Abstract
Automated mechanism of action studies are introducing the need for tailored compound delivery, which can be challenging for standard compound management procedures. Jump dilution assays investigating inhibitor reversibility require compound delivery at specific volumes to assay specific concentrations of 10 × IC50 for each inhibitor. Creating custom-made source plates with unique compound concentrations to dispense a uniform single volume can be prohibitively slow. A broadly applicable tool that enables on-the fly dispensing of variable amounts of stock concentrations was developed using the Acoustic Transfer System (ATS). The Dynamic Transfer Modification Program (DTMP) is an integrated LabVIEW program used to automate customized volume transfers from each well based on compound identity within a given source plate. A jump dilution investigating the time-dependent inhibition of the enzyme dipeptidyl peptidase-4 (DPP4) with multiple inhibitors is described here to demonstrate the delivery of specific volumes of various compounds in a high-throughput manner. The ability to automate this process allows for the characterization of inhibitor reversibility earlier in the drug discovery process, resulting in better informed lead candidate selection.
Keywords
Introduction
High-throughput screening (HTS) traditionally consists of assaying compounds at a single common concentration or standard titrations to identify lead candidates.1–3 Investigating compounds with this approach provides insight into activity and potency for the target but is limited in investigating the binding kinetics of the compound, which is necessary in understanding the pharmacological profile of a potential drug. 4 With greater awareness of the benefit of characterizing leads earlier in the discovery process, there is increased demand for testing each compound at individual concentrations. Automating these assays can be challenging because the preparation of source plates with different compounds in every well at unique concentrations is inefficient and requires increased time and resources. 5 The Acoustic Transfer System (ATS) (EDC Biosystems, Freemont, CA) was used to develop and implement a LabVIEW (National Instruments, Austin, TX) program, called the Dynamic Transfer Modification Program (DTMP). This application enables dynamic modification of volume transfers from a single-concentration source plate, creating customized volume transfers from each well based on compound identity within a given source plate. This capability helps to increase the automation of kinetic studies such as jump dilution analysis, which requires the transfer of specific volumes of each compound.
The DTMP application was automated using an internally developed screening platform. The screening platform uses control software, named Telios, and instrument drivers written in LabVIEW, to control the coordinated operations of integrated components such as third-party devices to execute a parallel process. The internal platform was an ideal resource to implement the DTMP solution because the program could be quickly and affordably integrated into the ATS driver as they were both written in LabVIEW ( Fig. 1A ). 6 The ATS is an acoustic droplet ejection system used in HTS compound delivery applications. 7 It was chosen to implement the DTMP application, as it has access to compound transfer information through an external comma-separated values (csv) file, referred to here as a plate map file. This characteristic enabled the creation of the DTMP, which was incorporated into the ATS driver and serves to customize this file based on compound identity immediately prior to each dispense. The automation concepts described here can be mirrored and used to integrate other programs to modify this external csv file prior to dispense in a similar fashion. Although in this scenario, the program was integrated directly into the ATS driver and functioned as an optional component of the driver, the program could have also been developed to customize the external file independently as a separate step in the protocol prior to the ATS dispense. Another advantage to using the ATS for complex dispensing of different volumes is the ability to use 2 drop volumes during acoustic dispense to each well using the variable drop volume (VDV) technology, which increases the dispense speed. 8
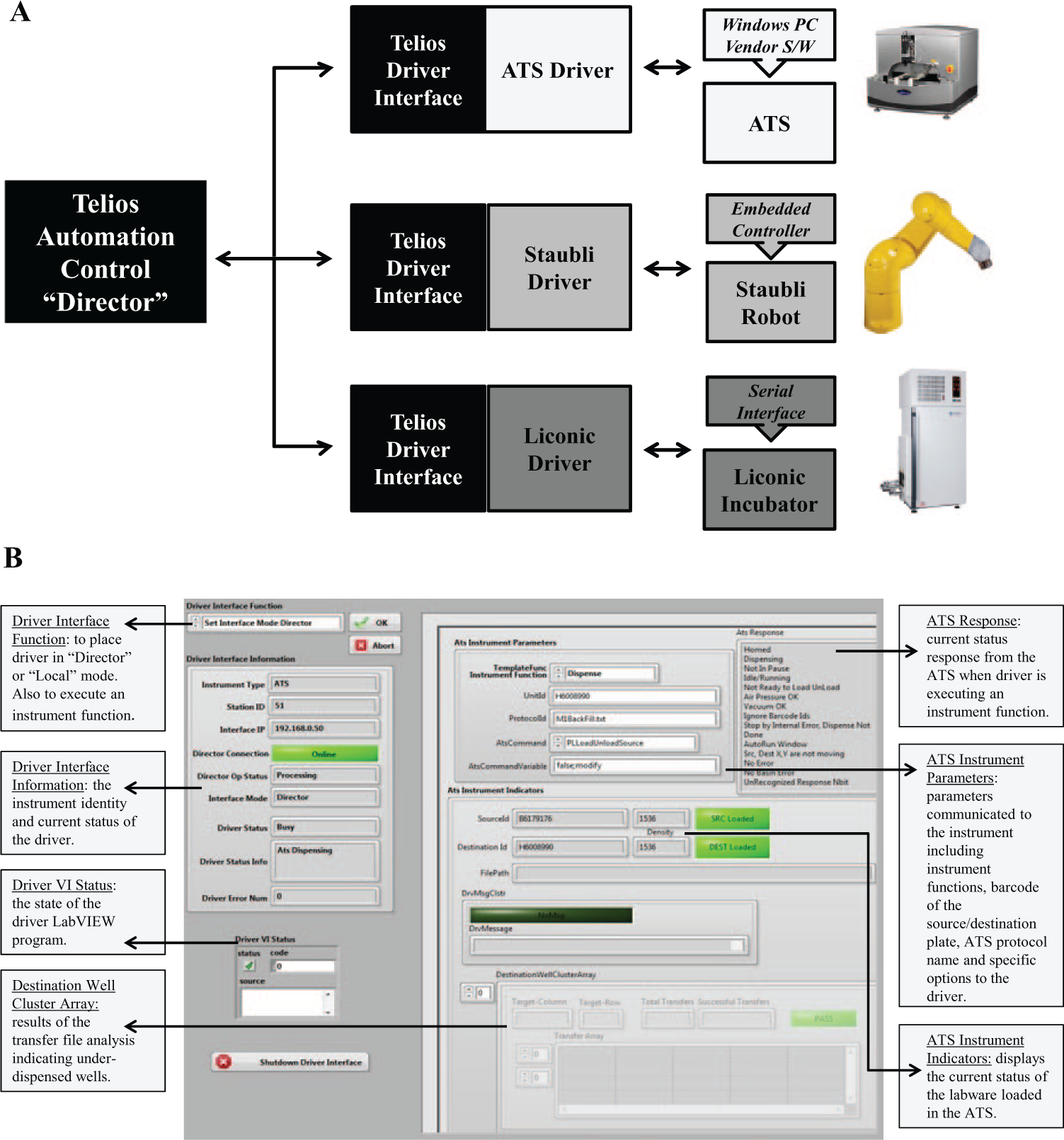
Map of communication between the Telios director software and integrated third-party equipment. (
The ability to recurrently modify compound transfer volumes facilitates an automated approach to complicated studies such as jump dilution analysis. As introduced and reviewed in Copeland et al.,9,10 jump dilutions evaluate the reversible binding of a compound of interest through the examination of enzyme recovery from compound inhibition. Following the formation of the inhibitor-enzyme complex, a large dilution is performed, during which enzyme activity is recovered according to the dissociation of the inhibitor. Conventional HTS paradigms include inhibitor titrations from which the specific potency values (IC50) necessary for jump dilutions can be obtained without altering the current workflow. By diluting the compound-enzyme complex by 100-fold or more so that the final compound concentration after dilution is far below IC50, it is possible to differentiate between reversible and irreversible inhibition based on the measurement of activity over time. This step is critical during hit characterization to determine if a compound exhibits the desired characteristics of a potential lead candidate. Investigations of compound reversibility are typically performed during more advanced stages of lead optimization due to the labor-intensive nature of these studies. Customized volume transfer allows for specific dispenses of compounds of interest from a single-concentration source plate, avoiding additional dilution steps for each inhibitor concentration.3,5
Dipeptidyl peptidase-4 (DPP4) was chosen as a model system to develop a high-throughput automated jump dilution assay due to the availability of known inhibitors with varying reversibility. Inhibitors of DPP4 are used in the treatment of type 2 diabetes to regulate glycemic control and insulin release.11,12 Several inhibitors, including vildagliptin (Galvus; Novartis, Basel, Switzerland), saxagliptin (Onglyza; Bristol-Myers Squibb/AstraZeneca, New York, NY), and linagliptin (Tradjenta; Boehringer Ingelheim, Ingelheim am Rhein, Germany), inhibit DPP4 with slow dissociation rates.13–19 These slow recovery inhibitors were used to demonstrate the capabilities of an automated jump dilution assay to identify compounds with slow dissociation rates. Additional DPP4 inhibitors from the MSD collection with shorter dissociation rates were evaluated to establish a range of dissociation rates that can be detected. Titrations of all inhibitors with DPP4 were initially performed using the internal standard of the HP D300 (Hewlett-Packard, Palo Alto, CA) for compound delivery as well as the ATS to verify that the ATS dispense was accurate with the customized volume transfer protocol. The jump dilutions were only performed using the ATS since the DTMP made it much better suited to these types of dispenses. Data presented here demonstrate rapid and robust differentiation of these diverse mechanisms of action.
Materials and Methods
Reagents
DPP4 activity was monitored through the cleavage of fluorescent substrate H-Gly-Pro-AMC purchased from Bachem (Bubendorf, Switzerland). Active recombinant human DPP4 enzyme derived from an NS0 cell line (residues Asp34-Pro766) was obtained from R&D Systems (Minneapolis, MD). DPP4 inhibitors were purchased from SelleckChem (Houston, TX) and reconstituted in 75% DMSO (Sigma Aldrich, Gillingham, UK) and 25% H2O, aliquoted, and stored at −20 °C until use. MSD DPP4 inhibitors were selected from the MSD compound library, 10 mM stocks were prepared in 75% DMSO, 25% H2O. Inhibitor titrations were performed in Corning 1536 high-base, white nonbinding plates (Corning, Corning, NY) using 1536 low-base plates for the inhibitor source plates (Brooks, Chelmsford, MA). Time-dependent inhibition assays were run in black nonbinding 384-well plates (Corning) using the same source plates as above (Brooks). Assay buffer comprised 25 mM Tris (Corning) at pH 8, as recommended by R&D Systems, and was used for all enzyme and substrate dilutions.
Schematic for DTMP
A custom LabVIEW program was developed and incorporated internally into the ATS driver to automate variable compound transfer volume efforts. When the ATS receives a source and destination plate from the screening platform, the Telios director software signals the ATS driver to perform the DTMP by parameters specified within the protocol ( Fig. 1B ). Before the volume transfer occurs, the program operates by scanning a designated csv file, defined as the Compound List (CL), containing a list of compounds with target volumes. The program compares the CL with the Source Plate Map (SPM) to determine if any compounds in the CL match the compounds listed in the SPM and would require modification ( Fig. 2A , B ). A custom csv file, the Well Plate Map (WPM), is then created on-the-fly for each transfer with volumes adjusted accordingly. The ATS accesses the WPM to perform the transfer, encompassing the updated acoustic transfer volume information for each well from source to destination, with unmodified wells set to the default volume and modified wells set to the volume specified ( Fig. 2C ). A Backfill Plate Map (BPM) is also created by the program, providing the option to backfill the modified well(s) with 75/25 DMSO/water. The backfill transfer can be arranged as a subsequent dispense using the same source plate following the initial transfer of the compounds. Designated DMSO well(s) from the source plate are used to backfill the modified wells to the default volume and establish consistent volume across the plate ( Fig. 2D ).
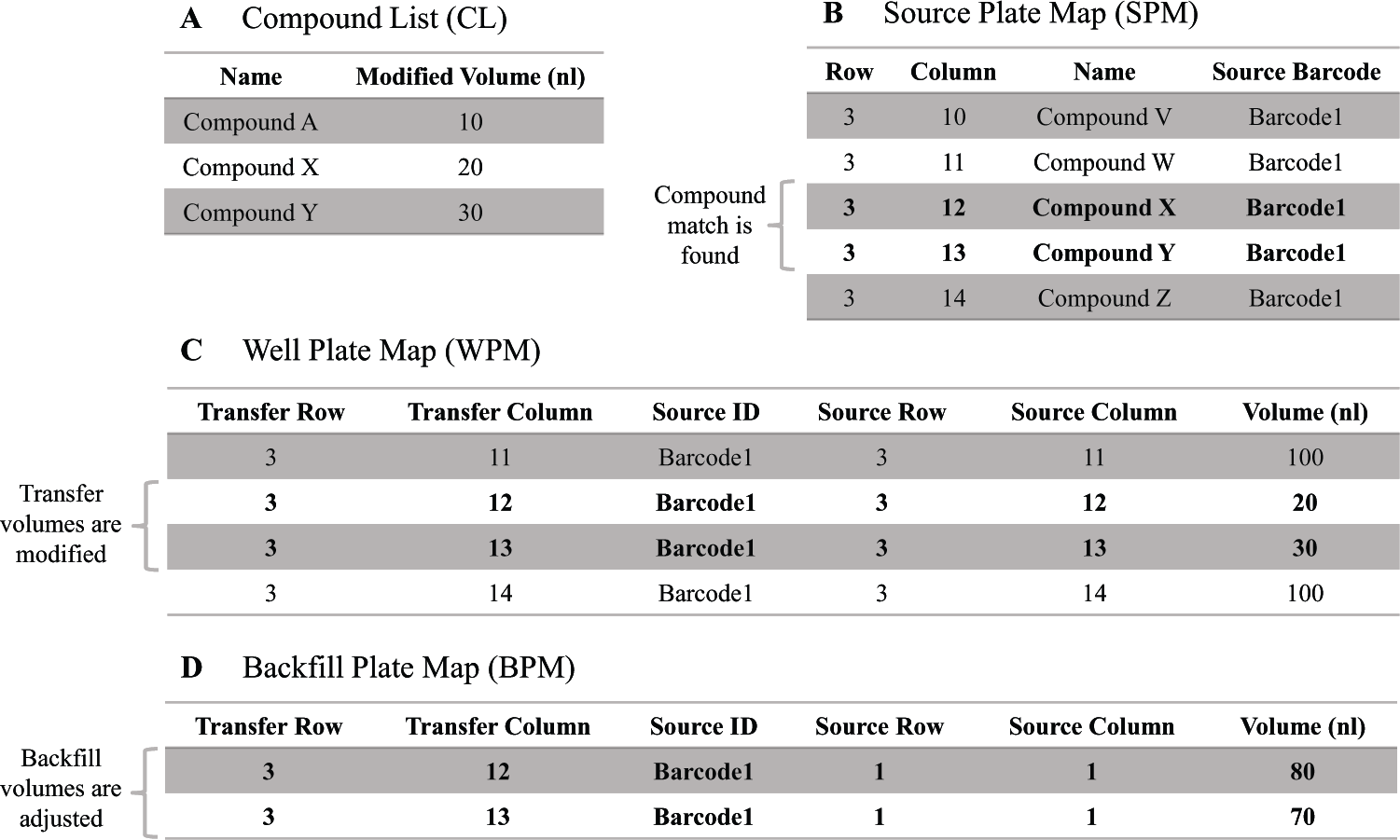
The Dynamic Transfer Modification Program (DTMP) operation schematic. (A) The Compound List (CL) created by the user is scanned by the program. (B) The Source Plate Map (SPM) is compared to the CL to determine if any compounds from the CL exist on the source plate. (C) The Well Plate Map (WPM) is created as a 1:1 transfer from the SPM with the volumes adjusted for the modified compounds. All other wells are transferred at the default volume, which is 100 nL in this example. (D) The Backfill Plate Map (BPM) is created to backfill the adjusted wells with DMSO up to the default volume using the same source plate.
Steady-State Inhibition of DPP4 (Inhibitor Titrations)
To evaluate the utility of the DTMP, inhibitor titrations were performed comparing inhibitor dispense with the HP D300 (Hewlett‑Packard, Palo Alto, CA) and the ATS using the DTMP program configuration as described above. Both dispense methods required two stock solutions of the inhibitors to achieve comprehensive titration curves. The concentrations were tailored to center the titration curves at the IC50 of each inhibitor. The inhibitor source plates were prepared by hand on the day of the experiment as well as stored at −20 °C and reused; no differences were noted with fresh versus frozen dilutions. Using the HP D300, stock concentrations of the inhibitors were dispensed from HP D300 cassettes ( Fig. 3A–D ). The ATS DTMP application required a source plate with inhibitors in two specific concentrations across several wells from which variable volumes were dispensed acoustically ( Fig. 3E–H ). The DTMP application required the SPM to be prepared using a unique naming convention for each well so the program would be able to recognize individual wells for modification ( Fig. 3I ). Backfill protocols were performed with both dispense methods to normalize for the % DMSO in all wells.
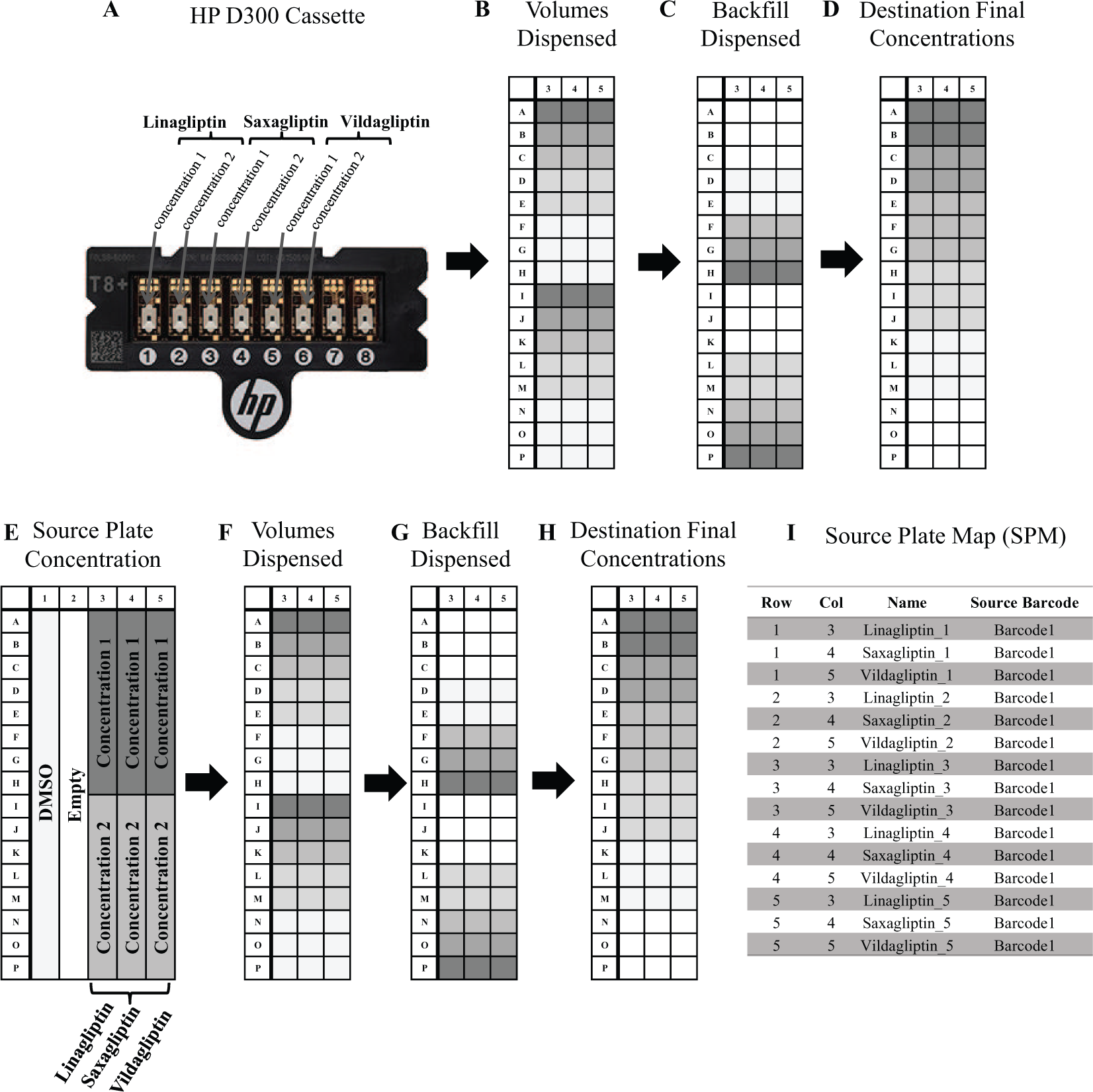
HP D300 inhibitor titration workflow and DTMP inhibitor titration workflow. (
The inhibitor titrations were dispensed by the HP D300 or the ATS using the DTMP into empty 1536-well plates. A BioRAPTR (Beckman Coulter, Brea, CA) was used to dispense 2 µL of 0.2 ng/mL DPP4 in assay buffer. The enzyme-inhibitor complex was allowed to form with a 30-min or 4-h room temperature incubation. DPP4 activity was monitored with the addition of 2 µL 20 µM H-Gly-Pro-AMC substrate with the BioRAPTR and incubating for 60 min at room temperature. Results were collected using 350-nm excitation and 460-nm emission settings on the Pherastar (BMG Labtech, Ortenberg, Germany). Three replicates for each data point were located on the assay plate to collect triplicate measurements for the titrations. Data were analyzed using GraphPad Prism v6 (GraphPad Software, La Jolla, CA), fitting the median of the triplicate values with nonlinear regression (curve fit), log[inhibitor] versus response, variable slope, and four parameters.
Time-Dependent Inhibition of DPP4 (Jump Dilution)
The DPP4 time-dependent inhibition protocol was developed to enable automation and increased throughput of jump dilution assays. The incubation times and concentrations are specific for DPP4. Substrate and enzyme titrations and stability studies were performed prior to the jump dilution experiment to ensure that neither was a limiting reagent for this experiment. The established automation will be applicable for all time-dependent inhibition studies. The ATS DTMP application was used to dispense DPP4 inhibitors using specific volumes to achieve a final concentration of 10 × IC50 of each inhibitor in 10 µL of 1 ng/mL DPP4. The solution was incubated for 4 h at room temperature to allow for complete equilibration of the enzyme. During that incubation, a separate assay plate was prepared using the Multidrop Combi (Thermo Scientific, Waltham, MA) to dispense 70 µL of 200 µM substrate to all of the wells in a 384-well plate. The jump dilution was performed with the Biomek FX (Beckman Coulter) by transferring 1 µL of the enzyme/inhibitor solution into the assay plate. The assay plate was imaged with a kinetic read on the Pherastar using the same settings as described above, measuring in 10-min intervals for 150 min. Data were collected in triplicate by including three replicates of each sample on the same assay plate. The median of each was normalized by subtracting the substrate alone median and then plotted using GraphPad Prism v6. Standard deviation was calculated using the triplicate measurements for each sample. Signal to background and Z′ calculations were performed comparing the triplicate median values of the enzyme + substrate wells to the substrate-only wells, both in the absence of any inhibitors.
Results and Discussion
While there are many liquid handling processes for DMSO-based test compound delivery, often times they require multiple dilution steps each using their own consumables, which is expensive and time-consuming. 3 In addition to using compound management infrastructure for compound plate preparation, the HP D300 has been used to dispense multiple volumes and concentrations of compounds. 20 The HP D300 is well suited to studies that require a broad range of volumes from 13 pL to 10 µL such as compound titrations. Applications such as jump dilutions, which require only a single concentration of each compound of interest, can benefit from the creation of a more widely applicable variable volume dispense. Prior to the development of the DTMP technology, specific 10 × IC50 dilutions for each inhibitor would need to be prepared manually or with significant effort from the compound management group so that a uniform volume transfer would result in the desired concentrations of each sample. The automated jump dilution workflow developed here uses 384-well plates as the final plate density. Dispensing to the designated sample region of a 384-well plate could require up to 320 different individual targeted compound concentrations ( Fig. 4 ). Initially, it was proposed to use the HP D300 system for this purpose due to the low-volume dispense capability. If this were attempted with the HP D300, it would require 40 different dispensing cassettes. Preparing a source plate of this nature is inefficient in an HTS environment, and therefore, identifying another application was essential to justify automating jump dilution studies.

Map of a high-throughput jump dilution using the Dynamic Transfer Modification Program (DTMP). (
The ATS DTMP streamlines variable volume transfers useful in studies that require compound-specific final concentrations. This process deviates from traditional screening experiments employing a source plate prepared with compounds at a single concentration for multiple dispense volumes or conditions. The automated jump dilution assay takes advantage of this simplified workflow by transferring diverse volumes from a single-stock concentration of the compound collection, demonstrating how this function can be used in a high-throughput setting ( Fig. 4 ). This feature expands the current HTS screening platform capabilities in addition to better utilizing compound management resources. For this effort, the IC50 values were determined as an initial experiment in studying the steady-state inhibition of DPP4 ( Fig. 5A–C ). Consistent with the typical mode of operation in screening, the preceding dose-response assays will provide these values for any compounds of interest. Comparing between the two methods, the IC50 values generated with the HP D300 and the ATS were consistent, indicating that the DTMP is a reliable application for compound delivery ( Table 1 ). In addition, the similarity between the IC50 values for the 30-min and 4-h incubation times indicates that slow binding does not occur with these inhibitors ( Table 1 ). 21 This type of verification must be performed prior to jump dilution studies or else slow binding inhibitors will appear to dissociate more rapidly due to incomplete inhibitor-enzyme complex formation.
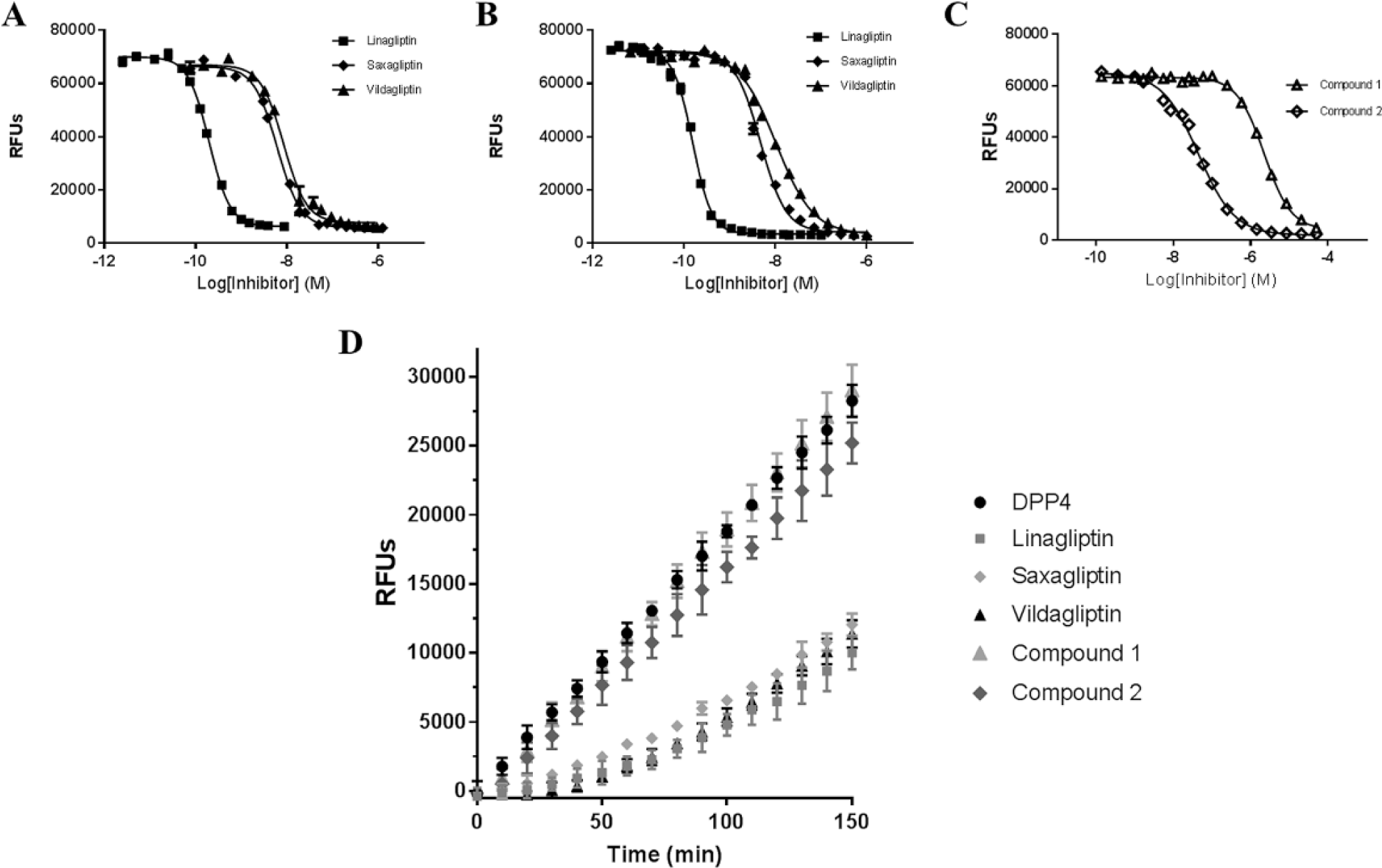
Inhibitor titrations and jump dilution results. Inhibitor titrations were performed in triplicate with compound dispense from the Acoustic Transfer System (ATS) (
IC50 Values Calculated in GraphPad Prism for the Inhibition of Dipeptidyl Peptidase-4 with Commercially Available Inhibitors.
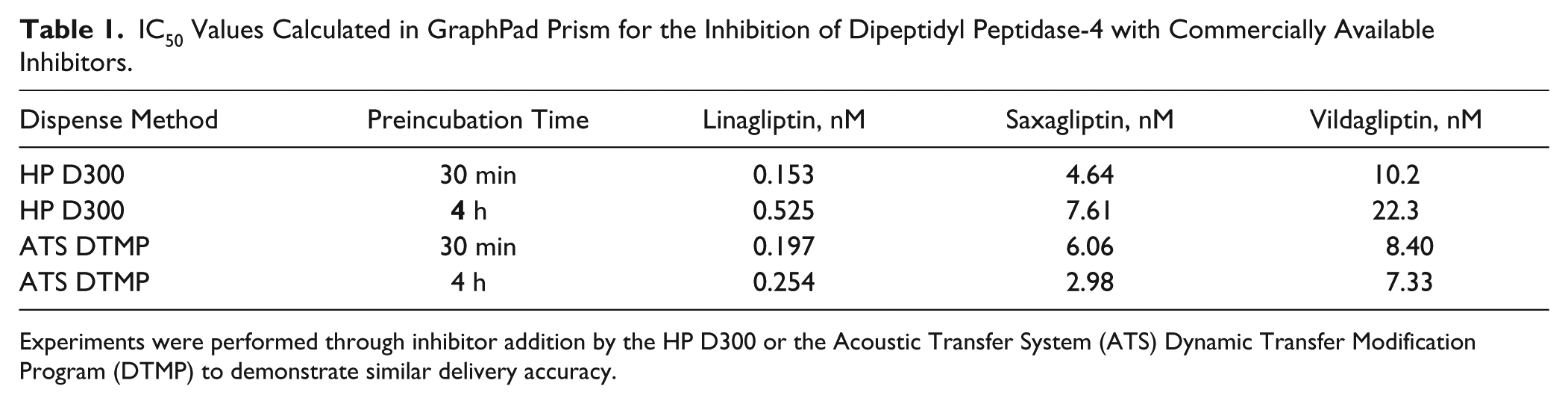
Experiments were performed through inhibitor addition by the HP D300 or the Acoustic Transfer System (ATS) Dynamic Transfer Modification Program (DTMP) to demonstrate similar delivery accuracy.
The DTMP application was used to deliver different volumes of DPP4-specific inhibitors to achieve final concentrations of 10 × IC50 for each inhibitor in a DPP4 solution. Jump dilutions were performed by diluting the enzyme-inhibitor complex and observing the percent recovery. The inhibitors were all located on the same assay plate for this experiment and had a Z′ value of 0.73 at 150 min. The recovered activity of DPP4 after 70-fold dilution from linagliptin, vildagliptin, and saxagliptin was reduced when compared to enzyme activity in the absence of inhibitors ( Fig. 5D ). The divergence of these slopes from the uninhibited enzyme activity correlates with the published slow-off rates of these DPP4 inhibitors. The deviation from linearity of these inhibitors indicates that they have reversible inhibition of DPP4 with slow dissociation of the inhibitor-enzyme complex.9,10 Establishing the upper limit for long-lived DPP4 inhibition allowed for the evaluation of other DPP4 inhibitors. The DPP4 inhibitors from the MSD collection display much shorter dissociation rates, highlighting the ability to differentiate a wide range of dissociation behavior ( Fig. 5D ). In an HTS setting, the fitted slopes of the dissociation curves could be used to rank order compounds based on their reversibility using preselected cutoff values. Often the treatment of reversibility has been more binary, bucketing compounds based on the presence of absence of reversibility. The addition of automation to these studies will allow for a more advanced approach to using the dissociation behavior due to the increased number of compounds that can be used to score lead compounds. In addition, larger mechanism of action data sets could allow for clustering and machine learning, which could produce better leads based on the enzyme-inhibitor interactions. More detailed binding studies could be subsequently performed to determine precise off rates, but the ability to analyze the reversibility of many compounds early in the drug discovery process can aid in candidate selection. DTMP compound dispense enables the investigation of compound reversibility in a high-throughput setting and will support additional mechanism of action studies with custom volume dispenses.
In addition to jump dilution studies, automation and customization of volume transfers on a well-to-well basis is technology that can streamline the downstream hit profiling process. The DTMP can be used to set the transfer volume to zero to disregard transfer of a specific compound, which is useful in situations where a compound has a strong fluorescent signal and is affecting neighboring wells. Conversely, single compounds can be selected for rescreening as needed, preserving stocks of other compounds from the same source plate for future use and responsibly using compound resources. Furthermore, the transfer volume can be set to a partial volume in cases where it is beneficial or necessary to test multiple concentrations of each compound, relevant to efforts such as quantitative high-throughput screening (qHTS). 3 As demonstrated above, the ability to evaluate multiple concentrations from a source plate with one concentration relieves pressure on compound management resources and provides assay scientists with more flexibility to incorporate complex studies during hit characterization screens.3,5
Earlier differentiation based on mechanism of action enables better prioritization of leads.9,10 Compounds are typically examined for their potency for the target protein, but compounds with the same potency can have different dissociation rates.
22
The known DPP4 inhibitors had potency values that correspond to their dissociation behavior. However, compound
Footnotes
Acknowledgements
We thank Rumin Zhang for his insights about jump dilution experimental design.
Declaration of Conflicting Interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Authors are employees of Merck & Co., Inc. (Kenilworth, NJ) and potentially own stock and/or stock options in the company.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
