitro, and it inhibited transverse aortic constriction (TAC)–induced cardiac hypertrophy in vivo. 89 Some studies reported that AA exerts beneficial effects on the heart; that is, it is able to inhibit pressure overload–induced cardiac hypertrophy by blocking p38 MAPK and ERK1/2 phosphorylation and by reducing the excessive production of TGF-β1 and nuclear factor NF-κB. 90 Interestingly, Huang et al. observed that AA attenuates myocardial ischemia/reperfusion injury via Akt/GSK-3/HIF-1 signaling in rat H9c2 cardiomyocytes. 91 In addition, AA protects cardiomyocytes against high-glucose-induced injury by attenuating oxidative stress. 92
Activity of AA in Neuroprotective Effects
Neuroprotection refers to the preservation of brain function and structure. Neurodegeneration can occur when the brain is exposed to high levels of oxidative stress, inflammation, mitochondrial dysfunction, protein deficiencies, or other various forms of neurotoxicity. To prevent or mitigate the effects of any neurodegeneration, tactical administration of a neuroprotective agent may be beneficial.93,94 AA possesses neuroprotection by maintaining the stability of the blood-brain barrier and by protecting the mitochondrial functions. 95 Lipid peroxidation and the inflammatory process take part in neuronal damage after spinal cord injury (SCI). AA administration immediately after the SCI reduces the level of lipid peroxidation products and some pro-inflammatory cytokines and improves functional motor recovery. 96 The administration of AA has clearly demonstrated an improvement in learning and memory in animal models, an effect that was correlated with the amplification in hippocampal neurogenesis. 97 Furthermore, AA has also shown the capability to improve hippocampal CA3 neuronal dendritic aborization in rats. Increasing the dendritic length of the hippocampal CA3 neurons may result in alterations in synaptic connectivity, 98 which may be a factor that improves learning and memory. AA might be valuable in preventing memory deficit in patients taking valproic acid. In the same study, Gadahad et al. also observed that the administration of AA is useful in enhancing cell proliferation in the subgranular zone (SGZ) of the hippocampus and spatial functioning memory. 98 Hence, AA might be helpful in increasing learning and memory in situations of cognitive decline.
AA can be regarded as a reasonable therapeutic candidate for Alzheimer’s disease, that is, a drug that protects neurons from β-amyloid toxicity. AA was used to describe the inhibitory effect on acetylcholinesterase (AChE) properties, excitatory postsynaptic potential (EPSP), and locomotor activity. 99 AA may benefit cognitive function with less effect on blood pressure. 100 Kim et al. found that AA derivatives might have therapeutic value in alleviating certain memory impairments observed in dementia. 101
Antimicrobial Activity of AA
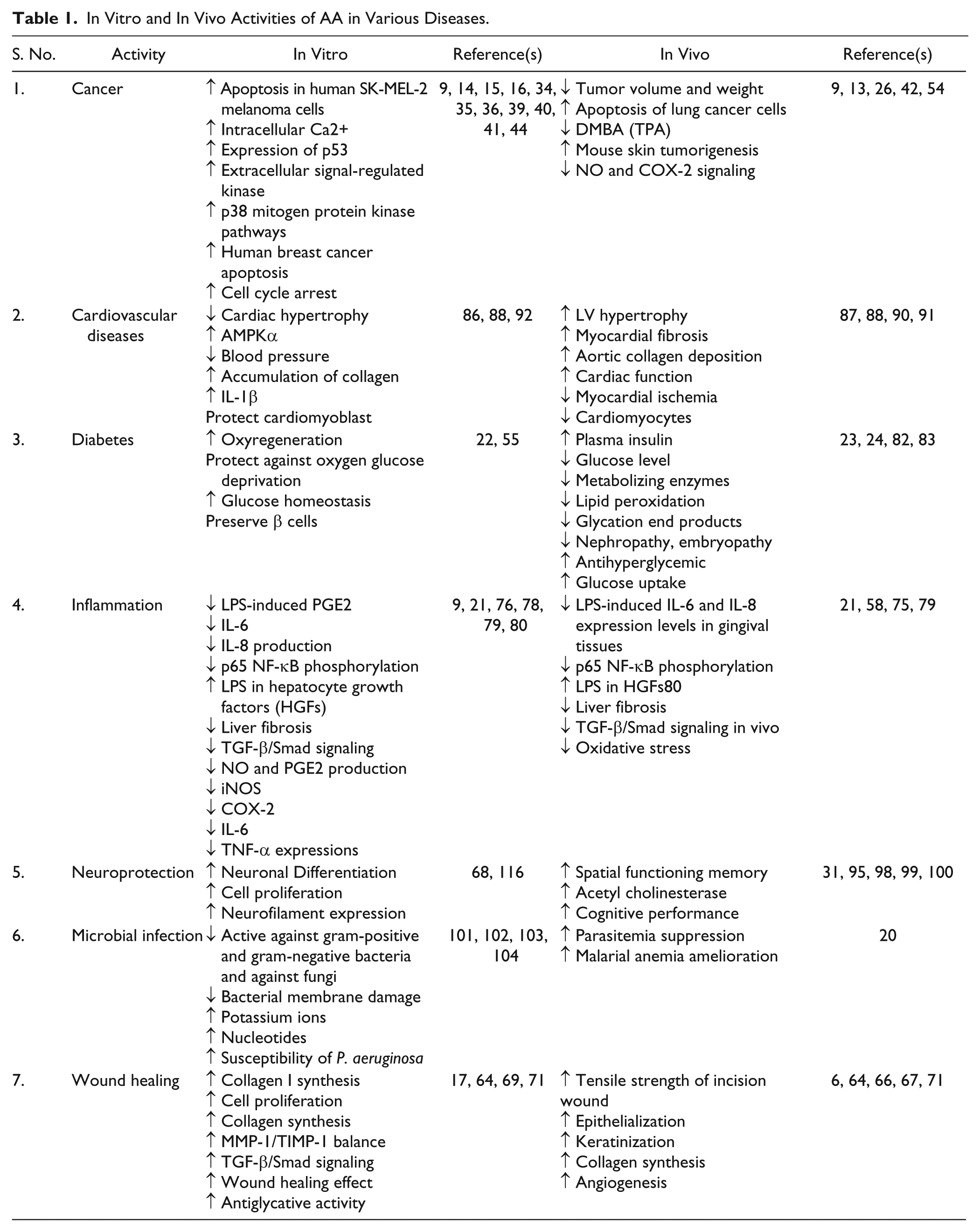
Inhibitory effects against Staphylococcus aureus, Bacillus cereus, Escherichia coli, Bacillus subtilis, and Shigella sonnei have been found with AA, as studied by agar diffusion methods. 102 Liu et al. 102 also reported that AA causes bacterial membrane damage and/or enhances the release of potassium ions and nucleotides. AA dose dependently inhibited the growth of E. coli O157:H7, Salmonella typhimurium DT104, Pseudomonas aeruginosa, Listeria monocytogenes, S. aureus, Enterococcus faecalis, and B. cereus in medium and in ground beef. It was also found that AA is a potent agent that prevents the contamination of foods by the aforementioned bacteria. 102 Garo et al. indicated that AA could enhance the susceptibility of P. aeruginosa biofilms to tobramycin. 103 The study of Masoko et al. revealed that AA benefited wound healing via its antifungal activity. 104 Ashella and Flemming 105 studied the potential effect of AA against bacterial and fungal strains. AA was found to be a therapeutically active agent against microbes and can be used for further preclinical investigations. Recently, Mavond et al. reported that AA influences parasitemia suppression, ameliorates malarial anemia, and significantly increases the biophysical properties of infected animals. 20 Table 1 describes the in vivo and in vitro effective mechanisms of AA in various disorders.
In Vitro and In Vivo Activities of AA in Various Diseases.
Pharmacological Activity of AA Derivatives
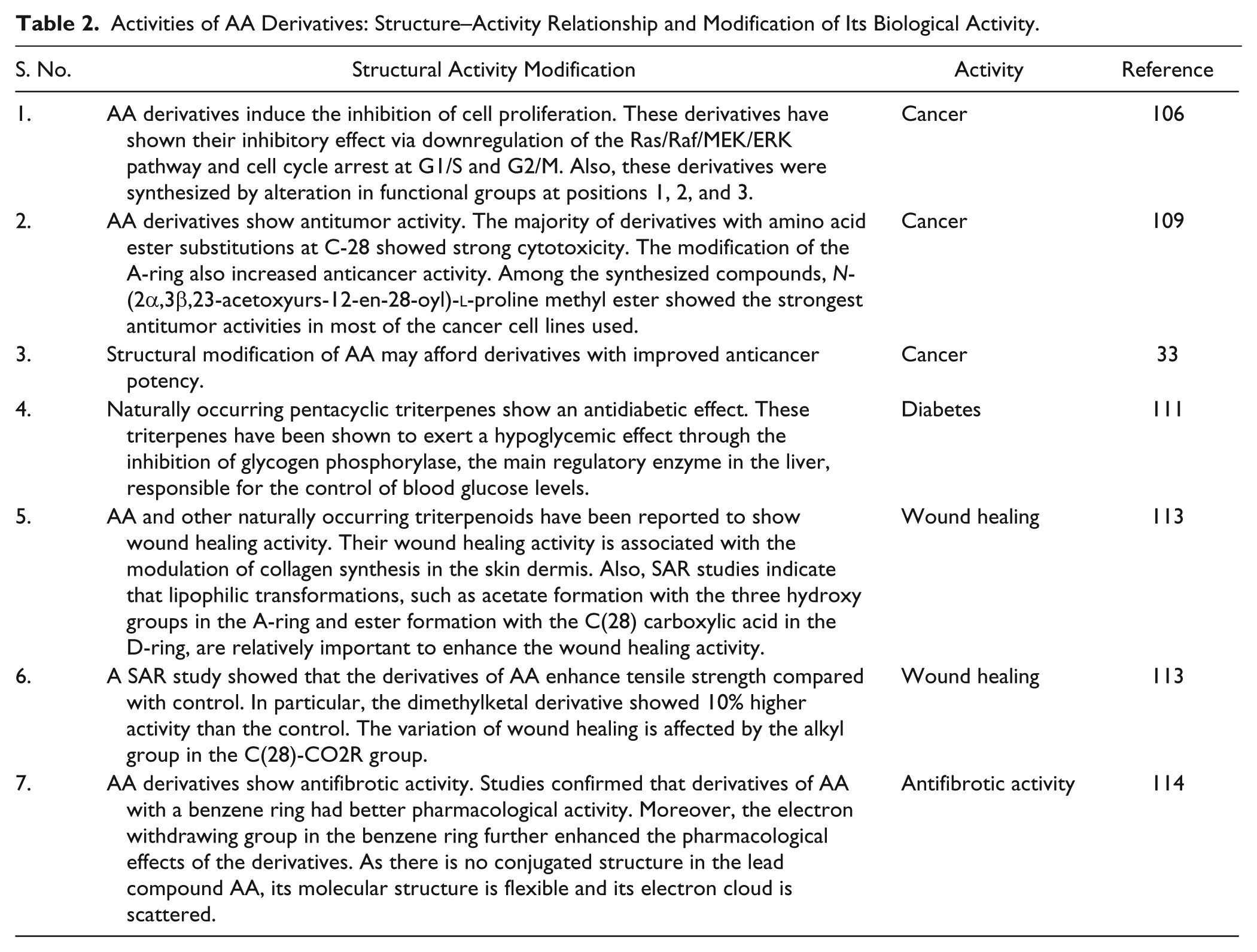
Although AA experimental work seems very promising, chemical modifications of the AA backbone would also have a high impact on its biological activity (Table 2). In recent years, various derivatives of AA were designed and synthesized based on chemical modifications of the AA backbone. This would have a high impact on its biological activity. Some AA derivatives have shown stronger cell growth inhibitory activity than AA. Wang et al. found that some AA derivatives induced inhibition of cell proliferation via downregulation of the Ras/Raf/MEK/ERK pathway and cell cycle arrest at G1/S and G2/M. 106 Meng et al. synthesized novel AA derivatives that showed stronger cell growth inhibitory activity than AA itself. 107 In another study, two AA derivatives, modified at the C-11 and C-28 positions, showed stronger antiproliferative activity than AA on the HepG2 cell line. The cell cycle analysis showed that the effective AA derivatives mainly arrested HepG2 cells in the G1 stage. 108
Activities of AA Derivatives: Structure–Activity Relationship and Modification of Its Biological Activity.
Jing et al. reported the synthesis of AA derivatives with modifications of the functional groups at C-2, C-3, C-23, and C-28. These derivatives were evaluated in vitro using seven cancer cell lines, and their antiangiogenic activities were evaluated using an in vivo larval zebrafish model. Their results illustrated that the majority of AA derivatives with amino acid ester substitutions at C-28, such as compounds 4–10, had stronger cytotoxicity than AA.
109
They reported that the introduction of amino moieties at C-28 of AA significantly improved the anticancer activity of the compound against several cancer cell lines. The modification of the A-ring of AA also increased its anticancer activity. Among the synthesized compounds, N-(2α,3β,23-acetoxyurs-12-en-28-oyl)-
Wang et al. 106 synthesized five AA derivatives and examined their inhibitory activities on growth in non-small-cell lung cancer cell lines A549 and PC9/G. Four derivatives were found to have stronger cell growth inhibitory activity than AA. Among them, one compound showed the most significant antiproliferative effects on tumor. The results collectively indicated that synthesized derivatives inhibited cell proliferation via downregulation of the MEK/ERK pathway and cell cycle arrest at G1/S and G2/M. 106
Gonçalves et al. claimed that structural modification of AA may afford derivatives with improved anticancer potency. 33 Treatment of HeLa cells lines with compound 24 led to cell cycle arrest at the G0/G1 phase. Interestingly, compound 24 induced apoptotic HeLa cell death via the activation of caspase-9, caspase-8, and caspase-3. One study suggested that the mitochondrial pathway is activated during the apoptotic process. 33
AA derivatives have also shown potential antidiabetic activity. Zhang et al. synthesized 24 AA derivatives and evaluated them as inhibitors of glycogen phosphorylase. 110 Among the synthesized compounds, AA benzyl ester exhibited stronger activity than its parent compound. Structure–activity relationship (SAR) analysis showed that AA possessing a 2α-OH function exhibited more potent glycogen phosphorylase inhibitory activity than the derivative, which possesses a 2β-OH function. They suggested that further lead optimization based on AA is needed in order to find more effective AA derivatives as antidiabetic agents with protective effects against ischemic diabetic complications.
Wen et al. revealed that pentacyclic triterpenes such as AA represent a promising class of antidiabetic agent that exerts hypoglycemic effects through glycogen phosphorylase inhibition. 111 They studied 25 naturally occurring pentacyclic triterpenes, including the pentacyclic triterpenes AA and maslinic acid, as inhibitors of rabbit muscle glycogen phosphorylase, the main regulatory enzyme in the liver responsible for the control of blood glucose levels. 111 Furthermore, AA derivatives with a modified A-ring displayed good cytotoxicity against neoplasm P388D1 and melanoma Malme-3M cells. 112
Jeong evaluated 10 semisynthetic derivatives of AA for their wound healing effects by employing a tensile strength assay and a wound area assay (Fig. 2). Among them, ethoxymethyl-2-oxo-3,23-isopropylidene-asiatate showed remarkable wound healing activity. He also studied the SAR of AA derivatives as wound healing agents. 113
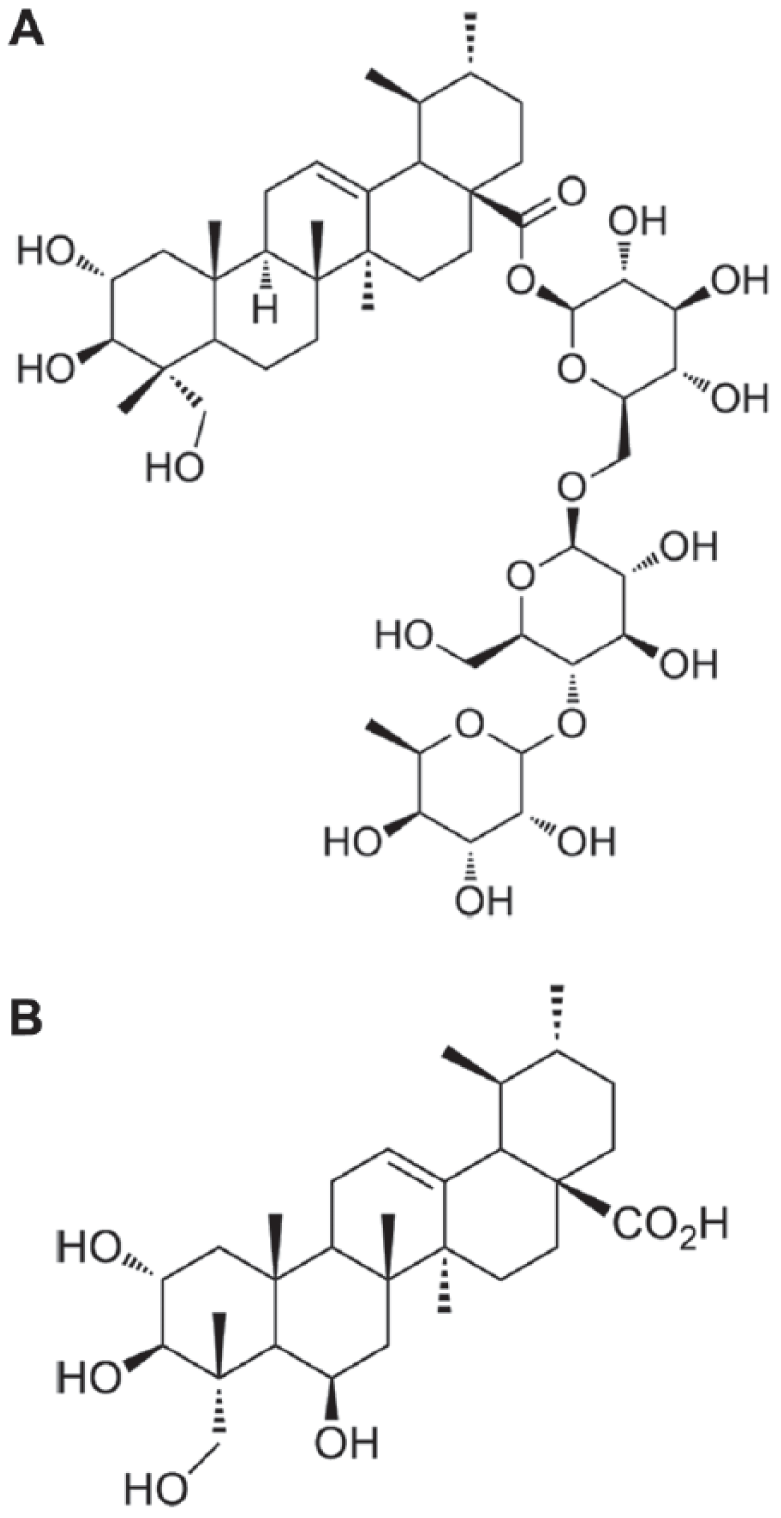
AA and other naturally occurring triterpenoids reported for wound healing activity. Their wound healing activity is associated with the modulation of collagen synthesis in the skin dermis. Also, a SAR study indicates that lipophilic transformations, such as acetate formation with the three hydroxy groups in the A-ring and ester formation with the C(28) carboxylic acid in the D-ring, are relatively important to enhance the wound healing activity. 113
Li et al. synthesized some AA derivatives and evaluated them as antifibrotic agents. The aglycone AA was chemically modified, and the resulting derivatives were evaluated for their potential as antifibrotic agents. The data were obtained from in vivo testing of these compounds in a rodent CCl4-induced liver injury mode. 114
Two AA derivatives, 3-β-23-dihydroxyurs-2-oxo-12-ene-28-oic acid and 3-β-23-dihydroxyurs-12-ene-28-oic acid, have shown hepatoprotective activity against CCl4-induced injury by preserving the cellular antioxidative defense system. 115
Lin et al. have reported the effectiveness of AA and related compounds for inducing neuronal differentiation. 116 Lin et al. further investigated and found that AA extensively promoted nerve differentiation in vitro, and it can induce nerve differentiation through the activation of the MEK signaling pathway. Their study supports the notion that the use of AA can be effective in neurodegenerative diseases. 116 The derivatives of AA, with their biological effects, are depicted in Figure 3.
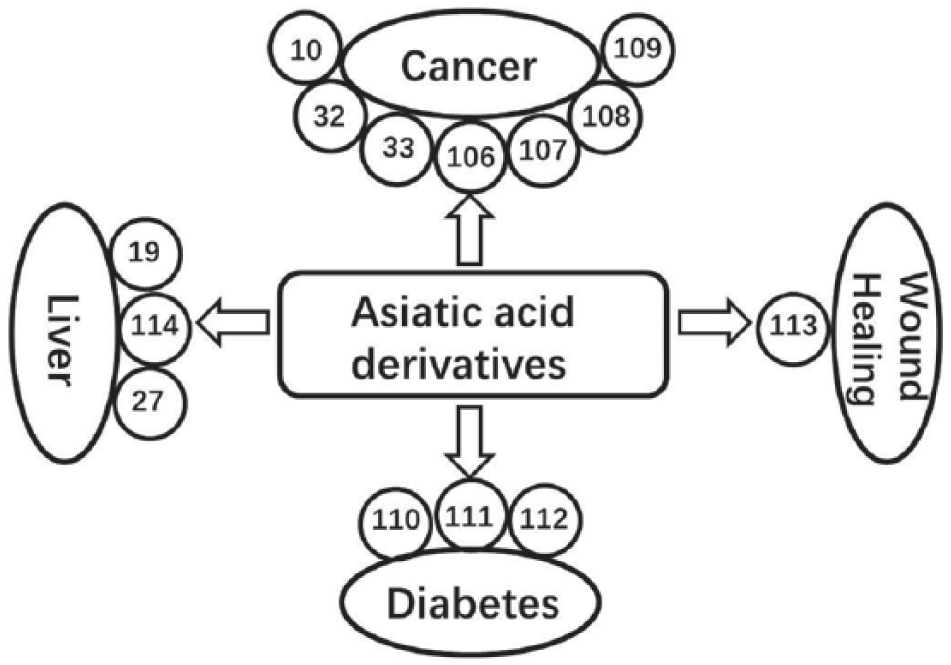
Potential effect of AA derivatives on the prevention of various diseases. However, various derivatives of AA were designed and synthesized based on chemical modifications and would have a high impact on its biological activity.
Pharmacological Activity of Other Potent Natural Compounds in Comparison with AA
In recent years, the use of natural plant compounds has attracted increased attention among clinicians and from the public for the prevention and treatment of various chronic diseases. Among them, polyphenols such as curcumin and resveratrol (3,5,4′-trihydroxy-trans-stilbene) (nonflavonoids) have received much attention for their ability to reduce cellular stress-induced injury. 117 Many successful pharmacological studies have demonstrated the use of curcumin and resveratrol in various diseases. Herein, the present work also considers the pharmacological studies of these two natural compounds. Furthermore, the pharmacological activities of AA are compared with those of curcumin and resveratrol based on their mechanisms of action.
Proposed Mechanisms of Action of Curcumin and Resveratrol in Various Diseases
Curcumin is a bright yellow compound found in turmeric, which is derived from the rhizomes of the plant Curcuma longa Linn of the Zingerberaceae family. 118 Resveratrol, chemically known as 3,5,4′-trihydroxystilbene, is a stilbenoid, a derivative of stilbene that exists as two geometric isomers: cis- (Z) and trans- (E) with the trans-isomer. 119
Extensive scientific work with curcumin and resveratrol has shown that these two potential natural compounds could alter multiple pathways via cell growth, apoptosis, and inflammation in cancer. It has also been proposed that curcumin exerts its anticancer effects through a variety of mechanisms, including the downregulation of NF-κB, the suppression of AP-1 activity, various effects on STAT proteins, and other mechanisms and biochemical pathways. These mechanisms, via pathways, are involved in the proliferation and survival of cancer cells by directly and indirectly binding to different targets.120–123 In contrast, resveratrol affects the processes underlying all three stages of carcinogenesis, and has been shown to suppress angiogenesis and metastasis. 124
Both natural compounds show a significant role in different liver diseases. 125 Curcumin demonstrated inhibitory effects on NF-κB, which is able to regulate different pro-inflammatory and pro-fibrotic cytokines. It also has a crucial role in the mitigation of several liver diseases.126,127 Studies have confirmed that resveratrol provides significant liver protection against chemical, cholestatic, and alcohol injury, and decreases liver fibrosis and steatosis by reducing hepatic fibrosis.128–130
There have been various experimental studies related to curcumin and resveratrol for their wound healing activity. Pharmacologically, curcumin stimulates the production of the growth factors involved in the wound healing process and accelerates the management of wound restoration.131–133 On the other hand, resveratrol may also have a beneficial effect on wound healing by significantly enhancing the endothelial function, tensile strength and inflammatory changes. 134
Interestingly, both curcumin and resveratrol could present an attractive alternative to current treatments for inflammation since curcumin’s anti-inflammatory effects are thought to be caused by lowering transendothelial monocyte migration by a decline in mRNA and involvement of protein expression.135–137 Research suggests that resveratrol demonstrates anti-inflammatory properties as it suppresses COX-2 by blocking NF-kB activation.138–141
A large number of studies have extensively investigated the cardiovascular system for curcumin in both animal and human subjects with detailed pharmacological action. 142 For resveratrol, several potential targets related to the beneficial effects in the cardiovascular system have also been studied. 143
Curcumin affects most of the leading aspects of diabetes, including insulin resistance, hyperglycemia, hyperlipidemia, and necrosis.144,145 Results show that resveratrol is also capable of inducing beneficial effects in diabetic animals and ameliorates metabolic disorders.146–148 Curcumin shows remarkable antioxidant activity, and it has been found to be an excellent free radical scavenger. 149 Published research has indicated that resveratrol decreases the extent of lung damage induced by nicotine and improves the antioxidant defense system. 150
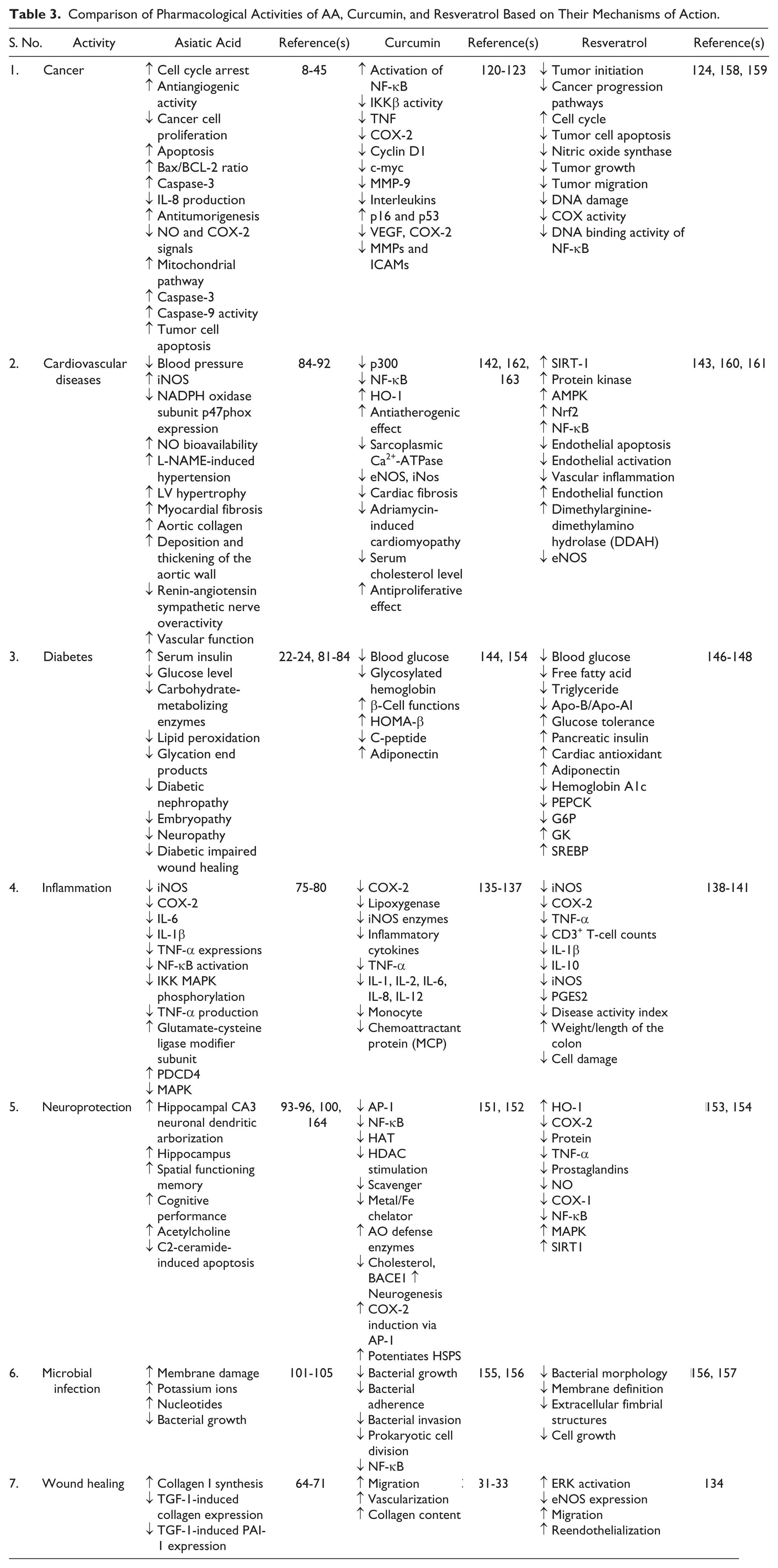
Curcumin is a strong candidate for use in the prevention or treatment of neurodegenerative diseases.151,152 Various studies, ranging from cell cultures to animal studies, have also confirmed that resveratrol inhibits β-amyloid protein aggregation and modulates intracellular effectors involved in neuronal cell survival/death.153,154 Cumulative findings in recent years have shown that curcumin and resveratrol are active against different bacteria, viruses, and fungi.155 –157 Table 3 summarizes the pharmacological activities of the comparison among AA, curcumin, and resveratrol based on their mechanisms.
Comparison of Pharmacological Activities of AA, Curcumin, and Resveratrol Based on Their Mechanisms of Action.
AA in Conjunction with Approved Targeted Therapies
AA has a wide variety of biological activities. Previously, it was mainly considered for anti-inflammatory, wound healing, antidiabetic, antioxidant, hepatoprotective, and some anticancer activities. However, in the recent past, the apoptosis-inducing activity of AA in various cancer cells has also been investigated. Extensive research has shown that AA can greatly alter the multiple pathways involved in the proliferation and survival of cancer cells by directly and indirectly binding to different targets; the involved mechanisms may affect cell growth or can induce apoptosis and inflammation (Fig. 4). Researchers have proposed that AA exerts its anticancer effects through a variety of mechanisms, including the downregulation of NF-κB, the suppression of AP-1 activity, and various effects on the STAT proteins. Hence, in conjunction with these findings and the compilation of this work, we speculate that the underlying mechanism of AA and its derivatives may be useful in various diseases, notably cancer.
Molecular targets of AA and its derivatives.
Current Findings and Future Prospects
This review provides important evidence that AA and its derivatives have promising therapeutic applications for the treatment of various chronic disorders. Because of its pharmacological activities, low toxicity, and commercial availability, AA has been receiving greater consideration in molecular biology. The outcomes of various experimental data, including in vitro and in vivo studies, have confirmed the potential epidemiological evidence of AA in various diseases. Although investigational data seem very promising, the clinical experiment of AA in the treatment of diseases is limited due to its modest therapeutic activity and poor bioavailability. Interestingly, derivatives of AA have also been investigated in a wide range of diseases and found significantly valuable for their immense medicinal and therapeutic benefits. Studies have demonstrated that chemical modification of the AA backbone has a high effect on its biological activity and may represent the solution to improve not only the antitumor activity of AA, but also its pharmacokinetic properties. More exhaustive studies are required to elucidate the molecular mechanisms and pharmacological effects of AA (analog and its derivatives) to establish it as an ideal therapeutic agent in a range of diseases focusing on cancer. Since the majority of therapeutic data on AA are based on in vitro and in vivo models, more extensive and well-controlled human trials should be established to accurately determine its optimal administration dose and route of administration with strong combating effects.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Natural Science Foundation of China (81301293) and the National Science and Technology Major Projects for “Major New Drugs Innovation and Develop-ment” (2014ZX09507008).
