Abstract
Acetaldehyde dehydrogenase (AdhE) is a bifunctional acetaldehyde–coenzyme A (CoA) dehydrogenase and alcohol dehydrogenase involved in anaerobic metabolism in gram-negative bacteria. This enzyme was recently found to be a key regulator of the type three secretion (T3S) system in Escherichia coli. AdhE inhibitors can be used as tools to study bacterial virulence and a starting point for discovery of novel antibacterial agents. We developed a robust enzymatic assay, based on the acetaldehyde-CoA dehydrogenase activity of AdhE using both absorption and fluorescence detection models (Z′ > 0.7). This assay was used to screen ~11,000 small molecules in 384-well format that resulted in three hits that were confirmed by resynthesis and validation. All three compounds are noncompetitive with respect to acetaldehyde and display a clear dose–response effect with hill slopes of 1–2. These new inhibitors will be used as chemical tools to study the interplay between metabolism and virulence and the role of AdhE in T3S regulation in gram-negative bacteria, and as starting points for the development of novel antibacterial agents.
Introduction
For decades, antibiotics have been extensively used to treat bacterial infections in humans and animals, leading to the development of resistance toward many or all antibiotics available today. This is a worldwide problem, and new therapies are urgently needed. One approach to address this challenge is to find compounds acting on bacterial virulence, the ability to cause disease, in contrast to conventional antibiotics that target bacterial growth without discrimination between pathogenic bacteria and the benign microflora. 1 The type three secretion (T3S) system is a conserved virulence system found in several gram-negative bacteria, including human, animal, and plant pathogens. 2 The gram-negative bacterium enterohemorrhagic Escherichia coli (EHEC) serotype O157:H7 causes severe foodborne disease, including symptoms like diarrhea, hemorrhagic colitis, and hemolytic uremic syndrome (HUS), with adolescents and the elderly being most at risk. 3 The Shiga toxin (Stx) production, responsible for HUS, can result in life-threatening renal failure. 3 Transmission to humans occurs via contaminated raw or undercooked meat, raw milk, and fecal contaminated raw vegetables, since cattle can asymptomatically be colonized by EHEC. Today there is no treatment available for EHEC infections, and treatments with conventional antibiotics exacerbate Stx-mediated cytotoxicity. 4 The salicylidene acylhydrazides are virulence inhibitors shown to be active against the T3S system in a number of pathogenic gram-negative bacteria, including Chlamydia sp., 5 Salmonella enterica serovar Typhimurium,6,7 Yersinia pseudotuberculosis,8,9 Shigella flexneri, 10 E. coli O157:H7, 11 Pseudomonas aeruginosa, 12 and Erwinia amylovora. 13 In a target identification study, several possible targets for this class of compounds were found in EHEC. 14 The salicylidene acylhydrazides are promiscuous, and the inhibition of T3S is the result of interactions with several proteins ultimately leading to the attenuation of the T3S system. However, in EHEC one of these target proteins, the metabolic enzyme acetaldehyde dehydrogenase (AdhE), was found to be a key regulator of T3S. 15 An EHEC adhE deletion mutant showed strong downregulation of the T3S system and overexpression of nonfunctional flagella without any effect on bacterial growth. Thus, the inability of EHEC to cause infection both in vitro and in vivo and these findings validate AdhE as a potential target for the development of novel antibacterial agents. AdhE is a bifunctional acetaldehyde–coenzyme A (CoA) dehydrogenase and alcohol dehydrogenase important for anaerobic metabolism. Previous studies report that AdhE acts as a virulence factor under alcoholic conditions in Streptococcus pneumoniae 16 and that AdhE is upregulated during infection in the gram-negative pathogen Salmonella typhimurium. 17 An adhE mutant alters the SPI-1 gene expression, supporting crosstalk between metabolism and virulence. 18 In humans, the aldehyde dehydrogenase (ALDH) superfamily includes 19 NAD(P+)-dependent enzymes that irreversibly catalyze the oxidation of endogenous and exogenous aldehydes. Out of these 19 isozymes, there are only drugs developed for 3 of them: ALDH2, involved in alcohol metabolism, and ALDH1A1 and ALDH3A1, involved in cancer. 19 Disulfiram, also known as Antabuse, is a prodrug resulting in metabolic products that irreversibly block ALDH. 20 Disulfiram is the first-line drug used to treat alcoholism and has been used since 1948. This makes the bacterial AdhE a promising target for the development of new antimicrobial drugs since adverse effects resulting from interaction with human ALDHs are well studied. To identify new virulence inhibitors and to study the interplay between metabolism and virulence based on AdhE in gram-negative bacteria, we have developed an enzymatic assay and screened ~11,000 small molecules in 384-well format, resulting in three novel inhibitors of EHEC AdhE.
Materials and Methods
The E. coli adhE gene was amplified from EHEC O157:H7, CCUG 29188, in conjunction with the primer pair GAGCATCATGACTGTTACTAATGTCGCTGAA and CGCGGATCCTTAAGCGGATTTTTTCG. The amplified product was cloned into the pETHis_1a expression vector, thereby fusing a hexahistidine tag and TEV cleavage site to the N-terminus of AdhE, called pCZ2.
Protein Expression and Purification
Competent E. coli BL21(DE3)pLysS (Novagen, Burlington, MA) cells were transformed with the pCZ2 plasmid and grown overnight on Lysogeny broth (LB) agar plates supplemented with kanamycin (30 µg mL–1). A preculture of 50 mL of LB medium supplemented with kanamycin (50 µg mL–1) and chloramphenicol (34 µg mL–1) was inoculated. The preculture was grown at 37 °C overnight and used for inoculation of LB medium (5 L) supplemented with kanamycin (50 µg mL–1) and chloramphenicol (34 µg mL–1). The bacterial culture was grown on an orbital shaker at 37 °C. When the absorption at 600 nm reached a value of 0.8, isopropyl β-
Enzymatic Assay and Screening
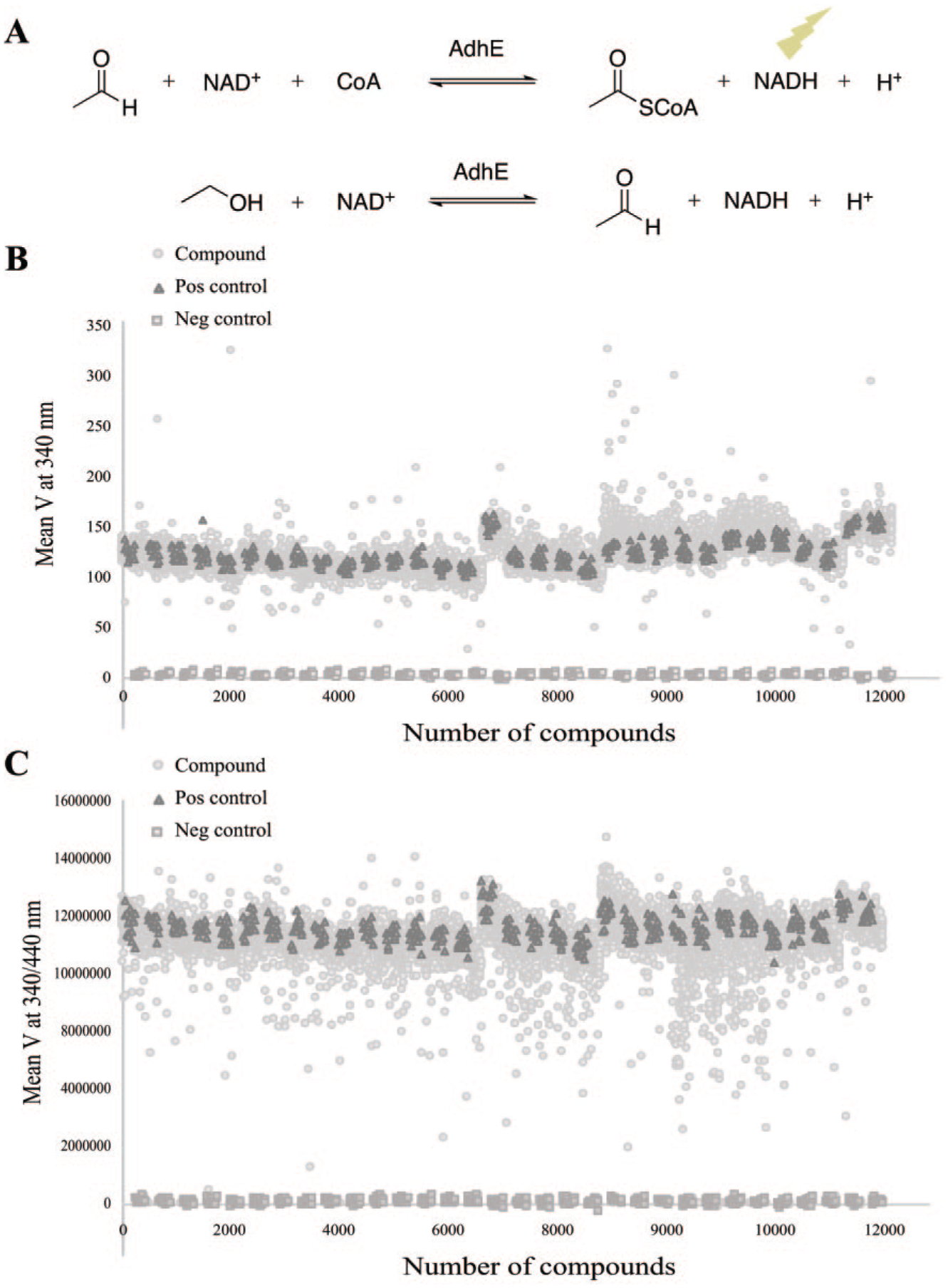
The catalytic activity of AdhE (
Fig. 1A
) was monitored by the reduction of β-nicotinamide adenine dinucleotide (NAD) as previously described.
21
The reduction was detected by absorbance at 340 nm and fluorescence emission at 440 nm when excited at 340 nm. Determination of Km was performed for acetaldehyde, CoA, and NAD, and finally DMSO tolerance was determined for the reaction. Substrate solution (10 µL N-cyclohexyl-2-aminoethanesulfonic acid [CHES], pH 9.3, NAD, CoA, acetaldehyde, and Tween 20) was added to enzyme solution (10 µL CHES, pH 9.3, AdhE,
(
Cell Toxicity
CMT-93 cells (mouse epithelial rectum cells, ATCC CCL-223) and HTC-8 cells (HRT-18; human epithelial colon cells, ATCC CCL-244) were seeded into 96-well plates (Nunc, flat bottom, transparent), 100 µL 1 × 85 cells/mL in colorless Dulbecco’s modified Eagle’s medium supplemented with 10% fetal calf serum (FCS) and glutaMAX. After 24 h incubation at 37 °C in 5% CO2, compounds
Compounds
A screening set from Chemical Biology Consortium Sweden (CBCS) was used in this campaign. The whole compound library was donated from the former Biovitrum AB, and around 6700 are the primary screening set consisting of Biovitrum AB’s in-house compounds. Around 1400 compounds were focused against kinases, 1500 compounds were focused against G-protein-coupled receptors, 200 nucleosides were from Berry & Associates, and around 1200 approved drugs were from Prestwick. The four disulfiram metabolites, diethyldithiocarbamate (DDTC), S-methyl-N,N-diethyldithiocarbamate (Me-DDTC), S-methyl-N,N-diethylthiocarbamate-sulfoxide (Me-DDTC-SO), and S-methyl-N,N-diethylthiocarbam-sulfone (Me-DDTC-SO2), are commercially available (Toronto Research Chemicals, Toronto, ON, Canada). Detailed protocols for synthesis of compounds
Results and Discussions
AdhE is one of the target proteins in E. coli of the promiscuous class of salicylidene acylhydrazides that block T3S in several gram-negative pathogens. 23 Unfortunately, these compounds are only weak inhibitors of AdhE (IC50 ~125 µM, data not shown), and the observed inhibition of T3S likely involves additional target proteins. The salicylidene acylhydrazides also have poor pharmacokinetic properties, which makes them unfavorable to work on further.24,25 To efficiently target AdhE in a selective manner, new inhibitors are needed. In this work, we developed a high-throughput screening (HTS) protocol and performed screening to identify putative inhibitors of the catalytic activity of AdhE from E. coli. In the screening assay, enzymatic activity was monitored by measuring the reduction of NAD to NADH using both fluorescence and absorbance readouts ( Fig. 1A ). Km values were determined for the substrates, and for acetaldehyde it was found to be 6.9 mM. The acetaldehyde assay concentration was set to 3 mM in order to allow identification of competitive inhibitors. 26 The assay tolerated DMSO concentrations up to 0.5% (data not shown), and the use of prespotted compound plates during screening allowed the final DMSO concentration to be kept as low as 0.1%. Tween 20 was included in the assay buffer to prevent compound aggregation. The stability of the enzymatic activity was found to decrease over time, and therefore fresh enzyme and substrate solution were prepared for every fifth plate during the screening. The final assay protocol includes a few pipetting steps, favoring small variations, and includes two detection methods where NADH formation is measured with both absorbance and fluorescence readouts, to reduce the number of false-positive hits ( Fig. 1B , C ). After protocol optimization, it was possible to run the assay in a final volume of 20 µL in 384-well plates with Z′ values above 0.7.
Disulfiram that targets human ALDH is a prodrug that requires metabolic activation to generate several metabolites, including Me-DTCC, that irreversibly block the catalytic Cys302 residue by carbamoylation. 20 The four commercially available disulfiram metabolites, DDTC, Me-DDTC, Me-DDTC-SO, and Me-DDTC-SO2, were tested in our assay, but we could not detect any reduction of enzymatic activity.
A library consisting of 10,981 molecules was screened according to the workflow in
Figure 2
. In total, thirty-two 384-well plates were processed with an average Z′ factor of 0.83 ± 0.03 for the absorbance readout and 0.89 ± 0.02 for the fluorescence readout (
Suppl. Fig. S1
). The enzymatic activity was followed kinetically, and the reaction’s mean velocity was calculated to further eliminate false positives in the screen. The screening identified 289 and 476 compounds as hits in the absorbance and fluorescence measurements, respectively. In total, 62 compounds inhibited both the absorbance and fluorescence readouts with three standard deviations from control corresponding typically to >15% inhibition (
Fig. 1B
,
C
). A detailed table of the compound origin can be found in supporting information (
Suppl. Table S1
). These compounds were selected for confirmation in the HTS assay using two concentrations in duplicates that resulted in nine compounds that were subjected to full dose–response analysis. For six of these nine compounds, the activity and identity were not confirmed or the compounds were considered pan-assay interference compounds (PAINS) (
Fig. 2
).
27
Finally, the three validated hit compounds,
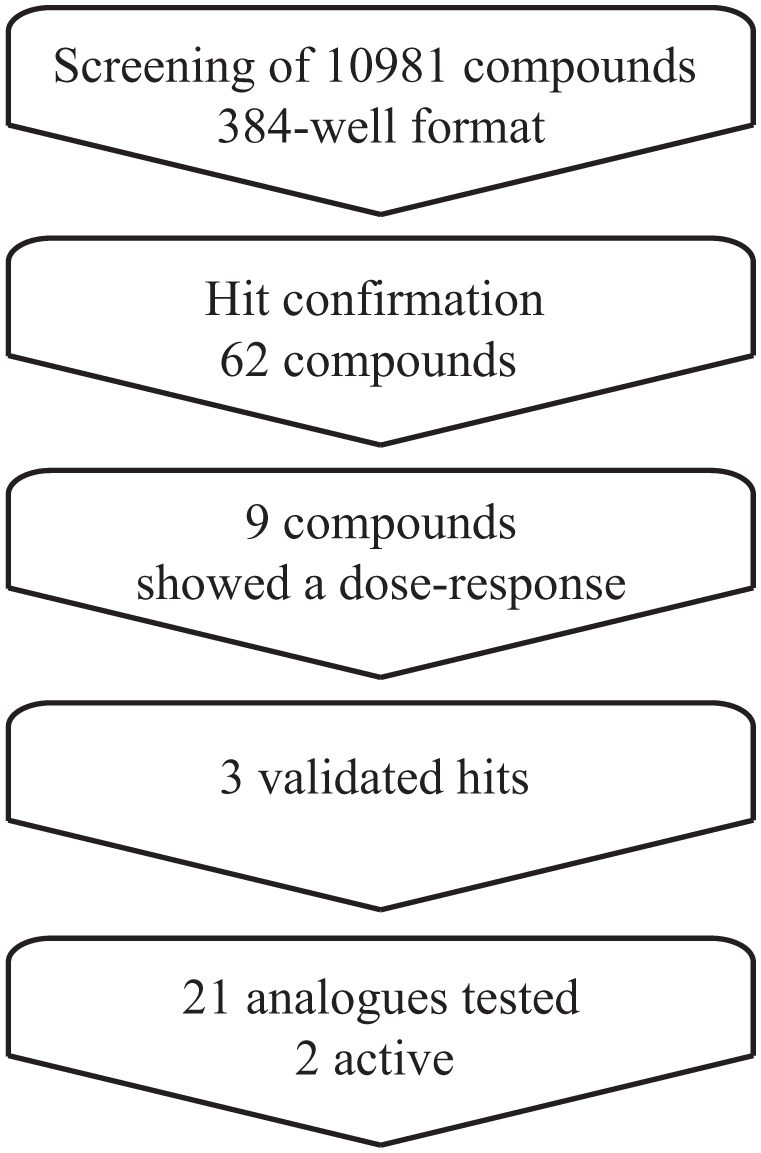
A summary of screening results and workflow.
Summary of Dose–Response Tests for the Three Confirmed Hits and Physicochemical Properties Calculated with MOE.
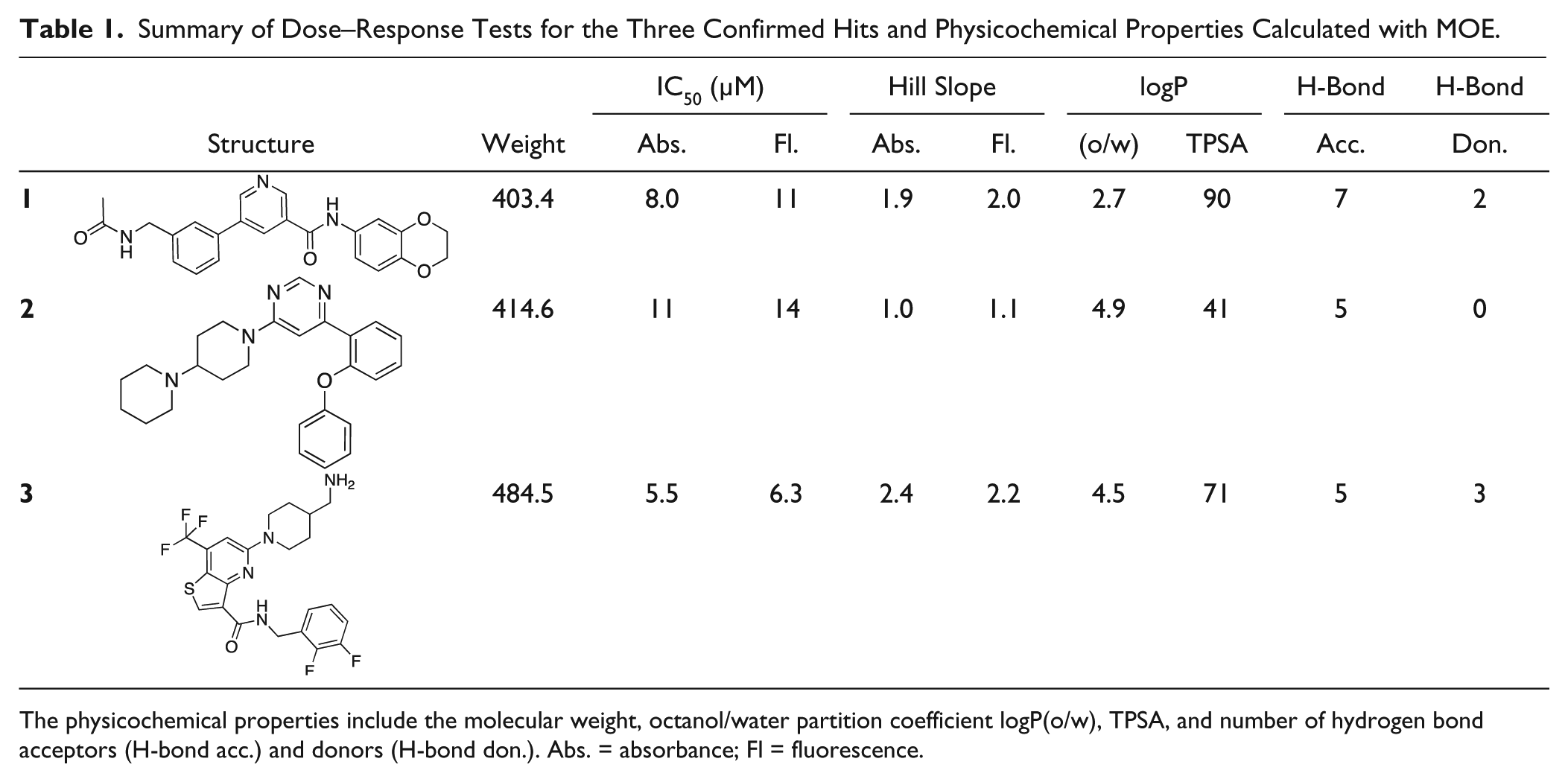
The physicochemical properties include the molecular weight, octanol/water partition coefficient logP(o/w), TPSA, and number of hydrogen bond acceptors (H-bond acc.) and donors (H-bond don.). Abs. = absorbance; Fl = fluorescence.
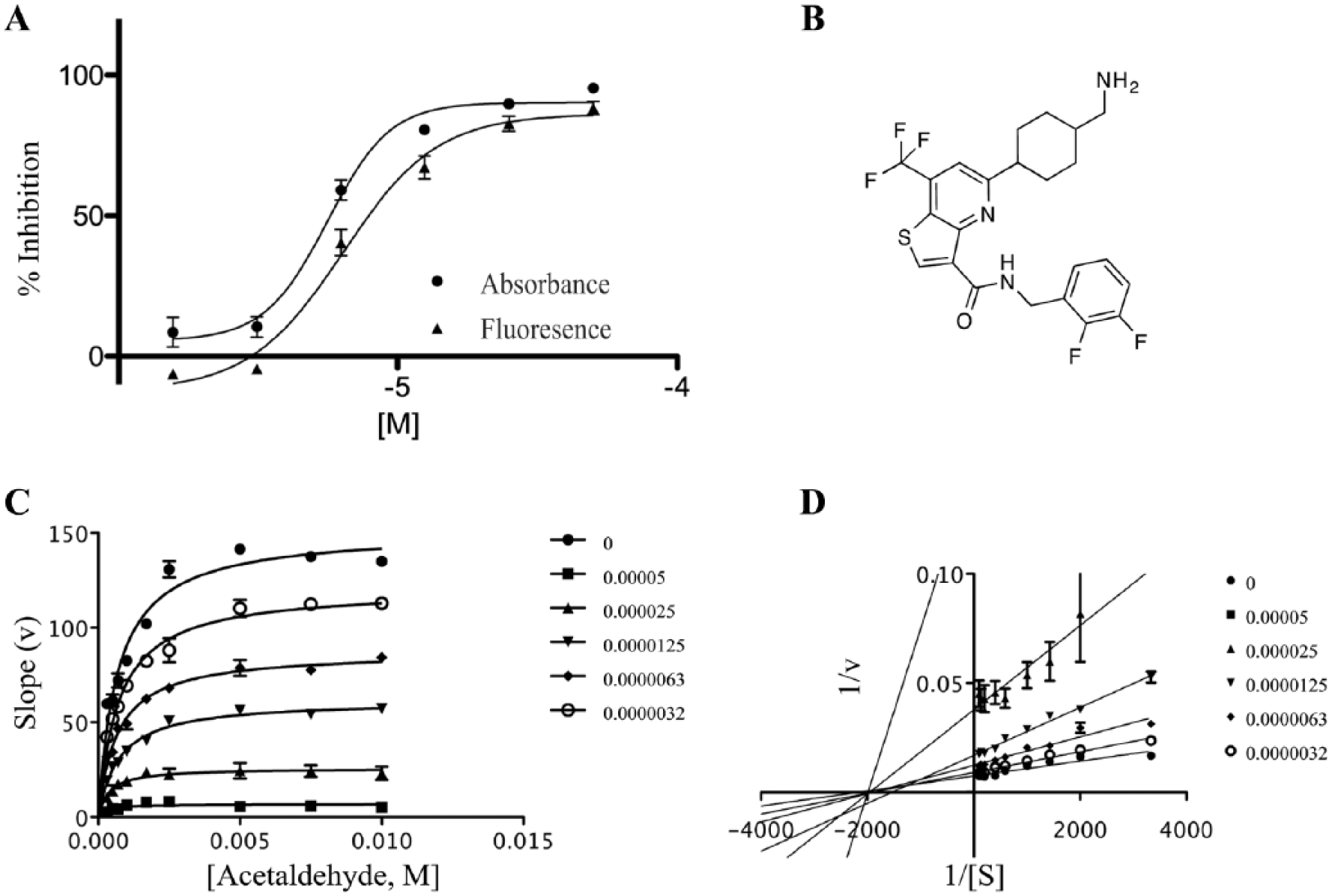
Data from compound
The toxicity against mouse epithelial rectum cells (CMT-93) and human epithelial colon cells (HTC-8) after 24 and 48 h incubation with compound (100, 50, 25, and 12.5 µM) was scored with the UptiBlue test. Compounds
Compounds
Compounds
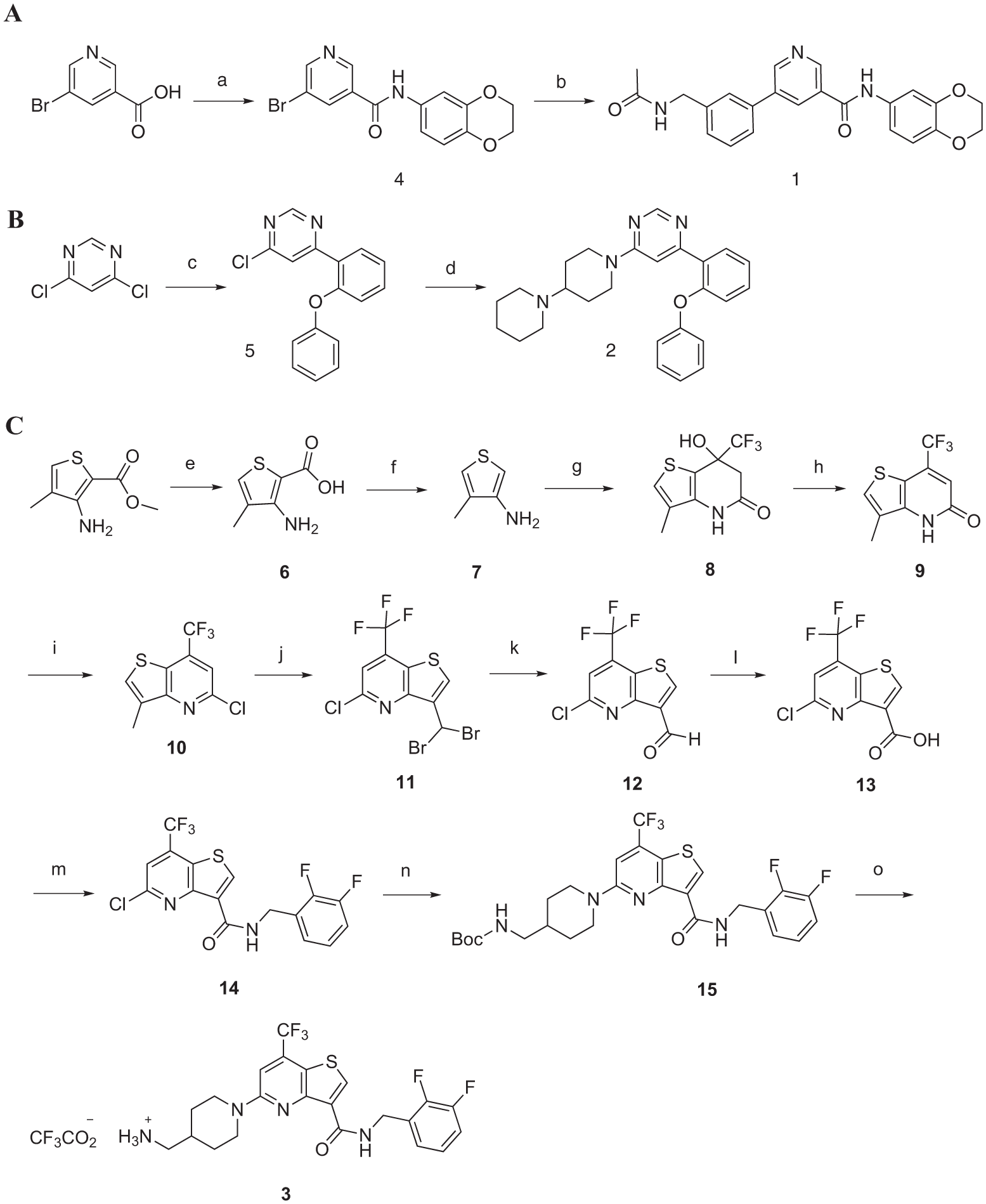
Synthetic schemes for the three hit compounds. Reagents and conditions: (
We also tested eight and six analogs to compounds
In summary, we screened 10,981 compounds in 384-well format resulting in 62 primary hits, of which 9 showed dose–response patterns with an IC50 of around 10 µM. From this set, resynthesis and validation identified three drug-like molecules,
Supplemental Material
DS_DISC768062 – Supplemental material for Screening for Inhibitors of Acetaldehyde Dehydrogenase (AdhE) from Enterohemorrhagic Escherichia coli (EHEC)
Supplemental material, DS_DISC768062 for Screening for Inhibitors of Acetaldehyde Dehydrogenase (AdhE) from Enterohemorrhagic Escherichia coli (EHEC) by Caroline E. Zetterström, Pia Uusitalo, Weixing Qian, Shannon Hinch, Rémi Caraballo, Christin Grundström, and Mikael Elofsson in SLAS Discovery
Footnotes
Acknowledgements
The authors would like to thank Maximilian Schneider for synthetic discussions and Charlotta Sundin for scientific discussions and technical assistance with cell toxicity measurements. They also would like to thank Chemical Biology Consortium Sweden (CBCS) for assistance during the screening and supply of screening compounds.
Supplemental material is available online with this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The authors thank Molecular Infection Medicine Sweden (MIMS), Umeå Centre for Microbial Research (UCMR), and the Swedish Research Council for financial support.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
