Abstract
The global pandemic caused by the SARS-CoV-2 infection is a health emergency that needs to be addressed immediately. The international scientific community, following World Health Organization (WHO) indications, launched different trials for testing drugs putatively able to block the SARS-CoV-2 infection or treat the COVID-19 disease symptoms. In parallel, studies devoted to a better understanding of SARS-CoV-2 biology are in the course for designing an effective vaccine. One of the human membrane proteins known to be docked by the virus is angiotensin-converting enzyme 2 (ACE2), proposed to be responsible for viral entry in target cells. Recently, the 3D structure of ACE2 has been obtained, showing its physical interaction with B0AT1 (SLC6A19), a plasma membrane transporter involved in the trafficking of amino acids in cells. The receptor targeted by SARS-CoV-2 is a supercomplex formed by a dimer of ACE2-B0AT1, in which ACE2 binds the viral protein and B0AT1 stabilizes the heterodimer. As a serendipity occurrence, nimesulide was shown to abolish the transport function of B0AT1. Here we suggest including nimesulide in the list of drugs to be tested for the identification of co-adjuvants in the treatment of COVID-19.
The emergency that the human population is currently experiencing calls for rapid identification of drugs to treat the COVID-19 disease caused by SARS-CoV-2. However, the design of novel drugs is a long process that cannot be pursued with immediate results. Indeed, a complete study starts with improvement of knowledge of the virus biology, moving to human target identification and drug design, then performing tests in in vitro and in vivo models, and concluding with human trials; in terms of time, this means more than 5 years. From the depicted scenario, it is evident that alternative, but much faster, paths should be followed, starting from actual knowledge on SARS-CoV-2. This virus belongs to the coronavirus family, is able to infect vertebrates, and is the seventh coronavirus acknowledged to infect humans; the outbreak of SARS-CoV-2, together with that of SARS-CoV and MERS-CoV, is the third passage of a Coronaviridae virus from an animal to humans causing a major epidemic event with severe and life-threatening disease.1,2 SARS-CoV-2, like other members of the family, is an enveloped positive-strand RNA virus that spreads quickly among humans. The rapid diffusion of the infections, together with the percentage of deaths, has boosted the efforts of the scientific community to identify the molecular mechanisms of infection. This primary knowledge is crucial for designing effective vaccines and drugs, able to prevent and block or reduce the infections, respectively. The approach proposed by the World Health Organization (WHO), to save time, is to find old approved and safe drugs to actively treat the SARS-CoV-2 infection, as a serendipity effect. In this respect, some drugs have already been highlighted as candidates for a megatrial called Solidarity (“Solidarity” clinical trial for COVID-19 treatments: https://www.who.int/emergencies/diseases/novel-coronavirus-2019/global-research-on-novel-coronavirus-2019-ncov/solidarity-clinical-trial-for-covid-19-treatments). The first list includes (1) remdesivir, already used against the Ebola virus and able to inhibit the key viral enzyme RNA-dependent RNA polymerase;
3
(2) the antimalaric chloroquine and hydroxychloroquine, for their capacity to block SARS-Cov-2 in vitro as well as in a group of COVID-19 patients;
4
(3) the anti-HIV complex ritonavir/lopinavir, which has protease activity and has been revealed to be effective with the coronavirus MERS;
5
and (4) the complex ritonavir/lopinavir in association with interferon-β, for modulation of the inflammatory response.
6
In the meantime, comparative analysis with other known coronaviruses has allowed us to conclude that a known SARS target, angiotensin-converting enzyme 2 (ACE2),
7
is one of the ways in which SARS-CoV-2 docking to human cells can occur.8,9 This hypothesis was demonstrated by the recently solved structure of the human ACE2, in complex with the viral receptor-binding domain (RBD), at 2.9 Å resolution by cryo-electron microscropy.
8
Interestingly, the 3D structure of the human receptor brought to light a more complex architecture in which ACE2 forms a heterodimer with B0AT1 (ACE2-B0AT1). ACE2 consists of an extracellular peptidase domain (PD) and a collectrin-like domain (CLD) that ends with a single transmembrane α-helix. This is the ACE2 moiety interacting with B0AT1 (SLC6A19), an integral membrane protein formed by 12 transmembrane α-helices. B0AT1 belongs to the SLC6 family and is responsible for the sodium-dependent transport of neutral amino acids, mainly in the intestine and kidney.
10
The interaction between ACE2 and B0AT1 is well documented; indeed, ACE2 plays a major role in trafficking B0AT1 at the plasma membrane in the intestine but not in the kidney, where collectrin is B0AT1’s chaperone. The complex reported by Yan et al. has a peculiar assembly in which two heterodimers, constituting ACE2-B0AT1, form a homodimeric superstructure (ACE2-B0AT1)2, as shown by
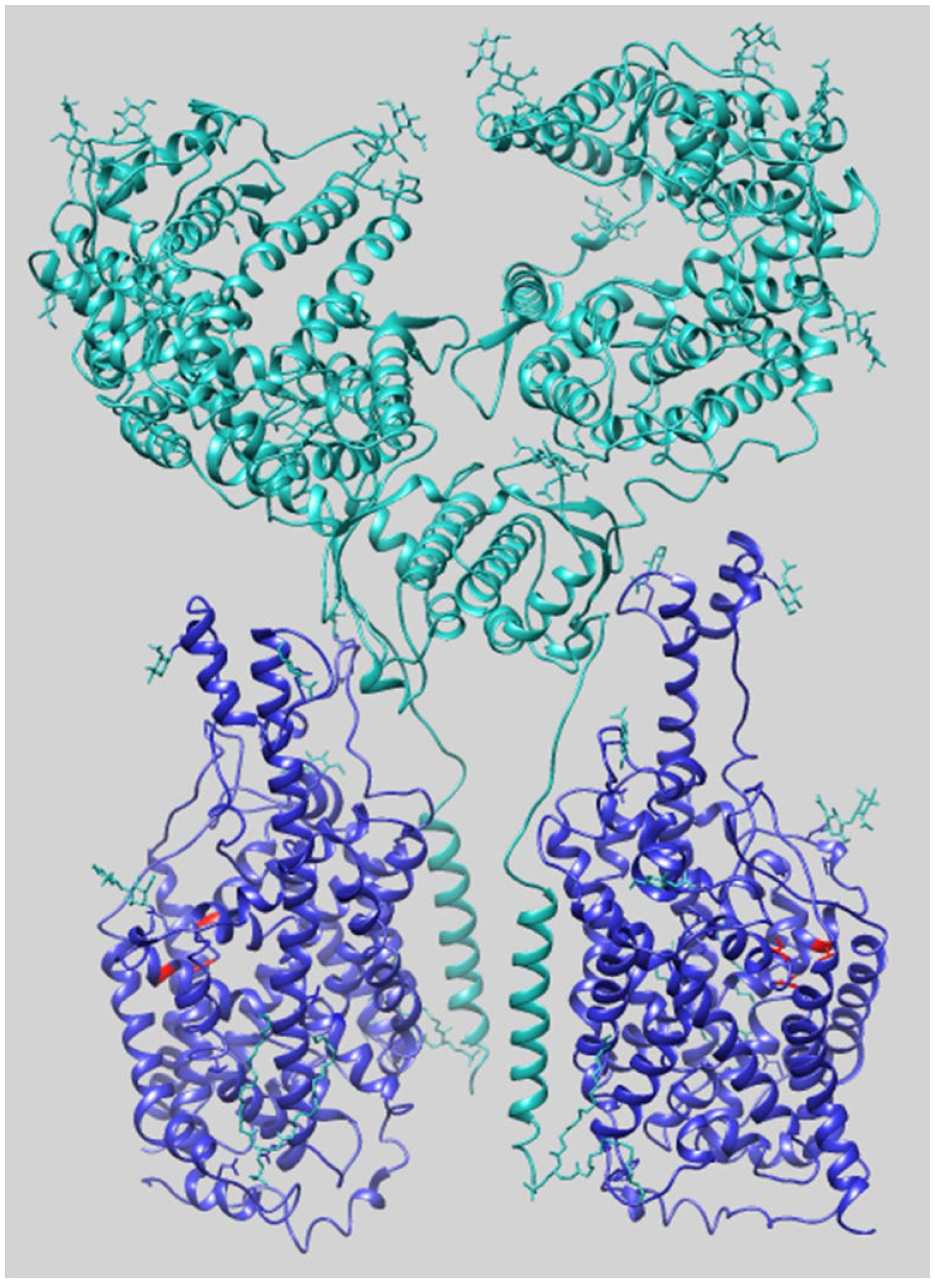
Overall 3D structure of the supercomplex (ACE2-B0AT1)2. The structure (PDB: 6M18) is represented in closed conformation as a ribbon. 8 The superstructure is composed of two heterodimers of ACE2-B0AT1; each heterodimer comprises one ACE2 subunit (cyan) and one B0AT1 subunit (blue). In red are shown residues predicted to form the nimesulide binding site on the B0AT1 subunit.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by PRIN (Progetti di Ricerca di Interesse Nazionale) project no. 2017PAB8EM MIUR (Italian Ministry of Instruction, University and Research) to C.I.
