Abstract
Human induced pluripotent stem cell–derived cardiomyocytes (hiPSC-CMs) have been widely used for the assessment of drug proarrhythmic potential through multielectrode array (MEA). HiPSC-CM cultures beat spontaneously with a wide range of frequencies, however, which could affect drug-induced changes in repolarization. Pacing hiPSC-CMs at a physiological heart rate more closely resembles the state of in vivo ventricular myocytes and permits the standardization of test conditions to improve consistency. In this study, we systematically investigated the time window of stable ion currents in high-purity hiPSC-derived ventricular cardiomyocytes (hiPSC-vCMs) and confirmed that these cells could be used to correctly predict the proarrhythmic risk of Comprehensive In Vitro Proarrhythmia Assay (CiPA) reference compounds. To evaluate drug proarrhythmic potentials at a physiological beating rate, we used a MEA to electrically pace hiPSC-vCMs, and we recorded regular field potential waveforms in hiPSC-vCMs treated with DMSO and 10 CiPA reference drugs. Prolongation of field potential duration was detected in cells after exposure to high- and intermediate-risk drugs; in addition, drug-induced arrhythmia-like events were observed. The results of this study provide a simple and feasible method to investigate drug proarrhythmic potentials in hiPSC-CMs at a physiological beating rate.
Keywords
Introduction
The Comprehensive In Vitro Proarrhythmia Assay (CiPA) initiative proposes use of human stem cell–derived cardiomyocytes to validate the proarrhythmic risks [e.g., torsades de pointes (TdP)] of new chemical entities. 1 Although CiPA validation results for reference compounds in spontaneously beating human induced pluripotent stem cell–derived cardiomyocytes (hiPSC-CMs) are promising, 2 the purity and spontaneous beating of ventricular cardiomyocytes, two important aspects of hiPSC-CMs with regard to drug cardiac safety assessment, should be considered. Typically, differentiated hiPSC-CMs comprise mixtures of ventricular-like (approximately 60–70%), atrial-like, and nodal-like cardiomyocytes, which have different electrophysiological characteristics3,4 that may cause variable results of drug effects on human pluripotent stem cell–derived cardiomyocyte (hPSC-CM) preparations. HiPSC-CM cultures spontaneously beat at various rates influenced by many factors, such as different differentiation protocols and cell sources,3–7 whereas in vivo adult ventricular myocardium beats under the control of the sinoatrial node. Beating rate is an important determinant of the field potential duration (FPD) of hiPSC-CMs, and different beating rates can affect both FPD and changes in drug-induced repolarization duration, as measured with a multielectrode array (MEA) system. 8 Examining the effects of drugs on hiPSC-CMs in conditions similar to the in vivo environment is expected to eliminate variations in testing caused by discrepancies of beating rates. Compared with optical stimulation, 8 electrical pacing is simpler and does not require transfection of light-sensitive protein (e.g., channel rhodopsin). The “T” waves in field potential (FP) signals corresponding to the ventricular repolarization in electrocardiogram, however, are too tiny to be detected under electrical pacing, which makes its application in assessment of drug proarrhythmic potential difficult. 9
Previously, we obtained highly homogeneous (>90%) hiPSC-derived ventricular cardiomyocytes (hiPSC-vCMs) using a direct ventricular differentiation protocol with chemically defined and albumin-free conditions. 7 Without fetal bovine serum or large amounts of albumin in the media, the efficiency and yield of cardiac differentiation were more consistent between batches. Due to the high purity of ventricular cardiomyocytes, these cells could be a good tool for cardiac safety evaluation. In addition, without the impact of the unknown component in the serum or albumin, the differentiation process can be further improved to better accommodate cardiac safety evaluation needs.
In this study, we systematically investigated the time window of stable ion currents of differentiated hiPSC-vCMs, and demonstrated that hiPSC-vCMs correctly predicted the proarrhythmic risk of CiPA reference compounds. Although these cells exhibited spontaneous beating, the beating rate was so slow that electrical pacing with a physiological rate (1~1.2 Hz) was feasible. Thus, we established a simple electrical pacing method that can be used for the assessment of drug proarrhythmic risk.
Materials and Methods
Cell Source
HiPSC-vCMs were differentiated according to a previously reported method.
7
The hiPSC-vCMs at day 30 after cardiac induction were cryopreserved as Cauliscell hiPSC-vCMs (cat. no. 300106; Cauliscell Company, Nanjing, China). The purity of cardiomyocytes was 97.3 ± 0.8% (
Manual Patch Clamp Recording
At the designated time points, beating colonies were dissociated into single cells according to a previously reported protocol. 10 Spontaneous action potentials (APs) and ionic currents were recorded at 35 ± 2 °C except for calcium current (ICa) measurements, which were performed at room temperature. Action potential, sodium current (INa), ICa, and E-4031-sensitive potassium current (IKr) recordings were carried out on single hiPSC-vCMs, as reported previously. 7 Perforated patch recording was used for pharmacological examinations of stimulated APs. 7
MEA Recordings
Cell culture
The cryopreserved hiPSC-vCMs were used in this study. After thawing, hiPSC-vCMs were plated into human recombinant vitronectin-coated six-well plates in 2 mL of cardiomyocyte plating medium at a density of 1 × 106 viable cells/mL. Twenty-four hours post thaw, the medium was replaced with cardiomyocyte maintenance medium and changed every 2 days. Five to seven days post thaw, the cardiomyocytes were digested, and 5–6 µL of cell suspension was plated in fibronectin-coated MEA 48-well plates (CytoView MEA 48; Axion BioSystems, Atlanta, GA) at a density of 5–6 × 104 cells per well. MEA recordings were performed 10–14 days post thaw at 37 °C. RPMI-1640 plus B27 supplement (cat. no. 17504044; Thermo Fisher Scientific, Waltham, MA) was used 2 h prior to recordings of acute drug effects.
Compound preparation
Thirteen CiPA reference drugs were chosen and purchased from Sigma-Aldrich (St. Louis, MO) or Toronto Research Chemical Inc. (North York, Canada), and five concentrations of each drug were examined (
Experimental protocol for spontaneously beating hiPSC-vCMs
The Maestro multiwell electrophysiology platform (Axion BioSystems) was used in this study. For each drug, five concentrations were added sequentially with 20 min of recording time for each concentration. An algorithm was used to identify the 30 most stable beats within the last 5 min of the 20-min recording, and these beats were used to measure FPD or Fridericia-corrected field potential duration (FPDc), spike amplitude (AMP), and beating period (BP) with the CiPA Analysis Tool software. The occurrence of arrhythmia-like events was marked manually through the whole 20-min recording. For clearer recognition of arrhythmia-like events, the Local Extracellular Action Potential (LEAP) assay, a functional module in the Maestro MEA system that allows acquisition of extracellular AP waveforms, was used alternatively after the 20-min FP recordings.
Experimental protocol for electrical pacing of hiPSC-vCMs
For electrical pacing, one electrode from each test well was selected, and a train of biphasic electrical stimulations (400 mV, 1 ms) was applied to the cells from low to high rates (0.5 Hz to 1.2 Hz). Three minutes of recording time were analyzed for each frequency.
Statistics
Results from patch clamp recording are presented as mean ± standard error, while other results are presented as mean ± standard deviation. Student t-test (two-tail) or one-way ANOVA with Scheffe’s test was used for statistical comparison between two or multiple groups with the software Origin 8 or GraphPad Prism 7. Statistical significance was assumed when p < 0.05.
Results
Developmental Changes in Electrophysiological Characteristics of hiPSC-vCMs
Because ion channel expression undergoes developmental changes during the differentiation of hiPSC-vCMs, it is important to investigate the time window during which their ion channels activities are stable. We first measured APs, INa, ICa, and E-4031-sensitive currents, which are most closely related to drug cardiac safety assessment,
11
at different time points during the course of 100 days. Typical APs of hiPSC-vCMs and statistical results of hiPSC-vCMs for days 15–100 are shown in
Figure 1A
. The percentage of cardiomyocytes exhibiting ventricular-like APs was greater than 90% after day 20 (
Fig. 1A
). Statistical results of APs, INa, ICa, and E-4031-sensitive currents revealed that ventricular APs and densities of ion channel currents in hiPSC-vCMs were stable after 30 days of differentiation (
Fig. 1A–1E
). At this stage, AP recordings showed that hiPSC-vCMs responded as we expected to tetrodotoxin (TTX), nifedipine, and E-4031, which are specific inhibitors of INa, ICa, and E-4031-sensitive currents, respectively. In addition, early afterdepolarization was observed after exposure to 100 nM E-40317 (
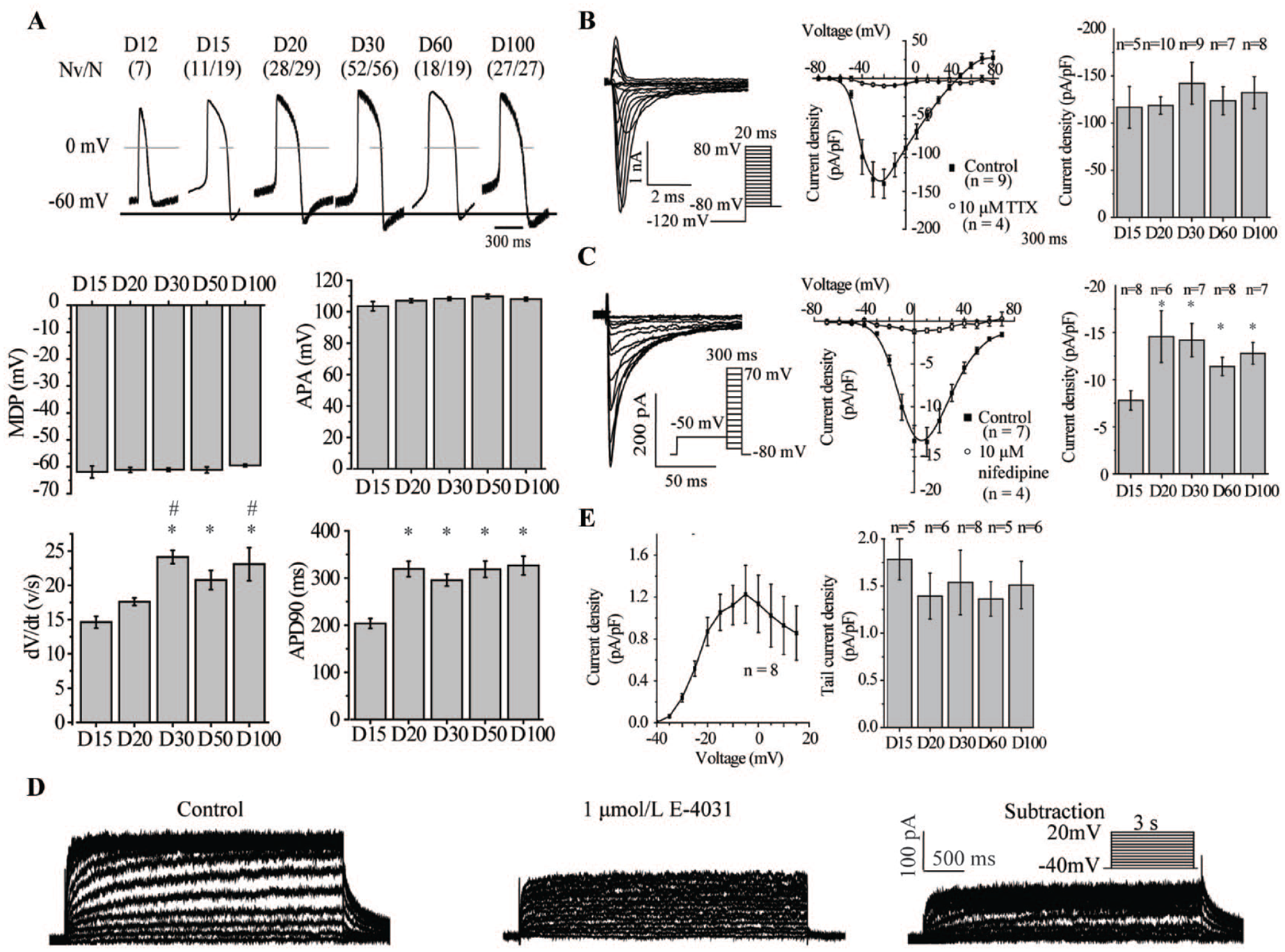
Determination of suitable hiPSC-vCM age for drug assessment. (
Drug Proarrhythmic Risk Analyses with Spontaneously Beating hiPSC-vCMs
To evaluate the drug proarrhythmic predictability of hiPSC-vCMs, reference validation drugs were tested using MEA with the CiPA paradigm. Thirteen CiPA validation drugs were selected, including four in the high TdP-risk category (quinidine, dofetilide, D,L-sotalol, and bepridil), four in the intermediate TdP-risk category (ondansetron, cisapride, terfenadine, and chlorpromazine), and five in the low TdP-risk category (ranolazine, mexiletine, diltiazem, nifedipine, and verapamil). The study outline is shown in
Figure 2A
. Representative FP signals and the four endpoints of FP (BP, FPD, FPDc, and AMP) are shown in
Figure 2B
. The spontaneous activities of hiPSC-vCMs are listed in
Table 1
. DMSO (≤ 0.1%, v/v) had no effect on the four FP endpoints, and hiPSC-vCMs properly responded to mexiletine, nifedipine, and dofetilide, which are specific inhibitors of sodium, calcium, and hERG channels, respectively (
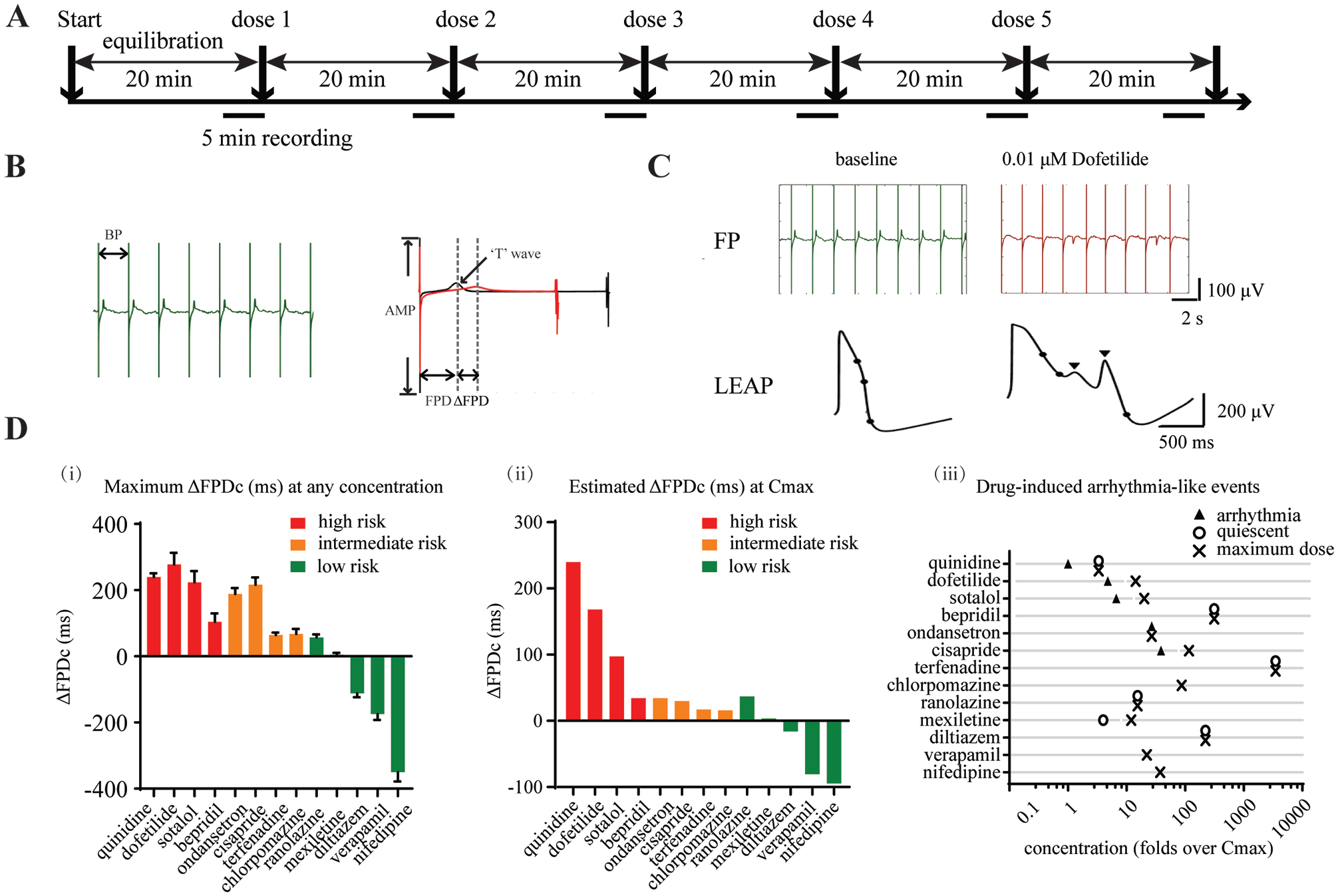
Proarrhythmic risk analysis of spontaneously beating hiPSC-vCMs cultured for 30 days. (
Average Electrophysiological Parameters of Spontaneously Beating hiPSC-vCMs Obtained Using Multielectrode Array.

AMP, Spike amplitude; BP, beat period; FPD, field potential duration; FPDc, Fridericia-corrected field potential duration; hiPSC-vCMs, human induced pluripotent stem cell–derived ventricular cardiomyocytes.
Summary of Effects of 13 Drugs on Spontaneously Beating hiPSC-vCMs.
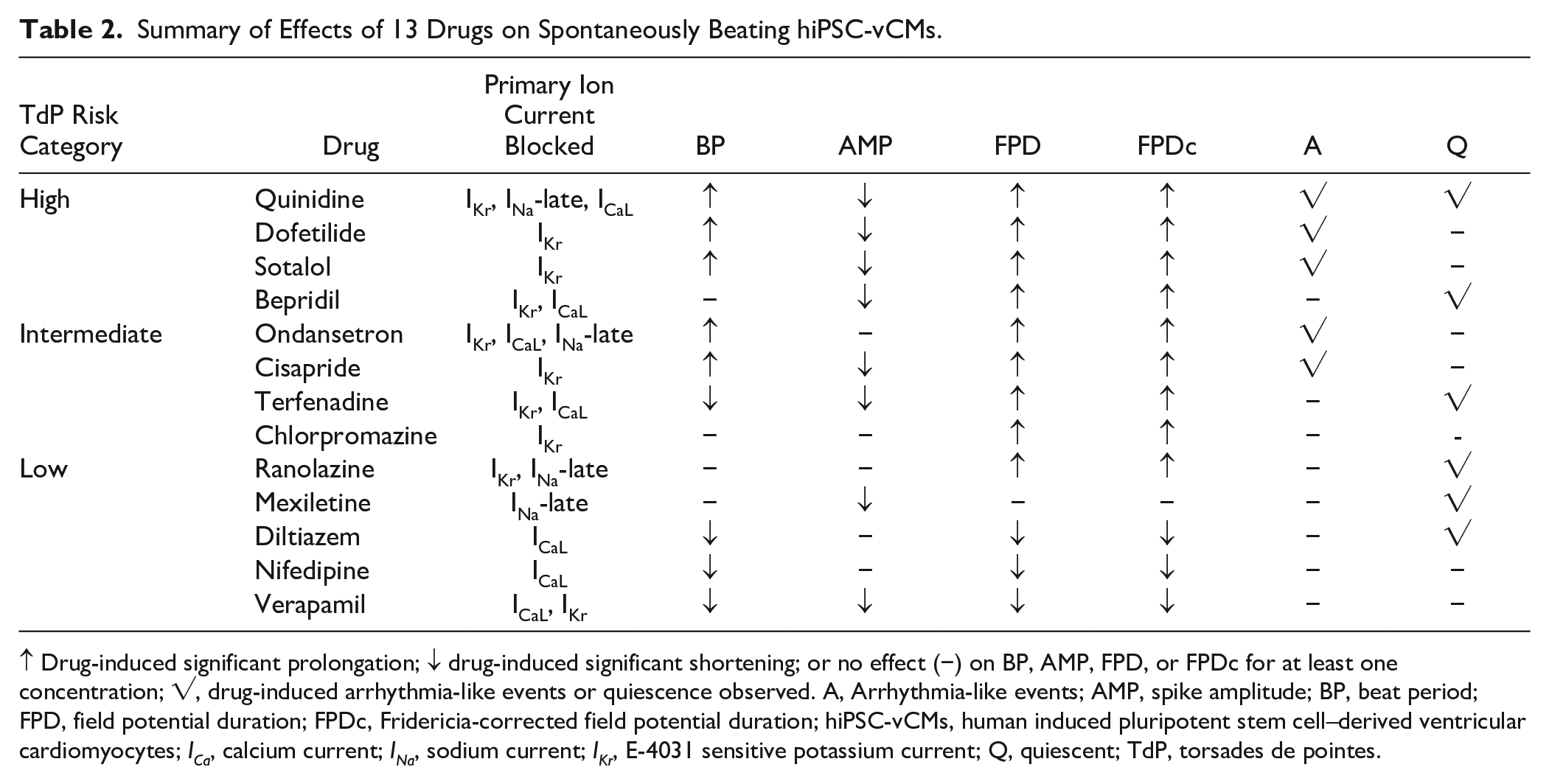
↑ Drug-induced significant prolongation; ↓ drug-induced significant shortening; or no effect (−) on BP, AMP, FPD, or FPDc for at least one concentration; √, drug-induced arrhythmia-like events or quiescence observed. A, Arrhythmia-like events; AMP, spike amplitude; BP, beat period; FPD, field potential duration; FPDc, Fridericia-corrected field potential duration; hiPSC-vCMs, human induced pluripotent stem cell–derived ventricular cardiomyocytes; ICa, calcium current; INa, sodium current; IKr, E-4031 sensitive potassium current; Q, quiescent; TdP, torsades de pointes.
Drug-Induced Prolongation of Repolarization in Electrically Paced hiPSC-vCMs
To investigate the proarrhythmic potential of drugs at a physiological heart rate (1~1.2 Hz), we explored the possibility of pacing hiPSC-vCMs with electrical stimuli and focused on drug-induced prolongation of repolarization and arrhythmia-like events.8,12 Ten drugs, including eight high and intermediate TdP-risk drugs and two low-risk drugs, were tested with electrically paced hiPSC-vCMs at 1 and 1.2 Hz. As shown in
Figure 3A
, electrical pulses (400 mV for 1 ms) successfully paced hiPSC-vCMs beating at 1~1.2 Hz. When the electrical stimulus was imposed on hiPSC-vCMs, “T” waves were obviously observed (
Fig. 3B
). Moreover, “T” waves were also clearly discriminated when the FPD was prolonged after exposure to dofetilide (
Fig. 3C
). Pacing increased AMP and decreased the FPD of FPs at 1 Hz and 1.2 Hz (
Fig. 3D
). Under 1-Hz and 1.2-Hz pacing, FPD prolongations in hiPSC-vCMs were detected in response to eight high- or intermediate-risk drugs (dofetilide, sotalol, quinidine, bepridil, ondansetron, cisapride, terfenadine, and chlorpromazine), exposure at the concentration that induced obvious FPD prolongation in spontaneously beating hiPSC-vCMs (
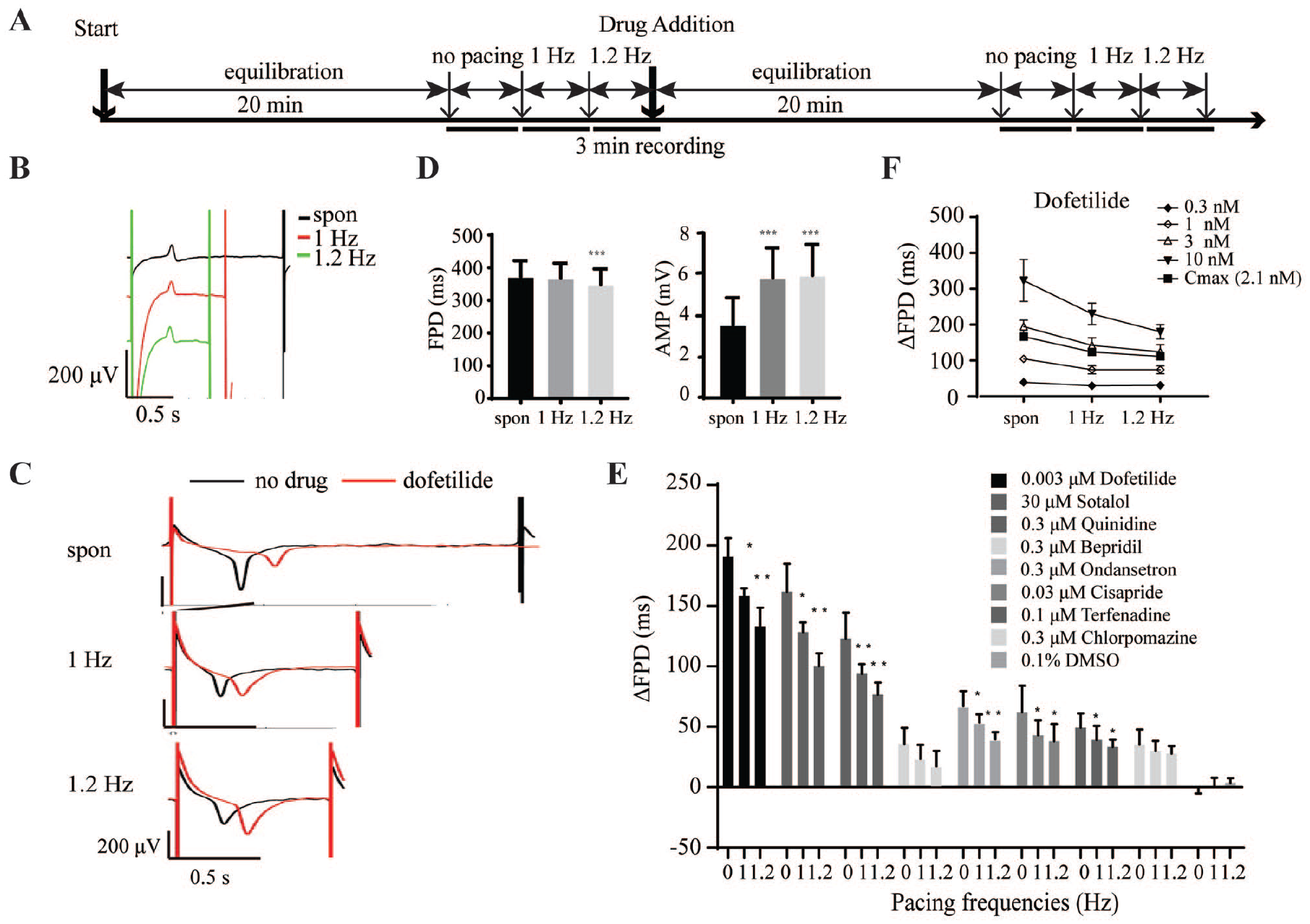
Application of MEA assay for drug testing of electrically paced hiPSC-vCMs. (
Drug-Induced Arrhythmic Events in Electrically Paced hiPSC-vCMs
To further explore the application of our MEA assay with electrically paced hiPSC-vCMs for detecting drug-induced arrhythmic events, and reveal the relationship between pacing rate and arrhythmia occurrence, 0.5–1.2 Hz pacing rates were applied to hiPSC-vCMs exposed to drug concentrations that induced arrhythmic events in spontaneously beating hiPSC-vCMs (
Fig. 4A
). For this study, we chose 0.01 µM dofetilide, 300 µM sotalol, and 0.1 µM cisapride. As shown in
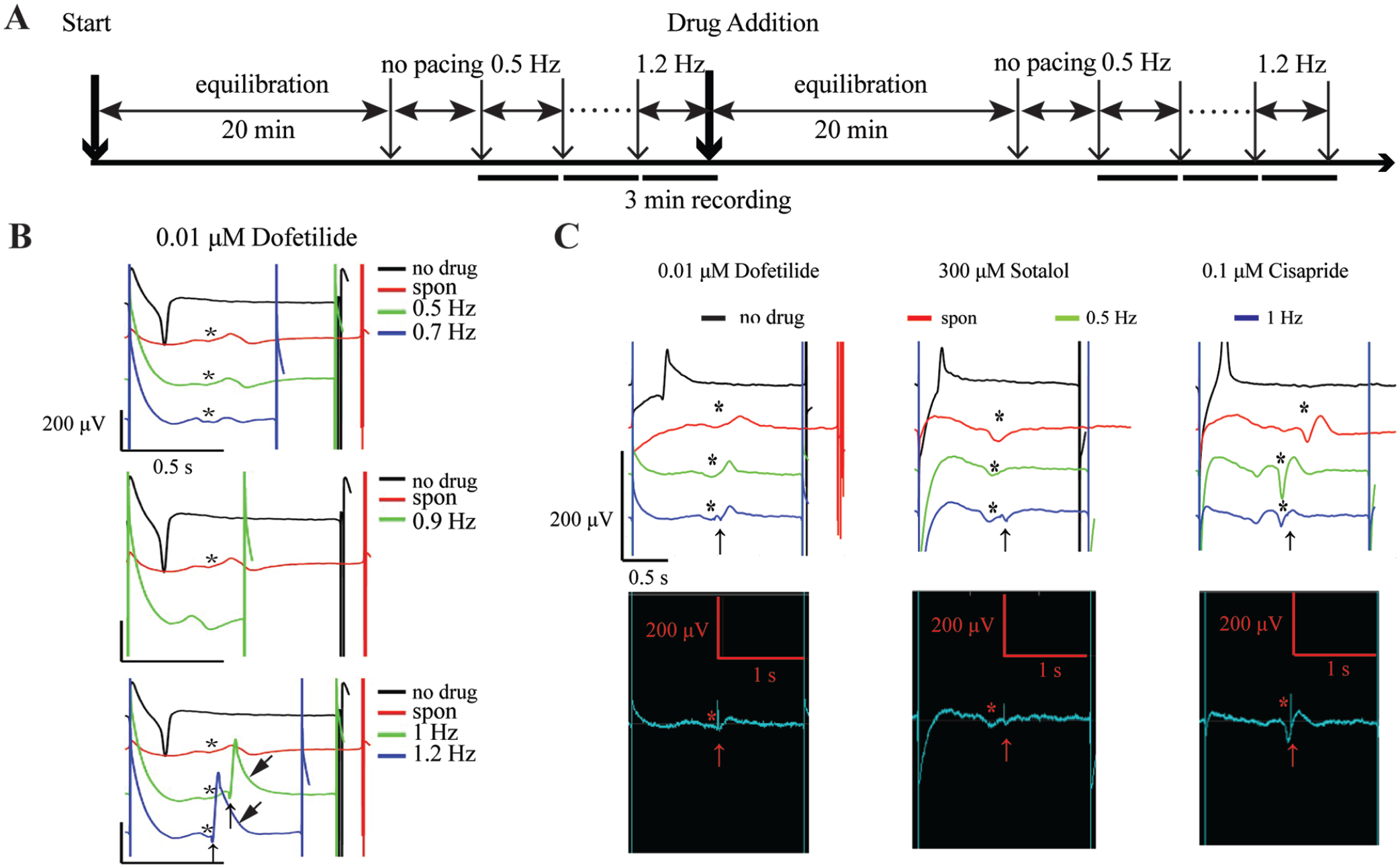
Proarrhythmic risk analysis using multielectrode array of electrically paced hiPSC-vCMs. (
Effects of Drugs on Beat Rates and Arrhythmia-Like Events at Different Pacing Rates.
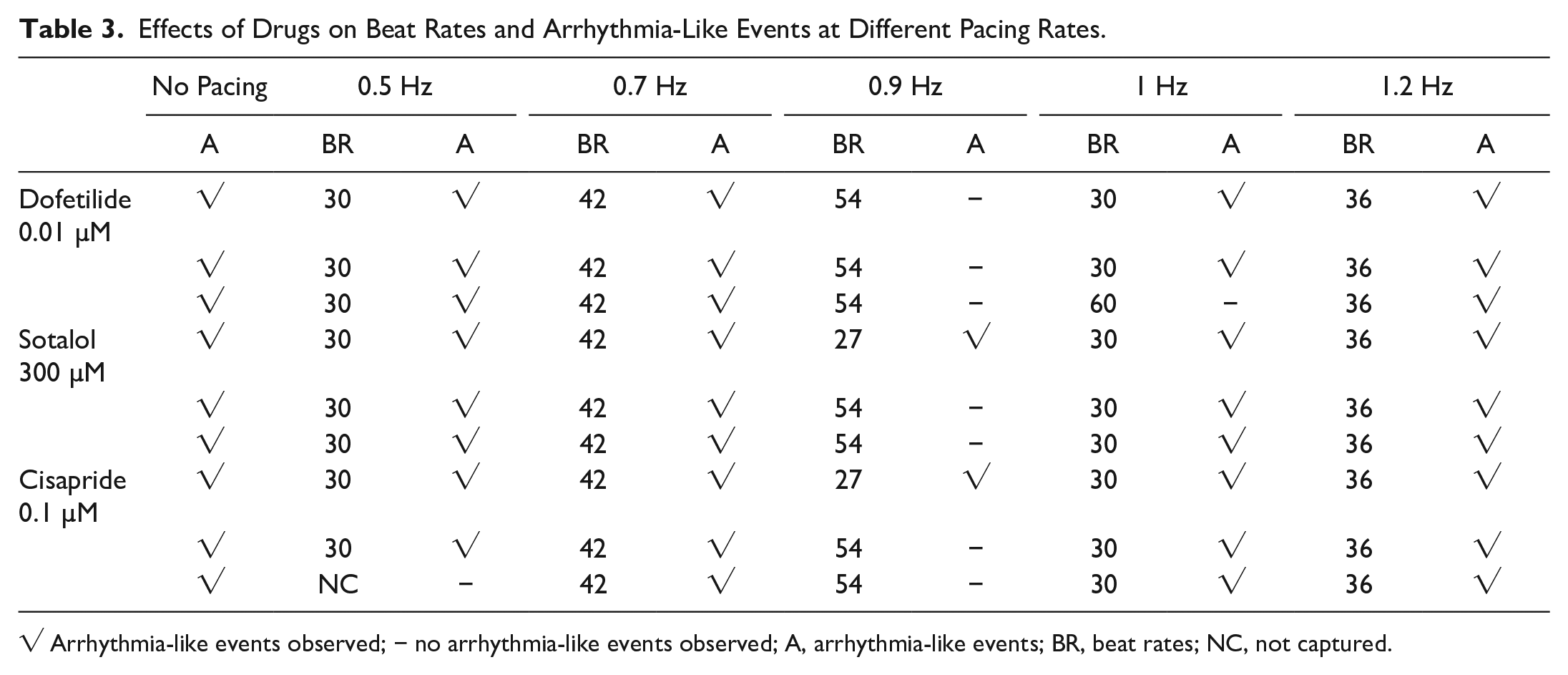
√ Arrhythmia-like events observed; − no arrhythmia-like events observed; A, arrhythmia-like events; BR, beat rates; NC, not captured.
Discussion
Ventricular cardiomyocytes are a good model for drug cardiac safety assessments. Considering the different electrophysiological properties of mixed atrial and nodal cardiomyocytes,3,13 homogeneous hiPSC-vCMs are preferred for drug TdP-risk prediction. Compared with other hiPSC-CMs, the hiPSC-vCMs used in this study were obtained through a direct ventricular differentiation protocol that yielded ventricular cardiomyocytes with more than 90% purity.
7
Through investigation of developmental changes in APs and densities of three ion channel currents in hiPSC-vCMs after the start of differentiation, we identified a suitable time window for using these cells for drug cardiac toxicity assessment. During this time window, spontaneously beating hiPSC-vCMs correctly predicted the drug TdP risk. Even so, it should be noted that the effects induced by bepridil and ranolazine in hiPSC-vCMs were also different from those of other drugs in the same TdP-risk category, as previously reported for other hiPSC-CMs.12,14 This discrepancy may predominantly arise from differences in ion channel gene expression levels of hiPSC-CMs compared with adult cardiomyocytes, especially higher CACNA1C and lower SCN5A gene expression in hiPSC-CMs (
Slow spontaneous beating of hiPSC-CMs is beneficial for their pacing at a physiological heart rate. Paced hiPSC-CMs beating at physiological rate can mimic in vivo ventricular myocytes beating under sinoatrial node innervation. When the pacing rate was close to or lower than the spontaneous beating rate, however, the internal pacemaker could not be suppressed, and doublets (a second beat within a normal beat) were induced. 9 Compared with hiPSC-derived atrial and nodal cardiomyocytes, ventricular cardiomyocytes beat slowly.3,4,7 The high-purity hiPSC-vCMs used here exhibit slower spontaneous beating (33 ± 5 beats per minute), which makes pacing at a physiological rate (1~1.2 Hz) feasible.
Compared with optical stimulation, electrical pacing is a simple, time-saving, and cost-effective way to pace hiPSC-CMs at a physiological heart rate because it does not require transfection of exogenous genes. 17 The adverse effects mentioned earlier in hiPSC-CMs caused by electrical stimulus, such as electrophysiological remodeling and structural changes, mainly occurred with long-term, rapid, and strong-intensity electrical stimulation (i.e., 3 V for 1 week at 3 Hz, or 3 V for 6 weeks at 1.2 Hz).18–20 One week of electrical stimulation with 3 V at 1 Hz did not, however, change the electrophysiological properties of hiPSC-CMs. 18 Considering current CiPA guidance on evaluating the acute TdP risk of a drug, the electrical stimulus (400 mV) used here should only have a minor influence on the activity of hiPSC-vCMs. We applied electrical stimuli to successfully pace hiPSC-vCMs beating at 1~1.2 Hz, and reliably identified “T” waves in FP signals. FP waveforms were similar to those observed in spontaneously beating and optogenetically paced hiPSC-CMs. 8 Our results, however, contrast with those of a previous study with no obvious “T-wave” in FP signals detected under electrical pacing; 9 differences in MEA instrument, stimulus mode, and intensity likely explain this discrepancy in outcomes. Under electrical pacing, we reliably detected the pivotal endpoints of FP signal in drug TdP-risk assessment: prolongation of repolarization induced by high and intermediate TdP-risk drugs, and the occurrence of arrhythmic-like events.
We also noticed the rate-dependent FPD changes in this study, which were consistent with previous reports.8,9 With the increase of the pacing rate, the drug-induced FPD prolongation was shortened. In addition, when the pacing rate increased to 0.8–0.9 Hz in this study, we started to see that the other pacing pulses failed to evoke the beating. These pulses might have fell into the effective refractory period of action potential evoked by previous pulses. At this level, if the pulse evoked beating, we didn’t observe the arrhythmia-like event. The shortened FPD at this level reduced the probability that the arrhythmia-like event generation could be the reason; it is also possible that the pulse directly interfered with the arrhythmia-like event generation. When the pacing rate was fixed at 1 Hz or 1.2 Hz, the drug-induced repolarization in hiPSC-vCMs was prolonged in a concentration-dependent manner. Similar to optogenetically paced hiPSC-CMs, 8 our results with electrically paced hiPSC-vCMs were consistent with the outcome of drug TdP risk assessment obtained with spontaneously beating hiPSC-CMs.
With the emergence of mature hiPSC-vCMs possessing low-rate or no autonomic beating, MEA assays in electrically paced hiPSC-vCMs may still have enormous potential for drug cardiac safety assessments at a physiological rate.
Supplemental Material
Final-Reversion_Supplemental_Material_for_Assessment_of_cardiac_safety_by_Bai_et_al – Supplemental material for Assessment of Drug Proarrhythmic Potential in Electrically Paced Human Induced Pluripotent Stem Cell–Derived Ventricular Cardiomyocytes Using Multielectrode Array
Supplemental material, Final-Reversion_Supplemental_Material_for_Assessment_of_cardiac_safety_by_Bai_et_al for Assessment of Drug Proarrhythmic Potential in Electrically Paced Human Induced Pluripotent Stem Cell–Derived Ventricular Cardiomyocytes Using Multielectrode Array by Shuyun Bai, Junjie Pei, Kan Chen, Ya Zhao, Henghua Cao, Luyang Tian, Yue Ma and Haiheng Dong in SLAS Discovery
Footnotes
Supplemental material is available online with this article.
Declaration of Conflicting Interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Yue Ma is the founder of Cauliscell Company.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Program on Key Basic Research Project (2019YFA0110400), Beijing Municipal Science and Technology Project (Y4DG021001), and China Postdoctoral Science Foundation.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
