Abstract
High-throughput screening for drug discovery is increasingly utilizing cellular systems of high physiological relevance, such as patient primary cells and organoid cultures. We used 3D-cultured cystic fibrosis patient bronchial epithelial cells to screen for new small-molecule correctors of the disease-causing F508del mutation in CFTR. Impaired mucociliary clearance due to insufficient airway hydration is a hallmark of cystic fibrosis and we used a simple measure of surface liquid levels to quantify F508del CFTR correction in cultured bronchial epithelial cells. Two robust assay formats were configured and used to screen more than 100,000 compounds as mixtures or individual compounds in 96-well format. The corrector discovery success rate, as measured by the number of hits confirmed by an electrophysiology assay on patient primary bronchial epithelial cells, was superior to screens in cell lines expressing recombinant F508del CFTR. Several novel corrector scaffolds were discovered that when combined with the clinical corrector VX-809 delivered combination responses greater than double that of VX-809 alone. This work exemplifies the advantages of a disease-relevant readout and 3D-cultured patient primary cells for the discovery of new drug candidates.
Introduction
High-throughput screening (HTS) approaches range from target-based assays using isolated proteins at one extreme to phenotypic assays with physiologically relevant organoids or tissues at the other.1,2 This work describes a phenotypic approach using patient-derived 3D-cultured primary bronchial epithelial cells to screen for new small-molecule correctors of a disease-causing mutation in cystic fibrosis (CF).
CF is a genetic disease caused by mutations in CFTR, a gene encoding an epithelial ion channel. 3 CFTR is a multidomain transmembrane protein that has so far proven difficult to purify and incorporate into target-based screens. Phenotypic approaches were used to discover the existing CFTR modulators, which are showing great promise in clinical trials and as marketed drugs.3–6
Deletion of Phe508 (F508del) is the most prevalent CFTR mutation in CF (90% of patients have at least one copy).7,8 F508del causes two main defects: (1) protein misfolding resulting in degradation and reduced channel numbers, and (2) a gating defect resulting in low open probability and reduced function for the remaining channels.9,10 CFTR-modulating small molecules called “correctors” partially reverse the F508del folding defect and increase the density of CFTR channels at the plasma membrane.4,5,11–14 “Potentiators” are small molecules that partially reverse the gating defect and increase channel open probability.6,15–21 Marketed combinations of a corrector plus a potentiator (ORKAMBI, SYMKEVI) have conferred modest (but significant) lung function improvements to patients. 22 In vitro, the combination of a corrector and a potentiator restored function in cells from F508del homozygote patients to approximately 35% of the wild-type level. 4 Substantially greater efficacy can be observed in CF bronchial epithelial cell cultures when a second corrector with a distinct mode of action is combined with a corrector and potentiator, and three-drug combinations are being successfully advanced in clinical trials.3,23,24 The promise of restoring F508del function to the level of carriers with no disease (50% of wild-type) with three-drug combinations has spurred the effort to discover new correctors with diverse mechanisms. Additional correctors would have utility if current triple combinations leave F508del patients with residual disease or are poorly efficacious in a subset of patients. New combinations based on different mechanisms of action might also have even greater efficacy than the current combinations.
F508del corrector compounds have been optimized using efficacy and potency data from F508del patient-derived primary bronchial epithelial cells cultured on filters at an air/liquid interface (ALI) to form a well-differentiated, three-dimensional, pseudostratified epithelium. 25 These cultures contain basal, goblet, and ciliated cells and express a full complement of ion channels and transporters that enable them to absorb or secrete liquid. CFTR function can be measured in these cultures using electrophysiology techniques, and the data have so far proven to be broadly predictive of clinical efficacy for both potentiators and correctors. 26 Progress has been made in increasing the ease and throughput of electrophysiology assays. 27 Nevertheless, most of this work is still done in 24-well filter plates, limiting the capacity for screening.
The discovery of new correctors has historically relied on HTS in cell lines expressing recombinant F508del CFTR, and this approach has yielded clinical compounds such as VX-809 (lumacaftor) and others.13,28–30 Unfortunately, only a small fraction of correctors identified using recombinant cell lines also show activity in F508del patient-derived bronchial cultures and are advanced further. A phenotypic screening approach based on primary bronchial epithelia would bypass the inefficient cell line screening step and should discover correctors more likely to show clinical activity.
While CF is a multisystem disease, most of the disease burden results from chronic airway dysfunction. Lack of CFTR function in the airways results in insufficient hydration, compromising the clearance of mucus and leading to cycles of infection, inflammation, and fibrosis, ultimately degrading lung function. 31 In ALI cultures of CF human bronchial epithelial (hBE) cells, the same processes that govern airway hydration in vivo control airway surface liquid (ASL) levels in vitro. ASL levels are governed by the competing processes of sodium absorption (through the epithelial sodium channel [ENaC]) and chloride secretion or absorption (mediated by CFTR) at the epithelial cell apical membrane, as well as other transporters that govern sodium and chloride entry at the basolateral membrane. 32 In F508del bronchial epithelial cultures, liquid secretion can be restored by clinical corrector and potentiator compounds. 5
In this report, we describe assays for surface liquid absorption or secretion based on CF hBE cell cultures in 96-well filter plates configured to identify F508del CFTR correctors. We directly compare the outcomes of screening using the liquid absorption assay with two recombinant cell line assays and find a superior success rate for the absorption assay. A liquid secretion assay format was used to screen more than 100,000 compounds, resulting in the identification of several novel corrector scaffolds with modes of action distinct from the clinical corrector VX-809 and which delivered combination responses greater than twice that of VX-809 alone.
Materials and Methods
Patient Primary Airway Cells
F508del/F508del patient bronchial epithelial cells at passage 1 were obtained from Tissue Procurement & Cell Culture at the Cystic Fibrosis Center of the University of North Carolina Chapel Hill (UNC-CH). The research conducted on human cells at UNC-CH has been verified as compliant with Pfizer policies, including institutional review board or institutional ethics committee approval. Cells from patient code R were used for all of the screening with ASL assays reported herein. Corrector activity was confirmed by an orthogonal electrophysiology assay in at least one patient code different from that used in the primary screen for 10 of the 12 confirmed screening hits shown in Figure 4 (see Suppl. Table S6 ). Cells from patient code Y were used for the Western blot in Figure 5B .
Compounds
VX-809 was prepared by a contract research organization (Ricerca Biosiences, Concord, OH; BioDuro, San Diego, CA; Peakdale Molecular, Manchester, UK; Wuxi Pharmatech, Shanghai, China; or Chembiotek Research International, Kolkata, India). VX-770 was prepared by a contract research organization (Bicoll GmbH, Planegg, Germany or Chembiotek Research International). Compound A was prepared by Peakdale Molecular Ltd.
Cell Culture and Differentiation
The procedure for bronchial epithelial cell expansion and differentiation was adapted from a published method. 25 Key modifications are described below.
Complete PneumaCult-Ex medium (StemCell Technol-ogies, Vancouver, Canada) with added hydrocortisone (100 ng/mL) was used for growth and expansion in flask culture. Passage 1 cells were expanded and cryopreserved at passage 2. Passage 2 cells were expanded to passage 4 before seeding onto 96-well filter plates (cat. 7369, Corning Inc., Corning, NY). A ViaFlo 384 electronic pipette with 96 tips (Integra Biosciences, Hudson, NH) or Multidrop Combi reagent dispenser (Thermo Fisher Scientific, Waltham, MA) was used to seed cells onto filters (69,000 cells per filter). Conditioned medium coating of filters was omitted.
The process of moving from seeded filters to an ALI culture was as follows. Day 0: Seeded filters with cells in growth medium. Day 1: Removed apical and basolateral medium and replaced with differentiation medium with added penicillin, streptomycin, and amphotericin (100 units/mL, 100 µg/mL, and 0.25 µg/mL, respectively). Day 3: Removed apical medium to begin ALI culture; replaced basolateral medium (differentiation medium with added penicillin, streptomycin, and amphotericin). After ALI culture was initiated, the basolateral medium was changed every 2–3 days. Voriconazole (1 µg/mL) was added as an antifungal agent starting at the first medium change after ALI.
Cultures were typically used 4–7 weeks after initiating ALI culture. Transepithelial resistance in parallel 24-well cultures typically showed resistances of 500–1000 ohm·cm2. Cells received a mucus wash after 3 weeks at ALI, again at week 5 or 6, and 72 h before the assay. To wash away mucus, 50 µL of Hanks Balanced Salt Solution (Life Technologies, Carlsbad, CA) was added apically, incubated for 60 min in a cell culture incubator, and then aspirated to dryness.
Ninety-six-well aspiration manifolds were used to facilitate medium changes and mucus washes. Medium changes were made using a 96-deep-well plate aspirator (VP 177A-1, V&P Scientific, San Diego, CA). Aspiration of apical medium or buffer was performed using a 96-well standard microplate aspirator (VP 178BJ, V&P Scientific).
Airway Surface Liquid Assay: Absorption Format
Washing away mucus 72 h before dosing compounds was critical for success. On the day of dosing, bright-field images of the apical surface were acquired on an InCell 6000 high-content imager (GE Healthcare Bio-Sciences, Pittsburgh, PA) with a 2× objective to identify wells with preexisting menisci, which were excluded from later analysis.
Compounds in DMSO were spotted into dry 96-well plates using an acoustic dispenser (0.4 µL per well). Assay medium (200 µL of differentiation medium containing vasoactive intestinal peptide [VIP; 30 nM; EMD Millipore, Billerica, MA] and VX-770 [1 µM]) was added using a Multidrop Combi dispenser (Thermo Fisher Scientific).
Positive control VX-809 (1–5 µM) and vehicle negative control (DMSO at 0.2% final concentration) were diluted in assay medium and added to control wells. A Biomek NX pipetting robot (Beckman Coulter) was used to mix and then transfer 200 µL of medium from compound plates to fresh empty receiver plates. Transwell filter inserts from the cell plates were then transferred to the receiver plates. The Biomek NX was used to add either 5 or 8 µL of phosphate-buffered saline to the apical surface of the filters. The plates were then returned to the incubator and scanned on the InCell imager (bright-field 2× objective) at 72 h postdosing.
Liquid on the surface of the culture forms a meniscus where the outer edge of the culture meets the wall of the Transwell insert (
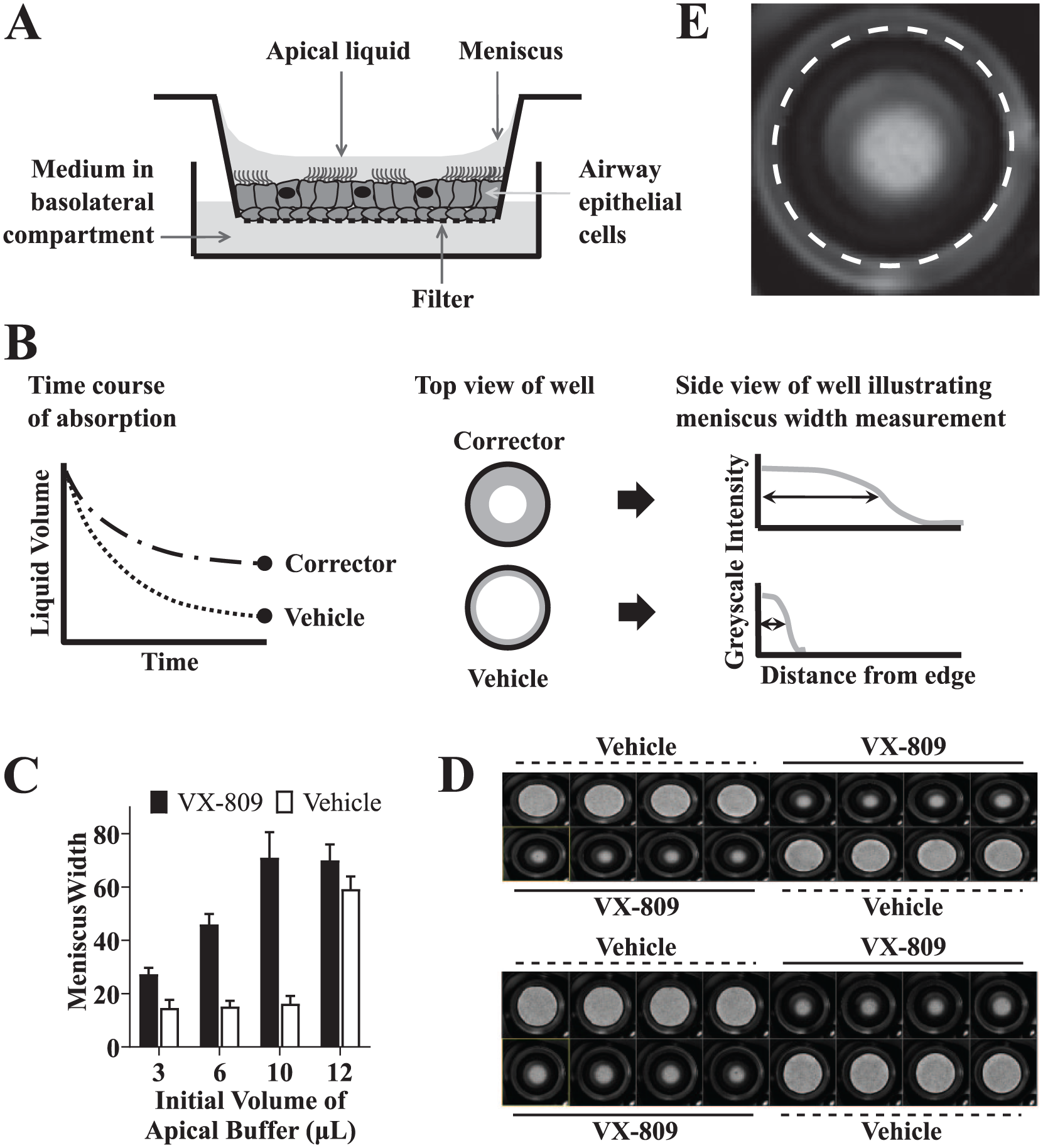
(
Airway Surface Liquid Assay: Secretion Format
The procedure was as described above for the absorption format except that no apical buffer was added to the filter inserts.
Airway Surface Liquid Assay: Secretion Combination Format
The procedure was as described above for the absorption format with the exception of the addition of compound A (200–300 nM) to the assay medium. No apical buffer was added to the filter inserts. An iPipette Pro pipettor (Apricot Designs, Covina, CA) was used to transfer medium.
CF hBE Equivalent Current Assay
Experimental details are given in the Supplemental Material.
CFTR Western Blot in HEK Cell Line and Primary Airway Epithelia
Experimental details are given in the Supplemental Material.
Results
Use of Patient Primary Bronchial Epithelial Cells at Passage 4
F508del/F508del CFTR primary hBE cells become available when CF patients undergo lung transplantation. They may be propagated and differentiated up to passage 525 but are typically used at passages 1–3 to preserve their phenotype. We routinely used passage 3 cells for in-house electrophysiology assays. Each passage results in about an eightfold increase in cell number. In order to generate larger quantities of cells for screening, we confirmed that passage 4 cells performed similarly to passage 3 cells with respect to their capacity to differentiate at an ALI, their amiloride responses (a measure of ENaC activity), and their responses to the clinical corrector VX-809 (described in the Supplemental Material). Use of passage 4 cells enabled the production of approximately seven hundred 96-well filter plates from 1 million passage 1 cells. Passage 4 cells expressed acetylated tubulin (ciliated cells), peanut agglutinin (basal cells), and mucin 5B (goblet cells) ( Suppl. Fig. S1A ).
Patient-to-Patient Variability and Normalization of Corrector Responses
During assay development, we evaluated cells from two patients and selected one based on acceptable Z′ values and consistency of responses across multiple experiments. To ensure consistency of responses, cells from one patient (patient code R) were expanded, banked, and used for all of the screening with surface liquid assays reported herein. Cells from patient code R as well as other patients were used in confirmatory electrophysiology assays. Corrector activity was confirmed in at least one patient code different from R for 10 of the 12 confirmed hits shown in Figure 4 (see Suppl. Table S6 ).
Variability in corrector responses between patient samples could arise if different proportions of the ciliated epithelial cells (thought to be the main source of CFTR) were harvested from lung tissue or if there were differences in the expression of other ion channels and transporters whose activities affect CFTR. These factors were not investigated. Rather, activity was expressed as a percentage of a positive control with the expectation that this would normalize for major factors affecting CFTR function. We chose a combination of the corrector VX-809 and the potentiator VX-770 (the components of ORKAMBI) as the positive control for scaling. Since VX-770 was included in all assay buffers, the positive control condition is hereafter designated VX-809 = 100. In published work, VX-809 plus VX-770, both at their maximally efficacious concentrations, gives activity corresponding to approximately 35% of wild-type CFTR. 4
Surface Liquid Levels Can Be Measured Using Meniscus Width
Surface liquid height can be measured by a number of techniques including confocal microscopy with fluorescently labeled dextran.
33
Liquid height can also be accurately measured from meniscus width in low-magnification images, and the results have been shown to correlate with results from confocal microscopy.
29
Bronchial epithelial cells cultured on Transwell filters develop a liquid meniscus where the outer edge of the culture meets the wall of the Transwell insert (
Fig. 1A
). We captured bright-field images of entire wells from 96-well Transwell plates using a 2× objective on a high-content platform (
Configuration of Bronchial Epithelial Cell Surface Liquid Assays for F508del Corrector Discovery
To measure F508del correction, we used VIP (a physiological cAMP agonist) and the clinical potentiator VX-770 (ivacaftor) to fully activate CFTR. The clinical corrector VX-809 was used as a positive control. Dose titrations demonstrated that maximal responses were seen with 30 nM VIP, 1 µM VX-770, and 1–5 µM VX-809, consistent with previous reports, 4 and these conditions were used in the work that follows.
Airway epithelia can either absorb or secrete liquid depending on physiological conditions. Airway epithelia are relatively permeable to water, and fluid flow across the airway is believed to be driven by osmotic gradients established by active ion transport. Sodium absorption through the ENaC increases when the surface liquid volume is high and liquid absorption increases accordingly. Conversely, when the epithelium is dry, sodium absorption is inhibited and chloride secretion increases, allowing for the secretion of liquid to maintain mucosal hydration. 29
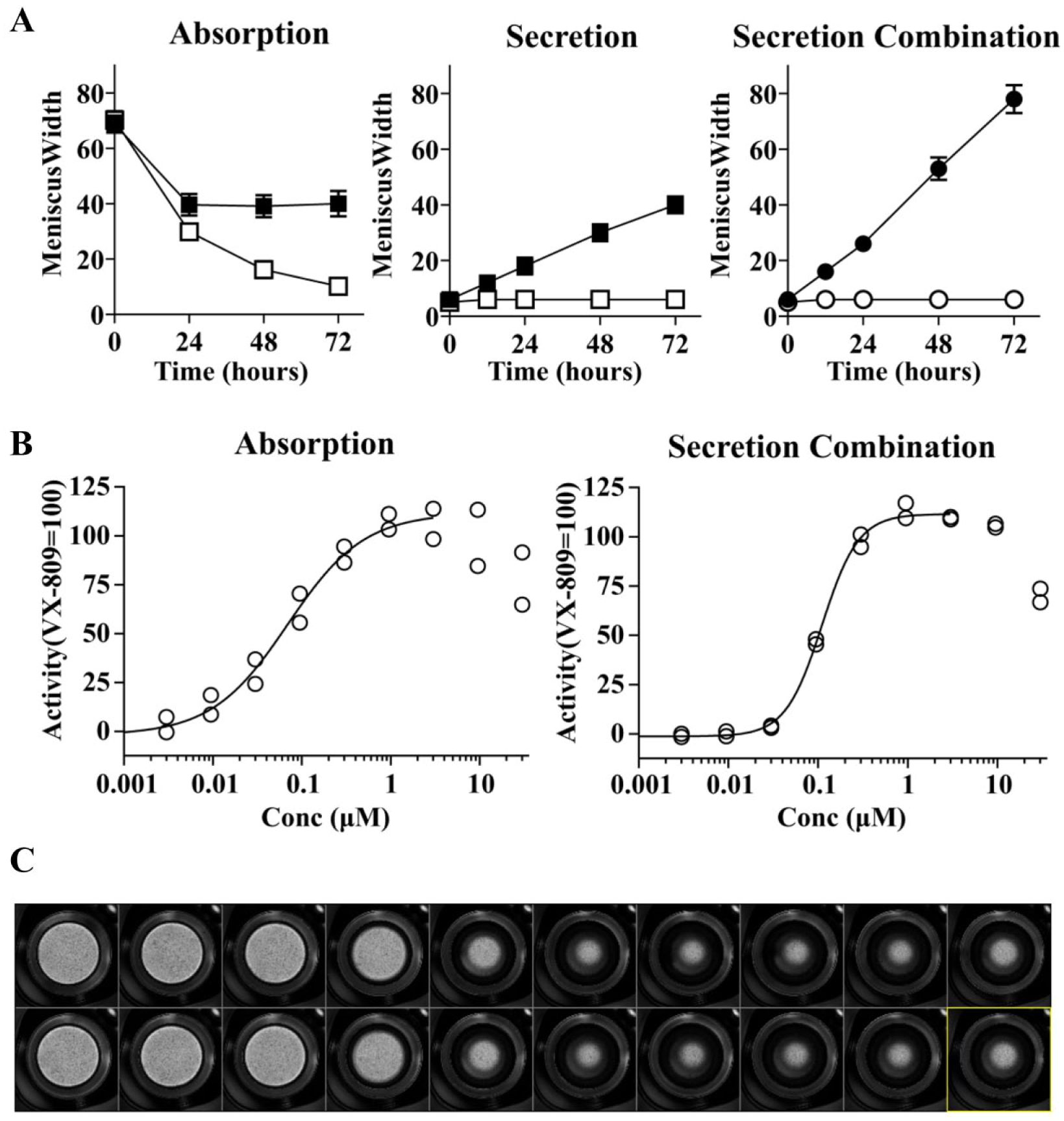
Both absorption and secretion formats were evaluated. In the absorption format, meniscus width decreased over time and the decrease was smaller for VX-809-treated epithelia than for vehicle controls (
Fig. 2A
, left). In secretion format, cultures with a dry apical surface were induced to secrete liquid by VX-809, while little or no secretion occurred in the absence of corrector (
(
A secretion combination assay format was also configured in which compounds were screened in combination with an in-house corrector, compound A, which had little ability to induce liquid secretion alone (details below). In this format, VX-809 also induced a robust increase of meniscus width over time ( Fig. 2A , right).
Configuration of a Surface Liquid Absorption Assay
Using VX-809 as a positive control, we evaluated assay performance with added volumes of 3–12 µL showing good performance up to 10 µL ( Fig. 1C ). The 12 µL result reflects flooding of the wells to an extent that absorption over 72 h did not lower the volume enough to allow the difference between VX-809 and the vehicle to be seen. An initial buffer volume of either 5 or 8 µL was used in all further work. We monitored meniscus width as a function of time and found that acceptable Z′ values of >0.3 were obtained at 72 h. We therefore used a fixed 72 h endpoint read for screening.
In screening work, VX-809 positive control and vehicle negative control wells were included in every plate. Representative positive and negative control data for 62 plates assayed over 9 weeks are shown in Figure 3A . Z′ values were greater than or equal to 0.3 for 79% of plates. We performed experiments to demonstrate acceptable within-plate uniformity using plates with VX-809 in all wells, acceptable reproducibility of responses between plates assayed on the same day, and acceptable agreement between responses from separate assay runs (not shown).
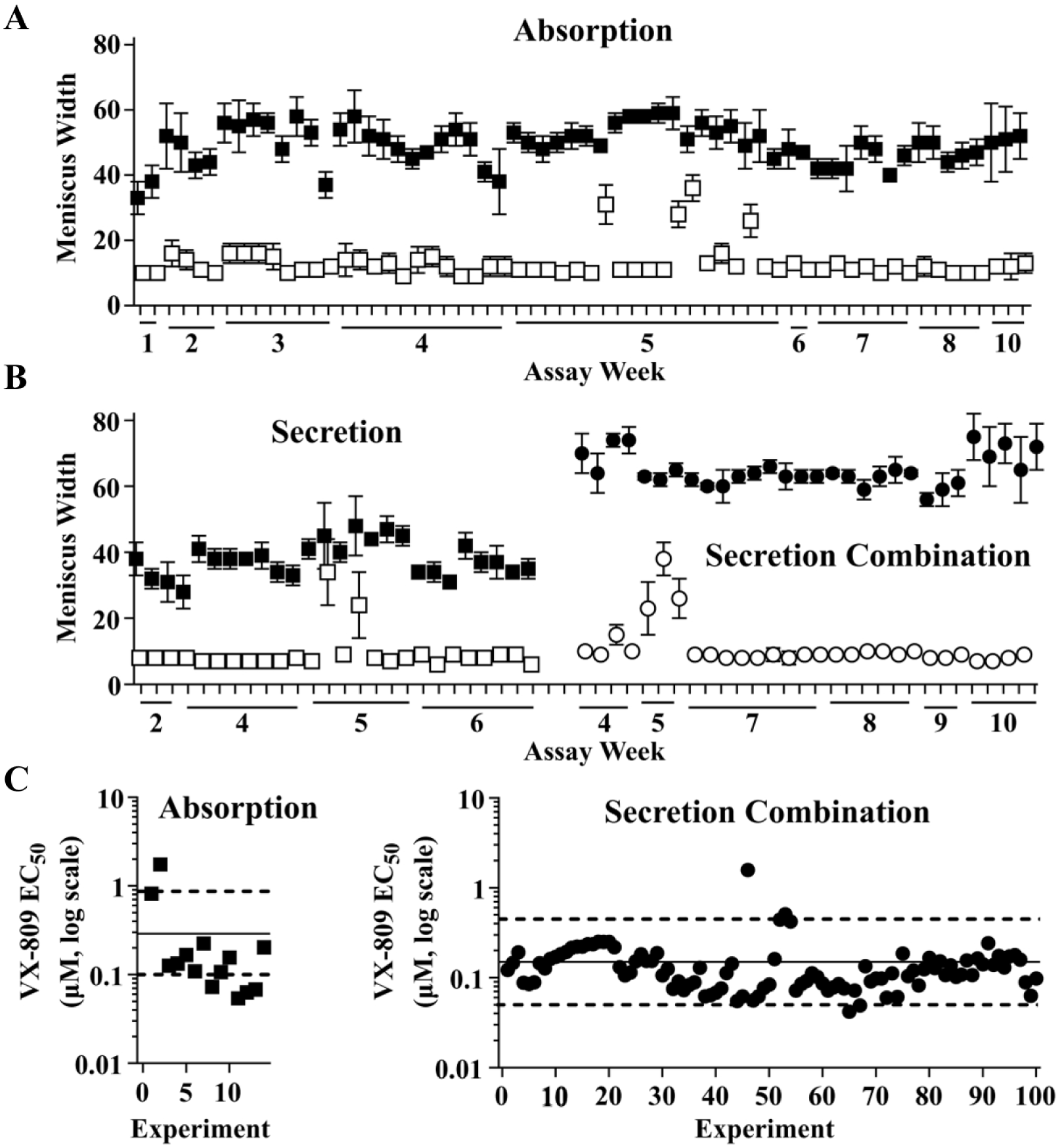
Comparison of assay performance for absorption, secretion, and secretion combination formats. For this data set, absorption assays were run with 5 µL of buffer added initially. (
Dose–response analysis of VX-809 was performed on a regular basis as another metric of assay performance. A representative VX-809 dose–response curve is shown in Figure 2B , left. A retrospective analysis of 14 dose–response curves for VX-809 ( Fig. 3C ) showed that EC50 values averaged 290 nM and 65% of values varied by no more than threefold from the mean (a range considered acceptable for most cell-based assays).34,35 This performance was considered modest but acceptable.
To maximize the number of compounds screened, we investigated the screening of mixtures of compounds as an alternative to one compound per well. This involved assessing both DMSO tolerance and the tolerance of increased compound burden. DMSO vehicle up to a level of 0.2% reduced VX-809 responses by less than 20%, and this level was used in subsequent experiments. Six compounds each at 10 µM (60 µM total compound burden) were reasonably well tolerated (see Suppl. Fig. S3 ) and sixfold compression was used for screening except where noted otherwise.
Configuration of a Surface Liquid Secretion Assay for Screening in Combination with a Corrector
Starting with dry cultures and using VX-809 as a positive control, we observed that liquid secretion increased over time and after 72 h reached a level similar to the steady state in the absorption assay (
We investigated a secretion assay format based on a combination with VX-809 in which the negative control was VX-809 and the positive control was VX-809 plus another corrector. Due to the robust efficacy of VX-809, it proved difficult to configure an acceptable assay for responses above the level of VX-809: the difference between corrector plus VX-809 and VX-809 alone was too small relative to the variability in the positive and negative controls leading to low Z′ values.
We therefore turned to a corrector from Pfizer’s CF discovery program, compound A (
(
We configured a secretion assay for correction in combination with compound A using VX-809 as a positive control. As shown in Figure 2A (right), compound A alone induced little or no secretion, but the VX-809 plus compound A combination reached levels nearly double that of VX-809 alone.
Representative positive and negative control data for 30 plates assayed over 6 weeks in the secretion combination format is shown in
Figure 3B
(right). Z′ values were greater than or equal to 0.3 for 97% of plates and the average Z′ was 0.7. We again performed repeated dose–response analysis of VX-809 (a representative curve is shown in
Pilot Screen Using Absorption Assay Showed a Good Discovery Rate for Correctors
We performed a pilot screen for new correctors using an in-house collection of 8414 compounds of diverse structures, good predicted physical properties and permeability, and acceptable predicted or measured cytotoxicity (referred to hereafter as the phenotypic screen library). This library was screened at an individual compound concentration of 10 µM with six compounds in each well and an initial buffer volume of 8 µL. Mixtures with activity >10% of the VX-809 positive control were identified and their single components tested individually, resulting in the identification of 90 hits. Supplemental Table S1 summarizes the screening, hit deconvolution, and hit confirmation results.
The confirmed hits were tested in F508del CF hBE cultures using an equivalent current electrophysiology assay that uses voltage and resistance measurements to quantify the CFTR-dependent response to the cAMP agonist forskolin and VX-770. The equivalent current electrophysiology assay and transepithelial current clamp (TECC) apparatus developed by Profs. R. J. Bridges and W. van Driessche allow the measurement of correction activity in differentiated CF hBE cells in 24-well filter plates.
27
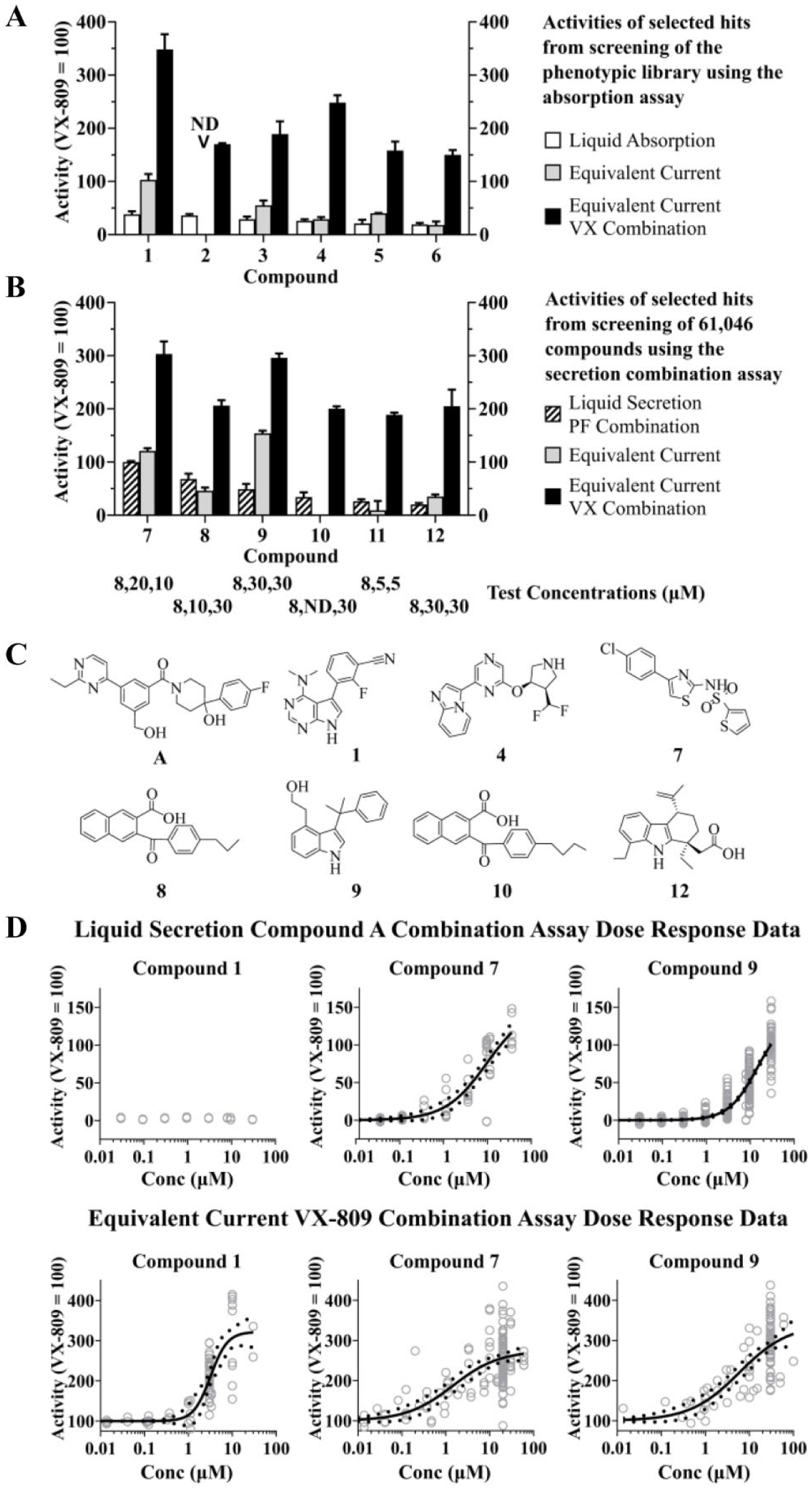
Equivalent current assays identified six compounds that when combined with VX-809 increased the combination response to ≥150% of the level of VX-809 alone (
CF hBE ASL Absorption Screen Identified More Correctors Than Recombinant F508del Cell Line Screens
Cell lines in which recombinant F508del CFTR is overexpressed have been used in HTS for corrector compounds using fluorescent sensors of ion flux, membrane potential dyes, or antibody detection of epitope-tagged cell surface CFTR.4,5,11,13 Screens of 150,000–164,000 compounds have yielded one or two correctors with activity in primary CF hBE cells.5,11 The discovery of six correctors with activity confirmed by electrophysiology assays in CF hBE cells from the ASL absorption screen of the 8414-compound phenotypic screen library was therefore notable, and we sought to determine whether this resulted from the custom library or the surface liquid assay.
The same phenotypic screen library was used to screen for corrector compounds using HEK cell lines expressing F508del R555K and F508del R1070W mutations. The presence of the second-site suppressor mutations R555K and R1070W is thought to bias for discovery of different classes of correctors that could then be combined to synergistically rescue F508del.36,37 Screening the library on both double mutants yielded two compounds that when combined with VX-809 increased the response to >150% of VX-809 alone in the F508del CF hBE equivalent current assay. One of these compounds, compound 1, was also identified in the liquid absorption screen. The other compound, compound 15, was a structural analog of compound 1 and likely has the same mechanism of action. The results are summarized in Supplemental Table S3 .
A similar screen of the phenotypic screen library in F508del 3S and F508del R1070W mutants in the CFBE41o- cell line 36 yielded no compounds that when combined with VX-809 increased the equivalent current response to >150% of VX-809 alone ( Suppl. Table S4 ). The discovery rate for CF hBE equivalent current-active correctors using the ASL assay was therefore at least three times greater than the HEK and CFBE41o- cell line assays when evaluated on the same compound collection. We concluded that the ASL assay and not the selection of compounds for the custom collection was responsible for the improved discovery rate.
Screening Using the Liquid Secretion Combination Assay
The secretion combination assay based on compound A was used to screen 61,046 compounds selected randomly from a large screening compound library as well as compounds from small, focused compound collections and hits from cell line screens for correctors. This resulted in the identification of six additional novel F508del correctors with robust activity in electrophysiology assays (when combined with VX-809, responses were ≥150% of VX-809 alone) (
Cumulatively, more than 100,000 compounds were screened sixfold compressed at a 10 µM individual concentration or as single compounds at 8 µM. The discovery rate for the secretion combination screen was lower than for the absorption screen, consistent with expectations from a pilot screen of the phenotypic library (see Supplemental Material). Nevertheless, several new correctors were discovered that when combined with VX-809 resulted in combined activity that was 200%–300% of VX-809 alone (the structures of compounds 7, 8, 9, 10, and 12 are shown in
The compounds with the highest equivalent current correction responses in combination with VX-809 (compounds 1, 7, and 9) were tested in dose–response format in the liquid secretion combination assay and the equivalent current combination assay (
ASL Hits Modulate CFTR Trafficking in Cell Lines and Primary Airway Cells
Some of the novel structural scaffolds discovered by the ASL screens became the subject of medicinal chemistry efforts to improve potency and efficacy with the goal of developing new clinical candidates. While F508del correction activity was a factor in the choice of which hit compounds to characterize further, other attributes, such as structural novelty and combination with VX-809 and compound A, were also considered. Further characterization of two selected examples, compounds 7 and 9, is described below.
Demonstration of F508del correction in a different cellular system by CFTR Western blot was used to provide evidence for an effect on CFTR trafficking. CFTR has different forms that vary in molecular weight due to changes in glycosylation. Core glycosylation in the endoplasmic reticulum (ER) gives the form known as band B, while further glycosylation in the Golgi apparatus results in a higher-molecular-weight band, C. Thus, trafficking of CFTR from the ER to post-Golgi compartments results in an increase in molecular weight, which is seen as conversion from band B to band C. It is believed that CFTR channel activity at the plasma membrane is due to post-Golgi band C protein. F508del corrector compounds typically increase the amount of band C and may also increase the C/(B + C) ratio.
Figure 5A
shows that compounds 7 and 9 increased the level of CFTR band C as well as the C/(B + C) ratio, both as single agents and in combination with VX-809 in an HEK cell line expressing recombinant F508del CFTR. In good agreement with the equivalent current functional responses, compound 9 also substantially increased the levels of band C as well as the C/(B + C) ratio in F508del patient airway epithelial cultures alone, in combination with VX-809, and in combination with compound A (
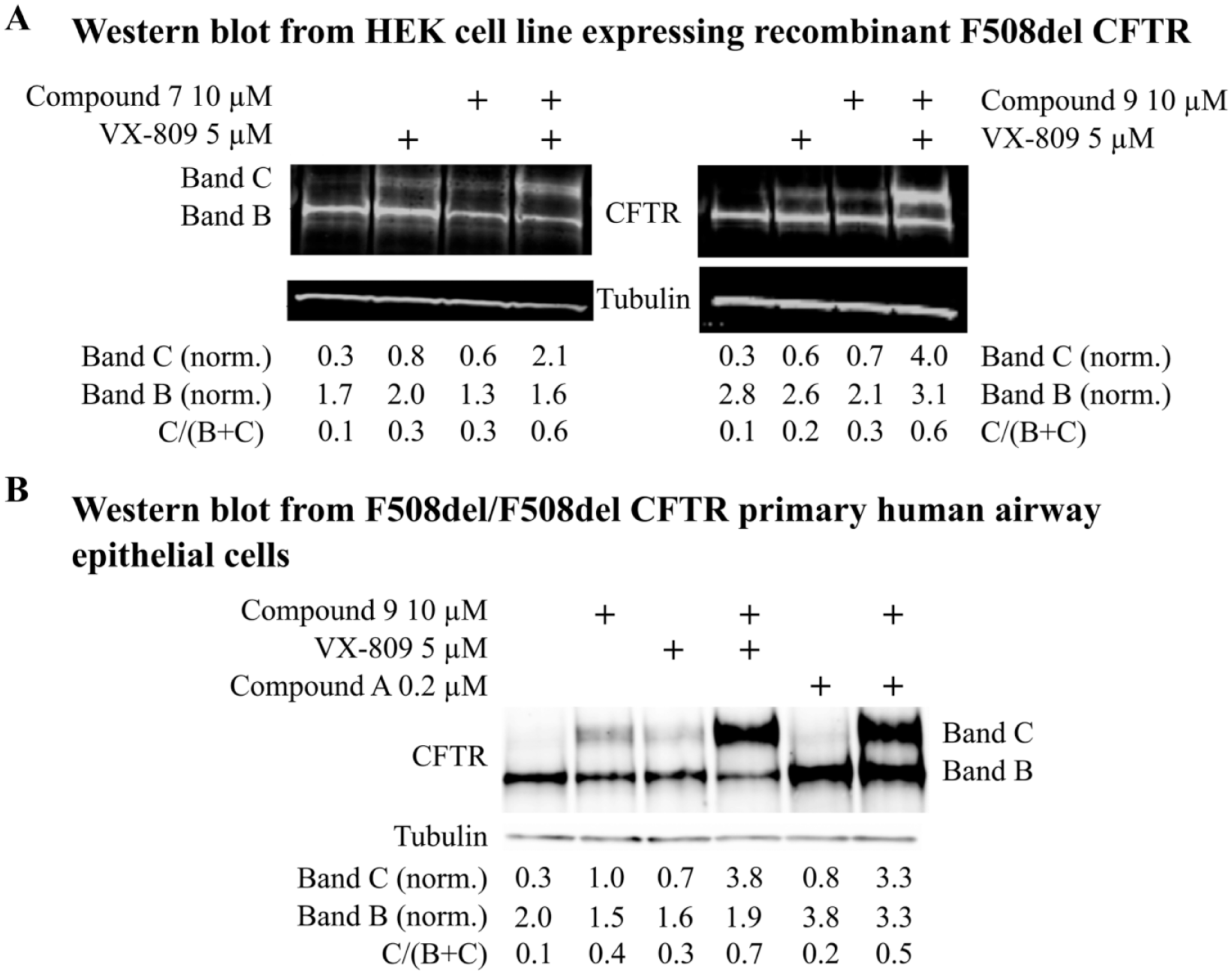
CFTR Western blots in an HEK cell line expressing recombinant F508del CFTR and F508del patient bronchial epithelia. (
Discussion
While F508del corrector compound efficacy and potency in patient-derived primary bronchial epithelial cultures are viewed as the data most likely to predict clinical responses, these assays have proven challenging to implement in formats with higher throughput than 24-well filter plates. Primary CF patient bronchial epithelial cells are not widely available, are limited in quantity, and lose CFTR expression upon passaging. Alternatives such as cell lines expressing recombinant F508del CFTR suffer from poor translation to the CF hBE assays, so HTS for new correctors has been an inefficient process. There was therefore an opportunity to develop improved methods for corrector discovery using patient bronchial epithelial cells.
We configured robust surface liquid absorption and secretion assays using CF bronchial epithelial cultures in 96-well filter plates. These cultures form a three-dimensional pseudostratified epithelium that secretes or absorbs liquid depending on the experimental conditions. Using an absorption assay format, we screened a library of 8414 compounds of diverse structures using mixtures of six compounds per well. Testing individual confirmed hits in F508del CF hBE cultures using a well-established electrophysiology assay, we identified six compounds that when combined with the clinical corrector VX-809 increased activity to a level more than 150% of the activity of VX-809 alone. Using the same activity threshold, we found only two CF hBE-active correctors when the same library was screened using either HEK or CFBE41o- cell lines expressing F508del together with second-site suppressor mutations. Based on this direct comparison, we concluded that the CF hBE liquid absorption screen was more efficient than the cell line screens, generating a greater number of confirmed corrector hits from a given number of compounds screened.
The ASL screens also yielded robust correctors that combined strongly with the clinical corrector VX-809, consistent with modes of action distinct from VX-809. Different mechanisms of action could include binding to different sites on CFTR or interaction with different proteins that modulate CFTR expression, folding, trafficking, or degradation. Such correctors may be useful in combination therapies based on VX-809-like correctors.
We evaluated several configurations of surface liquid assays and found that secretion formats had assay performance as good as or better than the absorption format. A secretion assay in combination with the in-house corrector compound A proved to be the most robust and was used to screen compounds selected randomly from a large screening compound collection as well as other compound collections. We cumulatively screened >100,000 additional compounds in this manner, finding six more correctors active in the CF hBE equivalent current assay. This represents the largest reported set of compounds to be screened directly in CF patient primary bronchial epithelial cells to date.
Screening for new correctors in combination with the in-house corrector involved a compromise. This assay configuration would be expected to poorly detect or miss correctors with a mode of action similar to compound A. Because compound A was used at its maximally efficacious concentration, compounds having a similar mechanism would be unlikely to further increase secretion and would not be detected. On the other hand, the presence of compound A would be expected to enhance the response to correctors with different modes of action, as it did with VX-809. Combination screening can therefore be useful to favor the discovery of compounds with mechanisms different from the compound used in the screen and likely to be complementary to that compound.
The translational potential of a phenotypic assay may be judged based on the physiological relevance of the cells or cell line, the stimulus, and the readout as summarized in the phenotypic screening “rule of 3.” 38 The present work employed patient-derived primary cells containing a disease-causing mutation in CFTR that had been allowed to differentiate into a tissue-like epithelium. VIP, a physiological stimulus, was used to initiate the assay. Surface liquid, the in vitro counterpart of airway epithelial lining fluid (which is affected in CF), was measured as the assay readout. The greater success of this approach relative to the cell line assays was likely due in part to the clinical relevance of the cell system, stimulus, and readout.
We confirmed that primary bronchial epithelial cells from F508del patients could be expanded to passage 4 while maintaining their capacity to differentiate and respond to corrector compounds. While co-culture methods using irradiated fibroblasts and Rho kinase inhibitor have been reported which allow increased passaging of CF hBE cells,39,40 we found that this approach was not needed to maintain corrector responses up to passage 4.
In contrast to typical cell line assays in which banked cells can sometimes be seeded directly into 384- or 1536-well plates, primary bronchial epithelial cell culture involved thawing, expansion in flasks over 2 weeks, seeding onto 96-well filter plates, and ALI culture for a further 4 weeks or more. In filter plates, basolateral medium changes were required three times per week and dedicated personnel were required to support screening. We noted differences in the behavior of cells derived from different patients, especially with regard to the rate of liquid absorption (data not shown). We chose to screen using cells from a single patient in the surface liquid assay but confirm activity in cells from one or more different patients using an orthogonal electrophysiology assay.
Our work demonstrated two ways in which it was possible to gain value within the throughput and labor constraints of a primary cell-based phenotypic assay. The first was through screening mixtures of compounds rather than one compound per well. The applicability of this approach depends on the tolerance of the assay to DMSO vehicle and compound burden. The second avenue was to test high-value compound sets that were enriched in active compounds or depleted of inactive or toxic compounds. For example, our custom phenotypic screen library prioritized cell-permeable compounds with good predicted physicochemical properties and low predicted toxicity; we also screened hits from a recombinant cell line screen for correctors.
In the case of F508del CFTR corrector discovery, screening using patient primary cells required a greater investment of effort and cost that was balanced by greater efficiency relative to cell line screens. Three-dimensional primary cell cultures that re-create a tissue-like system in vitro, such as organoids, are becoming increasingly available. Because the cell system used for screening likely strongly influences the kinds of mechanisms that are captured, we propose that 3D primary cell-based approaches should be considered as part of any broad-based discovery program. Their use for screening seems likely to identify compounds whose activity translates into the clinical setting.
Supplemental Material
Pregel__Suppl_revised3 – Supplemental material for High-Throughput Surface Liquid Absorption and Secretion Assays to Identify F508del CFTR Correctors Using Patient Primary Airway Epithelial Cultures
Supplemental material, Pregel__Suppl_revised3 for High-Throughput Surface Liquid Absorption and Secretion Assays to Identify F508del CFTR Correctors Using Patient Primary Airway Epithelial Cultures by Allison Berg, Shawn Hallowell, Mark Tibbetts, Chad Beasley, Tracy Brown-Phillips, Anita Healy, Leslie Pustilnik, Regis Doyonnas and Marko Pregel in SLAS Discovery
Footnotes
Acknowledgements
The authors wish to thank Cystic Fibrosis Foundation Therapeutics Inc. (Bethesda, MD) for support in the form of a research collaboration and for access to CF hBE cells; Prof. Scott Randell (University of North Carolina) for supplying CF hBE cells; Prof. Michael Myerburg (University of Pittsburgh) for the meniscus method and ASL assay advice; Dr. Martin Mense (CF Foundation Therapeutics Laboratory, Lexington, MA) for CFBE41o- data; Lucy Stevens for performing corrector screens using the HEK cell line iodide flux assay; Steven Hawrylik for leadership of cell culture support; David Gebhard for programming the Pipeline Pilot image analysis algorithm; Adam Gilbert, Markus Boehm, Rosalia Gonzales, Doug Johnson, Chris Keefer, Jacqueline Klug-McLeod, Erik Lachapelle, Rob Stanton, and Fabien Vincent for design of the phenotypic screen library; Yutian Zhan and Shawn O’Neil for immunofluorescence work; and Melissa McGill for organization and quality control of data for publication.
Supplemental material is available online with this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
