Abstract
The SLC1 family includes seven members divided into two groups, namely, EAATs and ASCTs, that share similar 3D architecture; the first one includes high-affinity glutamate transporters, and the second one includes SLC1A4 and SLC1A5, known as ASCT1 and ASCT2, respectively, responsible for the traffic of neutral amino acids across the cell plasma membrane. The physiological role of ASCT1 and ASCT2 has been investigated over the years, revealing different properties in terms of substrate specificities, affinities, and regulation by physiological effectors and posttranslational modifications. Furthermore, ASCT1 and ASCT2 are involved in pathological conditions, such as neurodegenerative disorders and cancer. This has driven research in the pharmaceutical field aimed to find drugs able to target the two proteins.
This review focuses on structural, functional, and regulatory aspects of ASCT1 and ASCT2, highlighting similarities and differences.
Introduction
The SLC1 family is a relatively small group of membrane transporters involved in amino acid traffic in cells. The family encompasses seven members, which are divided in two groups sharing substrate specificity and transport mode.1–3 The first group includes SLC1A1, A2, A3, A6, and A7, collectively known as EAATs (excitatory amino acid transporters), exhibiting high affinity for the negatively charged amino acids glutamate and aspartate and localized mainly in the brain; the second group includes SLC1A4 and A5, known as ASCTs (alanine, serine, cysteine transporters), involved in the traffic of several neutral amino acids in a broad set of tissues.1–3 The functional classification used in the pregenomic era has been later confirmed by the evolutionary analysis brought to the SLC classification. The seven members of the SLC1 family share a low percentage of identity, that is, 21% (
Gene Chromosome Location and Natural Variants
The human ASCT1 (SLC1A4) gene contains eight exons and maps on chromosome 2p14. 8 The ASCT1 gene is found in 341 organisms (https://www.ncbi.nlm.nih.gov/gene/6509), among which several are vertebrates. In the National Center for Biotechnology Information (NCBI) database, four different isoforms are listed. The first isoform is the longest one, encoding for a peptide of 532 amino acids (NM_003038.5), with a theoretical molecular mass of 55 KDa. The other three transcripts are shorter, being 312 (NM_001348407.2), 214 (NP_001180422.1), and 312 (NP_001335335.1) amino acids long. The isoforms differ in their 5′-UTR, coding sequences, and codon start. Several single-nucleotide polymorphisms (SNPs) have been found on the ASCT1 human gene, some of which have been associated with clinical conditions. In particular, rs201278558 and rs761533681 are responsible for the c766G-A and c.1369C-T transitions, respectively. These two mutations correspond to the substitution of the highly conserved residues E256 and R457 into Lys and Trp, respectively. Interestingly, these variants are associated with a disease called SPATCCM (OMIM 616657), that is, spastic tetraplegia, thin corpus callosum, and progressive microcephaly. 9
The human ASCT2 (SLC1A5) gene contains eight exons and maps on the chromosome 19q13.32. The ASCT2 gene is found in 56 different organisms (https://www.ncbi.nlm.nih.gov/gene/6510) and is virtually present in all vertebrates. In the NCBI database, three different isoforms are listed. The first isoform is the longest one, encoding for a peptide of 541 amino acids (NM_005628), with a theoretical molecular mass of 57 kDa. The second one differs in the 5′-UTR from the first variant and encodes for a shorter peptide of 313 amino acids (NM_001145144). The third isoform has a different translation start, lacks the first exon (NM_001145145), and encodes for a peptide of 339 amino acids. A longer transcript XM_005259167 has been identified by computational analysis and is only reported in the NCBI database. Several SNPs have been reported for the SLC1A5 gene, some of which have been characterized; as an example, the variants rs3027956 and rs11668878 are associated with breast cancer and chronic lymphocytic leukaemia, respectively.10,11 On the contrary, the other two variants, rs3027985 and rs1644343, have been linked to longevity. 12
Localization and Subcellular Localization in Physiological Conditions
The protein encoded by the SLC1A4 gene has also different aliases: ASCT1, SATT, and SPATCCM, with a unique localization at the plasma membrane of cells. The ASCT1 protein is widely distributed in the human body; high levels of ASCT1 are found in skeletal muscle, lung, kidney, ovaries, and heart and across the digestive tract. In particular, it is found at the basolateral membrane of stratified squamous epithelia from the oral cavity to the nonglandular stomach and in the basolateral membrane of Paneth’s cells.
13
Furthermore, ASCT1 is ubiquitously expressed in the brain, while its presence in the blood–brain barrier (BBB) has not been established yet. The physiological role of ASCT1 has been investigated since its first identification, even though no much information is available on this issue; the most acknowledged function of ASCT1 seems to be linked with brain homeostasis due to the transport of
The protein encoded by the SLC1A5 gene has different aliases, besides ASCT2, that is, AAAT, ATBO, M7V1, M7VS1, R16, and RDRC, that are much less used. The protein has a predominant localization at the plasma membrane of cells. However, the shortest ASCT2 variant (NM_001145145) has been localized at the inner membrane of mitochondria. 20 This finding opened an important perspective in the field of bioenergetics, considering that the search for a mitochondrial glutamine transporter has been a treasure hunt for decades. 21 However, the actual role of an ASCT2 variant in mitochondria has been questioned. 22 ASCT2 is widely distributed in the human body, with higher expression in lung, skeletal muscle, large intestine, kidney, testis, T cells, and adipose tissue.1,23 The described broad expression underlies the physiological role of the ASCT2 in harmonizing the amino acid pools in cells, being in line with its transport mechanism and with the functional and kinetic asymmetry of the catalyzed reaction. 21 This role is also relevant in pathological conditions in which the request for specific amino acids increases. 24 Finally, ASCT2 is also expressed in different areas of the brain and in the BBB, where it seems to regulate the flux of glutamate and aspartate, besides that of neutral amino acids.25,26 This alternative function, in line with the novel glutamate/proton transport recently demonstrated in vitro, 26 may underline a role for ASCT2 in neurological disorders and brain cancers.16,27
Structural and Functional Features
The main challenge in the study of structural properties of human membrane transporters is the scarcity of refined 3D structures, due to the intrinsic difficulties in handling such hydrophobic proteins. This paradigm also holds for ASCT1 and ASCT2 until the solution of the ASCT2 structure in 2018. 28 However, most of the available structure–function relationship information on the SLC1 family members, and hence for ASCTs, derives from comparisons with a bacterial glutamate transporter from Pyrococcus horikoshii, namely, GLTPh, whose structure was solved in 2004. 29 The overall architecture of GLTPh was clearly defined as a homotrimer with a concave aqueous basin and a triangular shape. Then, over almost two decades, other 3D structures of GLTPh in different conformations have been solved, together with those of other bacterial transporters for glutamate/aspartate, such as GLTtk.2,7,30 These studies confirmed the homotrimeric architecture and gave further insights into the molecular determinants of the transport mechanism and allowed studying the human members of the SLC1 family by homology modeling coupled to biochemical approaches.31–33 Later, in 2017, the 3D structure of the human isoform of EAAT1 was solved, demonstrating that the eukaryotic proteins show the same homotrimer organization as the bacterial counterparts; since then, more refined homology models have been built for the other members of the SLC1 family, including ASCT2.33,34 As stated in the introduction, the interest around ASCT2 increased a lot due to potential outcomes in human therapy, and its 3D structure was solved by cryo-EM in an inward occluded conformation in the presence of the favorite substrate, Gln (Protein Data Bank [PDB] ID: 6GCT). 28 Soon after, other structures were obtained in an outward open conformation, in the presence (6MPB) or absence (6MP6) of Gln, that helped in visualizing how the protein may move in the membrane during the transport cycle. 35 To solve the structure of ASCT2 in the inward open conformation, a specific mutation in the substrate binding site, namely, C467R, had to be introduced to obtain a suitable protein for the cryo-EM assay (6RVX and 6RVY). 36 Based on the information on the 3D ASCT2 and EAAT1 structures, some general features could be drawn for both ASCT1 and ASCT2: (1) the assembly of a homotrimer in the cell membrane, as for the prokaryotic proteins and EAATs ( Fig. 1A , side view); and (2) the presence, in each monomer, of a scaffold domain and a transport domain ( Fig. 1B , top view). In particular, the scaffold domain remains fixed and embedded in the membrane while, and the transport domain of each monomer slides over the scaffold, crossing the membrane, giving rise to a peculiar mechanism known as elevator mechanism, 37 described as such in 2011 for GLTPh. 38
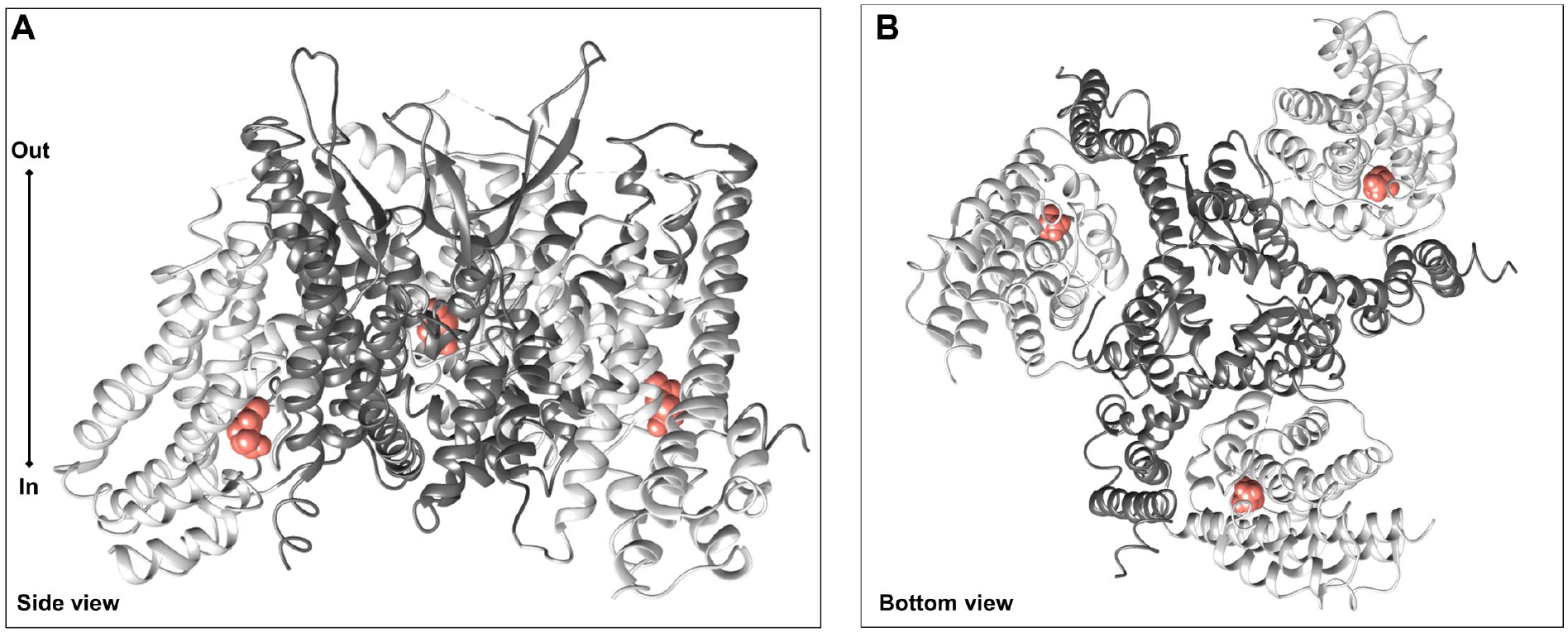
Cryo-EM structure of the human ASCT2. Homotrimers of human ASCT2 in the inward-facing conformation (PDB ID: 6GCT) are represented using Chimera v1.14 software (https://www.cgl.ucsf.edu/chimera). In dark gray is the scaffold domain, and in light gray, the transport domain. (
To better compare ASCT1 (SLC1A4) and ASCT2 (SLC1A5) in terms of structural properties, we built the homology model of ASCT1 based on the ASCT2 3D structure (
Fig. 2A–C
). The two proteins share a sequence identity of 58% (
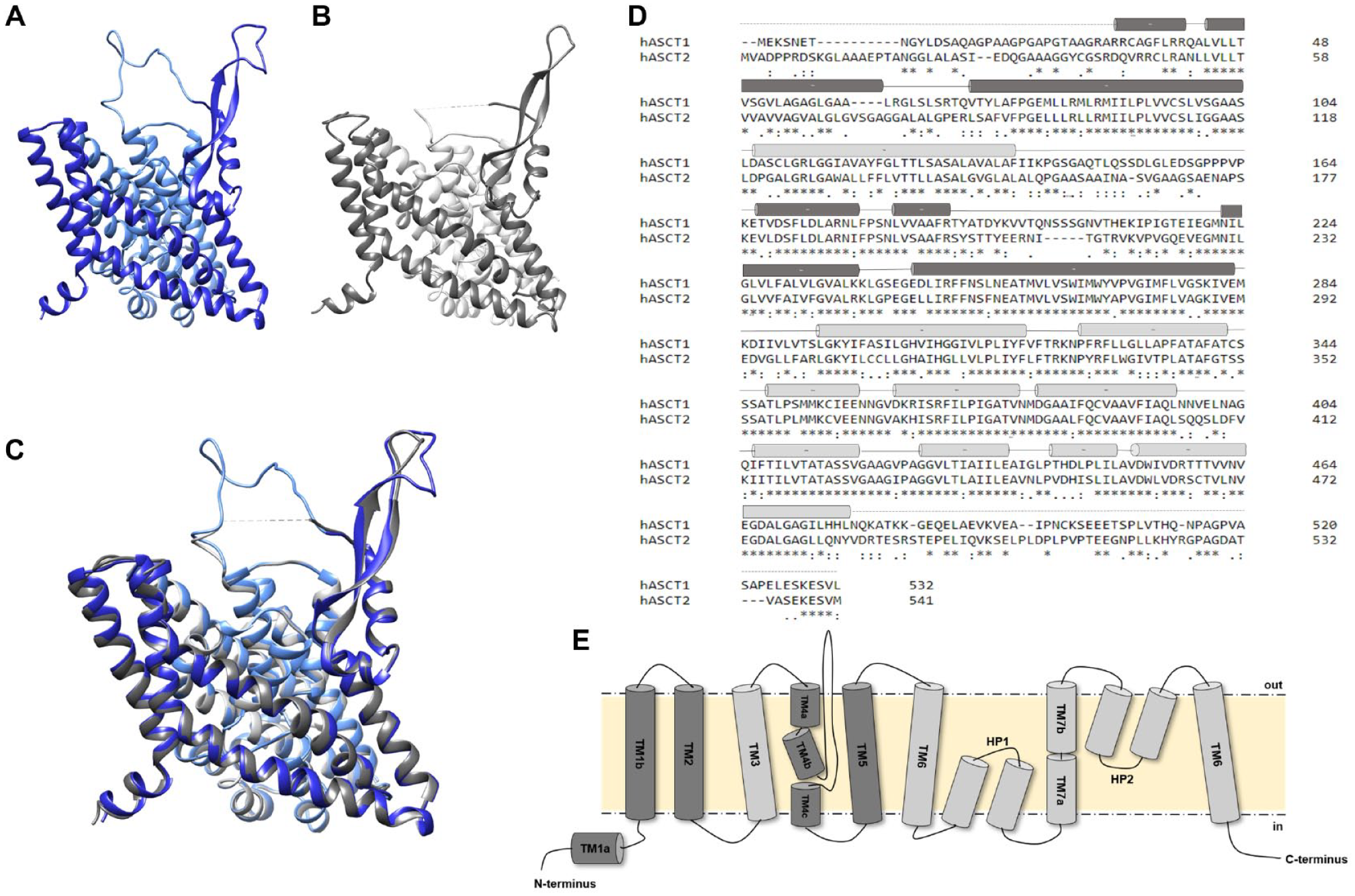
Human ASCT1 versus human ASCT2 structures. (
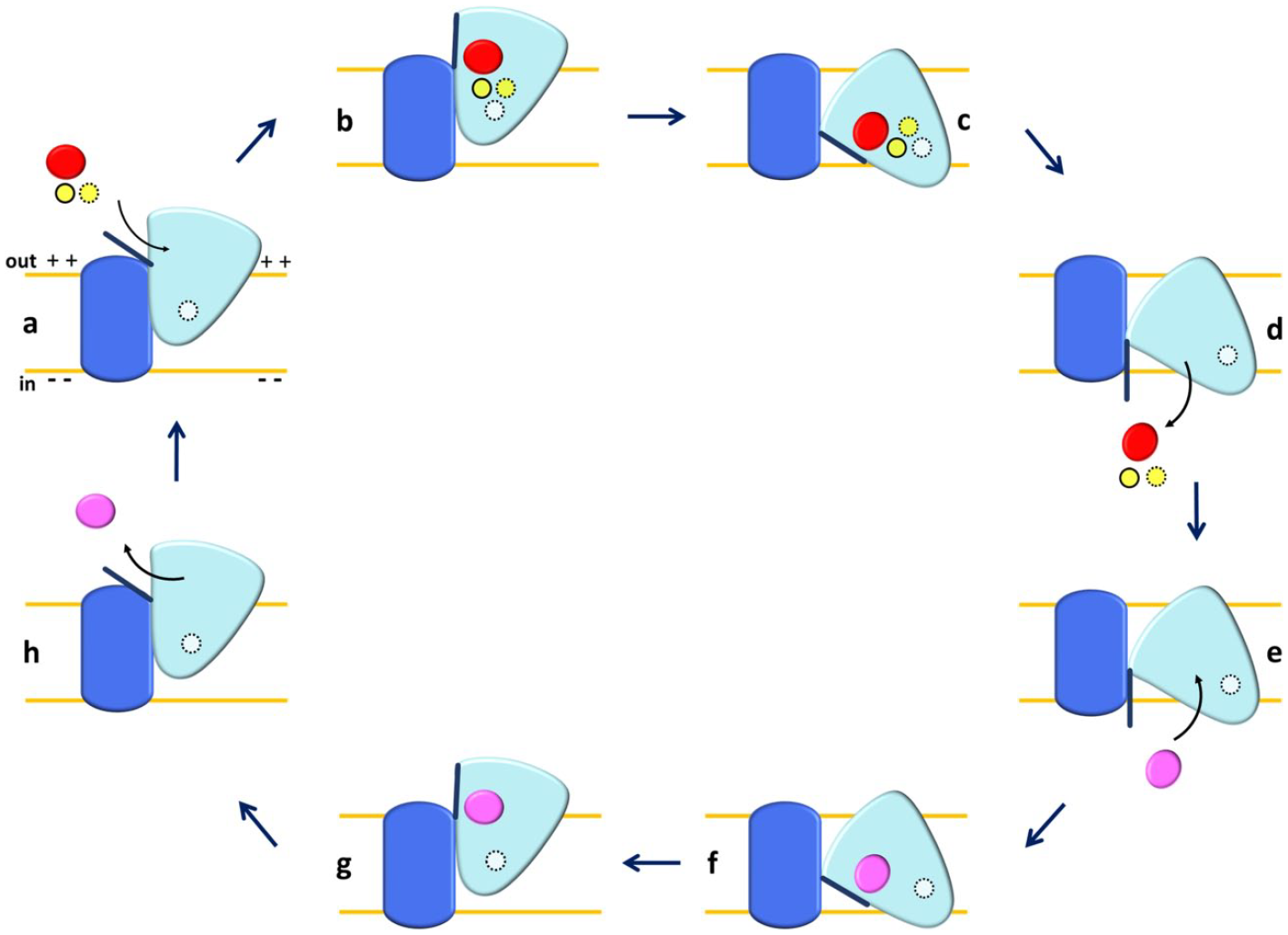
Schematic representation of the one-gate elevator mechanism in ASCT2. The scaffold domain is in dark blue, and the transport domain is in light blue. External substrate is depicted in red; internal substrate is depicted in pink; and Na+ ions are shown in a yellow solid line (Na3), yellow dotted line (Na1), and white dotted line (Na2). (
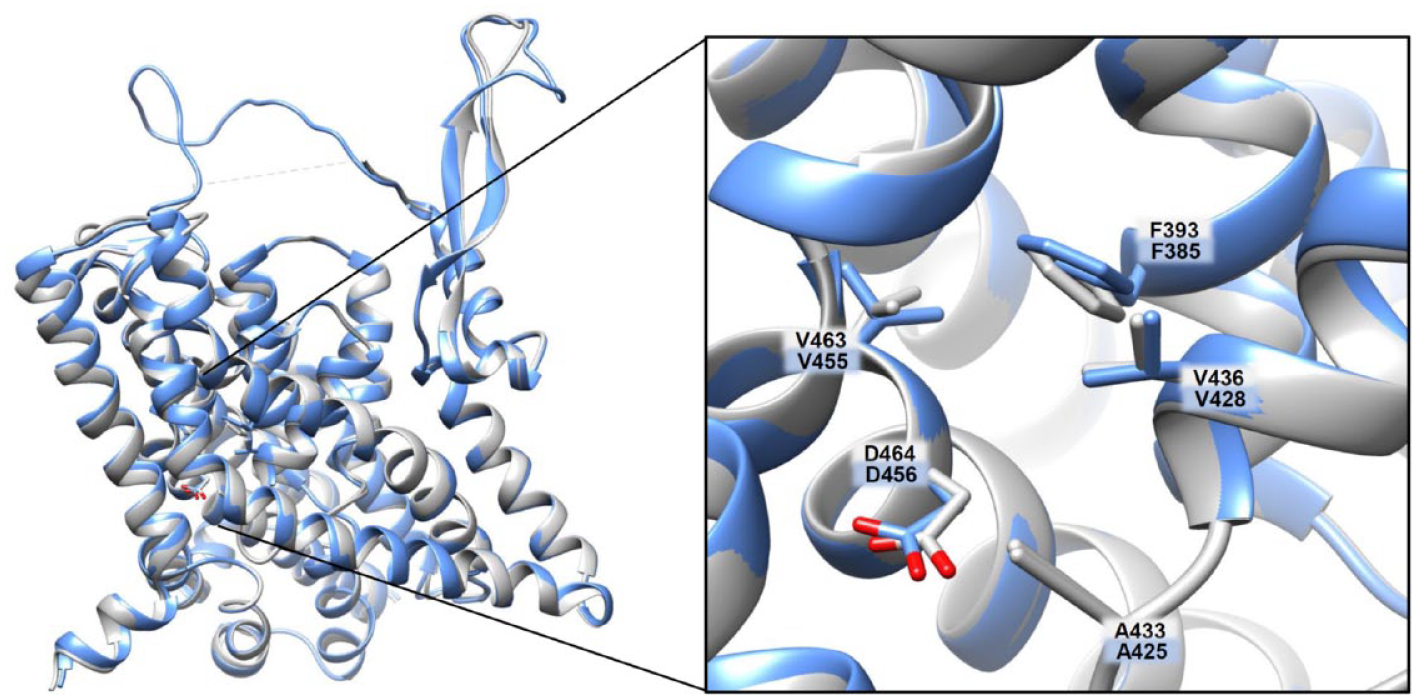
Human ASCT1 versus human ASCT2 structures: additional cavity. The homology model of ASCT1 and the 3D structure of ASCT2 have been superimposed using Clustal Omega (https://www.ebi.ac.uk/Tools/msa/clustalo). In the zoomed box, residues belonging to the additional cavity were compared between ASCT1 and ASCT2 and indicated as labeled stick. In blue are those of ASCT1, and in gray are those of ASCT2.
Substrate Binding Site
Identification of ASCT1 and ASCT2 substrate binding sites has been attempted over the years by employing combined approaches of homology modeling, molecular docking, and site-directed mutagenesis.2,31,33 These studies highlighted the crucial role of a single amino acid residue that, very intriguingly, determines the differences existing between EAAT and ASCT groups,2,21,31,33,34 namely, T459 in ASCT1 and C467 in ASCT2 (
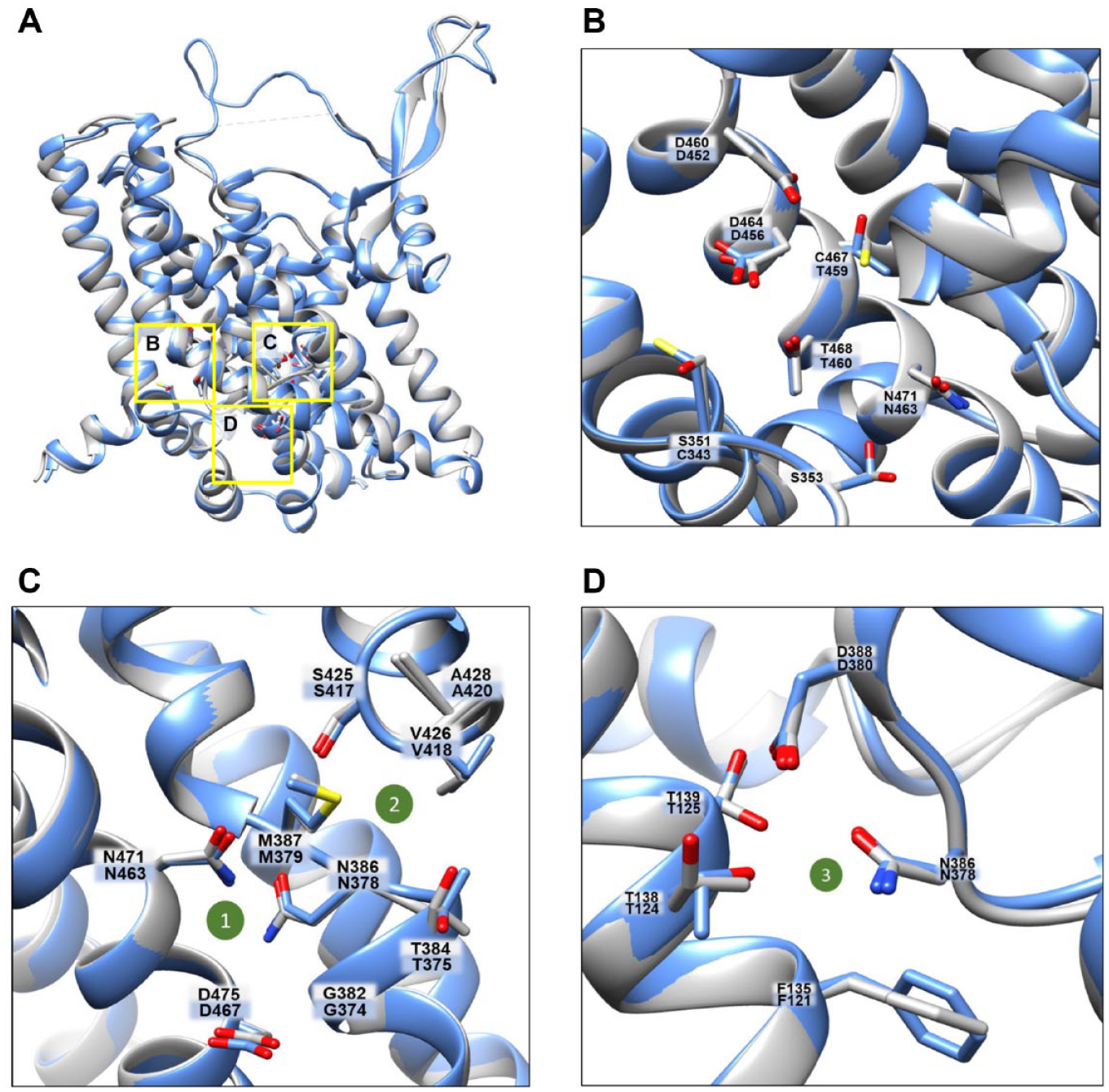
Human ASCT1 versus human ASCT2 structures: substrate binding site and sodium binding sites. (
Regarding studies on ASCT1 characterization, kinetics has been evaluated only for the external side, due to technical limitations of the intact cell model; Km values are in the micromolar range.13,61 Different from ASCT2, ASCT1 also recognizes
Sodium Binding Site
The classification of SLC1 family members in EAATs and ASCTs, besides the substrate specificity, is related to the different specificities for inorganic ions. The transport cycle of EAATs is based on the co-transport of a negatively charged amino acid with Na+ and H+ from the extracellular side and K+ from the intracellular side,1–3,64 while ASCTs were shown to obey an amino acid antiport cycle in which the exchange of neutral amino acids between the extracellular and intracellular sides is coupled to the co-transport of Na+ ions from the extracellular side with no involvement of K+ and H+.1,2 In line with this, site-directed mutagenesis on EAATs identified a Glu residue, not present in ASCTs, responsible for H+ coordination during the transport cycle and hence for pH dependence.
65
However, as above outlined, a proton can participate in the ASCT2-catalyzed reaction in the case of glutamate transport, probably coordinating other residues of the substrate binding site.
26
Over the years, the molecular basis of ions’ dependence of SLC1 family members has been investigated with site-directed mutagenesis coupled to functional analyses.1–3,31,64 When 3D structures of the bacterial ortholog GltPh became available,29,37 structural biology studies added further details on the interaction of EAATs and ASCTs with ions. In particular, a 3D structure of GltPh obtained in 200429 and then the EAAT structure in 201734 allowed for identifying two sodium binding sites in EAATs that are conserved in ASCTs (
A corollary to the actual role of sodium ions involved in the transport cycle from the extracellular and/or intracellular side is the electrogenicity of the entire process, that is, the net accumulation of positive charges linked to sodium entry. This issue has been overlooked for a long time for both EAATs and ASCTs, also due to controversial results collected with the bacterial GltPh that was shown to have a low electrogenicity.
70
Concerning ASCTs, the dependence on membrane potential has been observed only on ASCT2 with different and opposite results; indeed, using rat and murine ASCT2, an electroneutral exchange of amino acids has been postulated simultaneous to an electroneutral exchange of external Na+ with internal Na+.43,68 Then, a voltage-dependent electroneutral exchange was proposed for rat ASCT2, implying an electrogenic binding of Na+/amino acid on both sides of the membrane, resulting in a net electroneutral bidirectional transport;
69
since that study, however, no further analyses have been performed for defining this mechanism in entire cell systems due to the lack of a methodology allowing for direct measurement of the Na+ flux. Later on, using the proteoliposome tool, it has been shown that the transport cycle (Na+ex-Glnex/Glnin) catalyzed by the human ASCT2 is stimulated by membrane potential generated as a K+ diffusion potential in the presence of valinomycin. This correlates with an inwardly directed net flux of Na+.
50
Interestingly, Na+ is also needed in the intraliposomal compartment (intracellular side), but at a concentration much lower than the external one, corresponding to the physiological intracellular Na+ concentration. This effect is based on the allosteric regulation of ASCT2, not on Na+ transport from the intracellular side.
50
These data may also fit with a model in which three sodium ions bind to the protein, but not all of them are transported (
Anion Channel Conductance
One of the most intriguing aspects still lacking a definitive molecular explanation is the presence of an anion conductance in ASCT1 and ASCT2, similar to that observed in the other members of the SLC1 family.2,6,7 According to this property, the SLC1 family members reveal a unique double behavior of transporter and channel. The presence of these activities is acknowledged as a hallmark of the SLC1 family so that the permeation to anions has been used for both ASCT1 and ASCT2 as an indirect measure of the transport activity using patch-clamp methodology.1,3,47,68 The anion current, associated with the Na+-dependent uptake of neutral or negatively charged amino acids, showed the following preference: SCN– >> NO3– > I– > Cl– > F– >> gluconate.7,71 The anion conductance is evolutionary old, as proven by its presence in bacterial orthologs, such as in GltPh. 6 For the EAATs, this phenomenon seems to be linked to regulation of synaptic transmission and excitability with relevance to human pathologies such as ataxia and epilepsy, 72 opening important perspectives in therapeutic strategies for neurological disorders, while for the two ASCTs, the actual role of anion conductance is not clarified yet, considering that the preferred ion, SCN–, is unphysiological. 68 Based on investigations performed with GltPh, it has been assessed that the anion flux occurs at the interface between the scaffold and transport domains in a flexible area formed when the GltPh substrates (Na+ and aspartate) are bound to the transporter.6,73 Very recently, the chloride channel cavity has been solved by cryo-EM in an open-channel conformation, giving information on the other SLC1 family members. 74 It is worth noting that in ASCT1, different from the EAATs, the anion conductance is retained in all the mutants of the three sodium binding sites, although the amplitude of some substrate-activated currents is reduced compared to the wild type. 61
Regulatory Aspects
One of the still undeveloped aspects concerning transporter studies is the regulation of their function/stability through posttranslational modifications and interactions with other proteins and regulatory factors. The great importance of this area of investigation is testified to by the increasing number of in silico predictions and by some ex vivo experiments, even if the complete collections of post translational modifications (PTMs) and interactions for ASCT1 and ASCT2 are not deciphered yet. The state of the art on some of these aspects is given below.
PTMs: N-Glycosylation, Phosphorylation, Acetylation, and Methylation
N-glycosylation is acknowledged as the canonical pathway responsible for driving proteins to their definitive location in cell membranes.75–77 Based on sequence analysis, N-glycosylation sites are present, corresponding to N201 and N206 in ASCT1 and N163 and N212 in ASCT2 ( Fig. 6 ). The role of N-glycosylation in ASCT2 has been deeply investigated in the last decade; indeed, the two glycosylated Asn residues, identified in the 3D structure of human ASCT2, have been definitively confirmed by site-directed mutagenesis. Indeed, the substitution N163Q and N212Q of human ASCT2 prevents its localization to the plasma membrane and decreases the protein stability, 78 while the N163Q and N212Q mutants retain full intrinsic transport activity, as shown by parallel assays in intact cells and proteoliposomes demonstrating that glycosylation/deglycosylation status does not affect the capacity and specificity of ASCT2 to catalyze the physiological Na+-dependent amino acid antiport. Another study on N-glycosylation of human ASCT2 has been conducted using tunicamycin, an antibiotic that blocks the N-glycoprotein synthesis. Upon tunicamycin treatment, a link between the extent of ASCT2 N-glycosylation and glucose metabolism has been proposed, supporting the hypothesis that coordinated regulation of glucose and glutamine metabolism exists. 79 Much less is known about the role of the conserved N-glycosylation sites of human ASCT1. The only available data linked N-glycosylated ASCT1, 80 as well as ASCT2, to viral recognition at the cell membranes of a group of retroviruses named HERVs (human endogenous retrovirus). 81
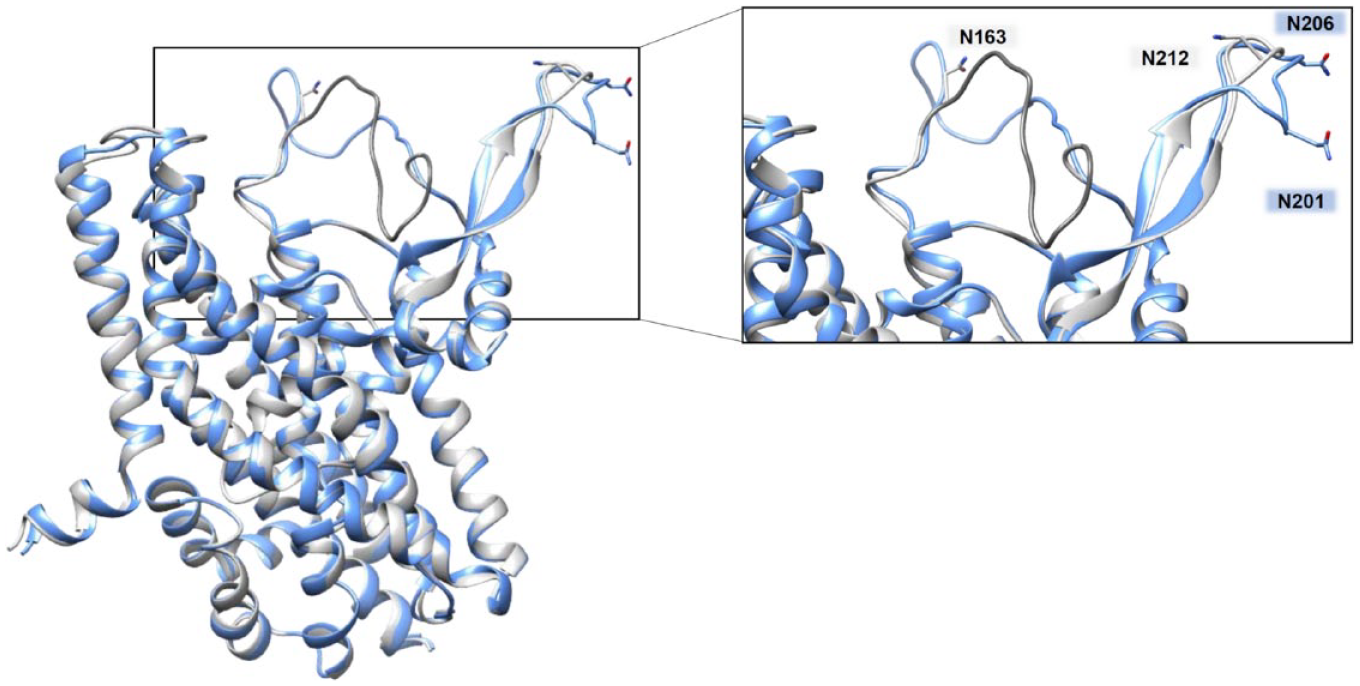
Human ASCT1 versus human ASCT2 structures: N-glycosylation sites. The homology model of ASCT1 and the 3D structure of ASCT2 have been superimposed using Clustal Omega (https://www.ebi.ac.uk/Tools/msa/clustalo). In the zoomed box, residues responsible for N-glycosylation were compared between ASCT1 and ASCT2 and are indicated as labeled sticks: in blue for those of ASCT1 and in gray for those of ASCT2.
Besides N-glycosylation, the phosphorylation of serine, threonine, and tyrosine residues is one of the most common PTMs linked to the regulation of protein functions being either an activation or inhibition message. 82 Proteomic analyses performed using different databases such as Phosphositeplus and iPTMnet showed that both ASCT1 and ASCT2 are subjected to phosphorylation at multiple sites ( Table 1 ). Concerning ASCT2, an old paper showed that phosphorylation of ASCT2 via SGK1, SGK3, and PKB kinase, activates ASCT2 in Xenopus laevis oocytes. 83 Moreover, it has been recently described that the extent of ASCT2 phosphorylation at position S503 decreased in a colon adenocarcinoma cell line after treatment with growth inhibitors. 84 Regarding ASCT1, a correlation between the increased levels of phosphorylated threonine and the decreased affinity of ASCT1 has been suggested as a result of leukotriene D4 (LTD4), an inflammatory mediator present in chronically inflamed intestine. 85 Moreover, the bioinformatics resource iPTMnet reports that ASCT1 is subjected to lysine ubiquitination and that ASCT2 is subjected to lysine ubiquitination, acetylation, sumoylation, and arginine methylation ( Table 1 ). Interestingly, iPTMnet includes the list of some ASCT1 and ASCT2 residues, subjected to PTMs, that are mutated in some human cancers. 86 For instance, the S502 of ASCT1 has been found mutated to Cys in breast cancer and the Ser535 of ASCT2 has been found mutated to Phe in melanoma. 86 Moreover, the methylation site R525 and the acetylation site K537 of ASCT2 are mutated in uterine cancer. 86
Post Translational Modifications of hASCT1 and hASCT2.

Protein–Protein Interactions and Cellular Effectors
Little detailed information is available regarding the regulation of ASCT1 and ASCT2 transport activity by interactions with other cellular proteins. Ubiquitin reconstruction proximity label–MS, affinity capture–MS, and the two-hybrid assay described some interactions of ASCT1 with cell proteins (https://thebiogrid.org/112400; https://www.ebi.ac.uk/intact/interactors/id:P43007*; https://mint.bio.uniroma2.it/index.php/results-interactions/?id=P43007; https://string-db.org/network/9606.ENSP00000234256). A few examples are the interactions of ASCT1 with (1) G-protein-coupled receptors, one of the most important protein classes involved in cell signaling in physiological and pathological conditions, 87 and (2) Nek 4, one of the largest members of the serine–threonine kinase Nek family, related to the primary cilia formation and in DNA damage response. 88 Moreover, in the mentioned databases, interactors of ASCT1 have been suggested, among other amino acid transporters related to brain homeostasis, such as SLC7A5, SLC7A11, and SLC38A2 (https://string-db.org/network/9606.ENSP00000234256). Regarding ASCT2, in silico predictions as well as immunoprecipitation or double-hybrid technologies described several interactions of ASCT2 with cell proteins (https://thebiogrid.org/112401; https://string-db.org/network/9606.ENSP00000444408; https://string-db.org/network/9606.ENSP00000444408; https://mint.bio.uniroma2.it/index.php/results-interactions/?id=slc1a5; https://www.ebi.ac.uk/intact/interactors/id:Q15758*). As an example, ASCT2 has been shown to interact with CD147/MCT1 and CD98/LAT1, forming a super-complex able to respond to metabolic changes. 89 Furthermore, ASCT2 physically interacts with PDZK1 and SNX27, two proteins containing a PDZ domain, one of the most common domains in the human genome.50,90–92 Recently, the serotonin transporter (SERT) has been shown to physically interact with ASCT2, stimulating the uptake of serotonin in cells and thus contributing to the homeostasis of neurons. 93 Furthermore, physical interaction of ASCT2 with EGFR in cancer has been described as a consequence of EGF-mediated regulation of ASCT2. 94
Regulation by Physiological Molecules
The regulation of ASCT1 and ASCT2 by endogenous effectors is still at the initial stage of the investigation. Human ASCT2 harbors in its primary structure eight Cys residues that have been substituted by alanine to evaluate their role in the sensitivity of ASCT2 to reducing and oxidizing reagents. Interestingly, the C467, crucial for substrate recognition, is also responsible for an ON/OFF regulation of ASCT2 by physiological cysteine-targeting reagents triggering redox responsiveness. In particular, it has been proposed that the C467 residue could be involved in the formation of a disulfide bridge with the vicinal C308 or C309 triggering the switch between an active (SH state) to an inactive (S–S state) protein.
33
The regulation of ASCT1 is much less studied, and an inhibitory effect by the potent oxidant peroxynitrite has been reported in chronic inflammation even though the molecular mechanism is still not clarified.
95
It is important to highlight that even if ASCT1 harbors seven Cys residues, only three are homologous with ASCT2 (
Interaction with Drugs
The involvement of the two proteins in human diseases together with the availability of the 3D structure of ASCT2 and the homology model of ASCT1 opened important perspectives in the field of drug design. Notwithstanding, the International Transporter Consortium has so far not included ASCT1 and ASCT2 as relevant for drug–transporter interactions.
97
It has to be stressed that almost no studies are available for ASCT1-targeting drugs, while a lot of effort has been made in the last 15 years to design novel molecules as drugs targeting ASCT2 transport activity. In particular, these studies have been conducted on both murine and human isoforms (
Fig. 7
). The first attempt, realized in a pre-3D era, was performed using substrate-mimicking molecules on the rat isoform identifying two molecules, benzylcysteine (
Fig. 7A
) and benzylserine (
Fig. 7B
), as competitive inhibitors able to block ASCT2 transport activity, measured as glutamine-induced anion currents, even if at high concentrations.
98
This study was conceived as a “proof of principle” to demonstrate that the competitive inhibitors are able to selectively block amino acid transport. Then, glutamine (
Fig. 7C
)
99
and serine (
Fig. 7D
)
100
structures were used as a scaffold for other substrate-like compounds, either substrates or inhibitors. In these cases, the efficacy of inhibitors has been tested as a block of anion currents as well. The serine analogs were revealed to be threefold stronger than glutamine analogs. More recently, other molecules based on the proline scaffold have been designed, able to activate or inhibit rat ASCT2 transport activity in intact cells.32,101 These findings are very intriguing because proline, per se, is not a substrate of ASCT2. Notwithstanding such a premise, the cis-3-hydroxyproline (
Fig. 7E
) revealed a small activator of ASCT2, while the proline fluorobenzyl substituent (g-FBP) was found to be a potent inhibitor with cytotoxic effects (
Fig. 7F
). Very interestingly, despite the differences existing between rat and human isoforms, these molecules were also active on the human melanoma cell line.
101
These observations triggered the development of drugs based on the scaffold of proline derivatives. Recently, a competitive inhibitor of human ASCT2 has been designed, namely, V-9302 (
Fig. 7F
), moving from a screening conducted on 2-amino-4-bis (aryloxybenzyl)aminobutanoic acid analogs.
102
The V-9302 reached a preclinical phase of investigation due to its ability in inhibiting ASCT2 transport in vitro and in mice harboring a patient-derived xenograft (PDX) tumor.
103
Besides the competitive inhibitors, an alternative strategy has been employed, using the rat isoform, for designing and testing covalent inhibitors able to irreversibly block ASCT2 transport activity, identifying six compounds with dithiazole (
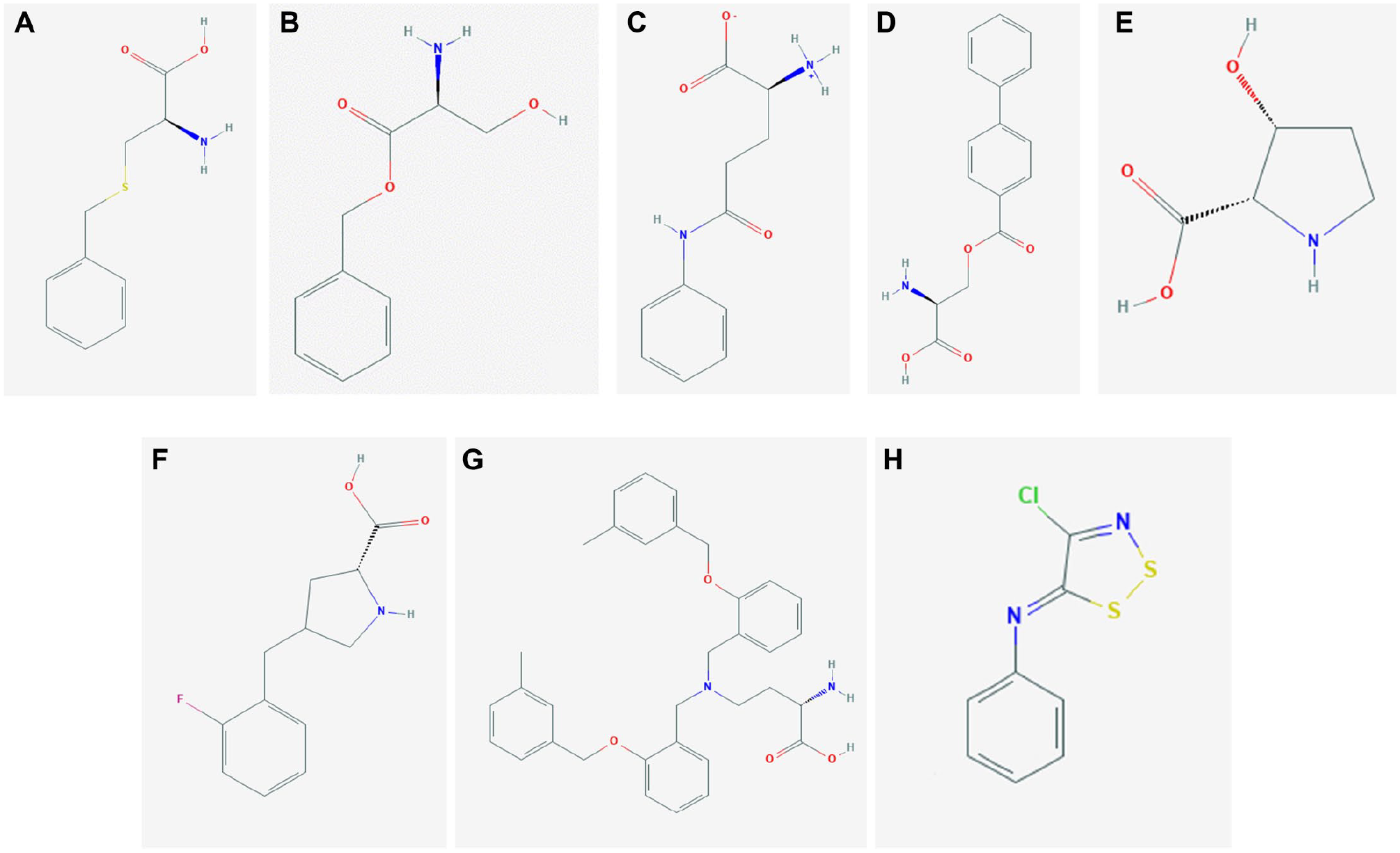
Conclusions
The proteins forming the ASCT group are intriguing players of cell biology due to their ability to mediate a Na+-dependent flux of amino acids in cells, despite their antiport mechanism (
Altogether, the available data on ASCT1 and ASCT2 may help to answer the question raised in the title: notwithstanding the structural similarities existing between ASCT1 and ASCT2, the actual differences in terms of substrate specificities and physiological roles suggest that the two proteins may be considered cousins rather than brother and sister.
Supplemental Material
sj-pdf-1-jbx-10.1177_24725552211030288 – Supplemental material for ASCT1 and ASCT2: Brother and Sister?
Supplemental material, sj-pdf-1-jbx-10.1177_24725552211030288 for ASCT1 and ASCT2: Brother and Sister? by Mariafrancesca Scalise, Lara Console, Jessica Cosco, Lorena Pochini, Michele Galluccio and Cesare Indiveri in SLAS Discovery
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by PRIN (Progetti di Ricerca di Interesse Nazionale) project no. 2017PAB8EM to CI granted by MIUR (Ministry of Education, University and Research) – Italy.
Supplemental Material
Supplemental material is available online with this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
