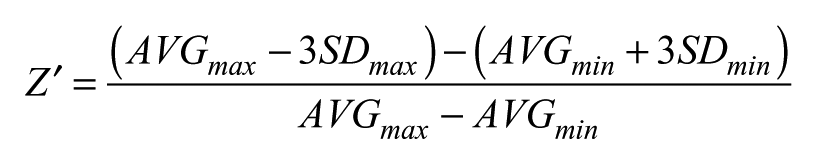Abstract
In this study we developed a new translational phenotypic in vitro model for high-throughput screening (HTS) of novel analgesics for treating neuropathic pain, in order to address the poor translation of traditional recombinant models. The immortalized dorsal root ganglia (DRG) neuron-like F11 cell line was selected based on its phenotype after differentiation. The acquisition of neuronal characteristics was evaluated by measuring the expression of TrkA as a DRG neuron marker (p < 0.01) as well as by measuring the global neurite length (p < 0.001). The response of F11 cells to ATP and KCl was obtained by measuring intracellular calcium concentration, dynamic mass redistribution, and membrane potential. A KCl-induced increase of intracellular calcium levels was chosen as the readout because of the better signal quality, higher reproducibility, and greater compatibility with HTS assay requirements compared with other methods. The response to KCl differed significantly between differentiated and undifferentiated cells (p < 0.05), with an EC50 value of 5 mM in differentiated cells. The model was validated by screening the Prestwick Chemical Library. Five hits already proposed for neuropathic-related pain were identified, with IC50 values between 1 and 7 µM. This cell model provides a new tool for screening novel analgesics for the relief of neuropathic pain.
Introduction
Neuropathic pain is caused by injury affecting either the central nervous system or the peripheral nervous system. It consists of spontaneous pain generated by nonpainful stimuli (allodynia) or of an exaggerated response to painful stimuli (hyperalgesia). 1 Neuropathic pain associated with peripheral nerve injury can be caused by metabolic disorders (e.g., diabetes), viruses (e.g., human immunodeficiency virus [HIV]), some drugs (e.g., cytostatic and antiretroviral drugs), mechanical nerve injury (e.g., carpal tunnel syndrome), immunological mechanisms (e.g., multiple sclerosis), or multiple neurotransmitter system dysfunction. 2
Neuropathic pain constitutes a health challenge because of the increasing number of patients suffering from the condition and the associated impaired quality of life. 3 Conventional analgesic drugs such as nonsteroidal anti-inflammatory drugs (NSAIDs) or opioids do not relieve neuropathic pain. Consequently, drugs such as tricyclic antidepressants, serotonin-specific reuptake inhibitors, and anticonvulsants (e.g., pregabalin or gabapentin) are prescribed in the treatment of this pathology, although these drugs are all rather ineffective and can have important adverse effects. 4 There is therefore a medical need for effective analgesics for the management of neuropathic pain.
Since the late 20th century, drug discovery programs have consisted of target-based assays, which evaluate the effects of molecules on a previously identified target. New analgesics thus share similar mechanisms of action and, moreover, the response of the target may not faithfully reflect what actually occurs in vivo. 5 A phenotypic approach would imply an important advance in the development of new analgesics to treat neuropathic pain. Thus, new drugs will be selected depending on their ability to reduce the symptoms of the disease, without the need to first determine the mechanism of action of the molecules. 6
Dorsal root ganglia (DRG) neurons are responsible for transmitting pain signals from the limbs to the central nervous system. 7 However, the use of primary DRG neurons in high-throughput screening (HTS) has several drawbacks, such as the need to sacrifice large numbers of animals, labor-intensive isolation procedures, the heterogeneity of cytochemical phenotypes, and the presence of nonneuronal cells that cause artifacts. 8
In recent years, several authors have described new methods for screening novel drugs for neuropathic pain by the employment of immortalized DRG neuron lines, like human induced pluripotent stem cell-derived sensory neurons, 9 or an immortalized mouse DRG neuron cell line. 8 In addition to these cell lines, immortalized DRG neuron lines that are hybrids of rat DRG neurons and mice neuroblastoma cells, such as F11 cells and ND7/23 cells, have been developed. 7 After exposure to different chemical substances, these cell lines undergo differentiation, thus acquiring a neuronal phenotype and responding to pain signals as DRG neurons do. Such cell lines could therefore be used to develop in vitro models for identifying new mechanisms of action or combinations of synergistic mechanisms of action that cannot be detected in formerly directed target-based assays.
Thus, our working hypothesis is that immortalized DRG neurons can be used as a simple translational phenotypic model for screening candidate drugs for the treatment/management of neuropathic pain. The final goal of the study was to develop an in vitro phenotypic model of analgesia based on DRG neuron-like cells for HTS to identify new drugs.
Materials and Methods
Cell Culture
F11 cells (08062601; ECCAC, Salisbury, England, UK) were grown in Dulbecco’s modified Eagle’s medium without sodium pyruvate (D5671; Sigma-Aldrich, Madrid, Spain) supplemented with 10% (v/v) nondialyzed fetal calf serum (F9665; Sigma-Aldrich), 2 mM glutamine (G7513; Sigma-Aldrich), and 100 units/mL penicillin and 100 µg/mL streptomycin (P0781; Sigma-Aldrich) in a humidified atmosphere containing 5% carbon dioxide at 37 °C. ND7/23 (92090903; ECCAC) cells were cultured in Dulbecco’s modified Eagle’s medium (D6546; Sigma-Aldrich) supplemented with 10% (v/v) fetal calf serum, 2 mM glutamine, and 100 units/mL penicillin and 100 µg/mL streptomycin in a humidified atmosphere containing 5% carbon dioxide at 37 °C.
Immunofluorescence Assays
Cells (80% confluence) were plated on clear-bottom, 96-well plates (6005558; PerkinElmer, Tres Cantos, Madrid, Spain) previously treated with 2 µg/mL laminin (L2020; Sigma-Aldrich) and 30 µg/mL poly-
Dibutyryl-cAMP (N 6 ,2′-O-dibutyryladenosine 3′,5′-cyclic monophosphate) (sc-201567; Santa Cruz Biotechnology, Heidelberg, Germany), forskolin (sc-3562; Santa Cruz Biotechnology), 1-methyl-3-isobutylxanthine (IBMX; I5879; Sigma-Aldrich), nerve growth factor (NGF; 480275; Millipore, Molsheim, France), retinoic acid (R2625; Sigma-Aldrich), and dialyzed fetal calf serum (FCSd; F0392; Sigma-Aldrich) were used as differentiation factors.
Both undifferentiated and differentiated F11 cells were fixed for 20 min with 4% paraformaldehyde (158127; Sigma-Aldrich) in phosphate-buffered saline (PBS) at 4 °C. The cells were then washed twice with PBS before being permeabilized with blocking buffer containing 5% bovine serum albumin (BSA) (10775835001; Sigma-Aldrich) and 0.1% Triton X-100 (T8787; Sigma-Aldrich) in PBS for 1 h at room temperature. The cells were then incubated overnight at 4 °C with 1:500 rabbit monoclonal anti-neurotrophic tyrosine kinase A (TrkA) receptor antibody (sc-118; Santa Cruz Biotechnology) diluted in blocking buffer. The cells were washed twice with blocking buffer and stained for 1 h with 1:250 CFL647 dye-conjugated goat anti-rabbit secondary antibody (sc-362292; Santa Cruz Biotechnology) in blocking buffer. Finally, the cells were stained with 1:1500 Hoechst 33342 (N21485; Thermo Fisher, Alcobendas, Madrid, Spain) in blocking buffer and washed two more times with blocking buffer. An Operetta High-Content Imaging System (PerkinElmer) was used to capture all fluorescence and bright-field images (12 fields per well, 20×). The images were analyzed with the Harmony High Content Imaging and Analysis Software (PerkinElmer). At least three independent sets of experiments, each including at least 24 replicates, were performed.
For quantification of neurite length, permeabilized cells were stained for 1 h with a 1:500 dilution of Alexa 488 dye-conjugated anti-β-tubulin mouse antibody (558605; Becton & Dickinson Biosciences, San Agustín de Guadalix, Madrid, Spain) and 2.5 µM nuclear stain DRAQ5 (108410; Abcam, Cambridge, UK) and washed twice with blocking buffer. An Operetta High-Content Imaging System was used to capture all fluorescence and bright-field images (12 fields per well, 20×). Image analysis was conducted with the Harmony High Content Imaging and Analysis Software (PerkinElmer). At least three independent sets of experiments, each with at least 24 replicates, were performed.
Dynamic Mass Redistribution Assays
F11 cells were seeded in 384-well EnSpire-LFC plates (6057408; PerkinElmer) pretreated with laminin and poly-
For differentiation of F11 cells, the culture medium was replaced with medium containing differentiation factors for another 72 h. Differentiated cells were then washed three times with assay buffer, in a JANUS Automated Workstation, and maintained in 40 µL of HBSS for 90 min at 37 °C before signal acquisition by DMR technology. The measurements were made at 37 °C. To obtain the DMR responses, an EnSpire Multimode Plate Reader (PerkinElmer) was used for whole-cell sensing. After recording a baseline for 20 min, 10 µL each of ATP (A2383; Sigma-Aldrich) and KCl (131494; PanReac-AppliChem, Castellar del Vallès, Barcelona, Spain) (dissolved in assay buffer) were transferred, in a JANUS Automated Workstation, and cellular responses induced by the compounds were monitored in real time. At least two independent sets of experiments were conducted, each with at least triplicate measurements.
Measurement of Intracellular Calcium Concentration
F11 cells were seeded in laminin and poly-D-lysine-coated clear, flat-bottomed, black-walled, 384-well plates (781091; Greiner Bio-One, Frickenhausen, Baden-Württemberg, Germany) at a density of 5000 cells/well. Culture medium and differentiation methods were as described for DMR assays. On the day of the assay, the medium was removed and replaced with 25 µL of fresh medium and incubated with 25 µL of FLIPR Calcium-6 dye (R8190; Molecular Devices, Sunnyvale, CA) diluted in HBSS containing 20 mM HEPES (pH 7.4) for 2 h at 37 °C. ATP and KCl were dissolved in assay buffer. Changes in intracellular calcium concentration ([Ca2+]i) were measured with a FDSS7000EX Functional Drug Screen System (Hamamatsu Photonics, Cerdanyola del Vallès, Barcelona, Spain) by excitation of the calcium-sensitive fluorescent dye with a light source at 470–495 nm and emission in the 515–575 nm range. FDSS tips were pretreated with a solution of 0.1% BSA diluted in the assay buffer.
Cell Membrane Potential Measurements
F11 cells were seeded at a density of 5000 cells/well in clear, flat-bottomed, black-walled, 384-well plates coated with laminin and poly-
Screening for Novel Drugs with Analgesic Effect in Neuropathic Pain of Peripheral Origin
The Prestwick Chemical Library of 1120 compounds was used for screening. Cells were seeded at a density of 5000 cells/well in clear, flat-bottomed, black-walled, 384-well plates coated with laminin and poly-
Data Analysis
The DMR data were background corrected by subtracting the response values elicited by assay buffer from the response values elicited by ATP and KCl dissolved in assay buffer. Data were normalized to the response of differentiated cells.
[Ca2+]i responses and cell membrane potential responses were obtained as the difference between the maximum and the minimum fluorescence values by considering the peak elicited by either ATP or KCl. Data were normalized to the maximum response to KCl of differentiated cells.
Data analysis was performed using GraphPad Prism 6.0 software (GraphPad Software, La Jolla, CA). A sigmoidal dose–response model was used to fit the concentration–response curves. The data obtained in immunofluorescence assays were compared using Student t tests. The data fits were compared using the extra sum-of-squares F test. Differences were considered statistically significant at p < 0.05.
Z′ factor was calculated as previously described using the following formula: 10
where AVG and SD are the average and the standard deviation of the maximum and minimum signal on the plate.
Results
F11 and ND7/23 Cells Acquired Neuronal Features and Expressed DRG Neuron Markers after Differentiation
The F11 and ND7/23 cell lines were exposed to the differentiation factors by supplementation of the growth medium. In F11 cells, three different combinations of differentiation factors were evaluated: (1) 30 µM forskolin with 1 mM dibutyryl-cAMP and 0.5% FCSd, (2) 0.5 mM dibutyryl-cAMP with 2 ng/mL NGF and 0.5% FCSd, and (3) 10 µM retinoic acid with 2% FCSd. In all three cases, the cells acquired neuronal characteristics after exposure to the differentiation factors ( Fig. 1A–C ), as revealed by comparison with undifferentiated cells ( Fig. 1D ). A neuronal phenotype, with large neurites and a swollen cell body ( Fig. 1A ) was observed when the medium was supplemented with forskolin and dibutyryl-cAMP as differentiation factors. The medium supplemented with dibutyryl-cAMP alone yielded fewer differentiated F11 cells ( Fig. 1B ), and the medium supplemented with retinoic acid induced the outgrowth of neurites in some cells but did not modify cell proliferation ( Fig. 1C ).
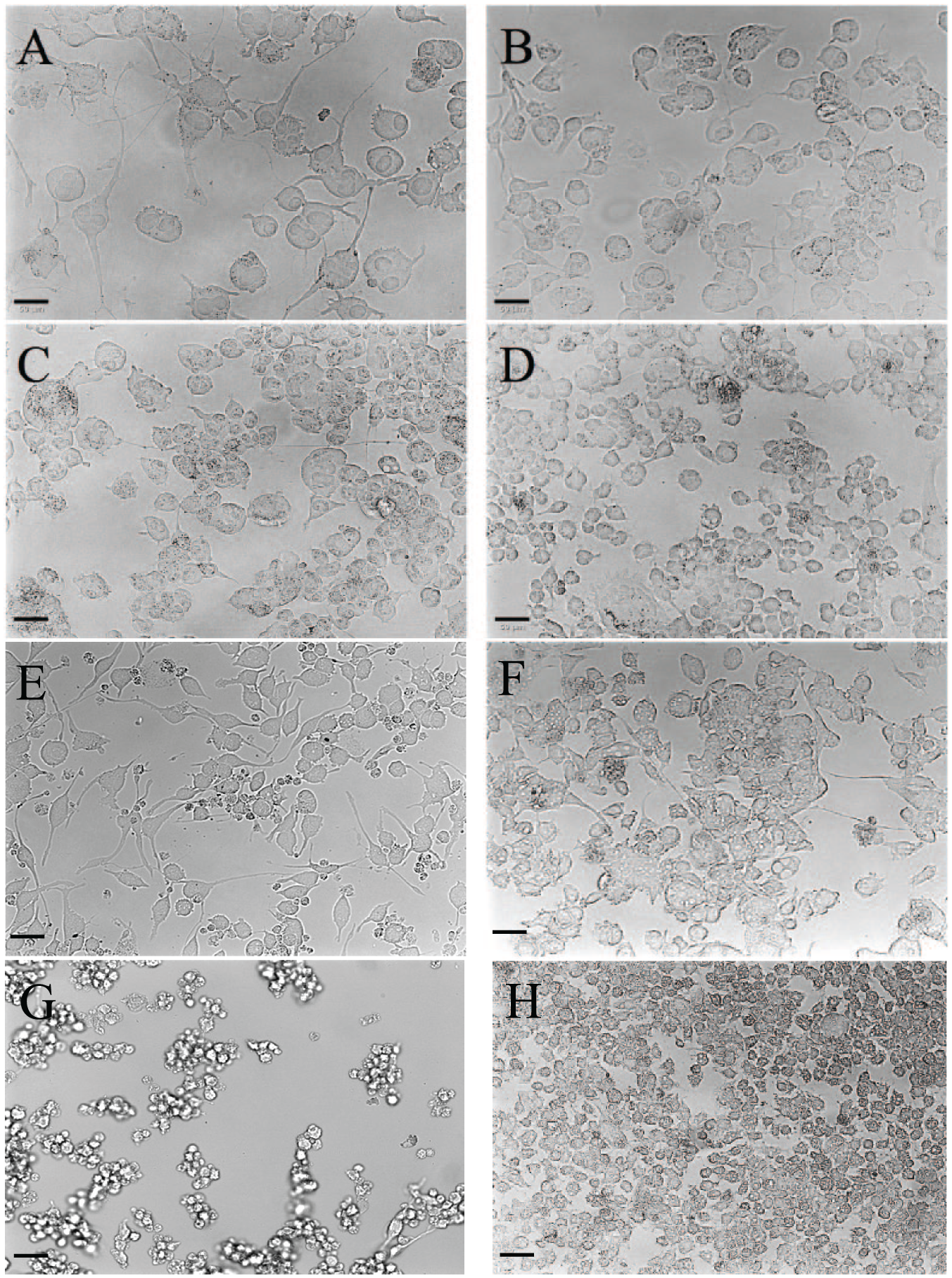
Differentiation induced the acquisition of neuronal characteristics in F11 cells but not in ND7/23 cells. Morphology of F11 cells differentiated by using (
In ND7/23 cells, three different combinations of differentiation factors were evaluated: (1) 60 µM forskolin with 1 mM dibutyryl-cAMP, 1.3 ng/mL NGF, and 0.5% FCSd; (2) 1 mM dibutyryl-cAMP with 2 ng/mL NGF and 0.5% FCSd; and (3) 20 µM retinoic acid with 100 µM IBMX, 2 ng/mL NGF, and 2% FCSd. Exposure of ND7/23 cells to differentiation factors yielded cells with different phenotypes depending on the treatment ( Fig. 1E–H ). The high variability in ND7/23 cells led us to focus on the F11 cell line.
The acquisition of neuronal characteristics in F11 cells was confirmed by measuring the expression of the TrkA receptor in differentiated cells relative to undifferentiated cells ( Fig. 2A–D ). Regardless of the method of differentiation, an increase in the expression of the TrkA receptor relative to undifferentiated cells was observed (p < 0.01) ( Fig. 2E ).
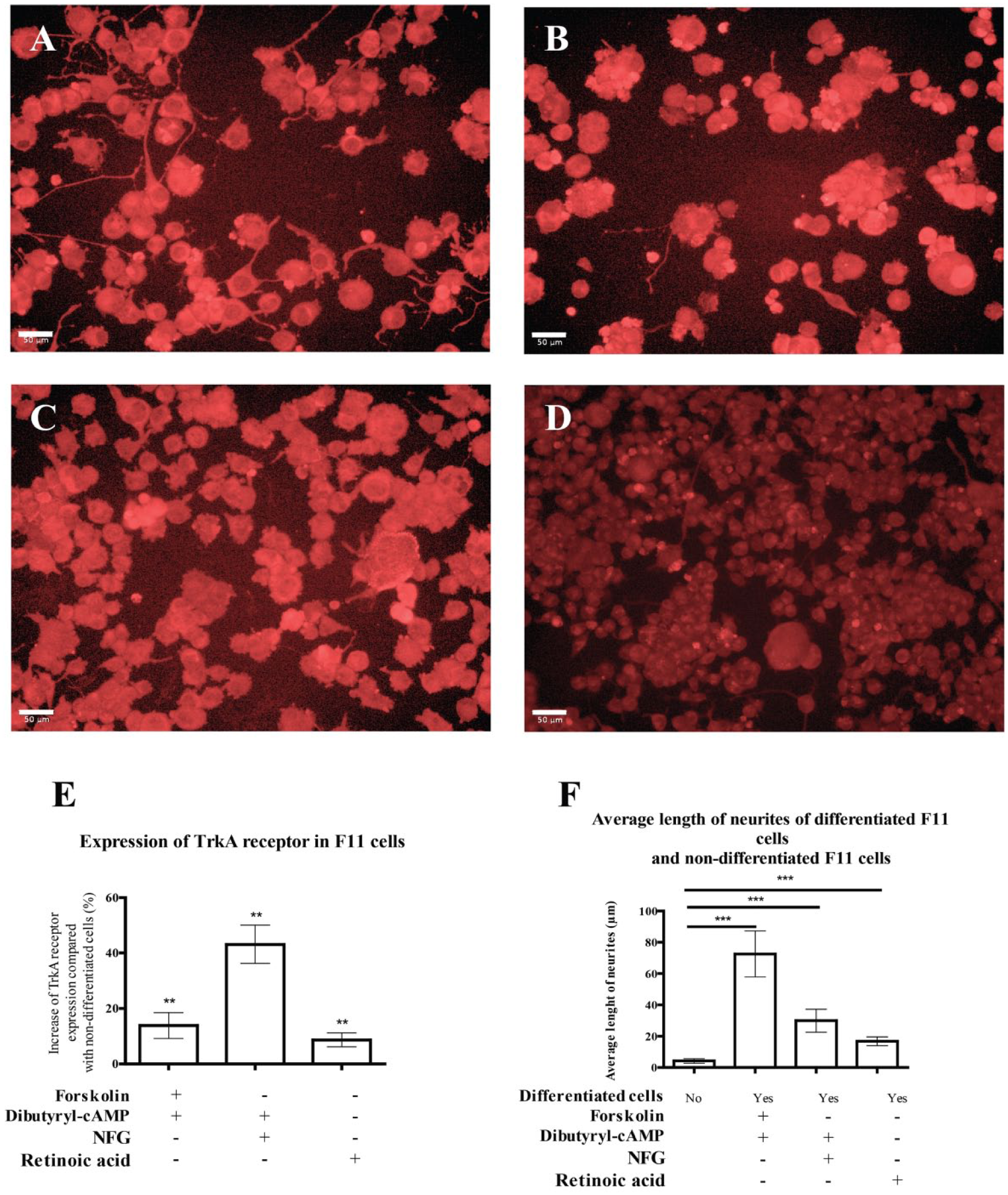
Differentiation increased the expression of DRG neuron markers and induced a greater increase in neurite length. Representative images of CFL647 stain of TrkA receptor in (
Acquisition of the DRG neuron phenotype was confirmed by measuring the average length of neurites in differentiated F11 cells relative to undifferentiated F11 cells. We confirmed a significant increase in the global length of neurites in differentiated F11 cells relative to undifferentiated cells (p < 0.001) ( Fig. 2F ).
KCl, but Not ATP, Induced a Dose-Dependent Effect Response of Differentiated F11 Cells
The response to increasing concentrations of ATP in F11 cells differentiated with forskolin and dibutyryl-cAMP was evaluated by using three different techniques: measurement of DMR, estimation of [Ca2+]i, and assessment of membrane potential. The differentiated F11 cells showed a concentration-dependent response to ATP in all three methods, but there were no statistically significant differences between the responses of differentiated cells and undifferentiated cells (
Fig. 3A–C
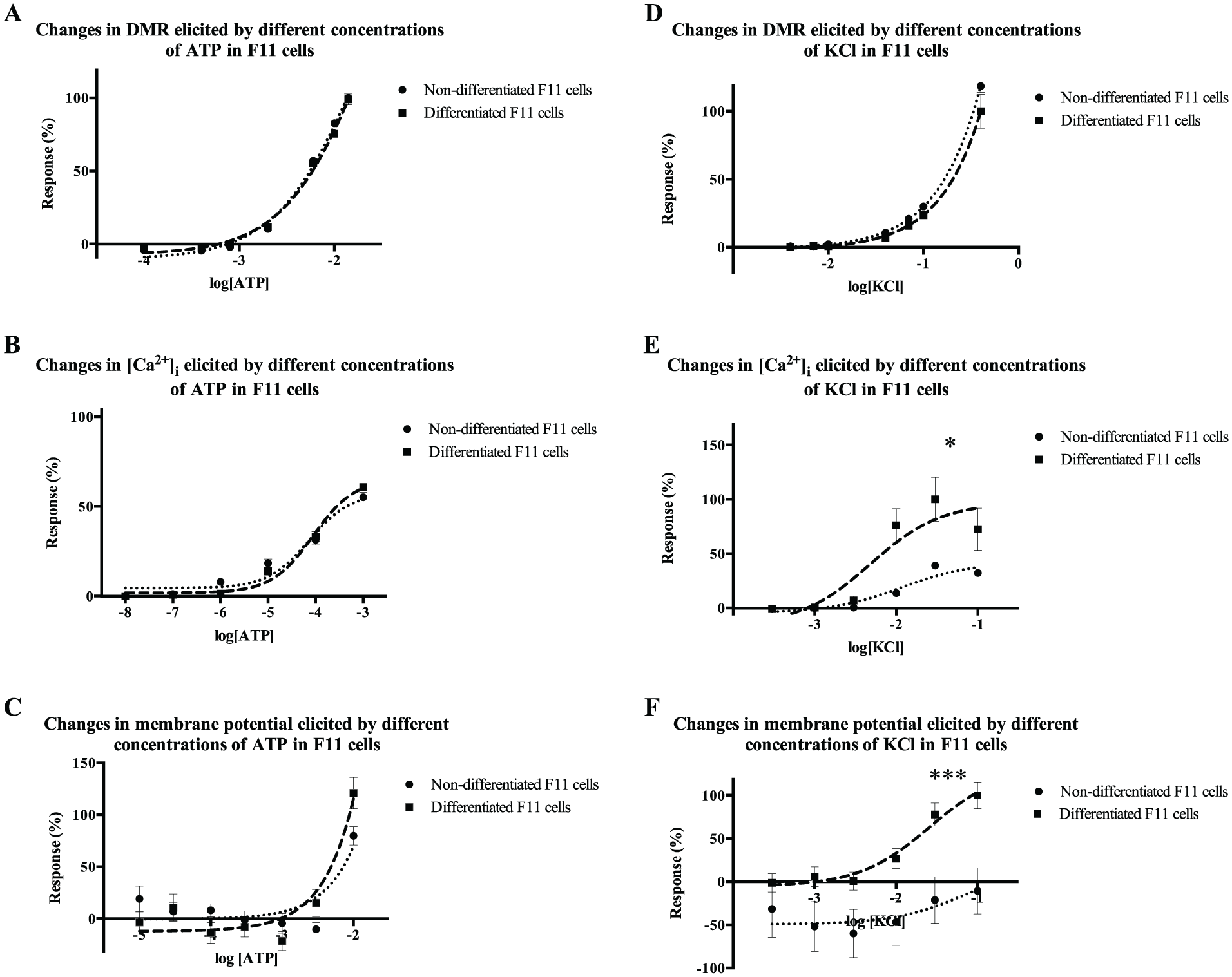
F11 cells responded to ATP before and after differentiation, but the response to KCl was different before and after differentiation. The response of F11 cells before and after differentiation with 1 mM dibutyryl-cAMP, 30 µM forskolin, and 0.5% FCSd to ATP and KCl was evaluated by three different techniques. (
Potency (pEC50) and Efficacy (Emax) of ATP and KCl in Differentiated and Undifferentiated Cells, as Evaluated by Three Different Methods.
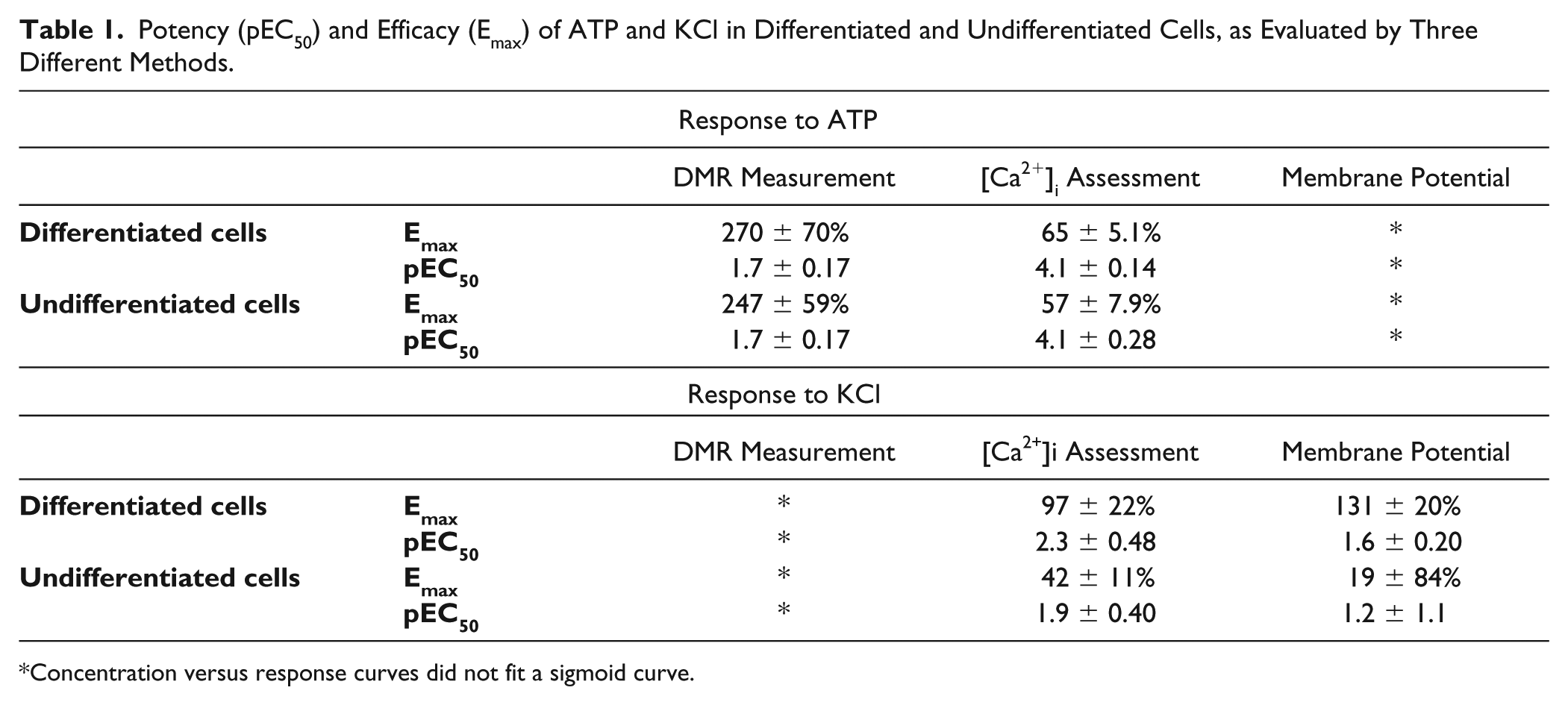
Concentration versus response curves did not fit a sigmoid curve.
The response to increasing concentrations of KCl of F11 cells differentiated with forskolin and dibutyryl-cAMP was assessed using the aforementioned techniques (measurement of DMR, estimation of [Ca2+]i, and assessment of membrane potential). Differentiated F11 cells showed a concentration-dependent response to KCl in all three techniques, and in addition, the variations in [Ca2+]i and membrane potential elicited by KCl in differentiated cells were significantly different from the responses elicited in undifferentiated cells (p < 0.05) (
Fig. 3D–F
Differentiated F11 Cells Are a Useful Model for HTS of Novel Analgesics
The Prestwick Chemical Library of 1120 compounds was used to validate the method described above in the presence of an inflammatory soup composed of 10 µM histamine, 10 µM serotonin, 1 µM bradykinin, and 10 µM prostaglandin E2. The expression of genes codifying the receptors for those mediators was confirmed by RNA-seq (
Suppl. Table S1
). The presence of these inflammatory mediators emulates a painful situation by enhancing DRG excitability. Indeed, the response to KCl was increased after incubation of cells in the inflammatory soup (
Fig. 4A
). Compounds were screened at a concentration of 10 µM. The hit threshold was established at mean + 2 SD, and nine hits were able to inhibit the response of differentiated F11 cells to 32 mM KCl higher than 65.5%: N
6
-methyladenosine, nicardipine, felodipine, nitrendipine, protriptyline, nimodipine, promethazine, piribedril, and prenylamine (
Fig. 4B
). The Z′ average value was 0.55 ± 0.035. Five hits were confirmed by concentration–response curve studies generating significant inhibition of the response to 32 mM KCl of more than 66% after exposure to the inflammatory soup (
Fig. 4C–G
,
Suppl. Fig. S1
,
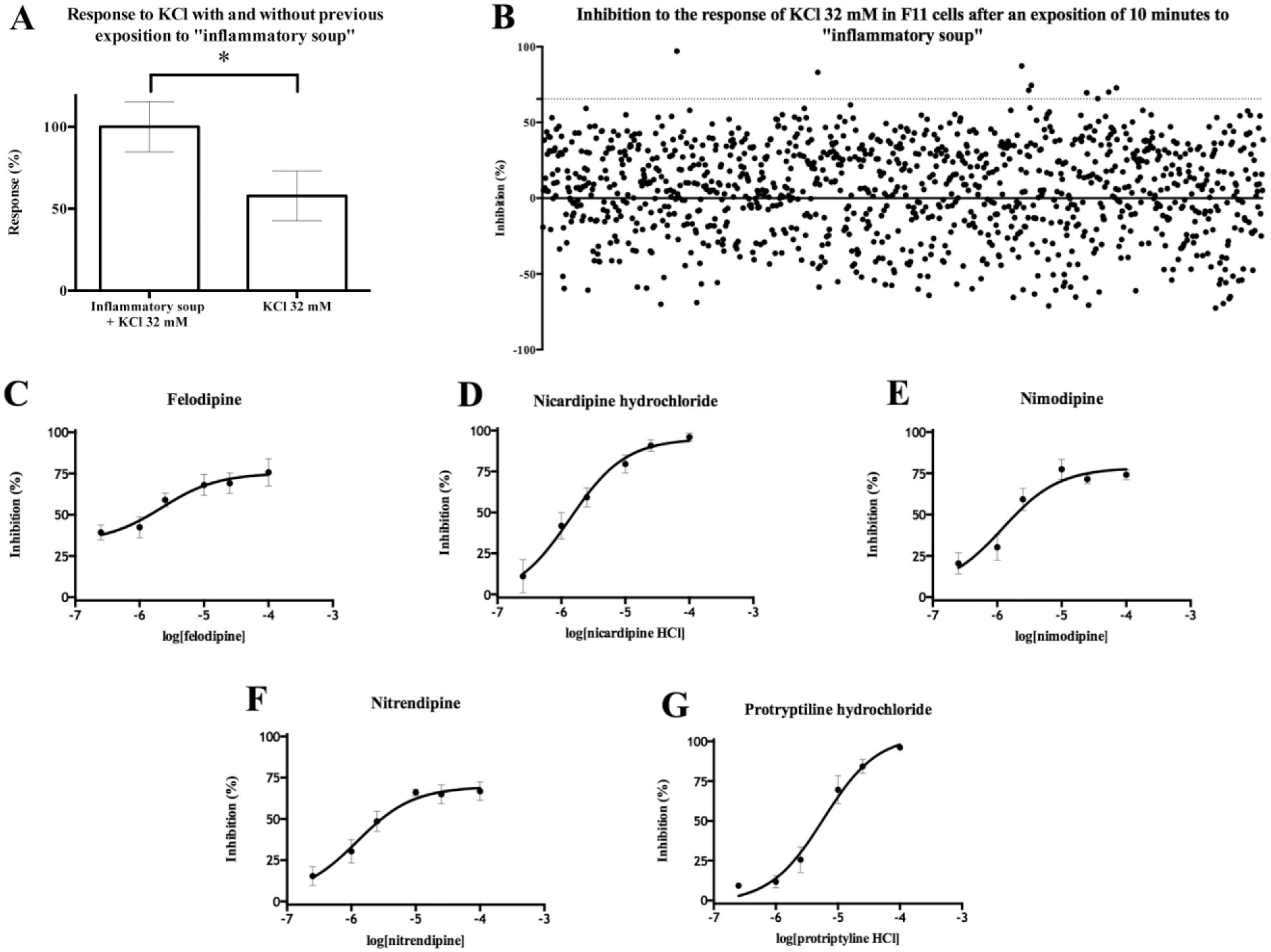
Five hits demonstrated activity reducing differentiated F11 cell excitability to 32 mM KCl after exposure to inflammatory soup. (
Potency (pIC50) Values for Compounds That Demonstrated Activity in Reducing the Excitability of Differentiated F11 Cells.

Discussion
The aim of this study was to establish and validate a new in vitro HTS model for testing novel analgesic drugs in DRG neuron-like cells.
HTS of novel analgesics is usually performed by studying the effects of thousands of compounds on well-characterized neuronal targets expressed in recombinant models (these cell lines are generally of neoplastic origin). 11 In an attempt to improve the translatability of these assays, we propose a new model for HTS of novel analgesics based on the use of immortalized DRG neuron-like cells. Damage to DRG neurons, which represent the neuroanatomical center of nociception, leads to peripheral neuropathic pain. 2
First, we evaluated the neuronal phenotype of two differentiated cell lines: F11 and ND7/23 cells. Both are immortalized DRG neuron hybrids of murine neuroblastoma cells and rat DRG neurons. F11 cells in particular have been shown to retain many characteristics of native DRG neurons, including the synthesis of neurotransmitters and expression of neuropeptides, chemokine and opioid receptors, and voltage-gated calcium channels (VGCCs). F11 cells have therefore been used as surrogates for primary DRG neurons in many studies, including studies involving the neurobiology of neuropathic pain. 12
Several treatments have been described to differentiate these cells to achieve a DRG neuron phenotype: the use of dibutyryl-cAMP, forskolin, 13 and retinoic acid. 14 Here, we compare the use of these substances to differentiate F11 and ND7/23 cells from cells with a neural phenotype.
Dibutyryl-cAMP is a cell membrane-permeable analog of cAMP. 13 Forskolin is a diterpene that activates the enzyme adenylate cyclase, inducing increased generation of intracellular cAMP. 13 Retinoic acid activates the β2 receptor, upregulating the transcription of neural genes. 15 IBMX supplementation was used to differentiate ND7/23 cells and inhibit phosphodiesterases, thus reducing intracellular degradation of cAMP. 16
An increase in intracellular cAMP stabilizes different transcription factors, increases the length of neurites, and induces morphological changes in cells, yielding neuronal characteristics. 13
For ND7/23 cells, after exposure to dibutyryl-cAMP and forskolin, cells with a neuronal phenotype were observed. In addition, the cell number and cell differentiation varied widely between wells.
By contrast, the three differentiation methods tested in F11 cells induced outgrowth of neurites and acquisition of neuronal features, as observed by comparison with undifferentiated cells. More specifically, differentiation increased TrkA receptor expression, which is a nociceptive neuron marker. The TrkA receptor interacts with NGF, which is very important in pain perception. 17 After measuring the global length of neurites, we observed that dibutyryl-cAMP plus forskolin yielded overall longer cell neurites. Thus, dibutyryl-cAMP and forskolin were finally selected as differentiation factors for further testing in the F11 cell line.
To ascertain the excitability of differentiated cells, the response of both cell lines to increasing concentrations of algogenic stimuli such as ATP and KCl was evaluated.18,19 For this purpose, we compared three different methods compatible with HTS of large compound libraries:20–22 (1) DMR measurement, which enables the assessment of small changes in the distribution of the cellular mass 23 and is suitable for detecting the redistribution of cell constituents triggered on receptor stimulation; 24 (2) estimation of [Ca2+]I, which is important in neurons due to its crucial role in pain transmission and the modulation of T-type calcium channel activity modifying neuronal excitability; 25 and (3) changes in membrane potential measured with Dibac4(3), a fluorescent dye that can modify fluorescence intensity in response to changes in membrane potential, which enables the detection of neuron depolarization in response to painful stimuli. 20
F11 cells were stimulated with either ATP or KCl. ATP exerts an algogenic effect by acting on two different types of purinoceptors: the P2X receptors coupled to ion channels that are selective for cations activated by ligand, and the P2Y G protein-coupled receptors.26,27 Both families of receptors are involved in pain transmission and have been found in DRG neurons, taking part in purinergic signaling of pain. 28 KCl acts as an algogenic stimulus exerting nonspecific depolarization in nociceptive neurons. 29 Depolarization induces the opening of VGCCs. 30 This event therefore leads to the activation of regulatory proteins and the phosphorylation of ionic channels and enzymes, prompting the transmission of painful signals by increasing neuronal excitability. 31
Differentiated F11 cells responded to increasing ATP concentrations in a concentration-dependent manner for the three evaluation methods. However, the response observed was not significantly different from the response elicited by the same concentrations of ATP in undifferentiated F11 cells. This lack of differences may be due to the fact that ATP is a ubiquitous mediator that exerts numerous roles in cells. 32 In fact, the presence of purinergic receptors has been extensively described, even in neuroblastoma cells. 33 Thus, the effect observed after stimulation with ATP may not directly reflect the DRG excitability.
When KCl was used to stimulate DRG neurons, it elicited changes in DMR in both undifferentiated and differentiated F11 cells. These changes were expected as this technique detects morphological changes, not only neuronal excitability. 23
On the other hand, significant differences between the Ca2+ levels and membrane potential modifications elicited by KCl in differentiated cells versus undifferentiated cells were observed. This observation is consistent with the fact that KCl evokes depolarization, which triggers an increase in cytoplasmic Ca2+ concentration in DRG neurons. 34 Therefore, measurement of intracellular calcium was selected as the evaluation method because regulation of its levels is directly involved in pain transmission. Moreover, measurement of intracellular calcium has several practical advantages, such as good signal quality, compatibility with HTS assay equipment, and high reproducibility. 35
In order to assess the suitability of this model for screening, an HTS trial was carried out with the Prestwick Chemical Library, which comprises 1120 off-patent drugs and some natural substances. 36 The trial was carried out as a proof of concept to ascertain whether the assay could identify active analgesics for the treatment of neuropathic pain of peripheral origin. For this purpose, differentiated cells were exposed to an inflammatory soup to enhance the excitability of differentiated F11 cells to KCl, mimicking the exposure to inflammatory mediators after nerve injury, which causes hyperalgesia and neuropathic pain. 37
The assay proved to be robust, with an average Z′ value of 0.55 ± 0.035. Under these conditions, five confirmed hits inhibited the response of differentiated F11 cells to 32 mM KCl by more than 66% (mean + 2 SD). This high threshold is ascribed to the heterogeneity of mechanisms of action present in the compounds of the Prestwick Chemical Library, which leads to a wide distribution of activities among all the compounds tested. All the drugs have known analgesic activity in neuropathic pain of peripheral origin. Felodipine, nicardipine, nitrendipine, and nimodipine all are calcium channel inhibitors belonging to the chemical group of the 1,4-dihydropiridines, extensively used as antihypertensive drugs inducing vascular muscle relaxation. 38 Use of these drugs as analgesics in neuropathic pain has also been described due to their inhibitory effect in neural L-type calcium channels. 14 Protriptyline, the fifth drug identified, is a tricyclic antidepressant belonging to a family of drugs that are used clinically as analgesics in neuropathic pain. 39
In summary, in this study we developed a novel DRG-like sensitive neuron model for screening novel analgesics for the relief of neuropathic pain. The model was validated using the Prestwick Chemical Library, identifying five hits already described as analgesics for neuropathic pain. The study findings contribute to developing a translational HTS model for the early identification of novel drugs for the relief of neuropathic pain.
Supplemental Material
Supplemental_Material_for_A_new_model_by_Martinez_et_al__(2)_revEsteve – Supplemental material for A New Model of Sensorial Neuron-Like Cells for HTS of Novel Analgesics for Neuropathic Pain
Supplemental material, Supplemental_Material_for_A_new_model_by_Martinez_et_al__(2)_revEsteve for A New Model of Sensorial Neuron-Like Cells for HTS of Novel Analgesics for Neuropathic Pain by Antón L. Martínez, José Brea, Xavier Monroy, Manuel Merlos, Javier Burgueño and María Isabel Loza in SLAS Discovery
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Joint R&D Unit Esteve-USC (IN853A-2014-08), co-financed by the Galician Innovation Agency and the Spanish Ministry of Economy and Competitiveness (MINECO) within the framework of the Spanish Strategy of Innovation in Galicia; and by the Phenopain project (RTC-2015-4207-1) of the RETOS-COLABORACIÓN program of the MINECO, co-financed by the European Union through the European Regional Development Fund (ERDF). A. L. Martínez-Rodríguez was in receipt of a predoctoral fellowship from the FPU program (Spanish Ministry of Education, Culture and Sports).
Supplemental material is available online with this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.