Abstract
The upstream mechanobiological pathways that regulate the downstream mineralization rates in periodontal tissues are limitedly understood. Herein, we spatially colocalized and correlated compression and tension strain profiles with the expressions of mechanosensory ion channels (MS-ion) TRPV4 and PIEZO1, biometal zinc, mitochondrial function marker (MFN2), cell senescence indicator (p16), and oxygen status marker hypoxia-inducible factor-1α (HIF-1α) in rats fed hard and soft foods. The observed zinc and related cellular homeostasis in vivo were ascertained by TRPV4 and PIEZO1 agonists and antagonists on human periodontal ligament fibroblasts ex vivo. Four-week-old male Sprague-Dawley rats were fed hard (n = 3) or soft (n = 3) foods for 4 wk (in vivo). Significant changes in alveolar socket and root shapes with decreased periodontal ligament space and increased cementum volume fraction were observed in maxillae on reduced loads (soft food). Reduced loads impaired distally localized compression-stimulated PIEZO1 and mesially localized tension-stimulated TRPV4, decreased mitochondrial function (MFN2), and increased cell senescence in mesial and distal periodontal regions. The switch in HIF-1α from hard food–distal to soft food–mesial indicated a plausible effect of shear-regulated blood and oxygen flows in the periodontal complex. Blunting or activating TRPV4 or PIEZO1 MS-ion channels by channel-specific antagonists or agonists in human periodontal ligament fibroblast cultures (in vitro) indicated a positive correlation between zinc levels and zinc transporters but not with MS-ion channel expressions. The effects of reduced chewing loads in vivo were analogous to TRPV4 and PIEZO1 antagonists in vitro. Study results collectively illustrated that tension-induced TRPV4 and compression-induced PIEZO1 activations are necessary for cell metabolism. An increased hypoxic state with reduced functional loads can be a conducive environment for cementum growth. From a practical standpoint, dose rate–controlled loads can modulate tension and compression-specific MS-ion channel activation, cellular zinc, and HIF-1α transcription. These mechanobiochemical events indicate the plausible catalytic role of biometal zinc in mineralization, periodontal maintenance, and dentoalveolar joint function.
Introduction
Sustained mechanical loads (chewing and therapeutic) on the periodontal complex affect the turnover rate of the innervated and vascularized periodontal ligament (PDL), and volume fractions and mineral densities of alveolar bone and cementum, and the PDL-space. These physicochemical changes significantly influence the overall shapes of the alveolar socket and root (Niver et al. 2011). Significant changes in shapes (forms) affect mechanically coupled fluid flow and overall biomechanics of the dentoalveolar joint. From a multiscale biomechanics point of view, the effects of these physicochemical changes are consistently observed as shifts in physical deformations and shear flow of blood and interstitial fluids within the periodontal complex (Jang et al. 2021). A plethora of literature illustrates uniform rates of mesial mineral formation and distal resorption in the periodontal complex of rodents (Sicher and Weinmann 1944; Wang, Ustriyana, et al. 2022). However, it is important to note that the biomechanically activated upstream signaling pathways that regulate downstream biological events, resulting in mineral formation and resorption in the periodontal complex, are not well understood.
Joint biomechanics triggers ultrarapid mechanotransduction through tissue-specific mechanosensory ion (MS-ion) channels (Botello-Smith et al. 2019; Cox et al. 2019). Two such MS-ion channels, PIEZO1 and transient receptor potential vanilloid 4 (TRPV4), were identified in the dentoalveolar joint (O’Conor et al. 2014; Zhang et al. 2022). MS-ion channels sensitive to divalent cations calcium (Ca2+), magnesium (Mg2+), and zinc (Zn2+) have been shown to control downstream effector molecules that affect nucleation and dissolution of minerals in bones (Mayer et al. 1994; Kara et al. 2009; Sun et al. 2019; Wang, You, et al. 2022), cartilage (O’Conor et al. 2014), tendons (Ji and McCulloch 2021), and the PDL (Jiang et al. 2021). As compared with the advanced research on Ca2+ and Mg2+ metabolism and their presence in mineralized tissues, if or how Zn2+ concentrations fluctuate in response to chewing magnitude and frequency and sequential downstream intracellular events in the periodontal complex are not known. In particular, the conversion of magnitude and frequency of chewing loads on the dentoalveolar joint into intracellular tissue–specific biochemical signals and anatomy-specific mineralization in a periodontal complex remains unknown.
Materials and Methods
For detailed materials and methods, see Appendix.
In Vivo: Specimens from Preclinical Animal Models
The chewing regimen of Sprague-Dawley male rats (Charles River Laboratories) was regulated as stated previously (Niver et al. 2011). Six 3-wk-old male rats (developing) were fed hard food (HF) pellets (PicoLab Rodent Diet 20 5053; LabDiet) for 1 wk and then randomly separated into 2 groups to be given either HF (n = 3) or soft food (SF; n = 3) for an additional 4 wk (Niver et al. 2011) (Fig. 1A). Rats were euthanized at 8 wk per the ARRIVE guidelines (Animal Research: Reporting of In Vivo Experiments; Institutional Animal Care and Use Committee protocol AN183106-03, University of California San Francisco).
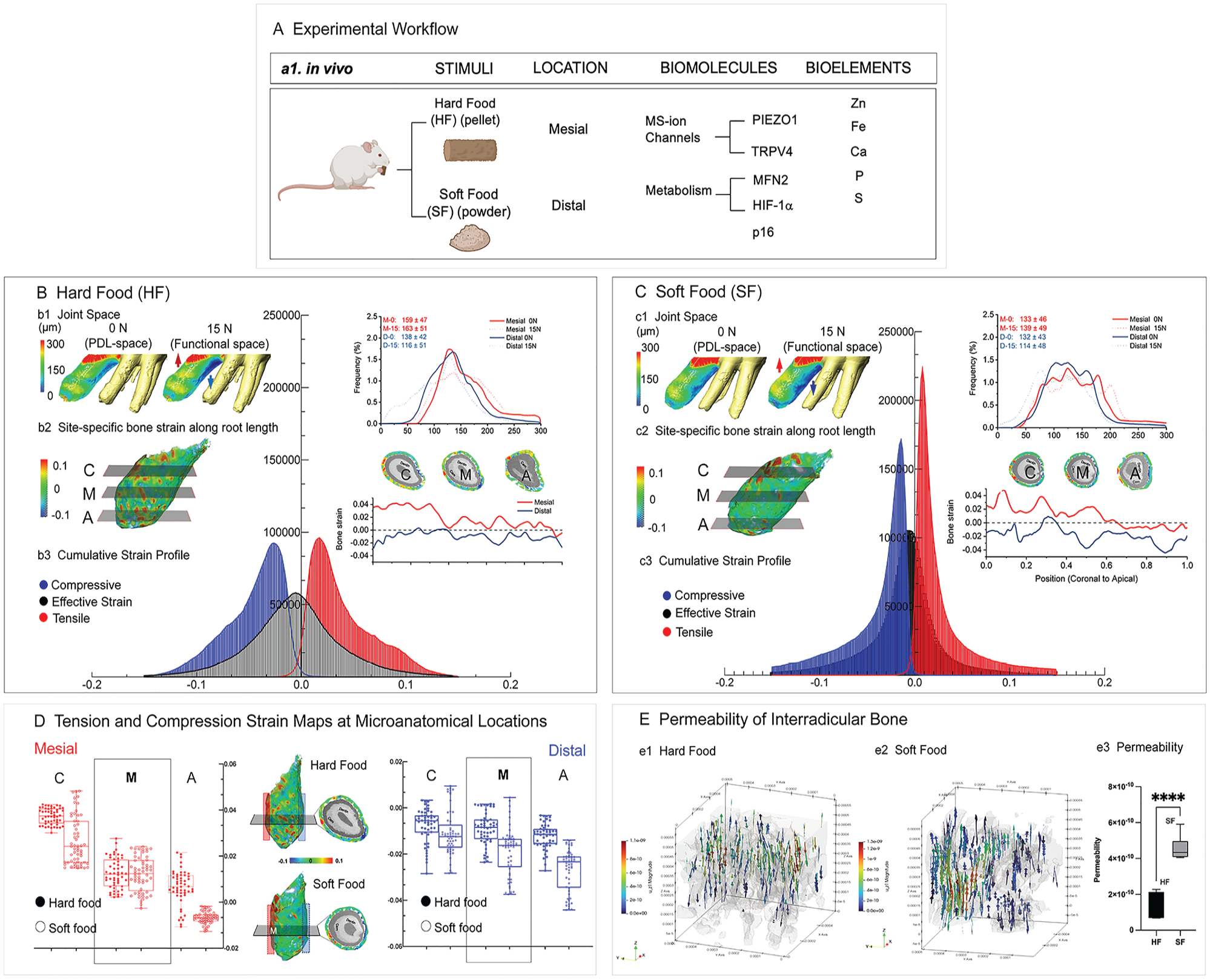
Physical properties of rat maxillary alveolar bones from hard and soft food groups. (
Dentoalveolar Joint Biomechanics, Stiffness, Bone Strain, and Permeability
Intact left maxillae were dissected for functional imaging (Wang et al. 2020) and processed for histology. Specimens (4 hemimaxilla: 2 each from HF and SF) were loaded to 15 N, and joint stiffness (Newton per millimeter) was estimated from the force-displacement curve (Lin et al. 2014) (Appendix Fig. 1). Digital computed tomography volumes at no load and under load were used to estimate anatomy-specific strains in the bone (Wang et al. 2020). Permeability in meters squared was computed with Porous Microstructure Analysis version 3.1.7.
Spatial Localization of Biomolecules in the Periodontal Complex
The 8-µm sections from fixed (10% neutral buffered formalin) and decalcified (20% EDTA) right maxillae were immunolocalized with the antibodies against TRPV4, PIEZO1, mitofusin 2, hypoxia-inducible factor 1α (HIF-1α), and p16. Some sections were further reacted with FluoZin3 and colocalized with PIEZO1 or TRPV4. Sections were counterstained with hematoxylin (immunohistochemistry) or DAPI (immunofluorescence). Three sections per animal per group were imaged by a light microscope (AXIO-Z3; Zeiss), and quantitative spatial maps of immunolocalized biomolecules (immunohistochemistry and immunofluorescence) were generated with ImageJ (Schneider et al. 2012).
Spatial Localization of Elements in the Periodontal Complex
Spatial elemental concentrations were collected from thin sections of rat maxillae with laser ablation inductively coupled plasma time-of-flight mass spectrometry. The laser spot was 6 µm with a 30-µm overlap; the single-pulse response was 5 milliseconds. Results were presented as parts per million. Box plots were generated, and an unpaired Student’s t test with P < 0.05 was used to identify differences across groups.
In Vitro: Agonist and Antagonist Treatments of MS-Ion Channels to Visualize and Quantify Intracellular Zn and Related Biomolecules in Human PDL Fibroblasts
Human PDL fibroblasts (Lonza Bioscience) were treated with agonists to activate, or antagonists to disable, PIEZO1 and TRPV4 (Appendix Table 1) at day 3 (D3; early stage), day 7 (D7; middle stage), and day 14 (D14; late stage). The concentrations (for dose, see Appendix Table 2) were selected according to MTT assay (Appendix Fig. 3A). Zn in cells was detected by treating them with FluoZin3 for 2 h. Following fixation with 4% paraformaldehyde/phosphate-buffered saline and counterstain with DAPI, cells were imaged with a light microscope. Total RNA was extracted from human PDL fibroblast cultures treated by vehicle or PIEZO1/TRPV4 agonists and antagonists with the RNeasy Mini Kit (Qiagen). Gene expressions of PIEZO1, TRPV4, ZIP8, ZnT1, MFN2, HIF-1α, and SOD2 were determined by real-time quantitative PCR (qPCR; for primers, see Appendix Table 2).
Statistical Analyses for Models In Vivo and In Vitro
An unpaired Student’s t test with P < 0.05 was used to identify anatomy-specific statistical significance in 1) mechanical strains in bone and permeability and 2) differential expression and abundance between all parameters and markers (metals, biomolecules) in HF and SF groups in vivo, and agonist and antagonist groups in vitro.
Immunolocalized Biomolecules In Vivo
Three animals from each group in vivo and 3 wells from each group in vitro were used to identify differences between the HF and SF groups and between the agonist and antagonist groups. Results from cell culture models in vitro and qPCR are expressed as a percentage of vehicle-treated controls. Results from histology sections taken from preclinical animal models are presented as mean ± SD for each group. Significant differences (P < 0.05) between groups were identified as stated previously.
Correlation and Hierarchical Clustering of Outcomes from In Vivo and In Vitro Models
After stratification by HF/SF status for in vivo replicates and agonist/antagonist status for in vitro replicates, biomarkers and metals were clustered, and Spearman correlation matrices were calculated to determine biomolecular and biometal associations. The PIEZO1/TRPV4 agonists and antagonists were separately clustered globally across all in vitro specimens. Hierarchical clustering was performed independently for D3, D7, and D14 for in vitro experiments, to note if the markers clustered by biological pathway.
Results
Alveolar Bones In Vivo: Higher Effective Strain and Lower Variance in SF but Lower Strain and Higher Variance in HF
Tooth displacement under load was significantly lower in rats that chewed SF than HF (Appendix Fig. 1C) (Niver et al. 2011). A bimodal distribution was observed from the distal (132 to 138 μm) and mesial (133 to 159 μm) PDL space in the HF group as compared with the SF group (Fig. 1B-b1, C-c1) (Yang et al. 2019).
Diverse effective strains (combined tensile, compression, and shear strain profiles) were observed within the alveolar bones from both groups (Fig. 1B-b2, C-c2). Bone strain along and across the root suggested mesial tension and distal compression in HF. Higher strains but a uniform profile was observed in the SF group (Fig. 1B-b3, C-c3). The effective strains varied from coronal, mid-, and apical locations in HF (Fig. 1B-b2, D) and SF (Fig. 1C-c2, D). The interradicular bone permeability increased significantly by 3 times in SF as compared with HF (Fig. 1E).
Blunting of TRPV4, PIEZO1, and Zn in SF Rats In Vivo
Experiments in vivo demonstrated biomolecular expression differences by food (HF, SF) and location (anatomically distinct mesial and distal regions). PIEZO1 expression (red, Fig. 2A) in HF was significantly higher (P < 0.05) in distal, compressive, and resorptive locations (79% cells) as compared with mesial, tensile, and formative locations (29%). PIEZO1 expressions in SF were impaired in resorptive (32%) and formative (13%) locations (red, Fig. 2A, C-c1). Contrasting PIEZO1, higher TRPV4 expressions were observed at the formative region (51%) than the resorptive region (16%). TRPV4 expression in formative (9%) and resorptive (2%) locations was impaired in animals that chewed SF (red, Fig. 2B, C-c2). Zn production in the PDL was accompanied by the PIEZO1 expression: higher expression in the distal, compressive, and resorptive region as compared with the mesial, tensile, and formative region in the HF group (green, Fig. 2A). Zn colocalized with PIEZO1 (Fig. 2A) and TRPV4 (Fig. 2B) in PDL cells, and its presence was impaired in the SF group.
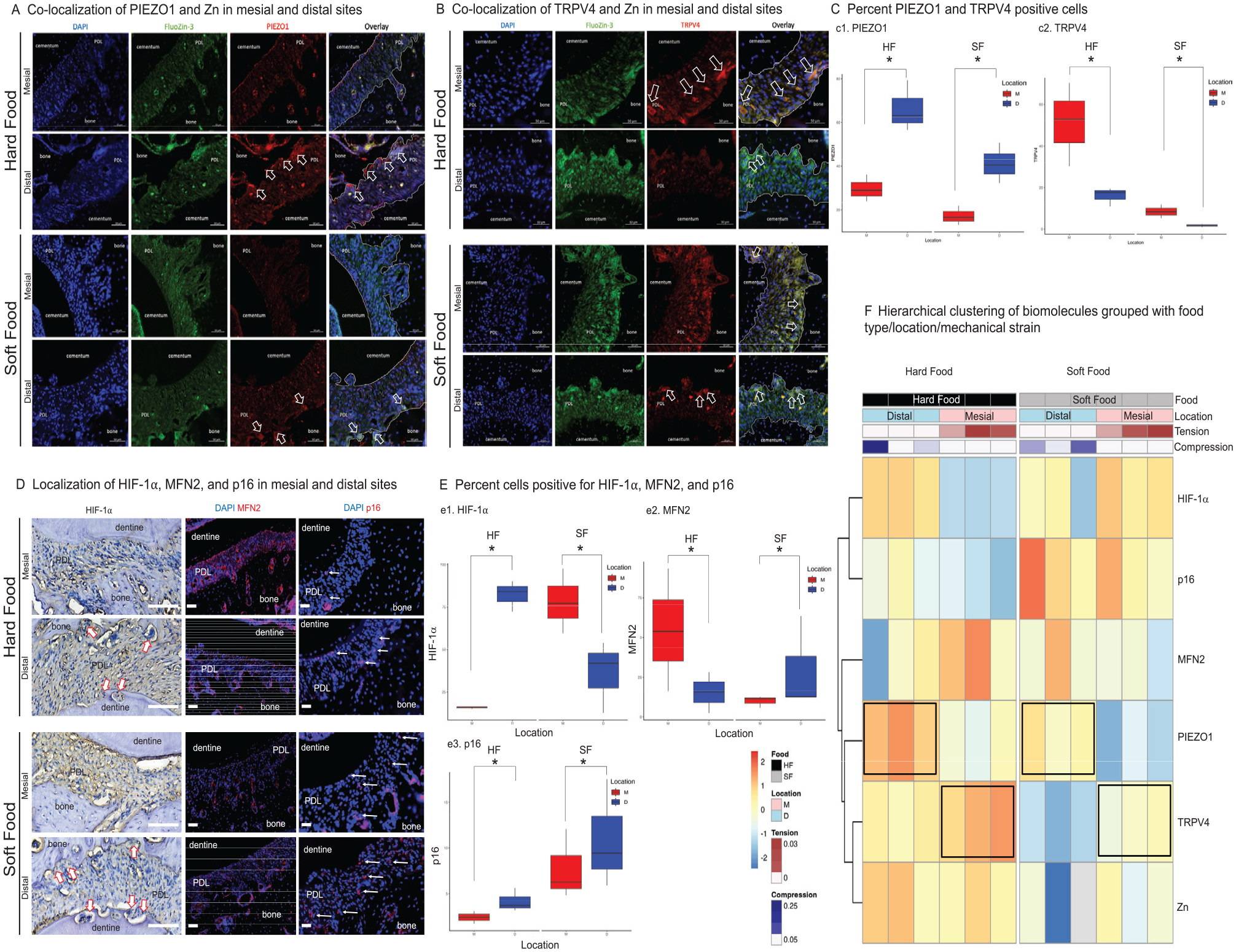
Site-specific mechanosensitive ion localization, mitochondrial function, hypoxia-inducible factor 1α (HIF-1α), and cell senescence in the rat periodontal complex can be regulated by food hardness. (
In Vivo: Altered Hypoxia, Impaired Mitochondrial Function, and Induced Cell Senescence
Altered Hypoxia
Increased PIEZO1 and HIF-1α (Fig. 2A, D, E-e1) were observed in distal, compressive, and resorptive cells (82%) than in mesial, tensile, and formative PDL cells (16%) in the HF group. Higher expression of HIF-1α in mesial (78%) as compared with distal (39%; P < 0.05) locations was observed in the SF group (Fig. 2D, E-e1).
Impaired Mitochondrial Function
MFN2 expression in the mesial and formative PDL (82%) was significantly higher than in the distal and resorptive PDL (52%), and this expression pattern was similar to TRPV4 (Fig. 2D). Chewing on softer foods dramatically decreased MFN2 in the mesial (53%) and distal (24%) sides (Fig. 2D, E-e2).
Increased Cell Senescence
Chewing on softer foods increased the number of senescent cells in mesial (8%) and distal (11%) regions (Fig. 2D, E-e3) (P < 0.05). In the HF group, p16-positive senescent cells were observed in the distal PDL (5%) as compared with the mesial PDL (3%) (Fig. 2D, E-e3). Significant increases in cell senescence were observed with SF, nonspecific to location.
Food- and Anatomy-Specific Correlation and Clustering of Biomolecular Expressions Suggested MS-Ion Channel Strain–Specific Axes
Spearman correlation matrices indicated relationships between Zn levels and the MS-ion channels, which varied depending on food hardness (Appendix Figs. 2, 4). For HF, Zn levels were negatively associated with TRPV4 expression while positively associated with PIEZO1 expression. For SF, Zn levels were positively associated with TRPV4 expression while negatively associated with PIEZO1 expression. Cluster analysis suggested distinct separation in biomolecular expressions in the periodontal complexes based on food type, with HF and SF diets forming distinct clusters of related biomolecules (Fig. 2F, HF-black, SF-gray). This suggests that the type of diet has a significant impact on the amount of Zn and gene expression (TRPV4, MFN2, PIEZO1, HIF-1α, and p16). Further stratification within each food group suggested a secondary level of separation corresponding to the anatomic location (Fig. 2F, mesial-pink, distal-blue). Notably, load-induced gene expression gradients were conserved across food types, although there were variations in the degree of expressions, as indicated by box plots (Fig. 2C, E). In clustering the genes, TRPV4 expression was closely related overall to metabolic programming through its clustering with MFN2, whereas PIEZO1 expression was related to HIF-1α and p16. Thus, the tension-TRPV4-MFN2 axis and the compression-PIEZO1-HIF-1α axis are regulated by HF.
Decreased Zn and Iron in the SF PDL In Vivo
Iron (Fe) was detected in the tensile and widened PDL-bone boundary with a rich blood supply (Fig. 3a1, a3) where relatively no HIF-1α was observed in the HF group in the first root of the first molar (R1M1), according to laser ablation inductively coupled plasma time-of-flight mass spectrometry (Fig. 3). Zn was detected in the compressive and narrow PDL of the R1M1 (Fig. 3a1, a3), as noted by histology (Fig. 2A, B). Fe and Zn concentrations dramatically decreased in the PDL of the R1M1 in the SF group in all locations (Fig. 3a2, a3).
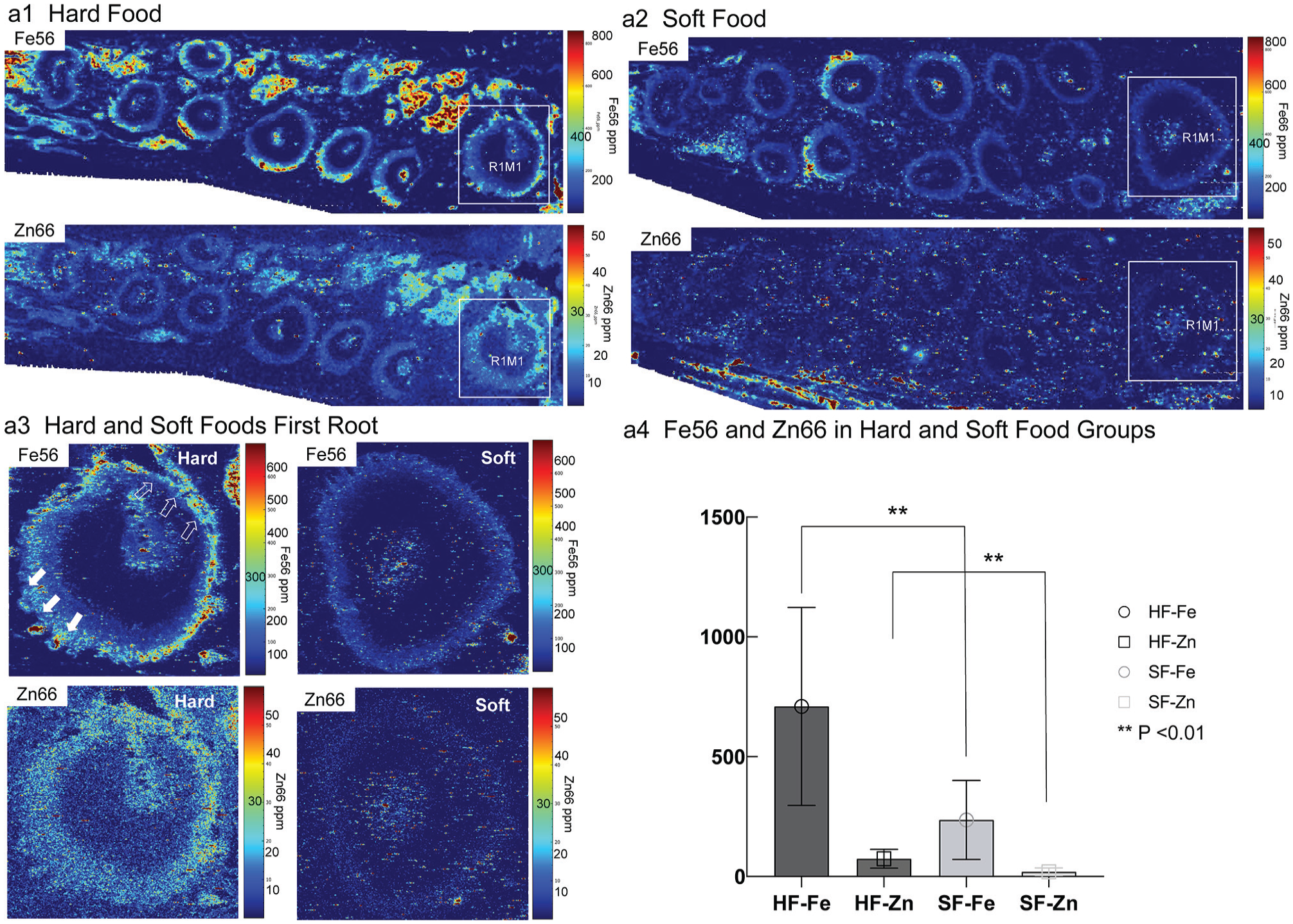
Fe56 and Zn66 concentrations were significantly higher in the HF group as compared with the SF group. Spatial maps of Fe66 (top row) and Zn66 (bottom row) in the hemimaxillae of rats fed HF (
In Vitro: PIEZO1 and TRPV4 Regulate PDL Cell Metabolism and Zn Homeostasis
PIEZO1 Regulates Intracellular Zn and Metabolism in Human PDL Fibroblast Cultures
Intracellular Zn (zincosomes; Appendix Fig. 3A, C-c1, c3) and the RNA levels of ZIP8 and ZnT1 (Appendix Figs. 3C-c2, c4, 4B-b2) increased in the human PDL fibroblast cells from the proliferating (D3 and D7) to fully differentiated (D14) stage with mineralization nodules observed in D14 cultures (Appendix Figs. 3A, 4B). Activation of the PIEZO1 channel by Yoda1 significantly increased intracellular Zn-positive vesicles while blunting the PIEZO1 channel by GsMTx4-decreased intracellular Zn at all stages of the cultures when compared with the vehicle-treated cultures (Fig. 4B-b1, b2). In addition, qPCR indicated that at D14 (Appendix Fig. 3C-c1), the effect of Yoda1 trended and significantly increased intracellular Zn and RNA levels of ZIP8, ZnT1, HIF-1α, and MFN2 but decreased RNA levels of SOD2 (Fig. 4B-b1, b2).
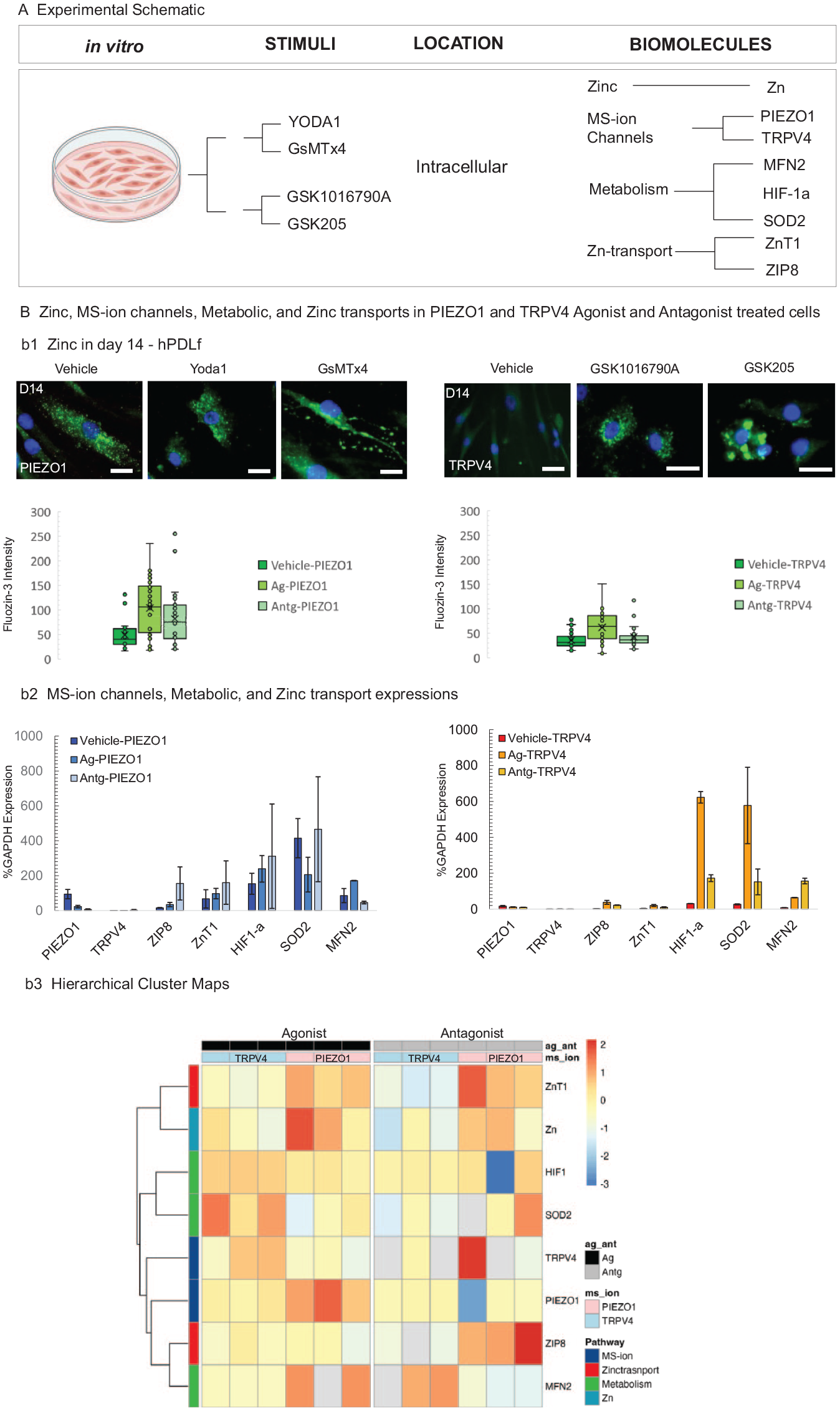
PIEZO1 and TRPV4 regulate intracellular zinc levels and cell metabolism of human PDL fibroblasts in vitro. (A) Experimental workflow of human PDL fibroblast cultures. (B) At day 14, the PIEZO1 channel in human PDL fibroblast cells was activated or disabled by agonist (Yoda1) or antagonist (GsMTx4), respectively. Intracellular zinc was identified by FluoZin3 (b1, green dots) and DAPI-counterstained nuclei. The number of intracellular zinc vesicles (green dots, box plots) are shown. The RNA levels of PIEZO1, TRPV4, ZIP8, ZnT1, HIF-1α, MFN2, and SOD2 (as measured by quantitative polymerase chain reaction) were calculated and are shown in panel b2, left panel. Similarly, the TRPV4 channel in the human PDL fibroblast cultures was activated or disabled by GSK1016790A or GSK205, respectively (b2, right panel). Scale bars = 20 µm. (b3) Hierarchical clustering suggested a clear differentiation in that the TRPV4 condition was closely associated with metabolic genes such as HIF-1α and SOD2. Conversely, PIEZO1 was closely associated with ZIP8 and the metabolic marker MFN2. Upon examination of the data focused solely on agonists and antagonists without the vehicle, the clustering shifted: TRPV4 clustered closer to ZIP8. Results on days 3 through 14 are in Appendix Figure 4. Data are presented as median, IQR, and 95% CI for box plots or mean (SD) otherwise. HIF-1α, hypoxia-inducible factor-1α; MS-ion, mechanosensory ion; PDL, periodontal ligament.
TRPV4 Channel in Regulating Intracellular Zn Levels and Metabolism in Human PDL Fibroblast Cultures
As shown in Fig. 4 stimulation of the TRPV4 channel by GSK1016790A increased intracellular Zn and RNA levels of ZIP8, ZnT1, HIF-1α, SOD2, and MFN2, while disabling TRPV4 channel by GSK205 did not significantly affect the intracellular zincosomes, ZIP8 and ZnT1 but increased the RNA levels of HIF-1α, SOD2, and MFN2 (Fig. 4B-b1, b2). However, the increased levels were not as dramatic as the treatment of the agonist GSK1016790A.
MS-Ion Channel and Zn Transport–Controlled Biomolecular Expressions In Vitro
Similar to our in vivo findings, Spearman correlation matrices—stratified by replicates treated by the agonist and antagonist—suggested positive relationships between Zn levels and Zn transporters but not with MS-ion channel expressions (Appendix Fig. 4A). By D14, hierarchical clustering (Fig. 4b3) suggested clear differences in gene expression profiles. TRPV4 expression showed proximity to metabolic genes such as HIF-1α and SOD2. Conversely, PIEZO1 was clustered near the Zn transporter gene ZIP8 and the metabolic marker MFN2. Upon examination of the data focused solely on agonists and antagonists without the vehicle, the clustering shifted: TRPV4 clustered closer to ZIP8 (Fig. 4b3). Results on D3-D14 are in Appendix Figure 4B.
Discussion
Followed by Fe, Zn is the second-most abundant trace metal in humans and animals. The importance of extracellular Zn and related bone phenotype (King 1990, 1996; King et al. 2000) at a tissue level is known. MS-ion channels sensitive to divalent cations including Zn2+ affect cellular function and nucleation and dissolution of minerals in bones and teeth (Mayer et al. 1994; King 1996; Kara et al. 2009; Sun et al. 2019; Wang, You, et al. 2022). We found that the hard and soft chewing regimens from our model in vivo potentially regulate cellular homeostasis of biometal Zn and its transports (ZnT1 and ZIP8); these results were recapitulated in analogous agonist and antagonist treatments within our cell model in vitro (Figs. 2–4). In response to mechanical strains, normal intracellular Zn levels are altered to initiate mitochondrial and cytoplasmic signaling events (Fukada et al. 2011) at the microanatomic periodontal locations. The Zn increase at D7 and mineralized nodules at D14 in fibroblast cultures (Appendix Figs. 3A, 4B) suggested the role of Zn in cell maturation and mineralization. Labile intracellular Zn to regulate organelle and cell functions (Bossy-Wetzel et al. 2004; Inoue et al. 2015; Kambe et al. 2015; Deng et al. 2021) and cell fate (Eron et al. 2018) are also known. Intracellular Zn homeostasis is controlled by membrane-bound Zn transporters (Slc30a family [Zn transporter]) that export or import (Slc39a family [ZIP]) (Cousins et al. 2006) and via storage in organelles (Lu et al. 2016), such as mitochondria and endoplasmic reticulum. Zn transporters tightly control reactive oxygen species (ROS) expression, Zn levels, cellular function, and fate (Maret 2019).
We postulate that the upstream, tension, compression, and shear-induced mechanical strains in vivo can stabilize the HIF-1α in a tissue- and cell-specific manner and control Zn and ROS (SOD2) production by mitochondrial function (MFN2) as a potential mediator. The HIF-1α–induced biochemical cascade can incite downstream oxidative phosphorylation, a necessary step to control tissue-specific cellular function (Sabini et al. 2023). Cellular homeostasis of ROS and resulting oxidative phosphorylation control mineral resorption and formation and maintenance of soft and hard tissue mechanical integrity (Jezek et al. 2010). In our study, compression of blood vessels in the PDL (decreased Fe signal in the narrowed PDL; Fig. 3) → PIEZO1 (Jin et al. 2015) stabilized HIF-1α (Solis et al. 2019) and decreased mitochondrial fusion (MFN2; Fig. 2) (Xiang et al. 2024), a cascade that resembles ischemia-induced hypoxia, SOD2 production (Xiang et al. 2024) → senescent cells (increased p16; Fig. 2) (Ren et al. 2022) but at the distal/compression regions. Mitochondrial function (MFN2) and hypoxia ( HIF-1α ) relate to cell senescence (p16) (Xing et al. 2018; Miwa et al. 2022). Based on results from this study and literature, we hypothesize that matrix tension → TRPV4 channel → MFN2 but reduced HIF-1α, and SOD2 (increased Fe signal) in the widened PDL → Zn (Figs. 2, 3). The lower magnitude imparted by SF, however, reduced overall fluid flow, decreasing overall tissue oxygenation (significantly decreased Fe signal in SF group; Fig. 3) and Zn levels, and increased region nonspecific HIF-1α expression in the periodontal complex (Fig. 2). Cementum growth was identified (Appendix Fig. 1) (Jang et al. 2015), alluding to the hypothesis stated by others (Choi et al. 2014) that a hypoxic environment encouraged by weak or no occlusal load promotes its growth. Elevated Zn in compressed PDL than in stretched PDL in the HF group contrasted impaired Zn levels in the SF group (Figs. 2, 3), adding to the unexplored role of biometal Zn in microenvironments and its effect on biomineralization in the periodontal complex. These data suggest that HF-induced mechanical strains can regulate intracellular Zn levels and cellular function.
So, how do chewing loads (biomechanical stimuli) and Zn concentrations affect PDL metabolism and cell fate? Our findings, as listed in Figure 5, illustrate that the downstream effects of the mechanical stimuli and Zn homeostasis on PDL cell metabolism and the oxidative state differ by mechanical strain type (stretch, compression, shear) and strain magnitude at the tissue and cell levels. Compression-activated PIEZO1 promotes the influx of extracellular Zn and releases the Zn from the vesicular storage to increase the intracellular labile Zn in a compressed PDL. Higher Zn in compressed PDL cells in a hypoxic environment inhibits mitochondria function (Bossy-Wetzel et al. 2004) and increases ROS production. Contrastingly, our findings suggest that tensile loads activate TRPV4 to regulate intracellular labile Zn to lower levels, stimulate mitochondria fusion and function, and reduce ROS production in the PDL cells. Thus, in addition to directing MS-ion channel activity, mechanical stimuli potentially maintain metabolism and regulate oxidative stress via the regulation of cellular Zn homeostasis. The mechanoregulation of MS-ion channels and ZnT expressions controlling Zn intracellular and cellular function also can result from 1) activation of PIEZO1 and TRPV4 to import Zn, the levels of which are affected by mechanical loading–coupled tissue oxygenation from the extracellular milieu (Inoue et al. 2015), and 2) mechanically stimulated PIEZO1 and TRPV4, which can affect Zn transporters ZIP8 or ZnT1 (as evidenced through qPCR analysis; Fig. 4) to regulate Zn influx and efflux. Consistent with the in vivo findings, PIEZO1 and TRPV4 exhibit opposing expression profiles in vitro, highlighting their potentially complementary yet opposing roles in cellular signaling pathways related to mechanical/chemical stimuli. These patterns of gene expressions suggest a complex interplay between mechanical and chemical stimuli in regulating metabolic and mineralization processes.
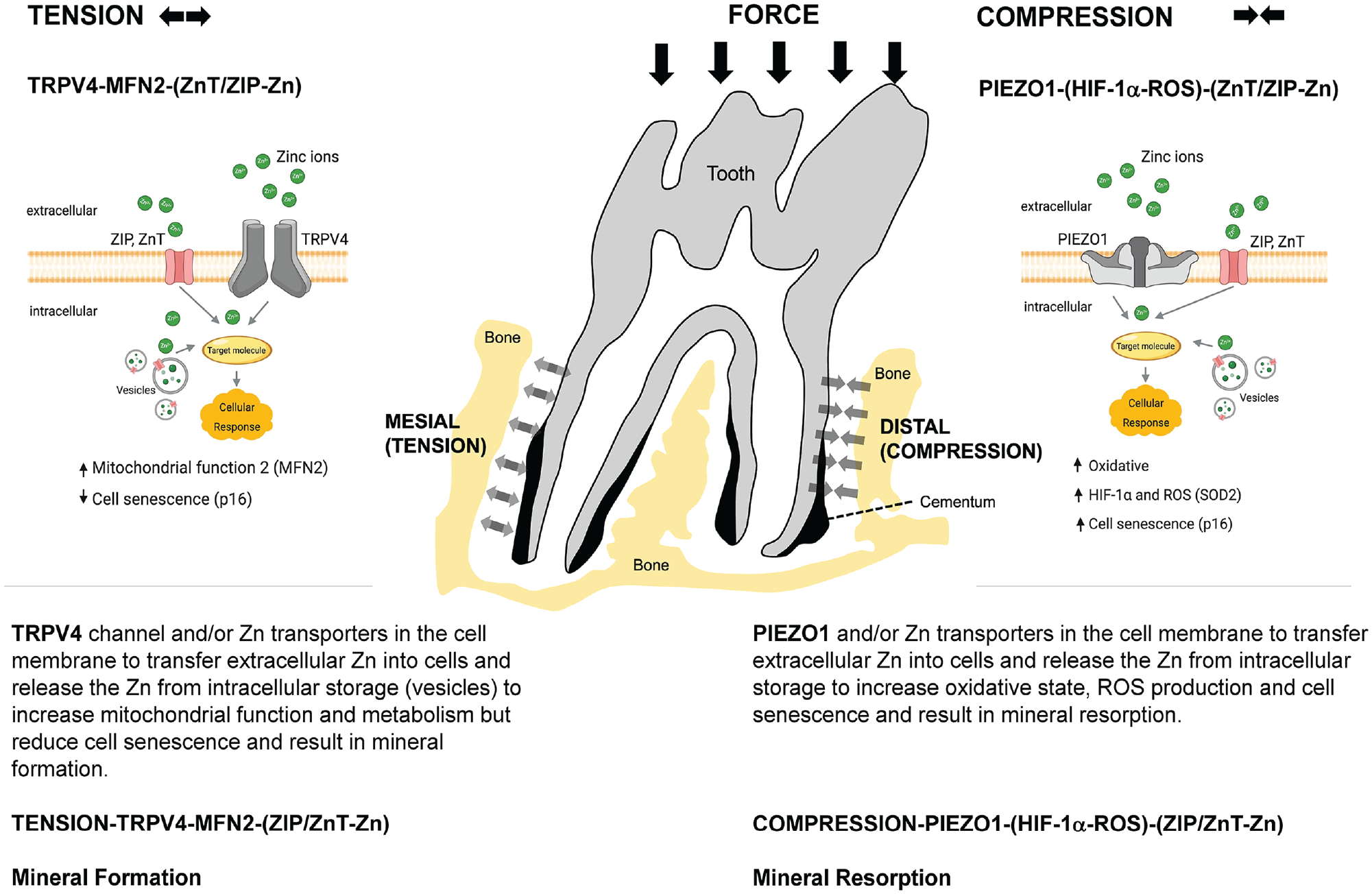
Hypothesized MS-ion channel–regulated biomineralization in the periodontal complex. Mineral formation is regulated by tension-mediated TRPV4 activation (MFN2-ZIP/Znt-Zn axis) and mineral resorption by compression-mediated PIEZO1 activation (HIF-1α–ROS-ZIP/Znt-Zn axis) within the periodontal complex when stimulated by chewing loads. HIF-1α, hypoxia-inducible factor 1α; MS-ion, mechanosensory ion; ROS, reactive oxygen species.
Observations revealed study limitations (Appendix) and provided plausible insights into the need for solid and fluid mechanics–induced shear flow to maintain Zn-mediated biological pathways in the periodontal complex (Fig. 5). These results corroborated with increased pore volume fraction, reduced alveolar bone mineral density, and increased cementum, which are observed physicochemical changes in a periodontal complex under disuse. In summary (Fig. 5), results suggest that modulation of short- and long-term prescription of a loading regimen (mechanotherapeutics) that constitutes chewing on harder and softer foods can influence MS-ion channels, cellular Zn, and HIF-1α transcription, as well as the ensuing biomineralization events, periodontal maintenance, and overall dentoalveolar joint function.
Author Contributions
Y. Wang, contributed to conception, design, data acquisition, analysis, and interpretation, drafted and critically revised the manuscript; B.H. Lee, contributed to data acquisition and analysis, worked on figures and tables of the manuscript, cell culture, MTT assays, histology; Z. Yang, contributed to data acquisition and analysis, physical measurements, and computed tomography analysis; T.J. Ho, contributed to data analysis, cell culture studies; H. Ci, contributed to data analysis, strain maps and permeability, and other physical measurements; B. Jackson, T. Punshon, contributed to data acquisition and analysis, worked on laser ablation inductively coupled plasma mass spectrometry; B. Wang, contributed to data acquisition and analysis, worked on mechanical testing and strain mapping; J. Levy, contributed to data analysis and interpretation, bioinformatics/higher-level data analyses; S.P. Ho, contributed to conception, design, data analysis and interpretation, drafted and critically revised the manuscript. All authors gave final approval and agree to be accountable for all aspects of the work.
Supplemental Material
sj-docx-1-jdr-10.1177_00220345241294001 – Supplemental material for Chewing-Activated TRPV4/PIEZO1–HIF-1α–Zn Axes in a Rat Periodontal Complex
Supplemental material, sj-docx-1-jdr-10.1177_00220345241294001 for Chewing-Activated TRPV4/PIEZO1–HIF-1α–Zn Axes in a Rat Periodontal Complex by Y. Wang, B.H. Lee, Z. Yang, T.J. Ho, H. Ci, B. Jackson, T. Punshon, B. Wang, J. Levy and S.P. Ho in Journal of Dental Research
Footnotes
Acknowledgements
The authors express their sincere thanks for the expert discussions and input from Wolfgang Maret, PhD, Professor of Metallomics, King’s College London, on zinc-related redox chemistry and cellular function. The authors thank Putu Ustriyana, Irene Hung, Arman Sheeler, Jasper Chang, Sukhmandeep Sidhu, and Lea Segdhi for their assistance with animal maintenance. The authors thank the Biomaterials and Bioengineering Correlative Microscopy Core, University of California San Francisco, for the use of MicroXCT-200 and biomechanical testing in situ. Laser ablation inductively coupled plasma mass spectrometry elemental imaging was performed at the Dartmouth Biomedical National Elemental Imaging Resource, as supported by the National Institute of General Medical Sciences (R24GM141194 and 1S10OD032352).
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was supported by the National Institutes of Health/National Institute of Dental and Craniofacial Research (R01 DE022032 to S.P.H.).
Data Availability Statement
The data that support the findings of this study are available from the corresponding author upon reasonable request.
A supplemental appendix to this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
