Abstract
Cancer is the consequence of aberrations in cell growth or cell death. In this scenario, mitochondria and ion channels play a critical role in regard to cell proliferation, malignant angiogenesis, migration, and metastasis. In this review, we focus on Kv1.3 and specifically on mitoKv1.3, which showed an aberrant expression in cancer cells compared with healthy tissues and which is involved in the apoptotic pathway.
In recent years, mitoKv1.3 has become an oncological target since its pharmacological modulation has been demonstrated to reduce tumor growth and progression both in vitro and in vivo using preclinical mouse models of different types of tumors.
Keywords
Introduction
Ion channels are involved in several cellular processes, such as cell proliferation 1 and death, 2 mitogenesis, 3 and oncogenic transformation. 4 Today, we know that both plasma membrane5,6 and intracellular ion channels 7 are involved in cancer, and that pharmacological targeting of some of them has been proved to reduce tumor development and progression in vivo. 8 In this context, potassium channels—the most heterogeneous group for both their structure and functions—have been shown to be involved in several types of cancer. Their activity is tissue specific and can be modulated by synthetic blockers or natural peptides, as already used for cardiac diseases. For the reason stated above, targeting potassium channels in cancer therapy is likely to be a successful strategy, as well as using them as prognostic factors.9,10
Mitochondria are important organelles considered to be the center of energy production, by generating the cellular energy currency, adenosine triphosphate (ATP), and signaling organelles in regulating signal transduction. Their function is wide ranging and they are involved in several pathophysiological processes ranging from cell life to cell death. 11 Mitochondria are composed of two membranes, the outer and the inner membrane separated by an intermembrane space. The respiratory chain complexes are located in the mitochondrial cristae formed by invaginations of the inner mitochondrial membrane, and their remodeling is crucial for several cellular functions. 12 Furthermore, mitochondria are also the main site for reactive oxygen species (ROS) production and they are involved in several intracellular signaling cascades. Importantly, mitochondria have been associated with the induction and regulation of programmed cell death; both the extrinsic and intrinsic apoptotic pathways are mediated by these organelles. 13
It has been demonstrated that mitochondrial ion channels are involved in several diseases, such as cancer,14–18 neurodegeneration,19–24 and ischemia,25–27 and their pharmacological targeting will be a promising way to treat these diseases. 28 In particular, for some of these ion channels in vivo evidence of their efficacy in preclinical models of cancers has been reported. 8 In this review, we focus on the mitochondrial potassium channel Kv1.3, which is known to have a role as an oncological target in cancer treatment. For the other mitochondrial ion channels, see the complete reviews in Pardo and Stühmer, 9 Bachmann et al., 14 and Szabo and Zoratti. 29
Mitochondrial Ion Channels
The mitochondrial inner membrane has long been considered to be poorly permeable to cations and anions, since the strict control of its permeability is necessary for efficient energy production. Various ion channels, along with exchangers and uniporters, are present in the mitochondrial inner membrane. They are present in extremely low abundance and are open for very short times to maintain low permeability to the ions required to exploit the proton-motive force for ATP generation. Indeed, an important role for mitochondrial function and regulation is mediated by mitochondrial ion channels, which are located in both the outer and inner mitochondrial membranes. 29
Concerning potassium fluxes, it has been shown that several potassium channels are found in the inner mitochondrial membrane: ATP-dependent potassium channel (KATP),29–31 big-conductance calcium-activated potassium channel (BKCa),32,33 intermediate-conductance calcium-activated potassium channel (IKCa),34,35 small-conductance calcium-activated potassium channel (SKCa), 36 two-pore potassium channel (TASK-3), 37 pH-sensitive potassium channel, 38 and finally, voltage-gated mitochondrial potassium channels Kv1.3 and Kv1.5.39–41 The above-listed mitochondrial potassium channels are also expressed in the plasma membrane, in different types of tissues and cells, even if the mechanism mediating the dual targeting has not been elucidated yet. 14
Mitochondrial Kv1.3
Voltage-gated potassium channels are the most abundant ion channels and are made up of six transmembrane domains. 42 They are divided into 12 families (Kv1–12). 43 For each family, the nomenclature refers to the homologous gene found in Drosophila melanogaster. The voltage-gated potassium channel Kv1.3 is encoded by the intron-less gene KCNA3, located on chromosome 1, p21-p13.3, and its product is 575 amino acids long. 44 Kv1.3 has been shown to have an altered expression in several cancer tissues compared with normal ones, like in mouse melanoma, or in human prostate cancer, breast cancer, B-cell lymphoma, B-cell chronic lymphocytic leukemia (B-CLL), gastric and pancreatic tumors, and lung cancer, where it could give an advantage to cancer cells, enhancing their proliferation, migration, and metastization. 9
The presence of a mitochondrial counterpart of the Kv1.3 channel has been seen in human B and T lymphocytes, macrophages, postsynaptic medial nucleus of the trapezoid body (MNTB) neurons, gerbil hippocampus, PC-3 prostate cancer, MCF-7 breast adenocarcinoma, SAOS-2 osteosarcoma, pancreatic ductal adenocarcinoma (PDAC) Colo357 cells, and B16F10 mouse melanoma cell lines. MitoKv1.3 shows the same electrophysiological properties as the correlated plasma membrane channel, suggesting that both plasma membrane and mitochondrial channels are encoded by the same gene ( Fig. 1 ). 13
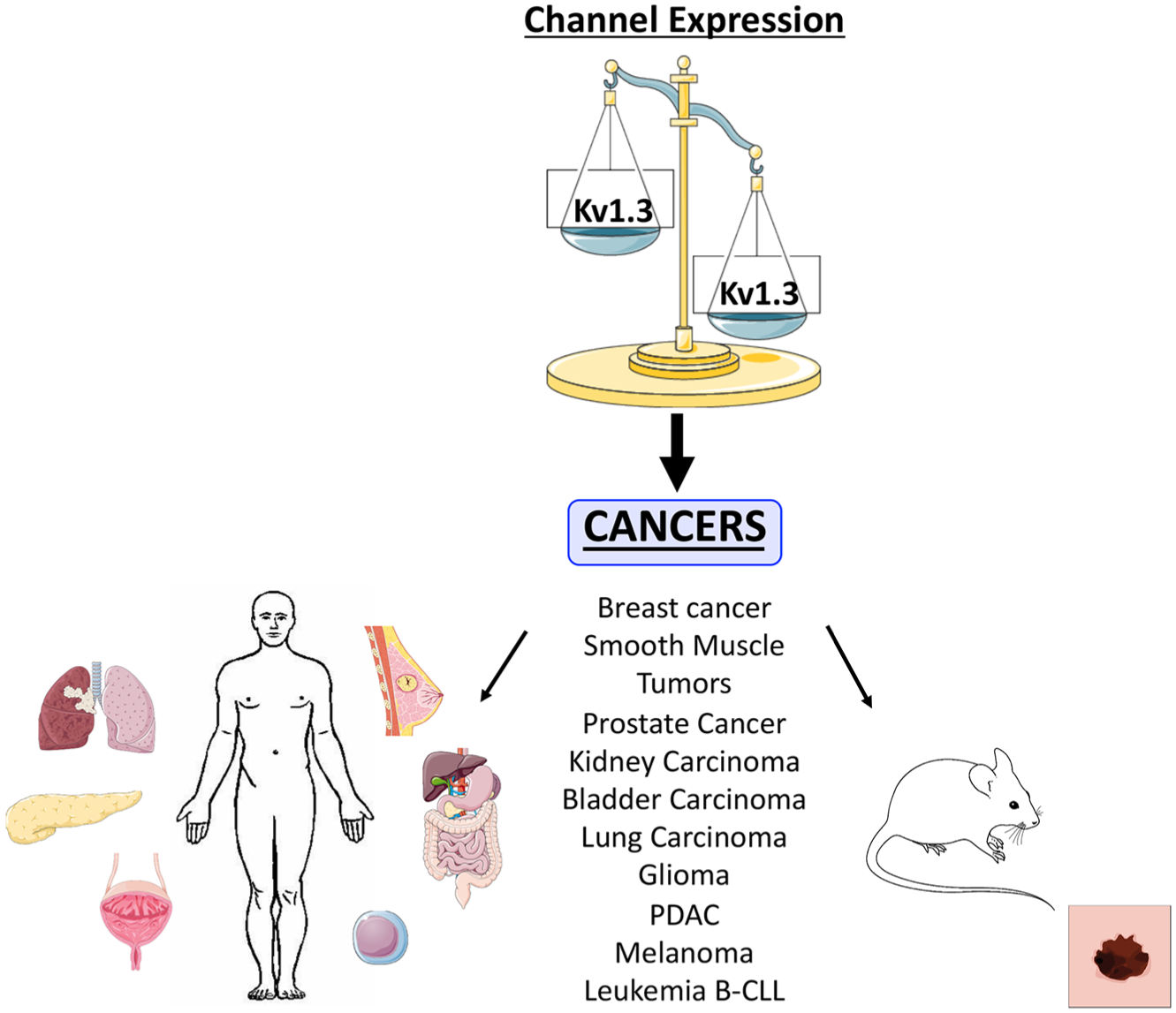
Kv1.3 expression in cancer. Potassium channel Kv1.3 shows an altered expression in several tumor tissues/cells compared with healthy ones.
MitoKv1.3 ( Fig. 2A ) is the most studied member of this family, since it has been discovered in the inner mitochondrial membrane, and its function seems to be associated with the regulation of organelle-mediated apoptosis.39,45–47 Indeed, mitoKv1.3 in lymphocytes is a target of the pro-apoptotic Bcl-2 family member Bax. Szabò and colleagues45,48 reported that Bax physically interacts with the mitoKv1.3 protein and inhibits channel activity via a toxin-like mechanism (as CTX, MgTx, or ShK).45,49–52 The interaction model suggests that the Bax monomer juts out from the outer mitochondrial membrane into the intermembrane space, 53 and the amino acid in position 128, a lysine residue, mimics the action of the critical lysine in Kv1.3-blocking toxins by binding to the ring of four aspartate residues of the channel vestibule. 54 The consequences of Bax-mediated inhibition include hyperpolarization, ROS release, depolarization, and cytochrome c release, 39 effects that are abrogated if this lysine is mutated to a glutamic acid ( Fig. 2B ). 46
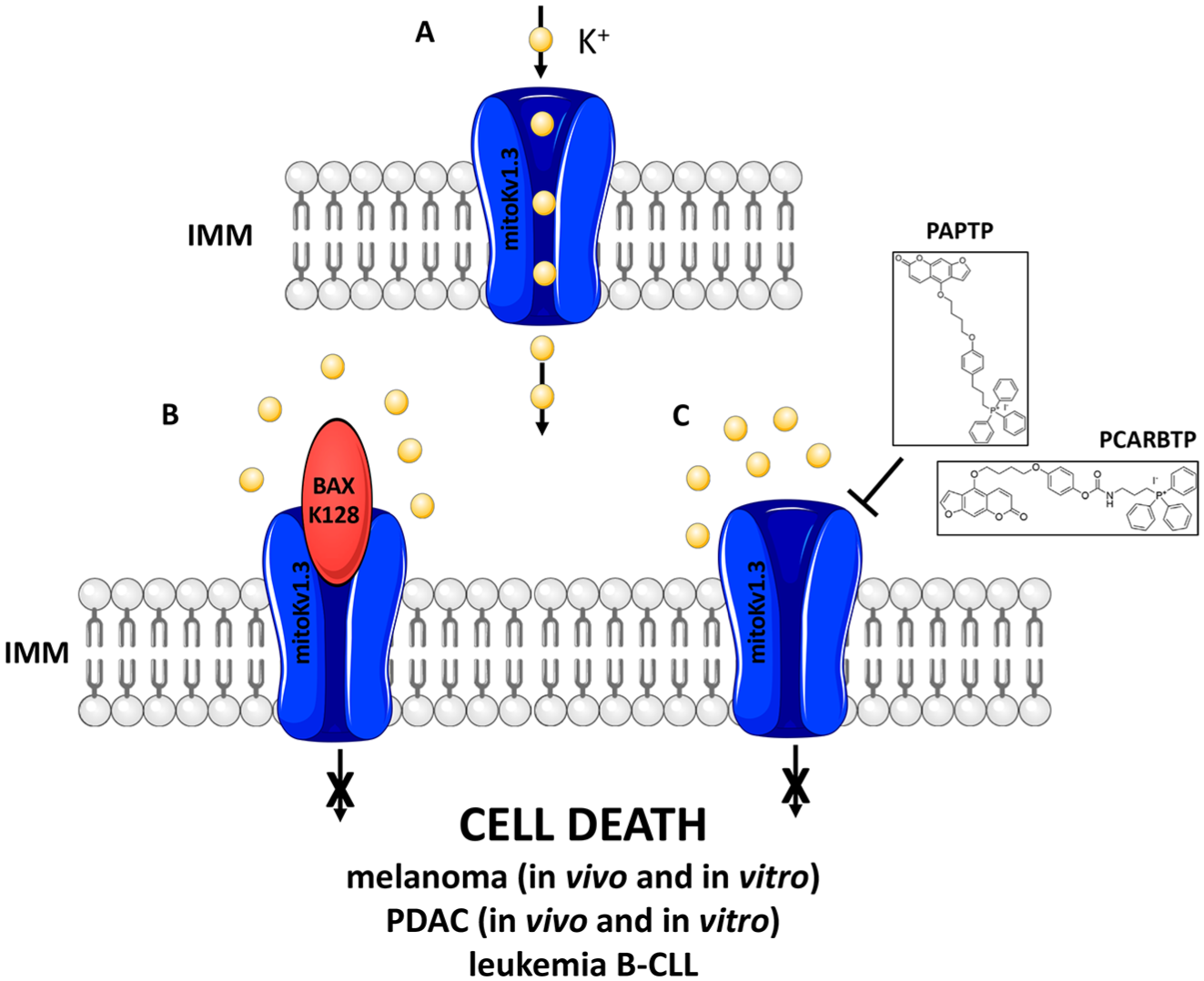
Targeting Kv1.3 to treat cancer. (
Mimicking this interaction with Bax, it has been demonstrated in vitro, ex vivo, and in vivo that pharmacological inhibition of mitoKv1.3 by membrane-permeant inhibitors Psora-4, PAP-1, and clofazimine can selectively kill cancer cells, sparing healthy ones in several tumors, such as mouse melanoma, human CLL, and human PDAC. After the potassium flux to the matrix was blocked, mitochondrial membrane hyperpolarization was reported and consequently increased ROS production was shown. ROS, in turn, can activate the mitochondrial permeability transition pore (mPTP), causing mitochondrial membrane depolarization and favoring cytochrome c mobilization from the cristae and its release from the intermembrane space to the cytosol, where it contributes to activating the apoptosome.55–58 Recently, to specifically target mitoKv1.3 without affecting the other subcellular isoforms, two new mitochondria-targeted Kv1.3 inhibitors, namely, PAPTP and PCARBTP, have been synthetized. These new drugs are lipophilic and positively charged due to the presence of a triphenylphosphonium group (TPP+), which drives them into the nega-tive mitochondrial matrix (–180 mV). In these two compounds TPP+ is linked to the molecule by a chemically stable C–C bond (PAPTP) or to the PAP-1 core via a carbamic ester bond O–C(O)–N (PCARBTP). Indeed, PAPTP was considered a drug, while PCARBTP was developed as a pro-drug. PCARBTP is liable to undergo hydrolysis in physiological conditions, releasing PAPOH, which differs from PAP-1 only by the presence of a hydroxyl group. Conversely, PAPTP is very stable and is not degraded in cells. 59 In addition to PAPTP and PCARBTP, other mitochondria-targeted derivatives were synthetized starting from either Psora-4 or PAP-1. 60 Their capability to kill pathological cells in vitro was lower compared with PAPTP and PCARBTP; those were instead used during the in vivo experiments in preclinical mouse models described below.
As expected, these compounds are rapidly taken up in the mitochondria and block mitoKv1.3, leading to mitochondrial dysfunction ( Fig. 2C ). These compounds are able to mediate the same cascade of events that was already demonstrated using the membrane-permeant inhibitors: mitochondrial membrane hyperpolarization, ROS production, mPTP opening favoring the release of cytochrome c, and induction of apoptosis in cancer cells. These drugs have shown an in vitro cytotoxic effect against Kv1.3-expressing cancer cell lines, such as mouse melanoma, human leukemia, human PDAC, and mouse and human glioma.59,61
Importantly, the specificity for cancer cells versus normal ones has also been demonstrated. Primary human skin fibroblasts as well as healthy human B cells did not undergo cell death. A synergistic effect has been shown to be involved in this specificity: mitoKv1.3 expression is necessary to induce the cascade of events downstream of mitochondrial potassium flux inhibition, but it has been illustrated that an altered redox state should be present in the cell, since the induced alteration of ROS production leads to activation of the apoptotic cascade. Indeed, normal cells have a lower ROS production, so an increase of ROS induced by mitoKv1.3 inhibition did not reach the critical threshold needed to trigger cell death. A slight increase in mitochondrial ROS production is known to favor cell survival and proliferation at low concentration. Conversely, cancer cells exhibit a higher redox state that reduces the necessary amount of ROS to be produced to reach the critical amount that leads to cell death.56,59
These compounds were also able to reduce tumor volume in vivo up to 90% in preclinical orthotopic mouse models for melanoma and PDAC. Importantly, no side effects have been observed after the treatment of animals with these drugs: the immune system subpopulations, cardiac function, and tissue histology did not display any alterations. 59
While these drugs at high concentrations kill cancer cells, lower concentrations (100 nM) of PAPTP and PCARBTP do not impact cell survival but are able to trigger an increase in cell cycle progression, especially affecting the percentage of cells in the S phase. 62
Other Ways to Modulate Kv1.3
Since the discovery of the important role that mitoKv1.3 plays in cell death regulation in cancer cells, this channel has gained recognition in the oncological field. As a result, targeting of the channel represents a real opportunity to selectively eliminate cancer cells. In fact, not only does the channel represent a good target, but also its interactors.
To our knowledge, little information is available about the intracellular targeting of this voltage-gated potassium channel. Modulation of Kv1.3 expression, as well as its potential to reach cellular membranes, could be a possible way to tune its activity. Recently, plasma membrane trafficking of Kv1.3 has been related to the caveolae. Caveolae are found in the plasma membrane of several cell types and tissues.63–66 They can be described as small, bulb-shaped invaginations of the plasma membrane, but the morphology depends on the physiological status of the cells; they can form transport vesicles67,68 or tubular structures 69 or be flat. 70 Caveolae regulate several important cellular processes, such as cellular signaling, cholesterol homeostasis, and mechanosensory response.71–76 These proteins are encoded by the caveolin gene family: caveolin-1, -2, and -3. Caveolin-1 and -2 are co-expressed and form a heteroligomeric complex in endothelial cells, fibroblasts, adipocytes, and smooth muscle cells, while caveolin-3 is specific for muscle, for example, skeletal muscle, diaphragm, and heart.77–80
These proteins form a hairpin-like structure with a hydrophobic domain and the hydrophilic N-terminus and C-terminus, which are on the cytosolic side of the plasma membrane. 81 The N-terminal portion is characterized by a caveolin scaffolding domain (CSD; residues 81–101) that is critical for caveolin oligomerization, caveola formation, and interaction with the putative caveolin binding domains (CBDs), a consensus sequence ФxФxxxxФxxФ (where Ф is an aromatic residue and x can be any amino acid) located in target proteins.82–85 Several studies have ascribed that caveolins interact with many different signaling proteins, such as endothelial nitric oxide synthase, Src kinases, or protein kinase C (PKC), targeting them into caveola platforms.85–87 Only recently, using co-immunoprecipitation and Förster resonance energy transfer (FRET) experiments, was a molecular interaction between Kv1.3 and Cav-1 found, and electron micrographs indicated that Kv1.3 was recruited into caveola-like structures in the presence of Cav-1. The data indicate that Cav-1 interacts with Kv1.3 via a CBD signature, located at the N-terminus of the channel in close proximity to the T1 tetramerization domain, and the Kvβ subunit interaction signature, and that this association is essential for the lipid raft targeting of the Kv1.3 and establishes, for the first time, that Kv1.3 lipid raft targeting happens via a direct interaction where Cav-1 recruits the channel inside caveola structures, altering channel membrane dynamics, stability, and activity. 88
Furthermore, two mechanisms can explain nonrandom distribution of ion channels. The first is the association with lipid rafts where receptor and protein kinase molecules reside; 89 the second is controlled by direct protein–protein interactions. Channels can be clustered by interacting with the extracellular matrix and/or by anchoring the channels to cytoskeletal elements.90,91 Panyi and colleagues described using two different methods, confocal laser scanning microscope (CLSM) images and FRET measurements, in the plasma membrane of human Jurkat cells and the colocalization of the Kv1.3 and CD3, an integral component of the T-cell antigen receptor (TCR) complex. 92 These results indicate that the triggering of the TCR/CD3 complex is directly linked to a change in the Kv1.3 channel, or that an alteration of ion passages and changes in membrane potential influence synaptic functions. Finally, Hajdu and colleagues 93 demonstrated that cortactin, a factor initiating the actin network that controls the surface expression of ion channels, is also able to regulate the membrane mobilization of Kv1.3 channels.94–97 Proximity ligation assay (PLA) experiments in T cells showed that the lack of a HS1 binding site does not preclude the channel from entering the immunological synapse (IS), suggesting that HS1 may not affect Kv1.3 diffusion into the IS. The results presented raise the possibility that the association between HS1 and Kv1.3 channels could shape the outcome of the signaling pathways during T-cell activation.
Besides the targeting of this channel, the known Kv1.3 partners can also be modulated to adjust Kv1.3 activity. Kv1.3, like the other Kv, forms both homotetrameric and heterotetrameric channels, composed of alpha subunits, which can be modulated by beta subunits. The assembly of these modulations (on both alpha and beta subunits) leads to a great assortment of potassium currents with distinct biophysical and pharmacological properties. Despite this variety, it was observed that the range of potassium currents, recorded mainly in neuronal preparations, did not match with the known Kv channels. The identification of hetero-oligomeric channels composed of different alpha subunits in mammalian brain98,99 justifies the huge amount of different currents recorded. Coleman and colleagues 100 identified the subunit composition of Kv channels in bovine cerebral cortex. Sequential immunoprecipitations allowed the identification of the first Kv heterotetramer of alpha subunits. It was a crucial step, since Kv channels, among the others, are involved in many neurological diseases, like episodic ataxia and epilepsy. A Kv1.3/1.4/1.1/1.2 tetramer was identified in the gray matter, and this combination is different from the previously identified Kv1.3/1.4/1.2/1.6. 101 The function of these heterotetramers is still unknown, but they are expected to show different electrophysiological profiles. The Kv subunit composition defines the activity, pharmacological characteristics, and membrane expression, but the function and structures of the heterotetrameric channel are still unknown.
A little more in this field is known about the interactions between Kv1.3 and Kv1.5. In the immune system, there are various Kv channels, but the most abundant is Kv1.3, which controls membrane resting potential, T-cell activation, and proliferation. Kv1.3, in the context of the plasma membrane of the cells, is located in a particular membrane domain: the lipid rafts that collect in immunological synapses during lymphocyte activation. Kv1.3 localization is well known, while Kv1.5 localization is still unconfirmed, but both proteins have been found in raft microdomains, and their functions can be influenced by lipid–protein interactions. They are able to co-assemble in heteromultimeric complexes channels that exhibit different locations and targeting. Kv1.3 and Kv1.5 co-assemble in heteromultimeric complexes in the plasma membrane and in the mitochondria, and the ratio between the two is modulated by cytokines, provoking different phenotypes. Immunoprecipitation showed that both Kv1.3 and heteromeric channels associate with caveolar raft domains. Homomeric Kv1.3 channels display higher association with caveolin traffic. Furthermore, FRAP analysis indicated higher mobility for hybrid Kv1.3/Kv1.5 than Kv1.3 homotetramers, suggesting that heteromers target distinct surface microdomains. In particular, a high ratio of Kv1.5 prevents lipid raft positioning of the Kv1.3. During macrophage activation the Kv1.3/Kv1.5 ratio increases and the heterotetramer accumulates on the lipid raft. 40 The different expression and modulation of the isoforms can affect the number of channels in the cell surface, modifying the membrane excitability and signaling. Using macrophages expressing both Kv1.3 and Kv1.5 channels, we demonstrated another possible way to target these ion channels. Indeed, we tried to target macrophages in order to eliminate tumor-associated macrophages, which support tumor-mediated inflammation and contribute to tumor progression. 102 However, we succeeded in inducing cell death only when we co-administrated both Kv1.3 inhibitors with Kv1.5 ones, namely, DPO-1.41,103 This combined treatment is a possible example in how it is possible to modulate ion channel activity.
Finally, KCNEs are regulatory proteins that interact as beta subunits with voltage gate potassium channels. They are composed of a single transmembrane domain and are usually associated with Kv7 channels. These beta subunits have a dominant negative role for various Kv channels, via gating inhibition. In this context, KCNE4, which is present in the immune system, when co-assembled with Kv1.3, exerts great influence by modulating the gating kinetics, trafficking, and stability of the channel complex.104,105 KCNE4, which is present in the immune system, might act as a very powerful dominant-negative regulatory subunit of Kv1.3 channels in leukocytes. 106 When Kv1.3 and KCNE4 interact, a marked retention within the endoplasmic reticulum (ER) is noted, impairing its surface expression. 104 It has been demonstrated that Kv1.3 is not a specific signature for the KCNE4 binding, while tertiary conformation of the C-terminus of the Kv1.3 channel is responsible for the association with this beta subunit. When Kv1.3 and KCNE4 interact, a marked ER retention is noted, which impairs membrane targeting. By interacting with the C-terminus of Kv1.3, this ancillary peptide masks a di-acidic forward trafficking motif, which is important for COPII anterograde transport to the plasma membrane. A major consequence of this association is the decrease in functional channels on the surface, leading to a massive intracellular retention. KCNE4 exerts important influences over Kv1.3, and the latter is involved in immune response. Therefore, the reasonable conclusion is that this ancillary peptide participates in the immune response modulation. 107
Kv1.3 is also present in the nuclear membrane of various cancer cell lines and in human brain tissue. The role of Kv1.3 in the nuclear membrane was examined by blocking the activity of the channel with a specific Kv1.3 inhibitor, MgTx. The inactivation of the channel induces the phosphorylation of CREB, an important transcriptional factor. Also, C-fos, an oncoprotein that is a component of the activator protein-1 transcription factor complex, is increased by blocking of the channel by PAP-1, a specific inhibitor of the Kv1.3 channel. CREB and C-fos are involved in cell growth and proliferation, and these observations confirm previous data, showing that silencing of the Kv1.3 or its inhibition has antiproliferative effects. In the nucleus, Kv1.3 also interacts with the UBF1 transcription factor, which is involved in RNA polymerase function, participating in ribosomal RNA synthesis. It seems that Kv1.3 has a role in the activation of gene expression of specific transcription factors, and these observations could be related to Kv1.3 involvement in both proliferation and apoptosis processes. 108
Human beta-defensin-2 (hBD-2) is a cysteine-rich antimicrobial peptide made by neutrophils, and it was identified as a toxin-like endogenous Kv1.3 inhibitor that can interact with the pore region. But hBD-2 is not only a pore-blocking toxin; it seems to act as a gating modifier and shifts the conductance–voltage relationship curve of the Kv1.3 channel. When hBD-2 interacts with the channel pore, it blocks the current through the channel and alters Kv1.3 activation kinetics, via voltage-dependent inhibition. 109 Notably, hBD-2 interacts with the pore domain of the channel to inhibit the current passage, but it was also noted that the S1–S2 linker of the channel is critically involved in the modifications of the Kv1.3 gating. The involvement of S1–S2 in toxin binding has never been observed. Interestingly, hBD-2 is a positive-charge peptide, which seems to change the Kv1.3 conformations through the electrostatic repulsion with voltage sensor domain S4.
Discussion and Conclusions
Among the potassium channels, Kv1.3, and in particular its mitochondrial counterpart mitoKv1.3, has been shown to have the classical feature of an oncological target: (1) its expression can regulate an important cellular process such as cell death; (2) its expression is altered in cancer cells versus healthy ones; (3) some specific modulators are available and have been tested in vivo in mouse preclinical models ( Table 1 ); and (4) several intracellular trafficking pathways and interactors could be targeted to regulate its function.
Membrane-Permeant Kv1.3 Inhibitors.
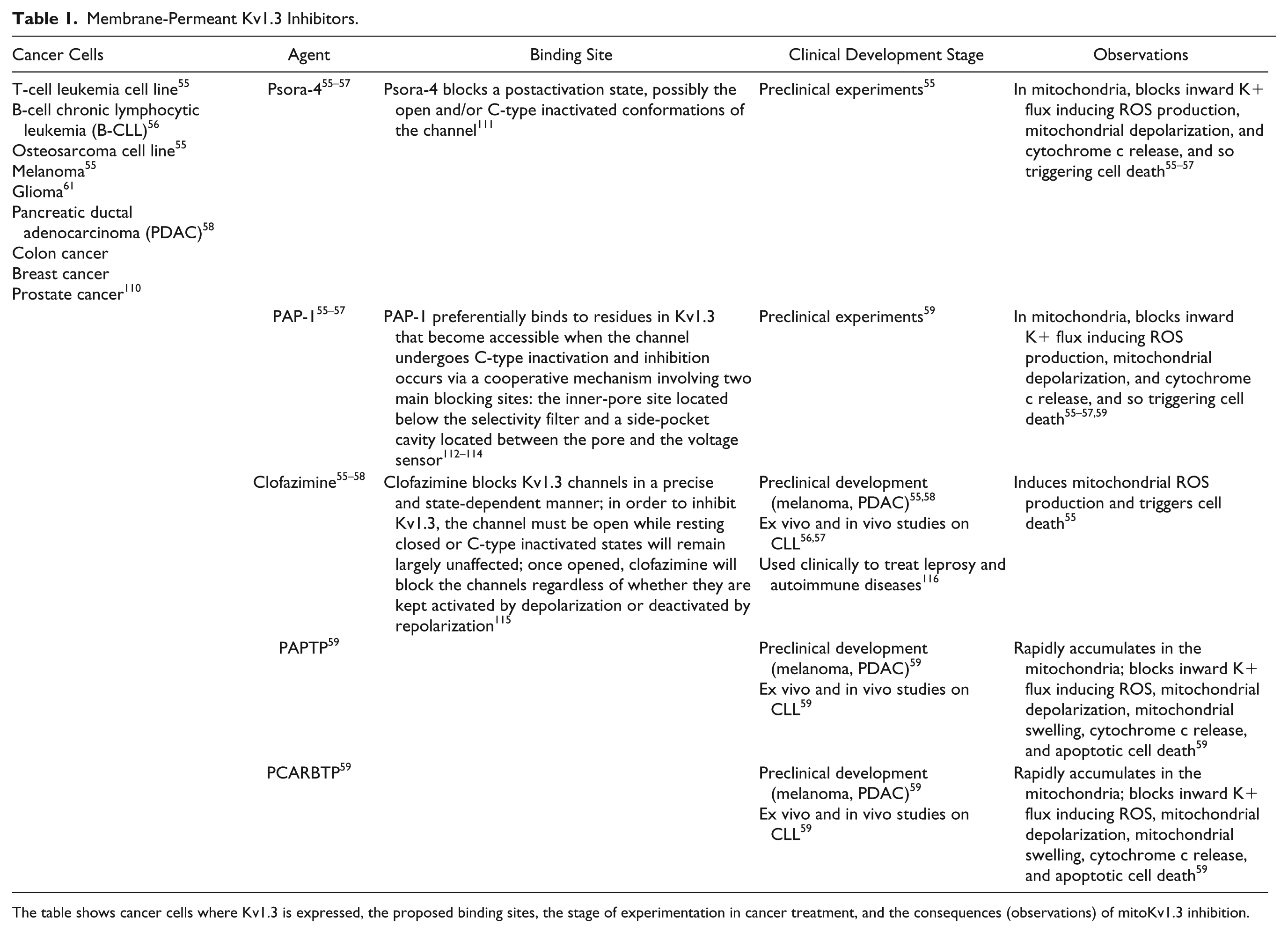
The table shows cancer cells where Kv1.3 is expressed, the proposed binding sites, the stage of experimentation in cancer treatment, and the consequences (observations) of mitoKv1.3 inhibition.
As discussed in this review, targeting mitochondrial Kv1.3 would lead to cancer cells death, as demonstrated with the new membrane-permeant inhibitors that we have recently developed. 59 In this context, blocking potassium fluxes through the inner mitochondrial membrane would impair mitochondrial respiratory chain activity, finally causing cell death. The synergistic action of the redox imbalance and the higher expression of this potassium channel in cancer cells prevented any side effect of mitoKv1.3 blockers on healthy cells. The strong expression of potassium channels in the immune cells, as well as the role of these ion channels in heart function, would suggest that we should clearly check whether possible adverse events are induced by any further developed drugs in this field, to not only reduce primary tumors but also impact the spreading of the metastasis, even if Kv1.3 expression is still debated in this field. Nevertheless, the data obtained up to date strongly encourage the effort to design new compounds able to modulate mitochondrial potassium fluxes and also to further study their possible use as prognostic factors in oncology. Conversely, it is important to consider that not only inhibition but also activation of mitochondrial potassium fluxes would have a great benefit in cancer biology. 13
In summary, mitoKv1.3 is the prototype of a good candidate to develop new and improved specific drugs or alternatively to use some of the drugs already available in the clinic. In this way, this research can be applied for the benefit of humans to hopefully eradicate tumors in cancer patients, as has been demonstrated with preclinical models.
Footnotes
Acknowledgements
The authors are grateful to Italian Association for Cancer Research (AIRC), to the EMBO, and to the Department of Biology of the University of Padova.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: L.L. is the recipient of a young researcher grant (no. GRIC12NN5G) and a PRID 2017 (no. BIRD162511) from the University of Padova and is grateful to EMBO for a short-term fellowship (ASTF 233-2014).
