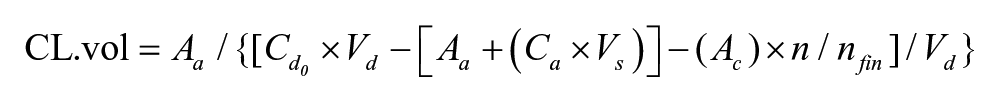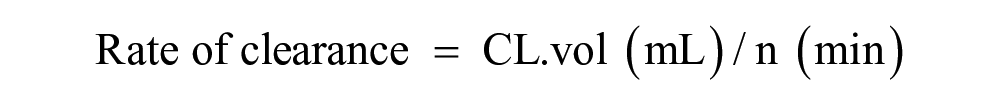Abstract
In drug discovery it is essential that one of the parameters tested for any new chemical entity is its affinity for human efflux systems, most notably P-glycoprotein (P-gp). These efflux systems affect not only rates of oral absorption but also rates of excretion through the liver, blood–brain barrier, and accumulation in potential target cells that upregulate efflux systems. Current methods to determine drugs’ P-gp transport potential include in vitro bidirectional transport studies, and the two most common cell lines used are Caco2 and MDR1-transfected MDCK models. Caco2 cells are human but slow growing and require more than 3 weeks to mature, while MDCK cells are canine, but when transfected with human P-gp become a rapid model of P-gp affinity. Our laboratory has generated a Caco2 subclone called CLEFF4 that is fully human, yet now approaches the rapid nature of the MDCK model. No special medium is required. We have shown, in as little as 5 days postseeding, high transepithelial electrical resistance values of more than 1000 Ω·cm2 plus P-gp expression more than threefold higher than that of 21-day-old cells. Currently tested drugs included rhodamine 123 (Rh123), vinblastine, and doxorubicin, and all drugs exhibited P-gp-mediated efflux that was inhibited by PSC833. By day 6, bidirectional transport of Rh123 was as potent as that of mature Caco2 cells, for use in comparative P-gp affinity studies. We now have a human P-gp model that is rapid and works without any need for special accelerating medium. We believe this could be a welcome addition to the testing regime of new chemical entities.
Introduction
The human Caco2 gastrointestinal cell line derived from a colon carcinoma has been used extensively by researchers and pharmaceutical companies for decades as an in vitro model to provide early estimates of novel drug oral absorption and drug efflux. The endogenous expression of the ABCB1 gene product also known as MDR1 or P-glycoprotein (P-gp) on the apical surface of this cell line, along with tight junction formation when grown on suspended inserts, has maintained the usefulness of this cell line in P-gp-mediated efflux research. Caco2 cells are able to spontaneously differentiate into columnar cells with microvilli on the apical surface with high apical expression of P-gp. In addition, they create tight junctions through expression of occludin, caudin-1, and zonula occluden (ZO-1), 1 which allows bidirectional transport studies to be conducted in vitro to test drugs’ affinity for P-gp with very little paracellular transport around the cells contaminating the transcellular transport results necessary to determine whether efflux proteins are pushing a test drug candidate back to the apical environment around the cell monolayers.
The Madin-Darby canine kidney (MDCK) cells transfected with human MDR1, 2 pioneered in the late 1990s, 3 are also a popular model. Early analysis of the MDCK model showed it to be different, though, from the Caco2 model, with a different composition of plasma lipids and possible orientation of P-gp in the plasma membrane being different from that of Caco2 monolayers, 4 along with functional canine P-gp still present within the transfected cell line. 4 Nevertheless, the MDCK-MDR1-transfected cell line has become a popular tool similar to the Caco2 model and is used as an in vitro marker of brain transport of P-gp substrates due to the very tight junctions available on this canine model. 5 These are the only two cell lines that currently provide tight junctions adequate enough for bidirectional drug transport where evidence of efflux can be recorded, usually expressed as an efflux ratio of drug moved out to drug moving into the cells that enables researchers to define the potential of a drug to be either a substrate or evidence of potential inhibitors’ effectiveness.
One of the benefits of using the transfected canine cells has been the speed of culture readiness from subculture to analysis of the test samples. This is between 3 and 5 days after culturing, depending on initial seeding density. The Caco2 cells, however, are much slower and normally require more than 3 weeks of growth, differentiation, tight junction formation, and ultimately P-gp apical localization before analysis can be conducted. A rapidly accelerating method using sodium butyrate as its base was developed 20 years ago to attempt to provide a similar 3- to 5-day accelerated mechanism for the Caco2 cells; however, tight junction development was quite poor, even though there was evidence of polarized cells and tight junction proteins. 6 Evident paracellular leakage around the cells only allows for identification of drugs effluxed more rapidly than the noise generated by nonspecific paracellular movement of the drugs between the compartments. 7 Nevertheless, for high-throughput screening such concessions were acceptable to get turnover of results within a week using the human Caco2 model.
In the current study, a new subclone of the Caco2 line has been created, called clone 4 (CLEFF4), that spontaneously accelerates differentiation and tight junction formation in 5–6 days, such that this new subclone cell line has all the advantages of the chemically induced model, yet with tight junctions even tighter than 3-week-old Caco2 cells, and may provide equivalent bidirectional transport studies to the 3-week-old model from the limited substrates tested to date, but now in 5–6 days instead.
Materials and Methods
Drugs and Chemicals
PSC833 was supplied by Novartis Pharma (Basel, Switzerland). Pierce Mini protease inhibitor tablets were supplied by Thermo Fisher Scientific (Waltham, MA). Ethanol and ultra-pure-grade Tris hydrochloride were supplied from Fisher Scientific (Fair Lawn, NJ). DMSO was purchased from Ajax Fine Chemicals (Taren Point, NSW, Australia). Sodium chloride, sodium potassium tartrate, and copper (II) sulfate were purchased from Chem-Supply (Gillman, South Australia). Rhodamine 123 (Rh123), sodium dodecyl sulfate, sodium carbonate, casein, potassium dihydrogen orthophosphate, trifluroacetic acid, phosphoric acid, formic acid, and nonidet P40 substitute were supplied by Sigma-Aldrich (North Ryde BC, NSW, Australia). Sodium hydroxide and Folin & Ciocalteu’s reagent were supplied by BDH Merck Pty Ltd (Bayswater, VIC, Australia). All other chemicals were of a minimum analytical grade.
Tissue Culture Reagents
The human colon carcinoma cell lines (Caco2) were obtained from the American Type Culture Collection (ATCC; Manassas, VA) at passage 18 and cultured for 1.5 years to reach a late passage state of 75
8
before being used to attempt subclone generation. High-glucose Dulbecco’s modified Eagle’s medium (DMEM), Dulbecco’s phosphate-buffered saline (PBS),
NuPAGE MOPS running buffer (20×), SeeBlue Plus2 Pre-stained Standard, Bolt Sample reducing agent (10×), Bolt 4-12% Bis-Tris Plus 15-well gels, NuPAGE transfer buffer (20×), NuPAGE LDS sample buffer (4×), and NuPAGE antioxidant were purchased from Novex by Life Technologies (Melbourne, Australia).
MDR1 mouse monoclonal antibody (sc13131), BCRP rabbit polyclonal antibody (sc130933), and MRP1 mouse monoclonal antibody (sc18835) were purchased from Santa Cruz Biotechnology (Santa Cruz, CA). Rabbit monoclonal antibody to MRP2 (ab172630) was purchased from Abcam (Melbourne, Australia). Mouse monoclonal anti-β-actin antibody was supplied by Sigma-Aldrich. Goat anti-mouse secondary antibodies were purchased from Jackson ImmunoResearch Laboratories (West Grove, PA). Clarity Western ECL substrate was supplied by Bio-Rad Laboratories (Gladesville, NSW, Australia).
Development of Cell Models
The parent Caco2 cell line was maintained in high-glucose DMEM supplemented with 10% FCS, penicillin, streptomycin, glutamine, and nonessential amino acid, incubated at 37 °C, 95% humidity, and 5% CO2. Medium was replaced twice a week and cell splitting was done using TrypLE Express, upon 90% confluency, approximately 1 week postseeding.
CLEFF4 Subclone Characterization
Our late passage (p70-80) parent Caco2 cell line was cloned via established dilution cloning techniques where trypsinized Caco2 cells were plated at one cell per 2 wells in 96-well plates, and subsequently manually monitored to ensure that eventual cell growth in a well came from a single source cell. As Caco2 cells are difficult to establish from a single cell, fewer than 20 subclones were able to be created from 6 months of dilution cloning. The clones with the greatest apparent P-gp as determined by densitometry analysis of P-gp bands from Western blotting studies were chosen for transport studies of known fluorescent P-gp substrates that had been used in our laboratory previously and had well-established 96-well plate fluorescence protocols for easy quantitation. CLEFF4 had the greatest activity when compared with parental Caco2 cells from the current subclone set and was subsequently analyzed for age-dependent changes in protein expression given the long lead time of more than 3 weeks to generally have the cell lines ready for efflux studies.
Bidirectional Transport Studies of Rh123, Doxorubicin, and Vinblastine
Cells were trypsinized from 25 cm2 flasks and plated onto 0.4 µm Millicell, 12 mm PCF cell culture inserts (PIHP01250; Merck-Millipore, Victoria, Australia) placed in each well of a 24-well culture plate, at approximately 75,000 cells/insert (growing surface of 0.6 mm2), which equated to a seeding cell density of 125,000 cells/cm2. Upon cell maturation, the transepithelial electrical resistance (TEER) across Caco2 and CLEFF4 cell monolayers was measured before and after experiments using an epithelial voltage/ohm meter (EVOM) and an ENDOHM-12 chamber (World Precision Instruments, Sarasota, FL). Transport studies were allowed to progress if TEER values above 300 Ω·cm2 were evident. TEER values were also taken at the completion of the study to ensure that membrane integrity was maintained during the 2 to 3 h time course of the study. In the determination of apical-to-basolateral (A-B) directional transport, the medium in the apical (donor) compartment was replaced with 300 µL of 5 µM Rh123 or 5 µM Rh123 plus 4 μM PSC833 in HBSS, while the basolateral (receiver) compartment had 600 µL of fresh HBSS or HBSS plus 4 μM PSC833. The apical chamber is narrower than the basolateral chamber, so to ensure that there is no pressure gradient between the chambers, it is required that half the volume be used in the upper chamber compared with the lower chamber to ensure that the meniscus level of HBSS is the same between the basolateral and apical chambers. The basolateral-to-apical (B-A) directional transport was determined by placing the drug in the basolateral compartment with the volumes as mentioned above (300 μL in the apical and 600 μL in the basolateral). For studies involving the other drugs, 25 μM doxorubicin or 10 μM vinblastine was used instead of Rh123. Transport medium was collected from the receiver compartment and was replaced with the same volume of fresh HBSS or HBSS plus inhibitors at 30, 60, 90, 120, and 180 min for all drugs. For A-B studies, 140 µL was taken from the basolateral chamber, and for B-A studies, 70 µL was taken from the smaller apical chamber. At the end of the experiment, 140 µL of sample was removed from the donor compartment and washed cells were lysed in protein lysis buffer containing protease inhibitors and collected for fluorescence analysis.
Rh123 and doxorubicin levels were quantified using a PerkinElmer Enspire multimode plate reader (Waltham, MA), and measurements were done using fluorescence detection. Rh123 was measured using excitation and emission wavelengths of 485 and 525 nm, respectively, while doxorubicin was measured using excitation and emission wavelengths of 480 and 590 nm, respectively. The apparent permeability (Papp) value was determined by the methods described by Crowe and Lemaire 9 with the mean and standard deviation values (SD using N = 3) calculated. Vinblastine’s fluorescence was too weak for plate reader analysis and instead was assayed using high-performance liquid chromatography (HPLC) as described below.
HPLC Analysis of Vinblastine
HPLC conditions were optimized in our laboratory based on the fluorescent characteristics of vinblastine. Samples collected in HBSS from transport studies were immediately collected into deep-well 96-well plates (Nunc). No extraction phases were required. The mobile phase consisted of 50 mM NaH2PO4 (BDH-Merck, Kilsyth, Victoria) (pH 3.0) containing 0.25% triethylamine; far-UV-grade acetonitrile and methanol were used as organic solvents (EM Science, Gibbstown, NJ) (55:24:21 v/v/v) for aqueous buffer, acetonitrile, and methanol, respectively. The HPLC system consisted of an Agilent 1100 series system run though the Agilent PC package “Chemstation” (Agilent, NSW, Australia). The quaternary pump ran at 1.2 mL/min and a PerkinElmer Series 200 autosampler injected 50 µL of sample through a Zorbax Stable bonded Aqueous (SB-Aq)-C18 column, with 5 µm pores and 15 cm × 4.6 mm ID with additional inlet frit (Agilent and Alltech, NSW, Australia, respectively). The effluent was detected on an Agilent 1100 variable-wavelength UV detector and in-line Agilent fluorescent detector. Vinblastine was measured concurrently at 214 nm with excitation and emission wavelengths of 270 and 357 mm, with a typical retention time of 4.0 min. The limit of quantification for vinblastine using fluorescence analysis was 25 nM.
Western Blotting
Late-passage Caco2 cells and clones 4 and 12 were grown concurrently with the cells used in the Rh123 studies in six-well plates using the above-described procedures. Cells were lysed on various days between days 5 and 28 and passed through a 21-gauge needle for maximum protein yield. The protein content was determined using the micro-Lowry protein assay and had electrophoresis undertaken by adding Novex sample buffer and reducing agent to the soluble protein sample. The reduced proteins were loaded in 4%–12% Bis-Tris 15-well Novex Bolt gels, along with SeeBlue precolored molecular weight markers. Gels were run using a Novex Bolt Mini Gel Tank (Life Technologies), and transfer was done in an Xcell II Bolt Module (Novex). The membrane was blocked using 2% casein in Tris buffered saline (TBS) and washed with TBS plus 0.05% Tween20 (TBST). The primary antibodies—P-gp (G-1) mouse monoclonal IgG2b for ABCB1, BCRP (B-25) rabbit polyclonal IgG, and MRP1 (QCRL-1) mouse monoclonal IgG1, all from SCBT, and Rab Mab MRP2 rabbit monoclonal antibody from Abcam—were all used with mouse anti-β-actin as the antibody for β-actin (Sigma-Aldrich), the reference protein. Horseradish peroxidase (HRP)-linked goat anti-mouse antibody or HRP-linked goat anti-rabbit antibody was used as the secondary antibody (Jackson ImmunoResearch). The washed membrane was incubated in Bio-Rad’s clarity chemiluminescent system and was semiquantified using a Chemidoc MPT imager with Image Lab software from Bio-Rad. The relative abundance of P-gp expression and other efflux proteins was given as a ratio of P-gp expression to β-actin.
Data Analysis
Drug transport through cell monolayers was calculated both as a simple amount passing the monolayer per minute, which would vary depending on the initial concentration used, and as an Papp coefficient as calculated previously.9,10 This calculation allows for a modification to the original Artursson equation, 11 where the concentration in the donor compartment (Co) is recalculated after every time point to compensate for that already present in the receiver chamber to ensure a greater accuracy in calculating the rate of movement into the opposing chamber. 12 The efflux ratio was defined as the value of the mean B-A direction permeability/A-B direction permeability (in nmol/s):
where CL.vol = clearance volume (mL), n = time (min), Aa = amount in acceptor compartment at time n (pmol),
Papp (cm/s) = Rate of clearance (mL/min)/Surface area (cm2)
The cleared volume was plotted against time and the slope of the regression line was divided by the surface area to obtain the Papp values. The mean Papp values for both the A-B (N = 3) and B-A (N = 3) directions plus standard deviation were presented as the bidirectional transport values of the study drugs. The mean Papp for the B-A direction was divided by the A-B direction to obtain the efflux ratio, and values above 2 were considered as drugs having true efflux transport. 13 At the end of the experiment, the total recovery/mass balance of the drug was calculated and results were considered valid if the recovery was >80%.
The results expressed in this study are presented as the mean ± SD, standardized to individual well protein concentrations. Significant differences between values were examined using the Student two-tailed unpaired t test or one-way analysis of variance (ANOVA). Results were considered significant if P < 0.05.
Results
Caco2 subclones isolated using the clonal dilution method (a concentration of one cell per 2 wells in 96-well plates) from passage 70 to 80 were tested for the presence of efflux proteins relative to the parent Caco2 cell line. Clones 5 and 12 had almost no detectable P-gp, which may be of interest in the future as P-gp-negative Caco2 cell lines, but of greater interest was the presence of P-gp in clone 4 ( Fig. 1 ), subsequently named CLEFF4, which sparked the most interest from our laboratory. Of the efflux proteins examined, only P-gp was highly expressed. There were no higher levels of BCRP or MRP2, while MRP1 appeared to be at very low levels ( Fig. 1 ) using Western blot analysis.
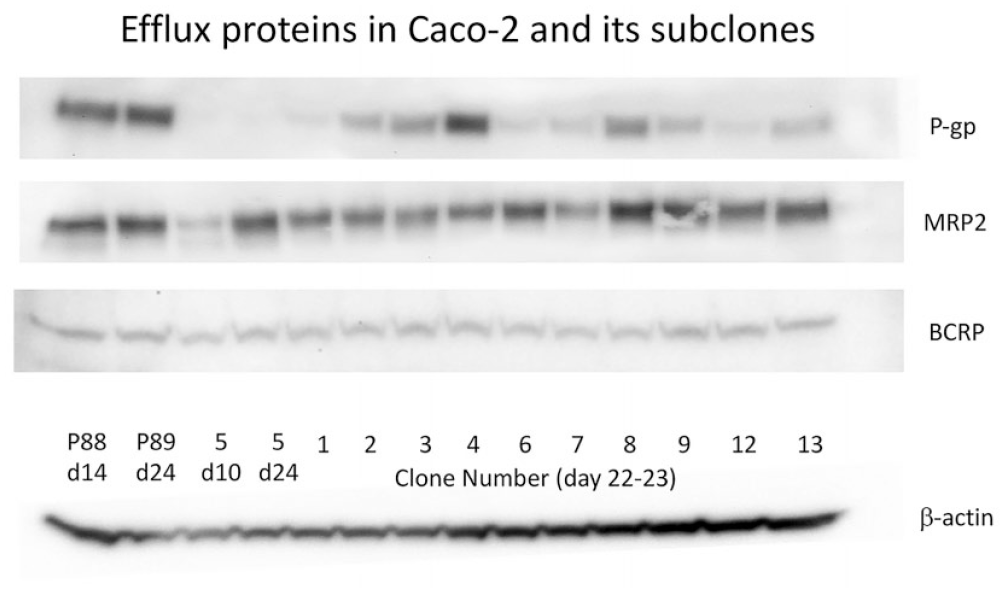
P-gp, BCRP, and MRP2 protein expression by Western blot in multiple Caco2 subclones. Most protein was collected from the mature age of 21–24 days postseeding.
In original studies with this subclone, protein had only been collected from day 14 onward to examine the amount of protein present in the cell monolayers, as days 21–24 has historically had the best expression of efflux proteins in Caco2 cells; however, as day 14 of culture had an unexpectedly high concentration of P-gp, this was explored by subsequently starting protein collection at day 5 postseeding (
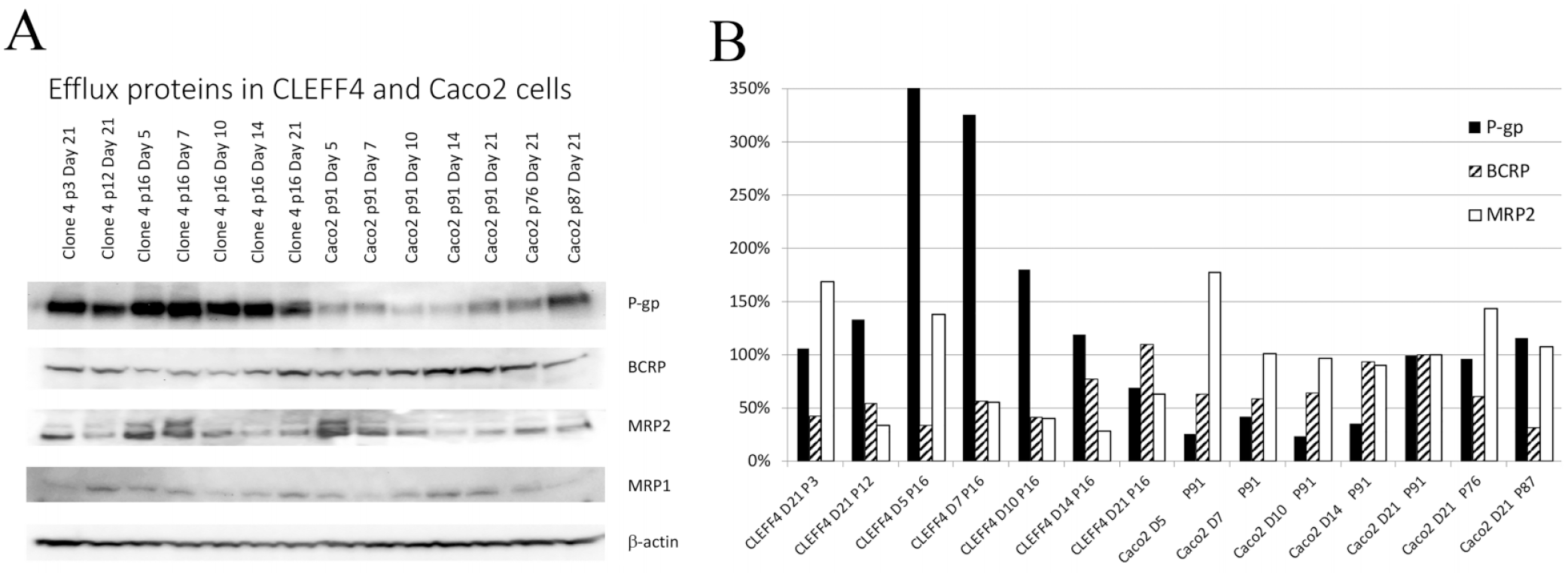
Protein changes with increasing postseeding age in CLEFF4 and parent Caco2 cells. (
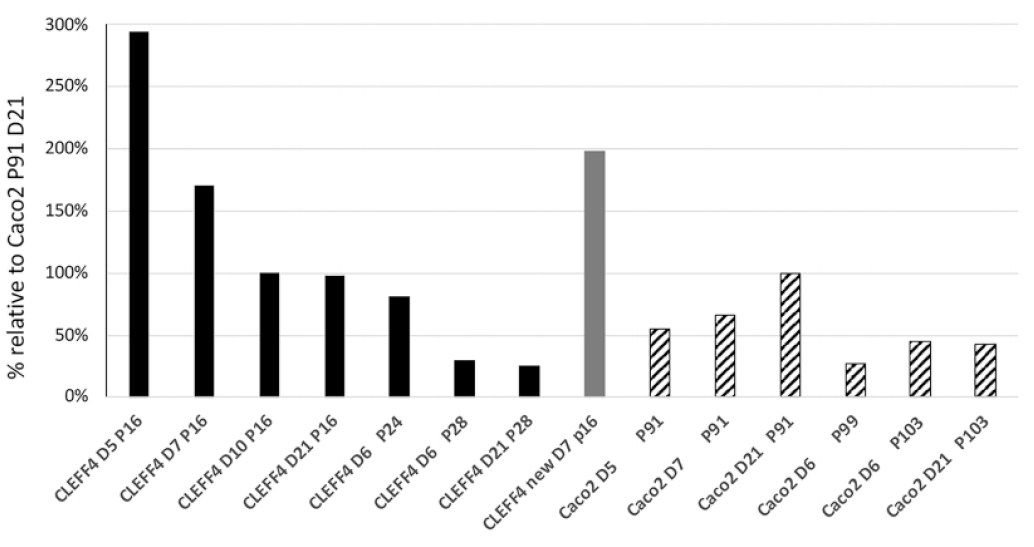
P-gp expression from passage 16 to 28 in CLEFF4 cells and passage 91 to 103 for Caco2 parent cells. Two distinct batches of passage 16 CLEFF4 cells cultured at different times are shown. P-gp is shown as relative to normal Caco2 cells at passage 91 and day 21 of culture. All data are normalized to β-actin expression.
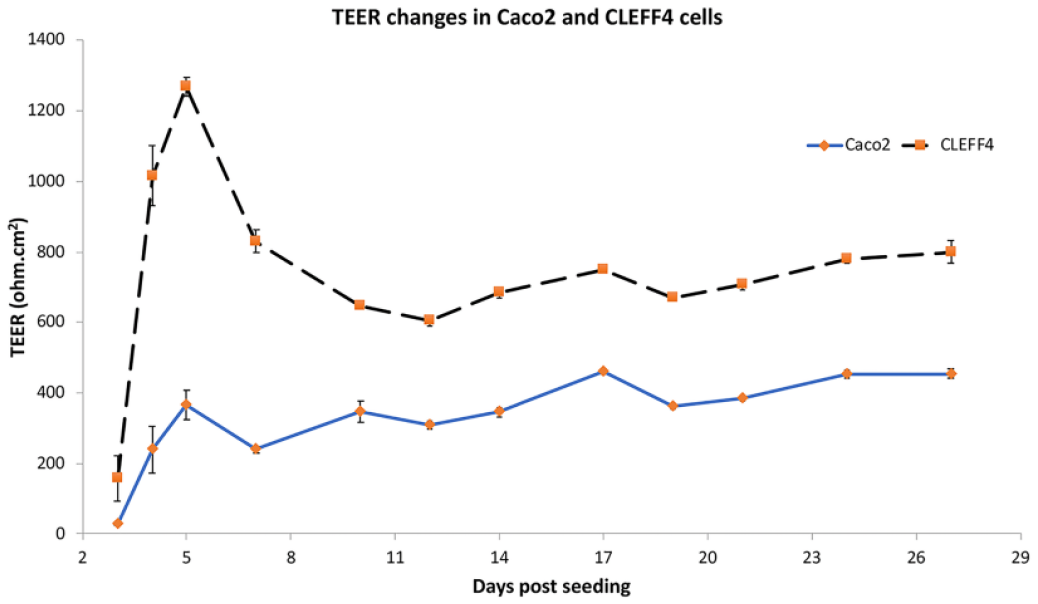
TEER changes with increasing culture age (parent Caco2 and CLEFF4). All results are shown in triplicate ± SD. Each well was followed from day 3 to day 27 with the TEER taken at the same time of the day for each day measured.
It was shown that CLEFF4 had high levels of P-gp expression from an early culture age, and although it maintained this during the 3 weeks of growth, it was highest within the first week of seeding, which was a completely different profile than that for standard Caco2 cells, where they were quite low even after 14 days postseeding, and in keeping with the literature,8,14,15 day 21 had the best expression for standard Caco2 cells, which is why the Caco2 cells are normally used at 3 weeks for efflux studies.
If P-gp was much higher at days 5–7 of culture for our CLEFF4 subclone, over threefold higher than 21-day P-gp in standard Caco2 cells, our next question related to tight junctions and whether they may also be higher in the first week of growth for the CLEFF4 subclone.
Tight junctions as measured with a ENDOHM-12 chamber and EVOM were able to be rapidly created within 4 days of culture at relatively modest seeding densities of approximately 125,000 cells/cm2. Although the CLEFF4 tight junctions did start to reduce from their peaks after week 1, they stayed higher than the parent Caco2 cells at all times, and recovered again for week 3 ( Fig. 4 ), showing that the CLEFF4 subclone was likely to be able to hinder the paracellular movement of drugs from as low as day 5 postseeding. Combined with the very high P-gp expression seen at the same time, we next explored the ability of the 5- to 6-day-old cell monolayers to show significant differences in bidirectional transport of some typical P-gp substrates.
Rh123 is a known P-gp substrate that due to its inherent fluorescent characteristics makes it an ideal P-gp substrate to examine whether functional P-gp is present on the apical sides of our polarized cells, as long as tight junctions are tight enough to ensure any P-gp-mediated efflux is not lost in potentially excessive paracellular leakage, as is evident for such young cultures of Caco2 cells (
Table 1
). However, bidirectional permeability studies using day 6 CLEFF4 cell monolayers showed significantly higher efflux than uptake (
Bidirectional Drug Transport in Parent Caco2 and the CLEFF4 Clone.
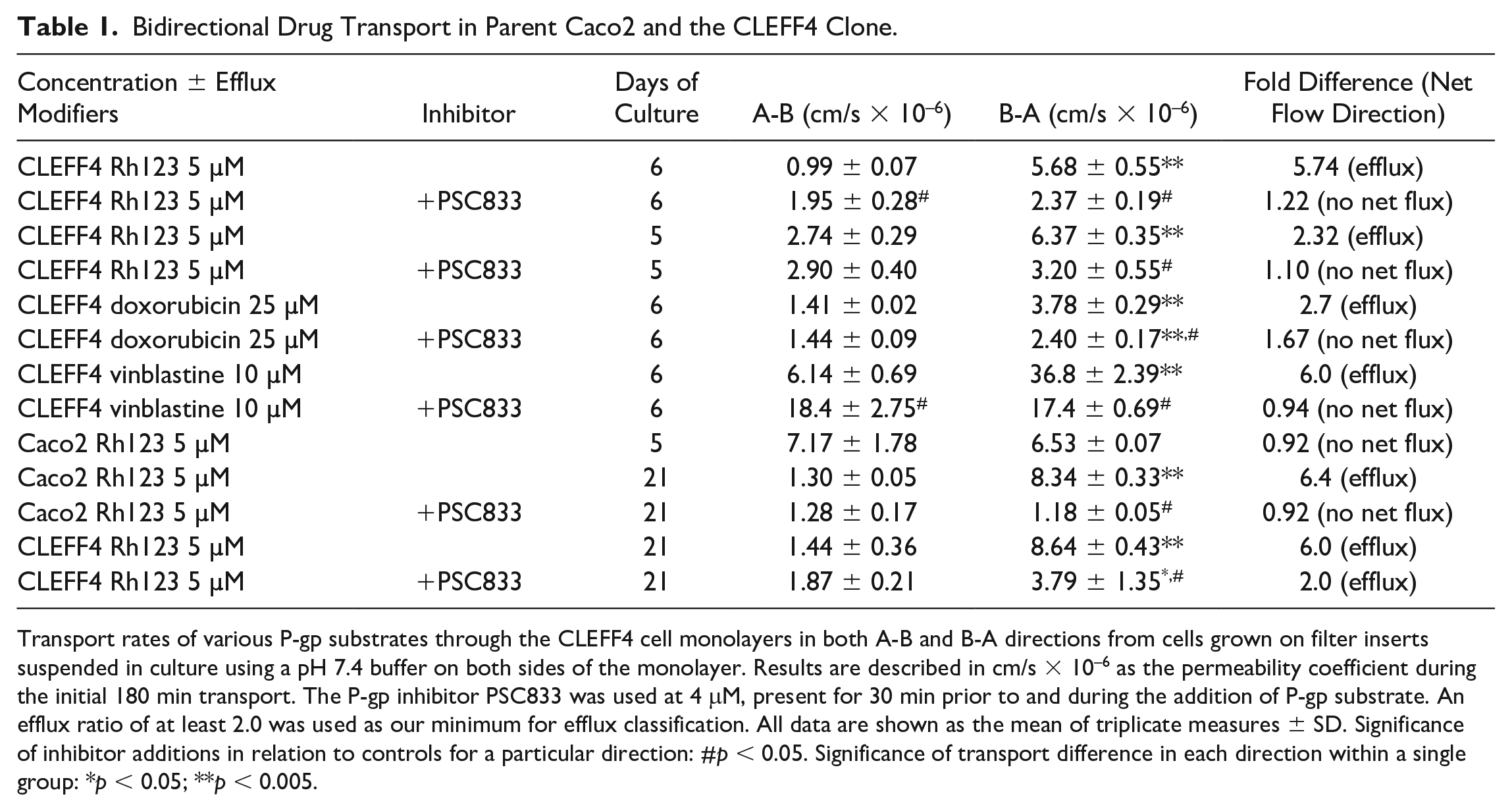
Transport rates of various P-gp substrates through the CLEFF4 cell monolayers in both A-B and B-A directions from cells grown on filter inserts suspended in culture using a pH 7.4 buffer on both sides of the monolayer. Results are described in cm/s × 10–6 as the permeability coefficient during the initial 180 min transport. The P-gp inhibitor PSC833 was used at 4 mM, present for 30 min prior to and during the addition of P-gp substrate. An efflux ratio of at least 2.0 was used as our minimum for efflux classification. All data are shown as the mean of triplicate measures ± SD. Significance of inhibitor additions in relation to controls for a particular direction: #p < 0.05. Significance of transport difference in each direction within a single group: *p < 0.05; **p < 0.005.
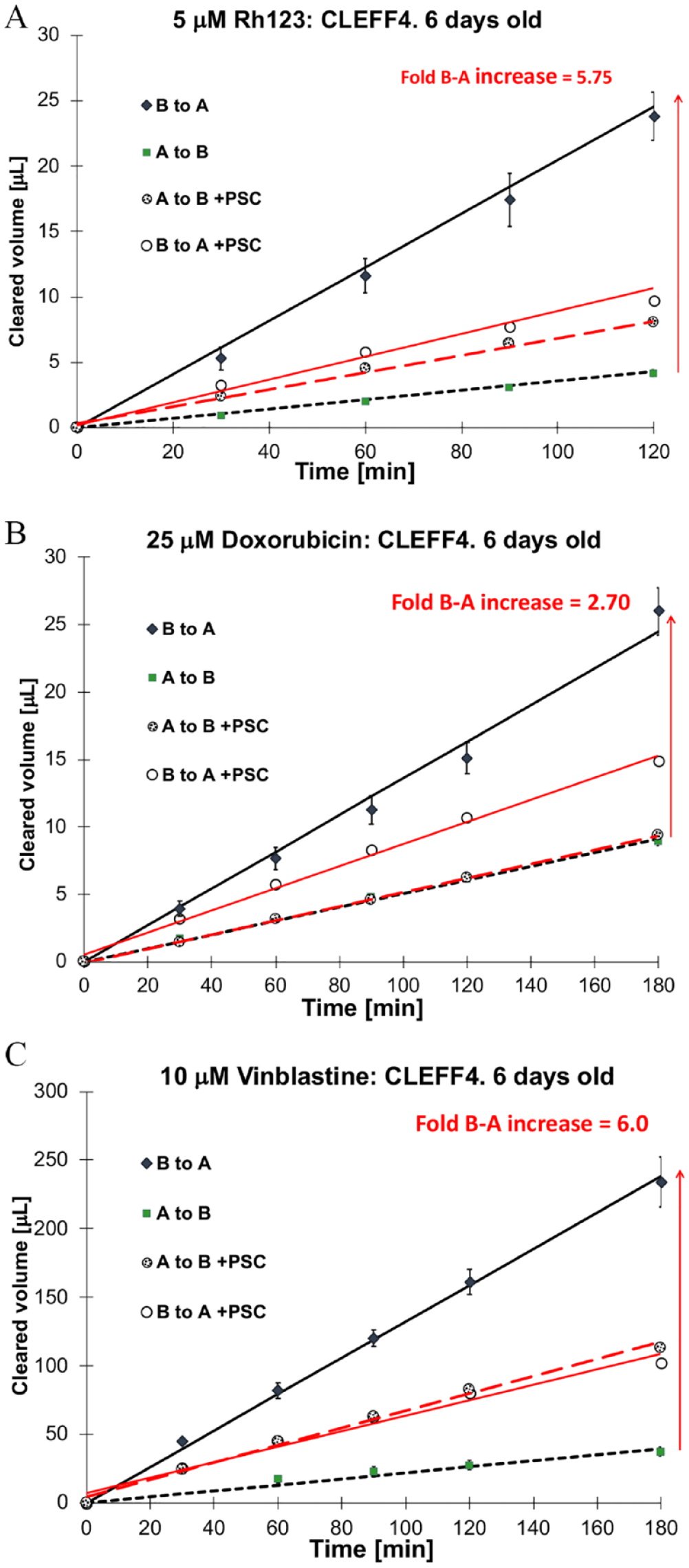
Bidirectional transport of P-gp substrates through Caco2 monolayers over 3 h. Each drug was examined with and without coadministration of a P-gp inhibitor (4 µM PSC833). (
To further validate the cell line, two more drugs were used, doxorubicin and vinblastine, both well-studied P-gp substrates.8,16,17
Doxorubicin has inherent fluorescence characteristics with excitation at 490 nm and emission at 590 nm, making it a useful second model drug after Rh123. It is weaker in its fluorescence characteristics than Rh123, but the PerkinElmer Enspire multimode plate reader was able to measure adequate drug passage into the receiver chambers for this to be a viable rapid data acquisition model to test. Nevertheless, the concentration of doxorubicin did need to increase to 25 µM in the donor chambers. Even at a relatively high concentration of 25 µM doxorubicin, the CLEFF4 model was able to show P-gp-mediated efflux of 2.7-fold, which had B-A transport significantly reduced upon coincubation with PSC833 (
Vinblastine is a strong P-gp substrate, as we have shown previously in our laboratory
20
and others more widely.21–23 Using 6-day-old CLEFF4 cell monolayers, it was evident that the efflux ratios detected were equivalent to that of Rh123 when vinblastine was used at 10 µM, being at least sixfold higher in the efflux direction. All of this was eliminated with the addition of PSC833, bringing transport back to equivalent in both directions (
All of the transport studies were done between passages 13 and 24 of the CLEFF4 clone, and as the results show in Table 1 , bidirectional transport was evident between this passage range. Although no transport data were taken above passage 24, protein was collected out to passage 28, and P-gp expression was determined relative to matched parent Caco2 passages ( Fig. 3 ). It was evident that as the passage number moved into the high 20s, P-gp expression did diminish, placing limits on the number of passages usable for transport analysis. Nevertheless, within the early passages there is consistency in P-gp expression as evidenced by the new passage 16 P-gp expression shown in Figure 3 that was regrown from cryopreserved CLEFF4 cells collected at a later time and provided similar high P-gp expression for that passage number. This is not dissimilar to standard practice with Caco2 cells where a 10-passage limit may be placed on transport studies before returning to cryopreserved stocks in individual laboratories.
Discussion
MDCK-MDR1-transfected cells tend to be used at a very high concentration of 300,000 cells/cm2 as seeding stock to get the cells to be able to generate an appropriately high TEER for bidirectional studies.24,25 Therefore, with this cell line and seeding concentration combination, studies are able to be done 3 days later to test P-gp-mediated efflux.24,25 One group used 96-well transwells, when using approximately 175,000 cells/cm2, so lower than previously, although still a high cell seeding density, and they used cells at 5 days postseeding for bidirectional transport studies. 5
In the early 2000s, Yamashita optimized the commercially available BIOCOAT method to accelerate Caco2 cell growth and was also able to get good tight junction formation and no passive permeability, as well as strong P-gp expression after 5 days of culture when using the BIOCOAT method with additional serum additions in normal basal medium while using Becton Dickinson’s collagen-coated wells. 6 In our study, we did not use collagen-coated inserts and instead just relied on the cells laying down their own collagen on the polycarbonate wells as they do when grown normally to 21 days under most protocol conditions. Other groups have also shown collagen coating to be unnecessary. 26
The current BIOCOAT method of accelerating Caco2 cells for use in less than a week uses butyrate and is harmful to the cells as it is considered to induce apoptosis27,28 and stresses the cells such that they are likely to start dying after a week of exposure. 6 Another group discovered that puromycin also was able to accelerate Caco2 growth, 29 but as puromycin is used as a tRNA mimetic that can disrupt wholesale protein translation in mammalian cells, it is unclear what additional stress-induced changes are occurring to the cells, and whether multiple passages can be used without the cells adapting to this different chemical addition to accelerate growth. In addition, the French laboratory was using seeding densities between 225,000 and 600,000 cells/cm2. 29 Our new subclone has been grown through to more than 24 days, yet is also able to be used at 5–6 days after seeding, with high TEER and P-gp expression, without the damage caused by other chemicals, and can be grown on uncoated Millicell polycarbonate inserts, just like standard Caco2 cells. Therefore, we consider this new subclone of Caco2 cells (CLEFF4) with endogenously accelerated differentiation to be superior in ease of setup and low cost to use in a more high-throughput fashion to determinate P-gp-mediated efflux. Compared with other attempts to create a rapid Caco2 bidirectional transport system, our model also does not require the very high levels of seeding densities of the BIOCOAT or puromycin models, allowing more replicates or test wells to be initiated from feeding stocks of Caco2 cells, which makes the whole process of cell culture consumable expenditure more cost-effective. Only with more drugs tested through this model, though, will we know how good a full replacement this cell line could be for the standard 3-week-old Caco2 system.
It is well known that P-gp expression can change over time with increasing passage number. Our laboratory has already shown high-passage Caco2 cells to show a much higher expression of P-gp than low-passage-number Caco2 cells, 8 and this is also evident in the MDCK-MDR1-transfected cell line, which needs a few passages after thawing stock from liquid nitrogen before P-gp expression stabilizes. 25 It is interesting to note that in a review from 2005 using MDR1-MDCKII-transfected cells, doxorubicin did not appear as a P-gp substrate, 30 yet doxorubicin resistance has commonly been associated with P-gp upregulation and Caco2 cells have always suggested doxorubicin to be transported by P-gp, 31 just as this new rapid CLEFF4 clone has done.
In 2006 we reported on a potent P-gp-expressing subclone, CLEFF9, which showed efflux rates of P-gp substrates much higher than late-passage parent Caco2 cells, for example, a 20-fold difference in Rh123 compared with 6.5-fold one for parent Caco2 cells. 20 Currently, we have established a new subclone, designated CLEFF4, and instead of elevated P-gp at 24 days, this subclone has elevated P-gp at 5–7 days with concurrent elevated TEER values, and bidirectional transport ability without the need for butyrate or puromycin-based accelerant mediums. These cells are able to generate an accelerated polarized development within 5 days with P-gp-mediated efflux clearly evident and inhibitable, similar to the use of current accelerating commercial products. Within a total of 6 days, not only is P-gp-mediated efflux evident, but also it approaches that of 24-day-old cells with very low paracellular leakage. Just as with Caco2 cell monolayers, though, there is some variability in P-gp expression as the passage number increases, and we would recommend staying between passages 5 and 24 for transport work, and ideally a model transporter could be used routinely as the passage number increases to allow comparisons between data generated at different passages.
Other groups have attempted to create co-cultures of Caco2 and HT29 cells (another human colon carcinoma, not normally able to induce its own tight junctions), but here tight junctions were very low and increased the leakiness of permeability markers. 32 This group also uses the terminology of “days postconfluence,” and their seeding densities are usually designed to have confluence by day 7 such that their T0 to T14 actually represent subculture days 7 to 21.
Of course, there is more to rapid Caco2 cultures than P-gp expression. Caco2 cells are still a good model for passive permeability and the estimation of likely gastrointestinal absorption. Miret et al., in the mid-2000s, examined the general permeability properties of the chemically induced rapidly accelerated Caco2 model with the 21-day model and concluded that compared with one another, the 21-day Caco2 model was much more predictive than the accelerated model. 33 Although P-gp substrates are the focus of this exploration of our new subclone, the P-gp-inhibited and therefore passive permeability data are very similar to the 21- to 24-day-old Caco2 data for the limited number of drugs tested. This suggests that in addition to P-gp-related studies, the CLEFF4 model could also be a rapid fully human in vitro model for general permeability work, and not just P-gp-mediated efflux. This would conform with the principles of being rapid and predictive as well as being cost-efficient, with far less medium consumed by the time of data acquisition. 34
This would not be the first subclone of Caco2 cells to have shown a clear distinction from the parent cell line. There is the TC7 subclone that has much higher CYP3A4 expression,35,36 and Borchart’s work in the early 2000s to increase P-gp expression with Caco2 subclones. 37 Certainly, though, I am unaware of any other group that has been able to generate a self-initiating rapid differentiator ready to have drug analysis conducted in 5–6 days with fully functional polarized P-gp expression and tight junctions mature enough for bidirectional analysis.
Footnotes
Acknowledgements
I would like to thank the School of Pharmacy and Biomedical Sciences, Curtin University, for assisting with internal school funding to purchase the consumables needed for this project. I would also like to thank Novartis Pharma, Basel, Switzerland, for donation of the PSC833.
Declaration of Conflicting Interests
The author declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.