Abstract
Malaria remains a major cause of morbidity and mortality worldwide with ~3.3 billion people at risk of contracting malaria and an estimated 450,000 deaths each year. While tools to reduce the infection prevalence to low levels are currently under development, additional efforts will be required to interrupt transmission. Transmission between human host and vector by the malaria parasite involves gametogenesis in the host and uptake of gametocytes by the mosquito vector. This stage is a bottleneck for reproduction of the parasite, making it a target for small-molecule drug discovery. Targeting this stage, we used whole Plasmodium falciparum gametocytes from in vitro culture and implemented them into 1536-well plates to create a live/dead phenotypic antigametocyte assay. Using specialized equipment and upon further validation, we screened ~150,000 compounds from the NIH repository currently housed at Scripps Florida. We identified 100 primary screening hits that were tested for concentration response. Additional follow-up studies to determine specificity, potency, and increased efficacy of the antigametocyte candidate compounds resulted in a starting point for initial medicinal chemistry intervention. From this, 13 chemical analogs were subsequently tested as de novo powders, which confirmed original activity from the initial analysis and now provide a point of future engagement.
Introduction
According to the World Health Organization (WHO), the parasites responsible for causing human malaria, Plasmodium falciparum, Plasmodium vivax, Plasmodium ovale, Plasmodium malariae, and Plasmodium knowlsei, infect up to 216 million people per year, resulting in an estimated 445,000 fatalities. 1 Current antimalarial regimens are not fully effective due to emerging drug resistance and their inability to prevent transmission.2,3 Approximately 40% of the world’s population lives in areas where the risk of malaria transmission is high, typically the tropic zones located closer to the equator. 4
The WHO, along with charitable organizations such as the Gates Foundation and the global ministries from highly affected countries, is now driving renewed efforts toward the eradication of malaria. While vector control strategies, including impregnated bed nets, and drug therapies, such as artemisinin combination therapies, have helped tremendously, their efficacy may have plateaued. Additionally, while at least one vaccine, Mosquirix or RTS,S/AS01, exists, it appears to have limited efficacy in inducing either robust antigenic responses or long-lasting immunity, thus leaving some vaccinated individuals unprotected. It remains under study to determine just how effective it is.5–7 Vector control and case management by chemotherapy remain the primary means of control for malaria. In the best scenario, while we may be able to achieve low levels of infection, additional measures are still necessary for complete eradication. This is true, in part, because drug-resistant parasite strains are evolving, thus providing a reservoir for transmission. 8 In other words, the route to complete elimination of malaria resides in our ability to not only block both the hepatic and erythrocytic stages of parasite replication within the human host, but also transmission of the parasite between the human host and the mosquito vector.
Hence, we are focused on identifying inhibitors that act on this late-stage of the parasite’s life cycle. Gametocytes are part of the sexual phase of the malaria parasite life cycle and are essential for transmission from one host to another via the mosquito. They are produced in the human host and once mature remain in a state of arrested cell development until ingested by a feeding female Anopheles mosquito where they undergo further development ( Fig. 1A ).9,10 P. falciparum gametocytes require 10–12 days to reach maturity, passing through five distinct stages (I–V) along the way ( Fig. 1B ). 11 As the gametocytes mature to stage V, they become relatively metabolically inert until they are taken up by a feeding mosquito, and they can remain in the host circulation for significant periods of time at subpatent levels. This lack of metabolic activity correlates to less druggable targets, making them insensitive to almost all commonly used antimalarial agents, and our own recent findings confirm and extend these results.12,13 However, some reports have found that late-stage gametocytes have increased lipid requirements, which is divergent from early-stage asexual parasites, making fatty acid metabolism a possible target. 14 Still, we have confirmed that late-stage gametocytes (stages IV and V) are largely speaking refractory to treatment by all the classes of antimalarial agents tested. These data indicate that even clinically effective antimalarial treatment of the host may not lead to the prevention of transmission. The metabolite of 8-aminoquinoline primaquine (PQ) is currently the only licensed antimalarial drug that is effective against late-stage gametocytes. 15 Unfortunately, there are side effects to treatment with PQ that decrease its usefulness. It is also known to cause acute hemolysis in patients with glucose-6-phosphate dehydrogenase (G6PD) deficiency, for whom PQ is contraindicated. G6PD deficiency is highly prevalent in malaria-endemic areas and severely limits the use of PQ. Early reports that tafenoquine has activity against gametocytes have not been confirmed, and it appears to have no in vitro activity against stage IV/V gametocytes. 16 It too is contraindicated in patients with G6PD deficiency. Therefore, blocking transmission of malaria by targeting late-stage gametocytes remains a top priority.
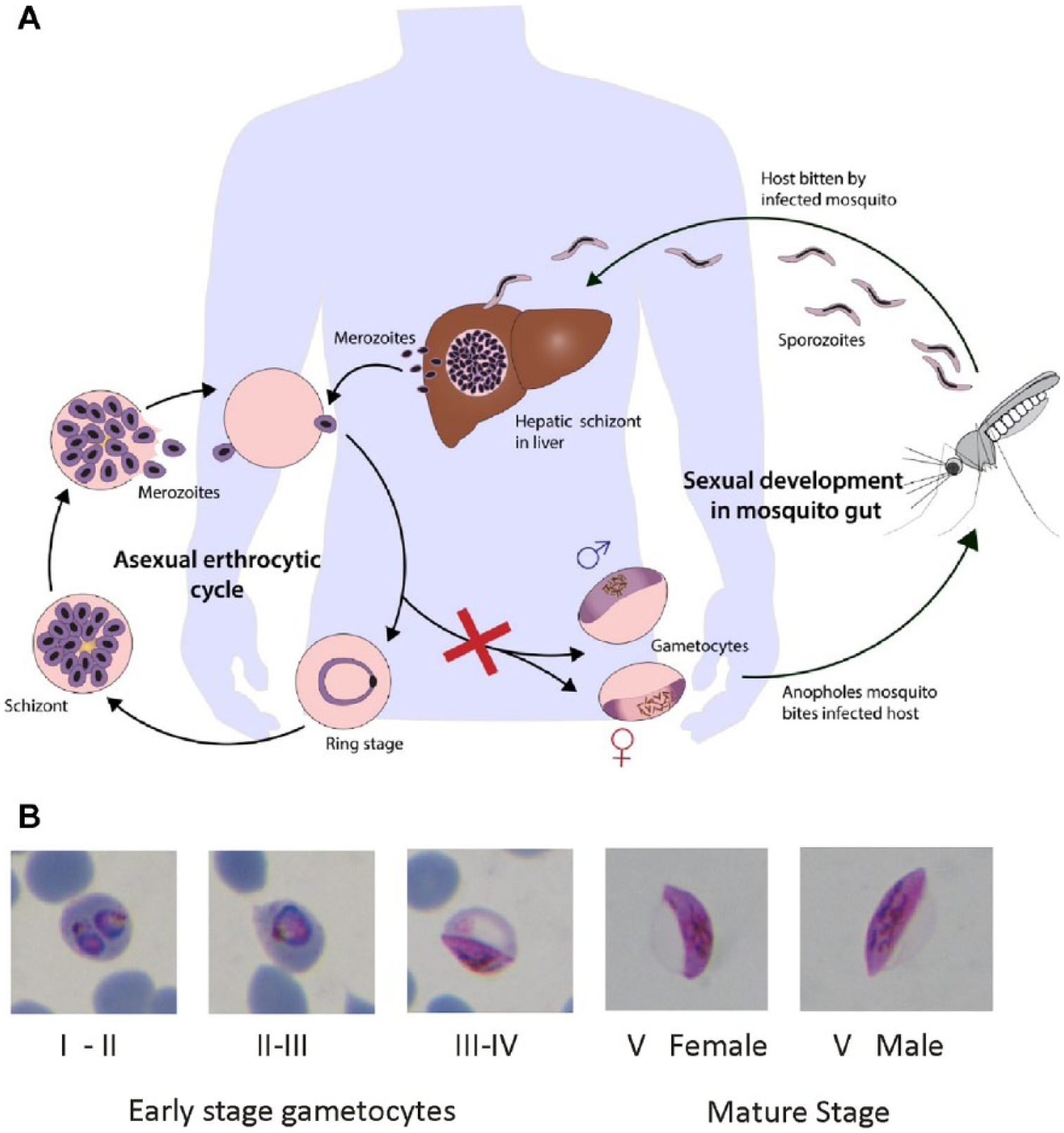
(
Traditionally, high-throughput screening (HTS) campaigns use one of two approaches to identify new agents against a given disease: cell based or target based. In contrast to the asexual stages, because late-stage gametocytes are essentially terminally differentiated, the effect of compounds on this stage cannot be monitored using cell multiplication as a marker. In addition, working with gametocytes is technically challenging and, until very recently, methods for the production of large numbers of gametocytes were not available.17,18 Consequently, we have little information with respect to the differences in protein expression and metabolism between late-stage gametocytes and asexually replicating parasites. As a result, neither a cell- nor a target-based HTS approach to identifying compounds with activity against gametocytes was feasible. However, as a result of improved gametocyte production methods and the development of tools for downstream analysis, by our group and others, large-scale antigametocidal HTS approaches are now feasible.19–21
Herein, we describe the miniaturization and completion of a large-scale screening agenda using a phenotypic approach incorporating a live/dead assay that monitors ATP, and hence the metabolic activity of gametocytes. Late-stage gametocytes were used in a rapid, cost-effective, highly sensitive luminescent detection assay in 1536-well format for the detection of phenotypic gametocyte inhibitors. We also report the results of a mammalian cell cytotoxicity assay that was employed as a counterscreen.
Materials and Methods
Assay reagents
BacTiter-Glo microbial cell viability assay detection reagents were purchased from Promega Corp. (Madison, WI, part G8231). Late-stage (stage V) gametocytes were isolated and cryopreserved as previously described. 17 These were subsequently shipped by overnight courier frozen on dry ice, to The Scripps Research Institute (TSRI) Florida, where they were stored at −130 °C until the time of use. Cryopreserved gametocytes were thawed and returned to culture via a dropwise saline solution revitalization procedure followed by resuspension at 62,500 gametocytes per milliliter in RPMI-1640 (Life Technologies, 11875) supplemented with 10% human serum (BioWorld, 30611043-1), 0.2% sodium bicarbonate, and 10 µg/mL gentamycin (Life Technologies, 15750). Four microliters of gametocyte cell suspension was then dispensed into each well of 1536-well microtiter plates (250 cells per well) (Corning, Corning, NY, part 7254) using the Flying Reagent Dispenser (FRD; Aurora Biosciences Corp.). Next, 44 nL of test compound in DMSO, low control (DMSO alone, 1.08% final concentration) or high control (media only, no gametocytes), was added to the appropriate wells using the GNF pintool (GNF Systems). Low controls in this sense are “vehicle-only” treated wells and will elicit an uninhibited luminescence response equivalent to the amount of viable gametocytes within the well, whereas high-control wells have no gametocytes and elicit no appreciable luminescence, which is anticipated to be corollary to a compound with maximal inhibitory effect. Plates were incubated for 24 h at 37 °C in 95% relative humidity (RH) at 5% CO2, whereupon 4 µL of BacTiter-Glo solution was added to all wells. Plates were centrifuged at 1000 rpm for 1 min and luminescence was measured using a 30 s read on the ViewLux microplate reader (PerkinElmer, Turku, Finland).
Compounds
The Molecular Libraries Small Molecule Repository (MLSMR) library was provided by the NIH’s Molecular Libraries Initiative. Briefly, the MLSMR library is a highly diversified collection of small molecules (more than 50% of compounds exhibit molecular weights between 350 and 410 g/mol) comprising both synthetic and natural products, from either commercial or academic sources, that can be grouped into the three following categories: specialty sets of known bioactive compounds such as drugs and toxins, focused libraries aimed at specific target classes, and diversity sets covering a large area of chemical space. Solid samples (powders) were purchased by the University of Kansas Specialized Screening Center from various commercial sources. The purity of all final compounds was confirmed by reverse-phase high-performance liquid chromatography/mass spectrometry (RP-HPLC/MS) analysis and determined to be >95%. 1H and 13C nuclear magnetic resonance (NMR) spectra were recorded on a Bruker AM 400 spectrometer (operating at 400 and 101 MHz, respectively) or a Bruker AVIII spectrometer (operating at 500 and 126 MHz, respectively). NMR spectra for the 13 purchased compounds used in the structure–activity relationship (SAR) analysis are shown in the Supplemental Materials.
Data Acquisition and Analysis
All screening assays were run on a Kalypsys/GNF robotic platform in 1536-well microtiter plates. Luminescence was measured by the ViewLux plate reader using a luminescence protocol that utilized a 30 s exposure time. Raw data were uploaded into an institutional HTS database (Symyx Technologies, Santa Clara, CA) for further processing. The activity of each well was normalized on a per-plate basis using the equation shown below.
The percent inhibition of each test compound was calculated as follows:
where Test Compound is defined as wells treated with test compound, Low Control is defined as wells treated with DMSO only, and High Control is defined as wells treated with media only and no gametocytes.
Each assay plate underwent a quality control check; a Z′ value greater than 0.5 was required for acceptance of data. 22 Any assay plate for which the Z′ value did not exceed 0.5 was rescheduled for another robotic procedure until an acceptable Z′ value was observed.
A mathematical algorithm was used to determine nominally inhibiting compounds in the primary screen. Percent inhibition is calculated for each individual well, including control wells, using the expression included above and applied here to determine an interval-based cutoff that was applied to take into account assay noise and general variability within compound activity to help preserve and identify more hits at the primary HTS phase. 23 Four values were calculated: (1) the average percent inhibition of all high controls tested plus three times the standard deviation of the high controls, (2) the average percent inhibition of all low controls tested minus three times the standard deviation of the low controls, (3) the average percent inhibition of all compounds tested between (1) and (2), and (4) three times their standard deviation. The sum of two of these values, (3) and (4), was used as a cutoff parameter; that is, any compound that exhibited greater percent inhibition/activity than the cutoff parameter was declared active.
For titration experiments, triplicate percent inhibition values were plotted against compound concentration. A four-parameter equation describing a sigmoidal concentration–response curve (CRC) was then fitted with an adjustable baseline using Assay Explorer software (Symyx). CRCs and IC50 values presented in this paper were generated by Prism (GraphPad Software, San Diego, CA). In cases where the highest concentration tested did not result in greater than 50% inhibition, the IC50 was determined manually as greater than the highest concentration. Compounds with an IC50 greater than 10 µM were considered inactive. Compounds with an IC50 equal to or less than 10 µM were considered active.
Counterscreen
The purpose of this cell-based assay is to determine if compounds that originally were found to inhibit viability of late-stage P. falciparum gametocytes in a primary HTS assay are also cytotoxic to mammalian cells, an undesirable feature. In the past, counterscreening utilizing this method when screening for other parasite targets has been extremely helpful in ruling out nonspecific cytotoxic compounds, and hence we sought to apply the same principle here. 24
This assay employs Jurkat cells, a human T-cell line originally isolated from an adolescent male with T-cell leukemia. The cells are grown in suspension that facilitates tissue culture and preparation for dispensing in the assay. The endpoint assay presented here employed a similar reagent to the primary assay, CellTiter-Glo luminescent reagent (Promega Corp., part G7573), which contains luciferase enzyme to catalyze the oxidation of beetle luciferin to oxyluciferin and light in the presence of ATP, in this case from Jurkat cells. Since metabolically active cells produce ATP, an increase in the number of dead or dying cells will correlate with a reduction in ATP levels. As designed, compounds that inhibit cell viability and reduce intracellular ATP will reduce the catalytic conversion of luciferin into oxyluciferin, resulting in decreased luciferase activity and well luminescence. This assay included doxorubicin as a positive control, an antibiotic used as an anticancer drug that elicits an appropriate antiproliferative effect on Jurkat cells. 25 Compounds are tested in triplicate using a 10-point 1:3 dilution series starting at a maximum nominal test concentration of 83.3 µM.
Jurkat cells (clone E6.1; ATCC cat. TIB-152) were routinely cultured in suspension in T-175 standing flasks at 37 °C in 95% RH at 5% CO2. Media consisted of RPMI-1640 containing 10% dialyzed fetal bovine serum, 0.1 mM NEAA, 1 mM sodium pyruvate, 25 mM HEPES, 5 mM
Prior to the start of the assay, cells were suspended to a concentration of 100,000 cells/mL in media. To start the assay, 5 µL of media was dispensed into the first two columns of a 1536-well plate and 5 µL of cell suspension was dispensed to the remaining wells (500 cells/well). The assay was started immediately by dispensing 42 nL of test compound in DMSO, doxorubicin (8 µM final concentration), or DMSO alone (0.6% final concentration) to the appropriate wells. The plates were then incubated for 48 h at 37 °C in 95% RH at 5% CO2.
Following the 2-day incubation, plates were equilibrated to room temperature (RT) for 10 min and 5 µL of CellTiter-Glo reagent was added to each well. Plates were centrifuged and incubated at RT for 10 min. Well luminescence was measured on the ViewLux plate reader. The percent inhibition for each compound was calculated exactly as it was for the primary HTS assay using the following controls: Test Compound is defined as wells containing test compound, Low Control is defined as wells containing DMSO only (0% inhibition), and High Control is defined as wells containing 8 µM doxorubicin (100% inhibition).
Results
Since all HTS at Scripps is done in 1536-well format, our former 96-well method was further miniaturized to optimize conditions based on the Z′ value and reproducible response to methylene blue, a known inhibitor of malaria ( Table 1 ).13,26 In particular, we optimized the number of gametocytes necessary per well to remain in the linear range of the detection modality, the incubation time, and further validated for HTS in the context of our fully automated screening system. This resulted in using 250 gametocytes per well at >90% viability, immediately adding test compound in 75:25 DMSO:H2O, and incubating for 24 h prior to adding BacTiter-Glo luminescence detection reagent. These conditions led to a reproducible Z′ factor averaging 0.53 ± 0.1 over the course of 124 assay test plates. After testing >153,000 compounds at ~11 µM, we identified 100 actives using a cutoff set at >38% inhibition ( Fig. 2 ). This effectively sets the hit cutoff such that it approaches the level of the sample field. Still, the hit rate was very low, observed as 0.1% of the compounds. This is further reflected by the average Z score assessed over the same course of plates equal to 0.60 ± 0.05. Here the Z factor is calculated using the method to generate Z′ but incorporates the use of the sample field in the expression used in place of the low controls. As such, achieving lower Z scores, such as those <0.5, would represent higher activity of the samples tested, which is not the case here.
Stepwise Protocol for the 1536-Well Plate Live/Dead Gametocyte Assay.

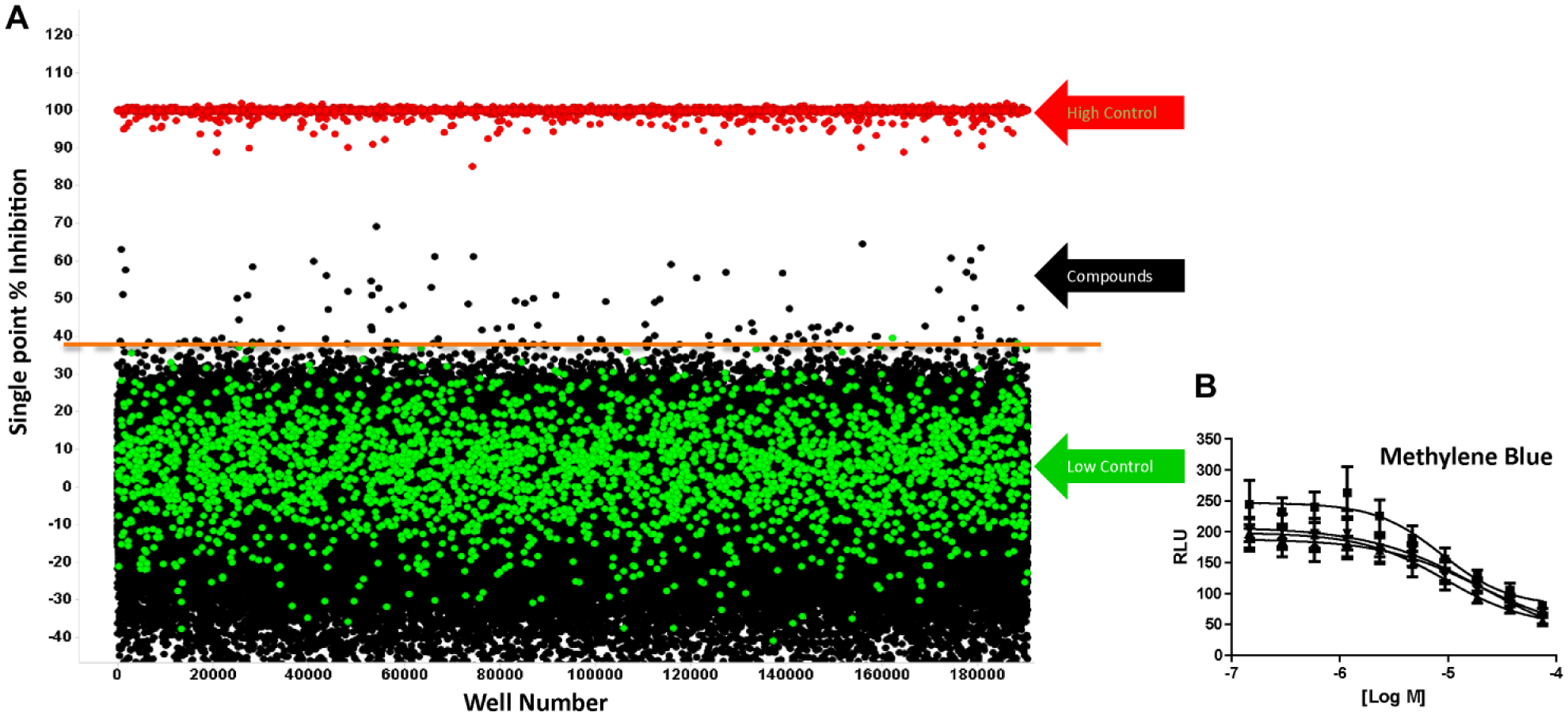
(
Considering the low number of actives identified, we proceeded immediately to the CRC determination phase, avoiding the expenditure of time and cost associated with the typical confirmation and counterscreen phase. Since this biological target is of major significance and difficult to hit, and the NIH repository allowed for up to 275 compounds to proceed to CRCs, we expanded the hit list to include the top 275 molecules for testing. Of those, 244 were available and subsequently tested in both the primary assay and counterscreen assay for CRC determination. Of the 244 molecules, only two compounds were identified with moderate potency and selectivity ( Table 2 ). These two molecules, SR-01000751192 and SR-01000471307, were found to have potencies <10 µM in the whole gametocyte assay ( Fig. 3 ). Only SR-01000751192 exhibited minimal cytotoxicity when tested in the Jurkat T-cell toxicity assay (>83 µM), an example of an HTS-qualified assay for cytotoxicity profiling. 24
Summary of the Ultra-HTS Campaign to Identify Inhibitors of Late-Stage P. falciparum Gametocytes.
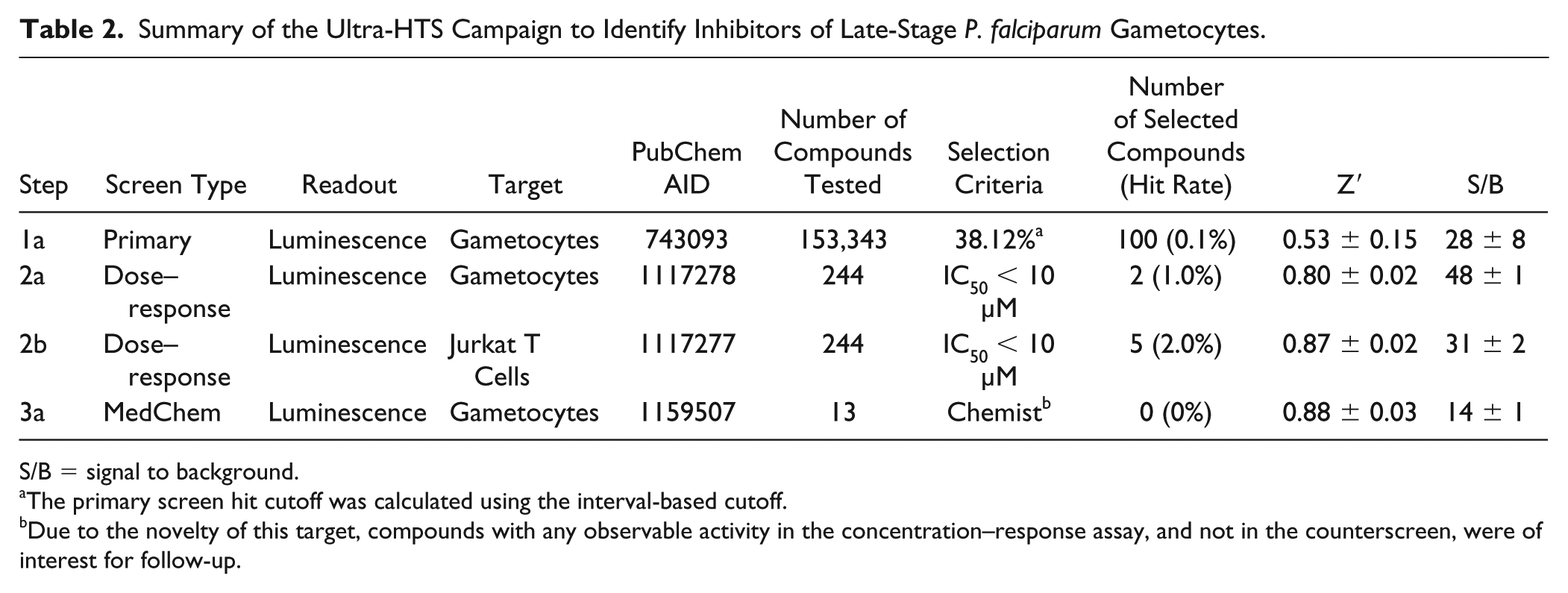
S/B = signal to background.
The primary screen hit cutoff was calculated using the interval-based cutoff.
Due to the novelty of this target, compounds with any observable activity in the concentration–response assay, and not in the counterscreen, were of interest for follow-up.
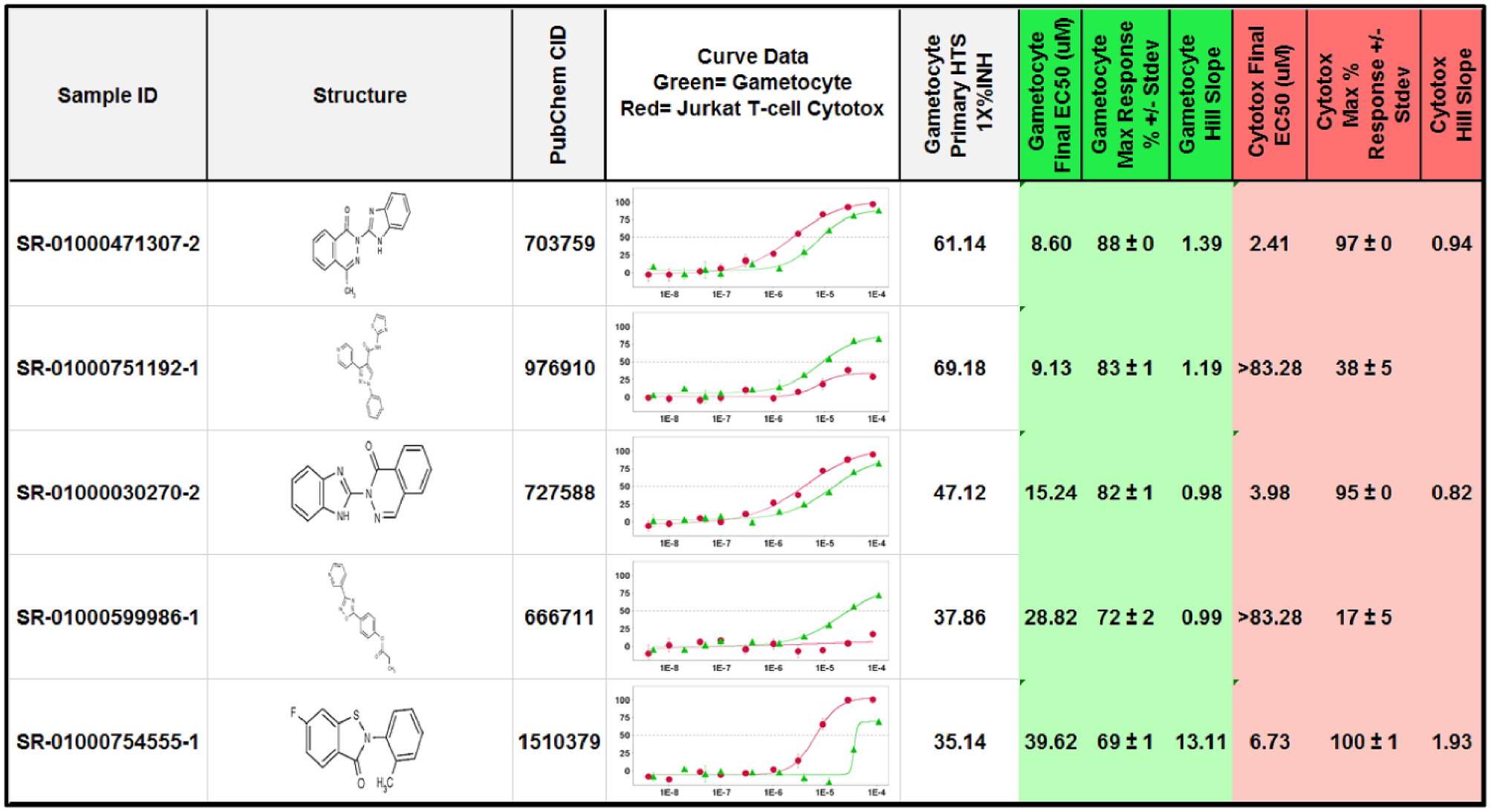
SAR of the top active molecules identified as hits in this HTS campaign. Compounds are ranked with the most potent first. Data have been normalized to “no gametocytes” as 100% inhibition versus “DMSO and gametocytes” only. Data are plotted as log M on the x axis versus percent inhibition on the y axis.
After validation of the hit compounds from dry powders that were originally identified from the HTS conditions and prior to moving into serious SAR optimization where analogs are typically obtained via laboratory synthesis, a routine procedure of analog-by-catalog (ABC) is usually performed to streamline the process. In this step, structurally close analogs are purchased from different commercially available sources that provide a basic idea of how to map the structural changes and their correlates with inhibitory activities. Often, this process provides useful insights regarding toward which directions the structural changes should be performed to improve the potency before the onset of SAR optimization using synthetic efforts. In this project, we carried out a quick ABC process by purchasing analogs that are close structural analogs of the hit compound SR-01000751192 with single-, double-, and triple-point changes (as summarized in the Suppl. Materials). Note that SR-01000751192 provided batch-to-batch reproducibility as identified from samples from the solid physical state. A raw four-parameter curve fit for this compound yields nearly identical IC50 values (7.1 µM batch2 vs 8 µM original hit). A small set of analogs (13) were obtained as fresh samples from the solid physical state. The late-stage SAR assay performed well with Z′ values averaging better than 0.6, and the methylene blue control worked as expected. The counterscreen assay also behaved as expected, with consistent Z′ scores, and response to the control doxorubicin. Unfortunately, improved antigametocydial activity was not observed for any of the 13 compounds purchased for initial SAR, although the compound sample from the solid physical state for the original compound hit did recapitulate the activity observed previously (see Suppl. Table S1 ). We therefore conclude that changing at position 3 of the pyrazole with phenyl, substituted phenyl, and cyclopropyl groups led to diminished activities. We also observed that changing the thiazolyl structure by adding substituents or an additional heteroatom was not tolerated.
Discussion
This work represents the first report of a large-scale HTS campaign completed in 1536-well format against late-stage P. falciparum gametocytes. Most HTS efforts targeting P. falciparum are directed at the liver and blood stages of the parasite life cycle ( Fig. 1 ). Some involve target-based approaches, such as isolation of purified proteins and screening for protease inhibitors that may block the active site for its respective substrate.27,28 Others that have the potential to identify transmission blocking molecules are low throughput and/or nonhomogenous in assay format, making them ill-suited for large-scale HTS. 21 As a critical biological target of unmet therapeutic need, we set the bar high in terms of accepting any compound hit of appreciable activity to proceed for further testing. This meant incorporating a conservative primary HTS cutoff followed by a rather relaxed 10 µM IC50 cutoff in the late-stage analysis. While our efforts were in progress, we overcame a few key hurdles. During the HTS of the gametocyte project there was an unavoidable but significant batch-to-batch difference in the gametocyte numbers and viability, which was overcome by pooling the batches at the point of HTS testing. This afforded us with acceptable Z′ scores. Methylene blue, while not a good antimalarial drug due to its side effects in humans, was used as a pintool-transferred control on each plate and was also tested for antigametocidal pharmacologic response as a CRC control during each experiment and during each run. It is notable that the Z and Z′ values are nearly overlapping, which simply indicates that the overall activity of the test compound was very low. The initial hits, while low in efficacy, do consistently reproduce activity in both labs (Scripps and QIMR Berghofer Medical Research Institute [QIMRB]). While it is unusual to obtain such a low hit rate against such a large number of diverse compounds tested, namely, the Molecular Library Probe Production Centers Network (MLPCN) library, which has yielded multiple clinical drugs, 29 this observation highlights the complexity and natural resistance, or refractiveness, of the gametocytes to the cytotoxic effects of small molecules, similar to what has been observed by others to date. Incorporating a cytotoxicity counterscreen was effective in focusing our attention on SR-01000751192 and SR-01000599986, but with the limited hit set may not be as useful at this stage as originally planned. Ultimately, we were successful in completing the HTS campaign against this difficult target, and there were no insurmountable challenges encountered.
This work involved a multi-institute and multicontent initiative that was supported by the MLPCN. One of the mains goals of the MLPCN was to identify novel molecular probes, that is, ligands that supersede the activity of all prior art in all assays of relevance to support such an achievement. So our goal was to obtain a series of small molecules that met these criteria. Based on this outcome, a probe was not identified for this project that would have been defined as a compound that elicited an antigametocidal effect <500 nM IC50 versus the primary assays and which produced reasonable potency in the low-throughput mechanism-of-action assays, such as the red blood cell infectivity assay performed at QIMRB.
An alternative approach that has shown promise by others incorporated fragment-based screening against biochemical targets in tandem with phenotypic assays.30,31 Perhaps, armed with a biochemical assay directly targeting gametocytes, this hybrid approach would have helped here, but to our knowledge none exist that are HTS amenable. We conclude that this method of HTS provides a path forward for future HTS, perhaps testing larger, more diverse libraries for antigametocidal inhibitor identification and development.
Supplemental Material
DISC796410_Suppl – Supplemental material for Identification of Antimalarial Inhibitors Using Late-Stage Gametocytes in a Phenotypic Live/Dead Assay
Supplemental material, DISC796410_Suppl for Identification of Antimalarial Inhibitors Using Late-Stage Gametocytes in a Phenotypic Live/Dead Assay by Timothy P. Spicer, Donald L. Gardiner, Frank J. Schoenen, Sudeshna Roy, Patrick R. Griffin, Peter Chase, Louis Scampavia, Peter Hodder and Katharine R. Trenholme in SLAS Discovery
Footnotes
Acknowledgements
We thank Pierre Baillargeon and Lina DeLuca (Scripps Florida) for compound management.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by Molecular Library Probe Production Centers Network (MLPCN) grant proposal number 1R21NS075609-01 and MLPCN funding for the University of Kansas Specialized Chemistry Center (U54HG005031).
Supplemental material is available online with this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.

