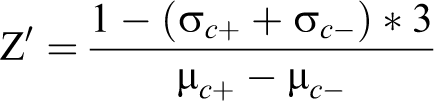Abstract
Interferon-γ (IFN-γ) is a critical cytokine in the defense against viral and bacterial infection. It is mainly produced by natural killer cells and activated T cells. Given its regulatory role in coordinating cellular and humoral immune responses, IFN-γ is considered to be an effective therapeutic agent in the treatment of viral infection. Here we established a fluorescence-based high-content screening model to find small molecules that can stimulate the production of IFN-γ in human Jurkat cells. After a primary screening of 267 natural products, two hits, Astragalus polyphenols and 6-shogaol, were identified to promote the activity of the IFN-γ promoter and subsequently validated by the flow cytometry assay. Obviously, both Astragalus polyphenols and 6-shogaol exhibited potential to induce the transcription and expression of IFN-γ in a dose-dependent manner. These results indicated that our high-content screening model could be a credible and useful platform to contribute to the discovery of novel molecules to promote the expression of IFN-γ and provide leading compounds for the treatment of viral infectious diseases.
Introduction
The interferon (IFN) families were identified for their protective functions against viral infection. 1,2 According to the structure of their receptors, IFNs were subdivided into three major classes: type I, type II, and type III. 3,4 In general, type I and type III IFNs were characterized by their contributions in the control of primary viral infections in epithelial cells, and type II IFN is known for its immunomodulatory effects. 5 -7 To date, IFN-γ is still the only member of type II IFN since the type was first identified in 1965. The IFN-γ gene is located on chromosome 12 in humans and is predominantly secreted by activated T cells and NK/NKT cells. 8 IFN-γ plays a central role in coordinating the innate and adaptive immune responses in recognizing and eliminating pathogens, such as the induction of major histocompatibility complex (MHC) class I/II and co-stimulatory molecule expression of antigen-presenting cells (APCs), 9 enhancement of antigen processing and presentation, promotion of CD4+ T-cell Th1 differentiation, 10 and blockade of IL-4-dependent isotype switching in B cells. 11,12 In clinical trials, IFN-γ is considered to be a potent administrative cytokine in the treatment of several infections and chronic granulomatous disease. 13 -15 Experimental data also show that IFN-γ administration can effectively inhibit the infections and replication ability of the Ebola virus in animal infection models. 16 Both clinical and experiment data demonstrate IFN-γ could be an attractive therapeutic option. 17
The human IFN-γ gene contains four exon regions and three intron regions, and the expression is tightly controlled by a complex regulation system at the transcriptional and translational level. Generally, the production of IFN-γ is controlled at the level of transcription, mRNA stability, and translation. 18 The promoter is responsible for gene transcription initiation, which is tightly regulated by transcriptional factors including activators and inhibitors. In the IFN-γ promoter, T-bet, NFAT, NF-κB, eomesodermin, AP-1, CREB-1/ATF, and STATs act as activators, while TGF-β, GATA3, DREAM, and YY1 are specific inhibitors. 8,19,20 Additionally, several long noncoding RNAs (LncRNAs) and microRNAs (miRNAs) have also been reported to negatively regulate the expression of IFN-γ by destabilizing the mRNA. 18,21,22 Except for the intracellular regulators, some chemicals could also affect the promoter activation. A region between –108 and –40 was identified to be an essential binding site for PMA/ionomycin, which would stimulate the transcription of IFN-γ. Meanwhile, the promoter activity was also inhibited by cyclosporine when binding to this region. 23,24 To study how extracellular signals triggered the production of IFN-γ in T cells and which elements would respond, a reporter gene such as firefly luciferase under the control of these transcriptional control elements was developed. 25
Reporter gene assay is a highly sensitive and specific bioassay. In this system, engineered promoters from genes of interest are used to drive reporter proteins. In general, bioluminescent reporters, such as firefly luciferase, and fluorescent reporters like green fluorescent protein (GFP) and red fluorescent protein (RFP) are two efficiently and widely used reporter proteins. 26,27 Reporter gene assay is a powerful tool used in high-content screening assays for novel drug discovery and studying some intracellular signaling pathways of interest, as well as cytokines and their induced genes in vitro. 28 What is more, the application of fluorescence-based cytokine reporter mice has also been well studied in vivo. 29 Stetson et al. have generated two cytokine knock-in reporter mice, named 4get (IL-4-eGFP) and Yeti (IFN-γ-eYFP), respectively. 30,31 These transgene mice contribute to faithfully evaluating the functions of endogenous cytokines and their dynamic distribution in vivo.
In this study, we established an IFN-γ-mCherry reporter model in human Jurkat cells. The promoter nucleic acid sequences from the –1.5 kb to þ35 bp sites of the IFN-γ gene were amplified and inserted into a lentivirus expression vector pCDH-CMV-EF1-copGFP to replace the cytomegalovirus (CMV) promoter, followed by the reporter gene mCherry. We named the reporter plasmid pCDH-pIFN-γ-mCherry-EF1-copGFP, in which the expression of mCherry was controlled by the IFN-γ promoter. Unless an endogenous or exogenous stimulation was received, the mCherry protein would not express in Jurkat cells, whereas the copGFP gene was constitutively expressed. To facilitate the screening study, we established a stable expression cell line, which was termed Jurkat-pIFN-γ-mCherry in this study. Afterward, a natural product library including 267 compounds was selected for primary screening in the reporter cells. We aimed to find the positive compounds that can induce the expression of IFN-γ. Fortunately, two hits, Astragalus polyphenols and 6-shogaol, were identified to be potent to promote the transcription of IFN-γ in this screening model. The activity was verified in wild-type Jurkat cells in mRNA and protein levels. Our screening model might provide a reliable platform for the discovery of novel compounds to stimulate the production of IFN-γ.
Materials and Methods
Cells and Culture
293FT and Jurkat cells were obtained from the Shanghai Institute of Biochemistry and Cell Biology, Chinese Academy of Sciences (Shanghai, China).
293FT cells were cultured in Dulbecco’s modified Eagle’s medium (DMEM; HyClone, Logan, UT) supplemented with 10% fetal bovine serum (HyClone). Jurkat cells were cultured in RPMI-1640 supplemented with 10% fetal bovine serum and 1% penicillin/streptomycin. All cells were cultured at 37 °C in a humidified atmosphere with 5% CO2.
Reagents
PMA, ionomycin, and propidum iodide (PI) were purchased from Sigma-Aldrich (St. Louis, MO). A library of 267 natural products was purchased from Selleckchem (L1400, Houston, TX). A human IFN-γ enzyme-linked immunosorbent assay (ELISA) kit was purchased from Abcam (ab46025, Cambridge, MA). The CellTiter 96 AQueous One Solution Cell Proliferation Assay (MTS) reagent was purchased from Promega (PR-G3580, Madison, WI). PI was purchased from Sigma-Aldrich (P4170). All restriction enzymes were purchased from Thermo Fisher Scientific (Waltham, MA), and T4 ligase was obtained from NEB (Ipswich, MA). Human IFN-γ promoter/enhancer plasmid was purchased from Addgene (cat. 17598, Cambridge, MA). 32
Plasmid Construction
pCDH-pIFN-γ-mCherry-EF1α-copGFP-puro vector was constructed on the basis of pCDH-CMV-MCV-EF1α-copGFP-puro plasmid (SBI, Palo Alto, CA). Briefly, mCherry from pmCherry-N1 (Clontech, Mountain View, CA) was inserted between the BamHI and NotI sites in the pCDH-CMV-MCV-EF1α-copGFP-puro vector to generate an intermediate vector pCDH-CMV-mCherry-EF1α-copGFP-puro.
The human IFN-γ promoter (–1.5 kb to þ35 bp) was amplified from pGL3-IFN-γ-Luc plasmid (cat. 17598, Addgene). Primer sequences (5′-3′) validated by Invitrogen (Shanghai, China) were as follows: pIFN-γ forward, CGG
Lentivirus and Infection
Lentivirus was packaged in a three-plasmid system. Briefly, 5 × 106 293FT cells were seeded in a 10 cm2 cell culture dish and cultured overnight until the cell density reached to 80%. A 5 µg expression vector pCDH-pIFN-γ-mCherry-EF1α-copGFP-puro, 2.5 µg package plasmid pCMVdR8.91, and 1 µg envelop plasmid pMD2.G mixed well with 17 µL P3000 reagent in 500 µL Opti-MEM medium in tube A. At the same time, Lipofectamine 3000 was diluted by 500 µL Opti-MEM medium in tube B. Then a vortex was gently performed between the mixtures in tubes A and B. After 15 min standing at room temperature, the mixture was added to 293FT cell culture medium without antibiotics. Forty-eight hours later, cultural supernatants containing lentiviral particles were collected and filtered through a 0.45 μM membrane.
Infection was performed in a 6 cm2 cell culture dish with 5 × 105 Jurkat cells. At 72 h postinfection, infected cells were exposed in 2 μg/mL puromycin for 7 days. After that, a single cell clone was selected by infinite dilution in a 96-well plate. After an expanded culture, the reporter cells, which we termed Jurkat-pIFN-γ-mCherry, were prepared for the screening assay.
High-Content Screening Assay
Jurkat-pIFN-γ-mCherry cells (5 × 103) were seeded in a poly-
The fluorescence intensity value of copGFP was used as an internal reference, and data were represented as the fluorescence intensity (mCherry/copGFP) ratio. A cutoff horizontal line was set, referred to as the positive sample. The Z′ factor was calculated as follows: 33
where σ is the standard deviation (SD), μ is the average value, c + is the positive control, and c – is the negative control.
Semiquantitative RT-PCR
Total RNA was extracted from Jurkat cells using Trizol reagent (Invitrogen, Camarillo, CA) following the manufacturer’s instructions. Reverse transcription (RT) was performed using the RevertAid H Minus First Strand cDNA Synthesis Kit (Thermo Fisher Scientific). The cDNA was used as a template for PCR assay by specific primers for human IFN-γ. The glyceraldehyde-3-phosphate dehydrogenase (GAPDH) housekeeping gene was used to standardize the levels of gene expression. The primer sequences (5′-3′) were as follows: IFN-γ forward, CATCCAAGTGATGGCTGA; IFN-γ reverse, GCAGGCAGGACAACCATT; GAPDH forward, TCACCACCATGGAGAA G; and GAPDH reverse, TAAGCAGTTGGTGGTGCA.
Enzyme-Linked Immunosorbent Assay
Jurkat cells were stimulated with 50 ng/mL PMA and 1 μM ionomycin or vehicle for 48 h; then the cell free supernatants were collected by centrifuging at 1000g for 10 min. The supernatant level of IFN-γ was measured by commercially available ELISA kits following the manufacturer’s instructions. Each sample was repeated three times, and results are shown after the background signal was subtracted (duplicates ± SD).
Flow Cytometry Analysis
Compounds or DMSO-treated Jurkat-pIFN-γ-mCherry cells were washed twice with cold phosphate-buffered saline (PBS) and then resuspended in 1× binding buffer at a concentration of 1 × 106 cells/mL. Then cells were stained with PI at a concentration of 5 μg/mL for 10 min in the dark at room temperature. Then cells were filtrated through 100 μM nylon. The fluorescence intensity was detected by flow cytometry (Calibur, BD Biosciences, Franklin Lakes, NJ). The data were analyzed using FlowJo7.6.1 analysis software.
Cell Viability Assay
The MTS assay was used to determine cell viability by CellTiter96 Aqueous One Solution Reagent (Promega). Briefly, 3 × 103 Jurkat cells were seeded in 96-well plates and treated with Astragalus polyphenols or 6-shogaol in triplicate at the same time, with Taxol as the positive control and DMSO as the negative control. Compounds were diluted five times from 40 μM as the highest concentration. After culturing for 48 h, 20 µL of MTS was added to each well and cells were incubated at 37 °C for 1–2 h. The absorbance was measured at 490 nm by a microplate reader (Bio-Rad, Hercules, CA). The IC50 value for each compound was calculated by the relative survival curves.
Statistical Analysis
A two-tailed Student t test was used to determine the statistical significance. All results were expressed as mean ± SD. A p value of less than 0.05 was considered to be statistically significant.
Results
Generation of pIFN-γ-mCherry Reporter Construct
IFN-γ plays a critical role in immunomodulation upon viral infection and cancer diseases. The recombinant human IFN-γ has been approved by the FDA in clinical trials for the treatment of renal carcinoma and rheumatoid arthritis in America. In that case, natural products that can induce the production of IFN-γ may also have therapeutic potential. To facilitate high-content screening by the Cellomics ArrayScan VTI HCS Reader, an mCherry reporter vector was constructed. First, the mCherry gene was cut from the pmCherry-N1 vector and inserted into the multiple cloning site (MCS) to generate an intermediate vector pCDH-CMV-mCherry-EF1α-copGFP-puro. Second, the IFN-γ promoter (–1.5 kb/+35 bp) amplified from the pGL3-pIFN-γ plasmid was inserted into the intermediate vector to replace the CMV promoter to obtain the pCDH-pIFN-γ-mCherry-EF1α-copGFP-puro vector ( Fig. 1A ). A diagrammatic sketch of the core elements for the reporter plasmid is shown in Figure 1B , and the nucleic acid sequence for the IFN-γ promoter is shown in Figure 1C . The regions from the IFN-γ promoter to the mCherry gene were confirmed by sequencing.
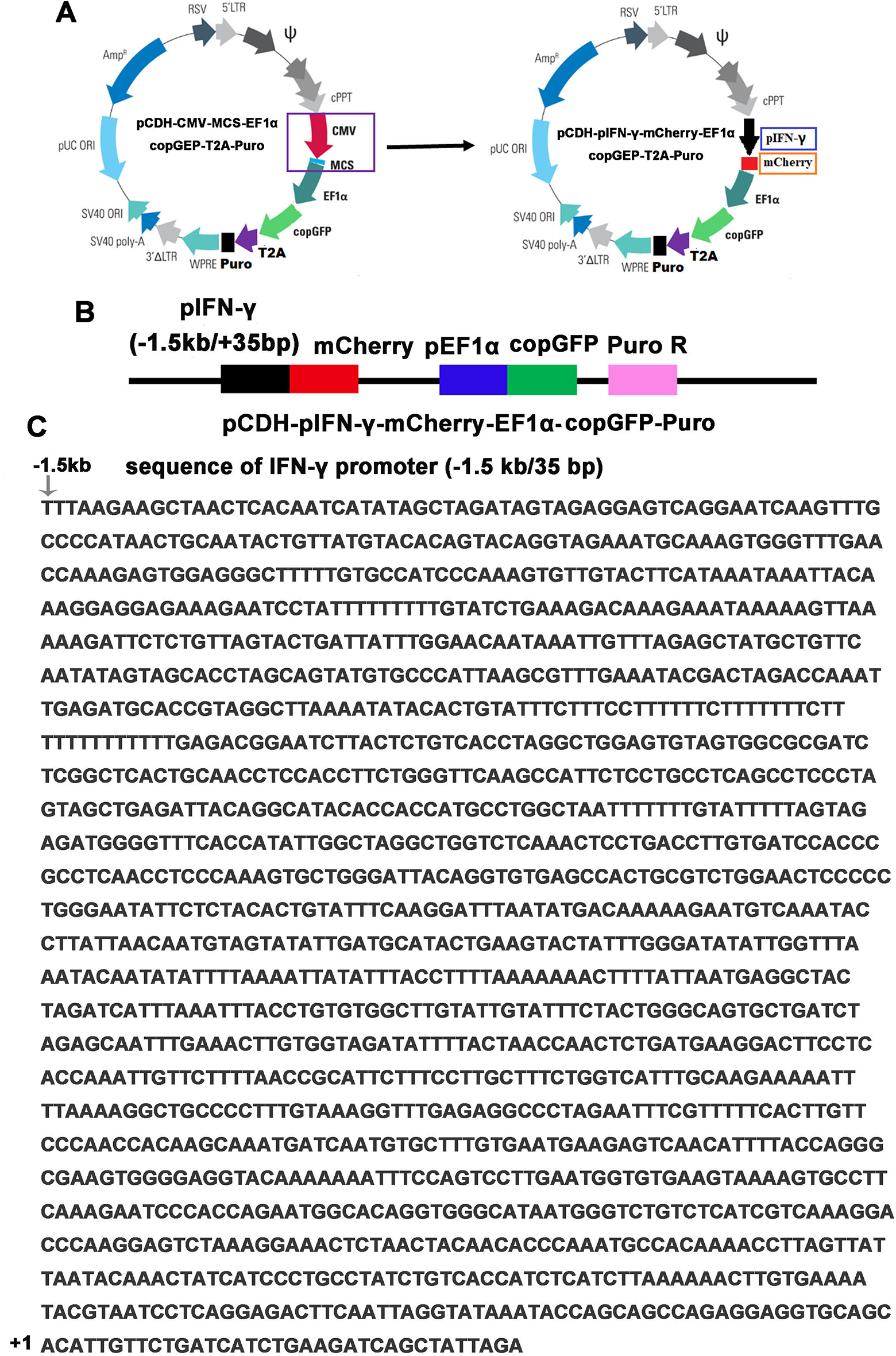
Construction of pIFN-γ-mCherry reporter lentivirus vector. (
Establishment of the Fluorescence Reporter Jurkat Cell Line
Lentivirus particles were produced by a three-plasmid package system. In short, the expression plasmid pCDH-pIFN-γ-mCherry-EF1α-copGFP-puro, packaging plasmid pCMV-dR8.91, and envelope plasmid pMD2.G were mixed gently and transfected into 293FT cells. As shown in Figure 2A , green fluorescence (copGFP) was constitutively expressed derived by the EF1α promoter, while red fluorescence (mCherry) under the control of the IFN-γ promoter was unobserved. At 48 h posttransfection, lentivirus was collected and used to infect Jurkat cells. At 72 h postinfection, 2 μg/mL puro was added into cell medium lacking penicillin/streptomycin to select lentivirus-infected Jurkat cells ( Fig. 2B ). Then a single-cell clone was picked up by infinite dilution assay, followed by an expansion culture to establish a stable expression cell line ( Fig. 2C ).
The detailed experimental procedure is shown as a schematic diagram in Figure 2D .
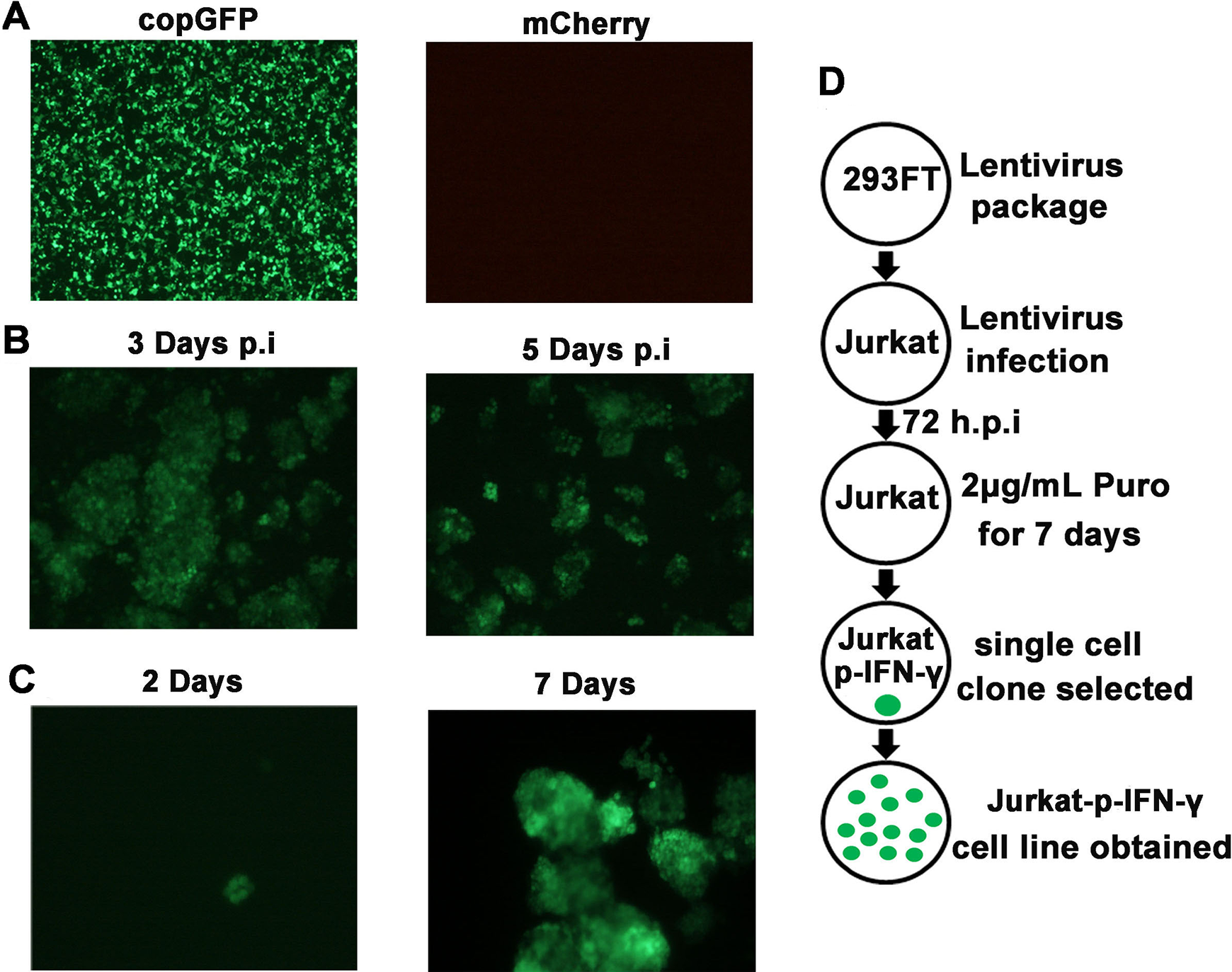
Establishment of fluorescence reporter Jurkat cell line. (
Performance of the High-Content Screening Assay
A screening effort based on the reporter cells was initiated to identify compounds that can stimulate the production of IFN-γ. Ninety-six-well cell culture plates were coated with 50 μg/mL PDL. A total of 5 × 103 cells were seeded in the PDL-coated plates and then treated with 20 μM compounds, with PMA and ionomycin as the positive control and DMSO as the negative control. Each compound was set in triplicate. The arrangement of compounds in the 96-well plate is shown in Figure 3A . After 24 h, the expression of the reporter gene mCherry and constitutive copGFP was monitored by the Cellomics ArrayScan VTI HCS Reader inautofocus mode. One hundred fields for each well were taken continuously, and the fluorescence intensities for mCherry and copGFP in each well were recorded automatically at the same time. Data including images and fluorescence intensity were collected and analyzed. The expression level of mCherry was normalized by copGFP. The Z′ factor 0.754745 was calculated based on the fluorescence intensity (mCherry/copGFP) ratio from the positive and negative controls. A Z′ value in the range of 1 ≦ Z′ < 0.5 predicates a quality screening model. 33 The cutoff horizontal line was set according to the result from the positive control, with dots above the line considered to be potent compounds. As shown in Figure 3B , two hits were captured in our model. The chemical structures for the two hits, Astragalus polyphenols and 6-shogaol, are shown in Figure 3C ,D . The fluorescent images from high-content screening assay are shown in Figure 3E . Obviously, red fluorescence (mCherry) was observed in the positive well, and Astragalus polyphenols and 6-shogaol in the treated wells, the results of which corresponded to the fluorescence intensity ratio.
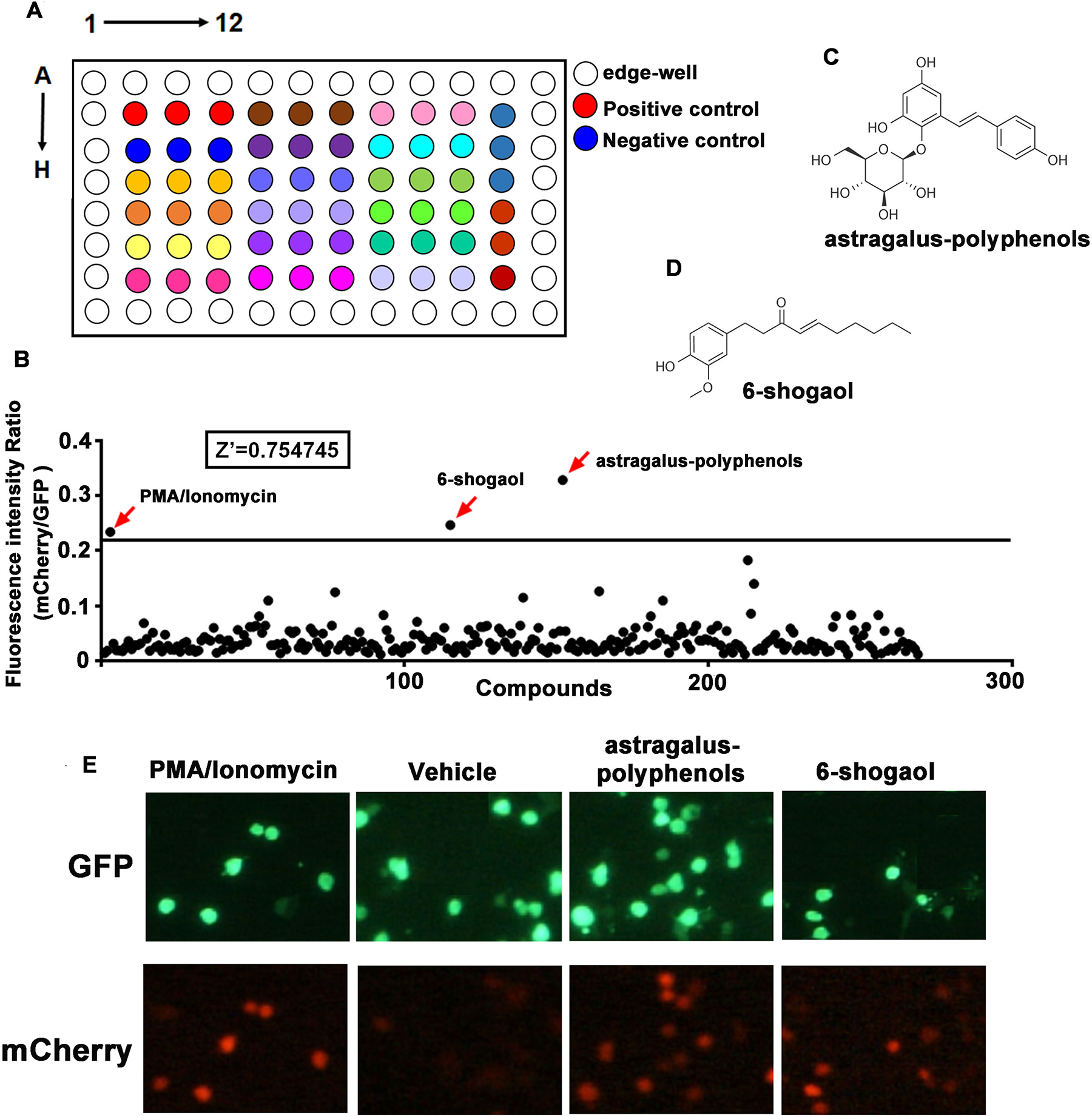
High-content screening for natural products in stimulating IFN-γ production. (
Astragalus Polyphenols and 6-Shogaol Stimulate the Production of IFN-γ
To further validate the activity of Astragalus polyphenols and 6-shogaol, a flow cytometry assay was performed to determine the mCherry expression level in live cells. As shown in Figure 4A , upon treatment of the indicated compounds, the positive ratio of mCherry live cells was 51.6% and 45.8% for Astragalus polyphenols and 6-shogaol treatment, respectively. Meanwhile, 22% and 38.5% dead cells were observed, among which 13.8% and 22.6% cells expressed or showed cross-talk with mCherry fluorescence upon treatment of Astragalus polyphenols and 6-shogaol, respectively. Thereafter, the cytotoxicity of Astragalus polyphenols and 6-shogaol was determined by MTS assay. As shown in Figure 4B , both compounds exhibited a slight cytotoxic activity in Jurkat, and the IC50 value of the two compounds was over 40 μM. As shown in the curve, the cell viabilities were approximately 86% and 75% at the concentration of 20 μM for the two compounds, respectively, the results of which corresponded with those of flow cytometry assay. Moreover, the transcription and expression level of IFN-γ were further detected by semiquantitative RT-PCR and ELISA. As shown in Figure 4C ,D , in wild-type Jurkat cells, both Astragalus polyphenols and 6-shogaol exhibited the potential to stimulate the expression of IFN-γ in a dose-dependent manner, indicating the real activity of the two hits from our screening model.
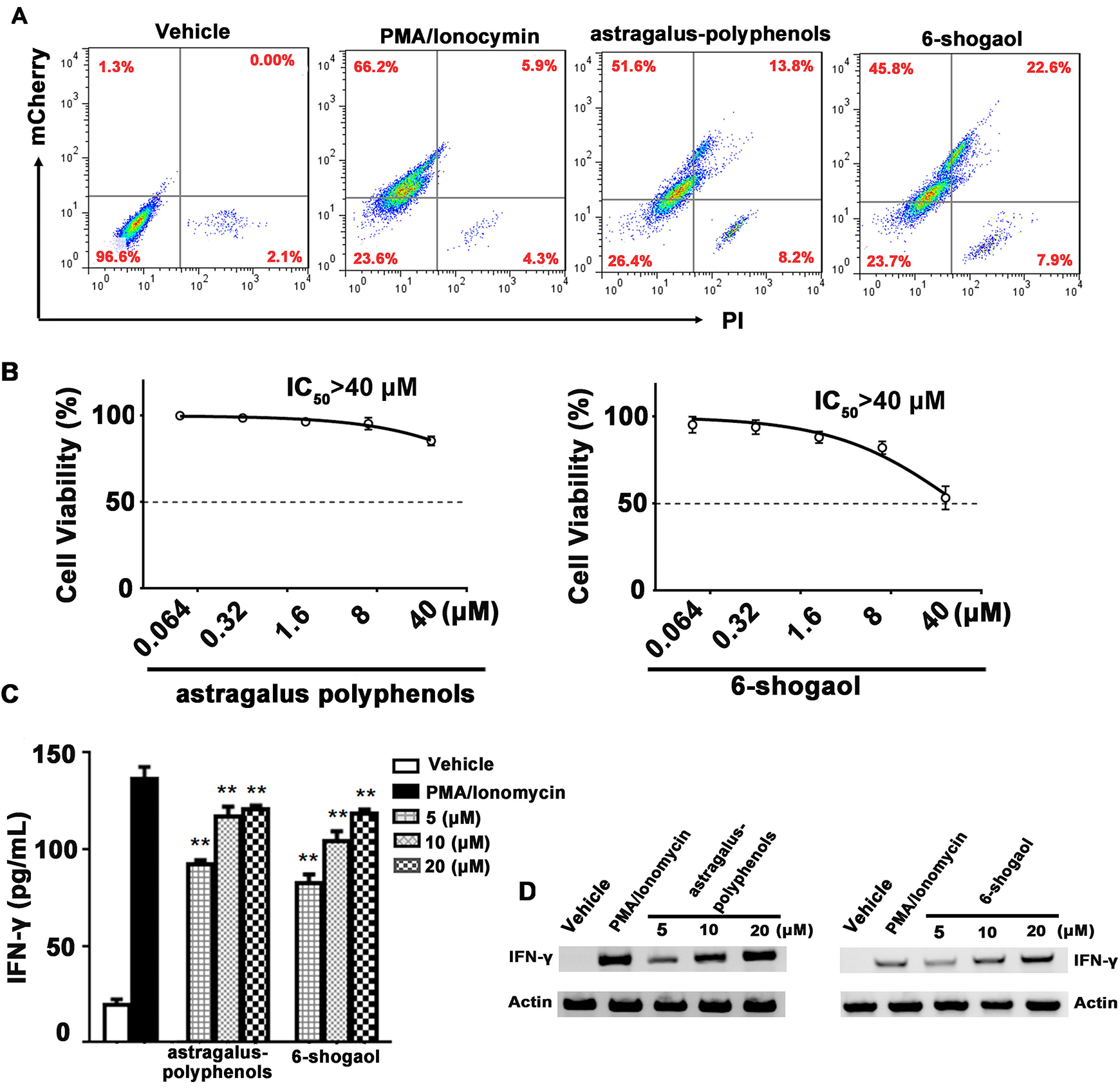
Astragalus polyphenols and 6-shogaol stimulate the expression of IFN-γ in a dose-dependent manner. (
Discussion
Small natural molecules are potential candidates for the discovery of novel drugs, especially for immunomodulatory medicine due to their relatively low side effects. As an immunoregulatory cytokine, IFN-γ plays an essential role in coordinating the innate and acquired immune responses. In animal models and clinical trials, IFN-γ showed great potential to enhance the host immune responses and therapeutic effects in vivo. 34,35 The recombinant human IFN-γ has been approved in clinical trials for the treatment of renal carcinoma and rheumatoid arthritis, and acts as an adjuvant for vaccine or chemotherapies. Therefore, natural products that can induce the production of IFN-γ may also serve as therapeutic agents.
Jurkat is an immortalized human T lymphocyte cell 36 and it has been widely utilized as a cell model to understand the immune responses associated with the signaling pathway for T cells in vitro. 37,38 Notably, Jurkat cells have also been used to develop the endogenous interleukin-2 reporter system based on the luciferase reporter system, the activity of which could be evoked by PMA/ionomycin as well. 39 In this study, we established a novel image-based, high-content screening technique for the discovery of novel natural products that can stimulate the expression of IFN-γ in Jurkat cells. The specific characteristic for the model was the application of dual fluorescent proteins, in which the mCherry gene was under the control of the IFN-γ promoter, with the copGFP gene constitutively expressed. In that case, the cell number can be normalized by copGFP, which made the data from digitalized fluorescence intensity more creditable.
After a primary screening, two hits, Astragalus polyphenols and 6-shogaol, were discovered. The activity of the two compounds was confirmed by flow cytometry assay. Both Astragalus polyphenols and 6-shogaol showed potential to promote the expression of IFN-γ in a dose-dependent manner. At the same time, the cytotoxicity of Astragalus polyphenols and 6-shogaol was also measured. Data suggested that both chemicals exhibited a slight cytotoxic activity, which was in accord with the results from the flow cytometry assay. It is noteworthy that the cytotoxic activity should be determined to exclude the false-positive chemicals, because parts of dead cells of unexpressed mCherry might also be detected due to a cross-talk effect.
Astragalus polyphenols are a natural product derived from the Astragalus herb, which exhibits good pharmacological and immunomodulatory effects. 40 It was reported that Astragalus polyphenols are a potent adjuvant for both the hepatitis B subunit and DNA vaccines. Multiple cytokines including IL-2, IL-4, and IFN-γ were evoked in immunized mice models, leading to the enhancement of both humoral and cellular immune responses. 41,42 As for 6-shogaol, it is a natural product from ginger, whose immunoregulatory effects, including anti-inflammation and anticancer properties, have also been widely reported. 43,44 Evidence shows that 6-shogaol can inhibit breast cancer cell invasion and exhibit an antagonistic effect in high-glucose-induced NKRP3 inflammasomes in artery smooth muscle cells. 45,46 Although both hits were not novel chemicals, and their immunomodulatory role as well as their capacities to induce the expression of IFN-γ have been extensively studied, it was still fortunate for us to detect them, which might help to validate the effectiveness of our screening model. In order to find novel potent chemicals, we need to expand the screening scale, but as least our primary screening data had encouraged us to continue the screening work.
Footnotes
Acknowledgments
We would like to thank Liang Gong for professional suggestion regarding the analysis of screening data.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported financially by the funding of the Key Research Project of Gannan Medical University (ZD201906), the Project of Jiangxi Provincial Department of Education (GJJ160988), the Project of Science and Technology plan of the Jiangxi Health Committee (20204503), and the Program of Science and Technology Guided plan of Ganzhou (GZ2019ZSF019).