Abstract
In the recent past, there has been a growing interest in developing nonalcoholic fatty liver disease (NAFLD) and nonalcoholic steatohepatitis (NASH) therapeutics. As a result, a need for in vitro cell models of human hepatic steatosis and high-throughput assays to measure intracellular lipid levels has arisen. To address this growing need, we optimized the conditions based on the current literature to fatten HepG2 hepatocytes by adding a mixture of saturated and unsaturated fatty acids (oleate/palmitate, 2:1 molar ratio) without inducing any overt cytotoxicity. Our results indicate that hepatocytes fatten in a concentration- (0.75–1.5 mM of fatty acids) and time-dependent manner, with a substantial increase in intracellular lipid levels seen within 6 h. Additionally, a method to quantify lipid levels in cells using a fluorescent reagent that is more sensitive than that in conventional assays and adaptable for high-throughput screening is presented. Lastly, the utility of the in vitro cell model and an assay based on AdipoRed to measure hypolipidemic effects of therapeutic drugs is demonstrated using fenofibrate, a molecule that was previously shown to lower lipid levels in the liver.
Introduction
Nonalcoholic fatty liver disease (NAFLD) refers to a clinical condition in which the liver is overloaded with fat and is not a result of excess alcohol consumption or other liver diseases. 1 The underlying mechanisms leading to the pathogenesis of NAFLD are unclear. However, elevated levels of plasma free fatty acids (FFAs) have been linked to the pathogenesis of insulin resistance (IR), which is considered a major determinant of NAFLD.2,3 In agreement with this hypothesis, NAFLD is widely prevalent in obese and type 2 diabetes patients who are generally IR and have high circulating levels of FFAs due to unabated antilipolytic insulin action.4,5 The high levels of FFAs lead to impaired postreceptor insulin signaling, 3 thereby contributing to existing IR, which results in a further increase in FFAs and worsening of fatty liver condition. Several lines of evidence also indicate the importance of dietary fatty acid composition (e.g., saturated vs. unsaturated) as an impetus for the incidence of NAFLD and nonalcoholic steatohepatitis (NASH).2,6 –8 HepG2 hepatocytes, when cultured in the presence of oleate and palmitate (2:1 molar ratio), the two most abundant dietary fatty acids, led to significant fat accumulation, IR, and low-grade inflammation, 9 consistent with the observations seen in human NAFLD. 1 Similar to these observations, primary human hepatocytes also showed fattening following treatment with a mixture of oleate and palmitate (2:1 molar ratio).10,11 The extent of fattening was in fact higher in primary hepatocytes than in HepG2 cells. 10 A high proportion of palmitic acid (oleate/palmitate, 0:3 molar ratio), on the other hand, represented a cellular model of steatosis in which saturated FFAs promoted fibrogenesis, and hence are possibly more suited for studying NASH.7,12,13
Given that NAFLD/NASH has emerged as a serious health issue worldwide, several drugs are in development to treat the condition.14 –16 Along with this surge is the growing need to develop high-throughput screening assays. Typically, lipid levels within cells are quantified using oil-red-o staining or biochemical estimation of free glycerol levels released following lipase digestion of lipids. 12 These methods, however, are not suitable for medium- or high-throughput assaying platforms. To overcome this limitation, we developed an assay based on fluorescence readout using the AdipoRed reagent, which requires minimal handling of cells and does not involve the laborious steps, such as cell lysis, lipid extraction, or image analysis. Fluorescence-based assays are widely used in high-throughput screening due to their high sensitivity, diverse selection of fluorophores, ease of operation, and various readout modes. 17 Typically, the use of fluorophores increases the assay signal one to two orders of magnitude without increasing background. This gives a meaningful and transformative improvement in the signal-to-noise ratio, providing lower limits of detection, improved reproducibility, and reduced assay “grey zones.” Additionally, a few steps are required and timing is not critical, so the assays can be adapted for automated handling in high-throughput applications.
In the current report, development of an in vitro model system based on fatting of HepG2 hepatocytes using FFAs (oleate/palmitate, 2:1 ratio) is described. Additionally, a novel assay based on AdipoRed fluorophore to measure intracellular lipid levels is presented. Finally, the utility of this hepatic steatosis model and the AdipoRed-based assay to investigate the hypolipidemic effects of test agents is demonstrated.
Materials and Methods
Cell Culture
HepG2 cells were purchased from ATCC (Manassas, VA, cat. HB-8065) and cultured according to the supplier’s instructions. In brief, the cells were regularly grown in a T-75 flask containing Eagle’s minimum essential medium (EMEM, ATCC, cat. 30-2003) in the presence of 10% fetal bovine serum (ATCC, cat. 30-2020) and 1× pen-strep (cat. 15140-122, Life Technologies, Waltham, MA) at 37 °C, 95% humidity, and 5% CO2. The cells, upon reaching 80% confluency, were trypsinized and reseeded onto a fresh flask or 96-well plates. Once the cells were plated and ready for use in the experiments, the plates were sealed with membranes that permit gas exchange. In all instances, cells grown in the edges of plates were not used for conducting experiments.
Free Fatty Acid Mixture Preparation
A 100 mM stock solution of oleic acid (cat. O7501, Sigma, St. Louis, MO) and palmitic acid (cat. P9767, Sigma) was prepared in 99% methanol (cat. 494437, Sigma). In order to bring palmitic acid into solution, the stock solution was placed in a water bath warmed to 50 °C for 10 min. A mixture of these FFAs was then prepared by combining oleic acid and palmitic acid at a 2:1 molar ratio in fatty acid–free 10% bovine serum albumin (BSA, cat. A1595, Sigma) (prewarmed to 45–50 °C). The BSA complexed fatty acids were diluted in EMEM containing 10% charcoal-stripped fetal bovine serum (cat. 12676-011, Life Technologies) and 1× pen-strep was added to prepare high (HC, 2 mM), medium (MC, 1.5 mM), low (LC, 1 mM), and very low (VLC, 0.75 mM) concentrations of fatty acids as needed. Since the fatty acids are dissolved in methanol, the final concentrations of methanol are 0.75%, 1%, 1.5%, and 2% in VLC, LC, MC, and HC of FFA mixture preparations.
Triglyceride Assay to Estimate Lipid Levels
A method was developed based on the 96-well plate format to quantify intracellular lipid levels. The cells were washed twice with phosphate-buffered saline (PBS) (without Ca2+ and Mg2+) and then incubated at room temperature for 20–30 min in the presence of 50 μL of 0.2% NP-40 (cat. PI85124, Thermo Fisher Scientific, Waltham, MA) or RIPA buffer (cat. 89900, Thermo Fisher Scientific) to break open the cells. Next, 100 μL of Infinity Triglyceride reagent (cat. TR-22421, Thermo Fisher Scientific) was added and incubated at 37 °C for 15 min. The absorbance was measured at 570 nm using a multiwell plate reader (PerkinElmer, Waltham, MA). In order to adjust for well-to-well variability, the absorbance values were normalized using protein levels estimated using bicinchoninic acid assay (BCA) reagent (cat. PI-23227, Thermo Fisher Scientific). It is important to note that the estimated protein levels using this reagent can sometimes be skewed in the presence of phospholipids. 18 This is not an issue as long as it is used for internal reference only.
AdipoRed Staining to Quantify Lipid Levels
The fattened cells in the 96-well black plate were washed once with PBS. Staining solution (100 µL) that was prepared by mixing 2.5 µL of AdipoRed reagent and 10 µL of Hoechst 33342 stain (cat. R37605, Thermo Fisher Scientific) in 1 mL of PBS was added to each well. After incubating the plate at 37 °C for 10 min, fluorescence was read using a FlexStation multiwell plate reader (Molecular Devices, Sunnyvale, CA) set at excitation/emission wavelengths of 485/572 and 360/460 nm.
Triglyceride Standard Curve Using AdipoRed
The triglyceride standard (cat. 17811-1AMP, Sigma) was initially dissolved in DMSO at a concentration of 10,000 mg/dL by warming the contents to 55 °C for 10 min. This stock was further serially diluted in 0.2% NP-40 PBS to prepare concentrations ranging from 125 to 0 mg/dL of triglyceride standard. The lipid levels were then quantified using AdipoRed and Infinity Triglyceride regent as described earlier.
Testing Defatting Effects of Fenofibrate
A 100 mM stock solution of fenofibrate (CAS 49562-28-9, Tocris, Minneapolis, MN) was prepared in DMSO. To test defatting effects, the small molecule was diluted to appropriate concentrations in culture media and added to the cells.
Results
Treatment with Free Fatty Acids Increased Fatting of HepG2 Cells in a Concentration-Dependent Manner
Toward establishing an in vitro model of hepatic steatosis, the effect of varying concentrations of a mixture of FFAs (oleate/palmitate, 2:1 molar ratio) on the extent of triglyceride accumulation in HepG2 cells was studied. The HepG2 cells were initially grown in a T-75 flask and later plated at a density of 40,000 cells per well in a 96-well plate. The total concentration of FFA mixture added to the cells ranged from 0.75 to 2 mM. After 24 h, the triglyceride levels in cells were quantified using an absorbance-based triglyceride assay as described in Materials and Methods. The well-to-well variability was normalized using protein levels in each of the wells (BCA assay, Thermo Fisher Scientific). The extent of fatting is shown in Figure 1 . A statistically significant increase in lipid levels is seen in the presence of added FFA mixture. Moreover, the extent of fatting increased with an increase in FFA concentrations from 0.75 to 2 mM. A basal level of lipid accumulation is also observed in the absence of any added FFAs to the culture media. This is due to the inherent nature of HepG2 cells to convert and store some of the carbohydrates as triglycerides.
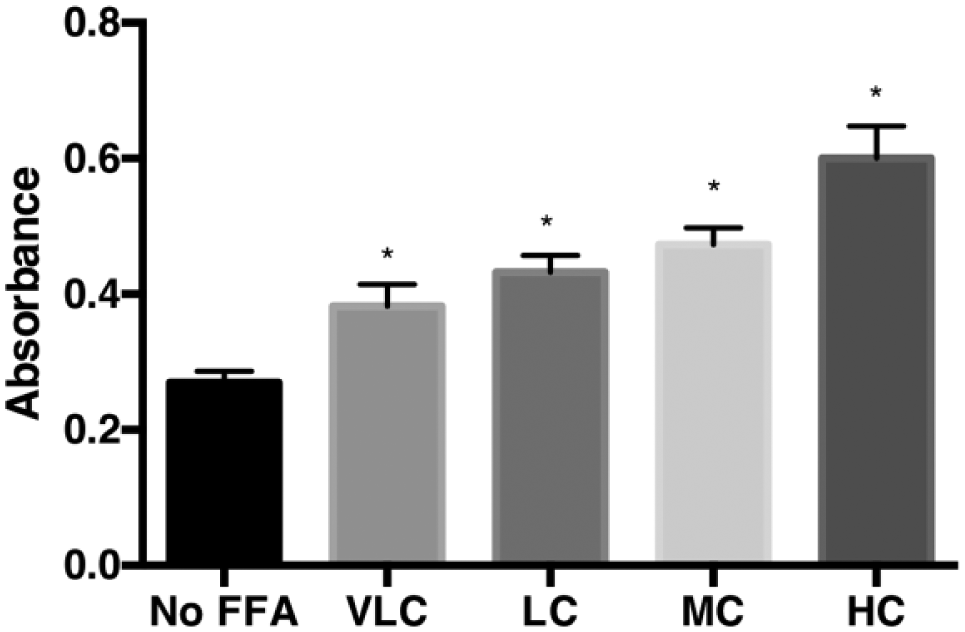
Fatting of HepG2 cells with FFA treatment. The extent of hepatic steatosis in HepG2 24 h after treatment with 0.75 mM (VLC), 1 mM (LC), 1.5 mM (MC), or 2 mM (HC) FFA mixture (oleate/palmitate, 2:1 molar ratio) is shown. Results are summarized from three experiments conducted in duplicate. The mean and range of absorbance (Abs) values measured using the triglyceride assay and corrected for well-to-well variability with absorbance from the BCA assay (protein levels) are presented. The statistical significance between the means observed in the control (vehicle control) and each of the treatments (varying concentration of FFAs) was determined using one-way ANOVA, followed by a post-Dunnett’s test. *p < 0.05.
Cell Viability Is Reduced with an Increase in Concentration of FFA Treatment
Published reports indicated that exposure of cells to exogenous FFAs can cause cell death.20,21 This was evident in our experiments as well. The phase contrast images of HepG2 cells showed a marked reduction in cell number when treated with an MC or HC of FFAs for a period of 24 h ( Fig. 2A–E ). A similar reduction in cell number, however, following a VLC or LC of FFAs was not apparent. In order to quantify the extent of reduction in cell viability, a luminescence assay was performed following the manufacturer’s instructions. In brief, the assay involves adding a single reagent (CellTiter-Glo Reagent, Promega, Madison, WI) directly to cells cultured in serum-supplemented medium. The reagent causes cell lysis and generation of a luminescent signal proportional to the amount of ATP present. The amount of ATP is directly proportional to the number of cells present in culture. The assay results confirmed that viability is significantly reduced by 31% under MC and 90% under HC of FFA treatment, and that the viability is not affected under VLC and LC FFA treatments when compared with corresponding vehicle controls ( Fig. 2F ). It is worth noting that the contribution of vehicle controls toward the overall reduction in cell viability has an increasing trend with increased levels of methanol (used to bring FFAs into solution) in the cell culture media. However, a one-way analysis of variance (ANOVA) followed by a Dunnett’s test (p < 0.05) did not identify any significant differences between the various vehicle controls.
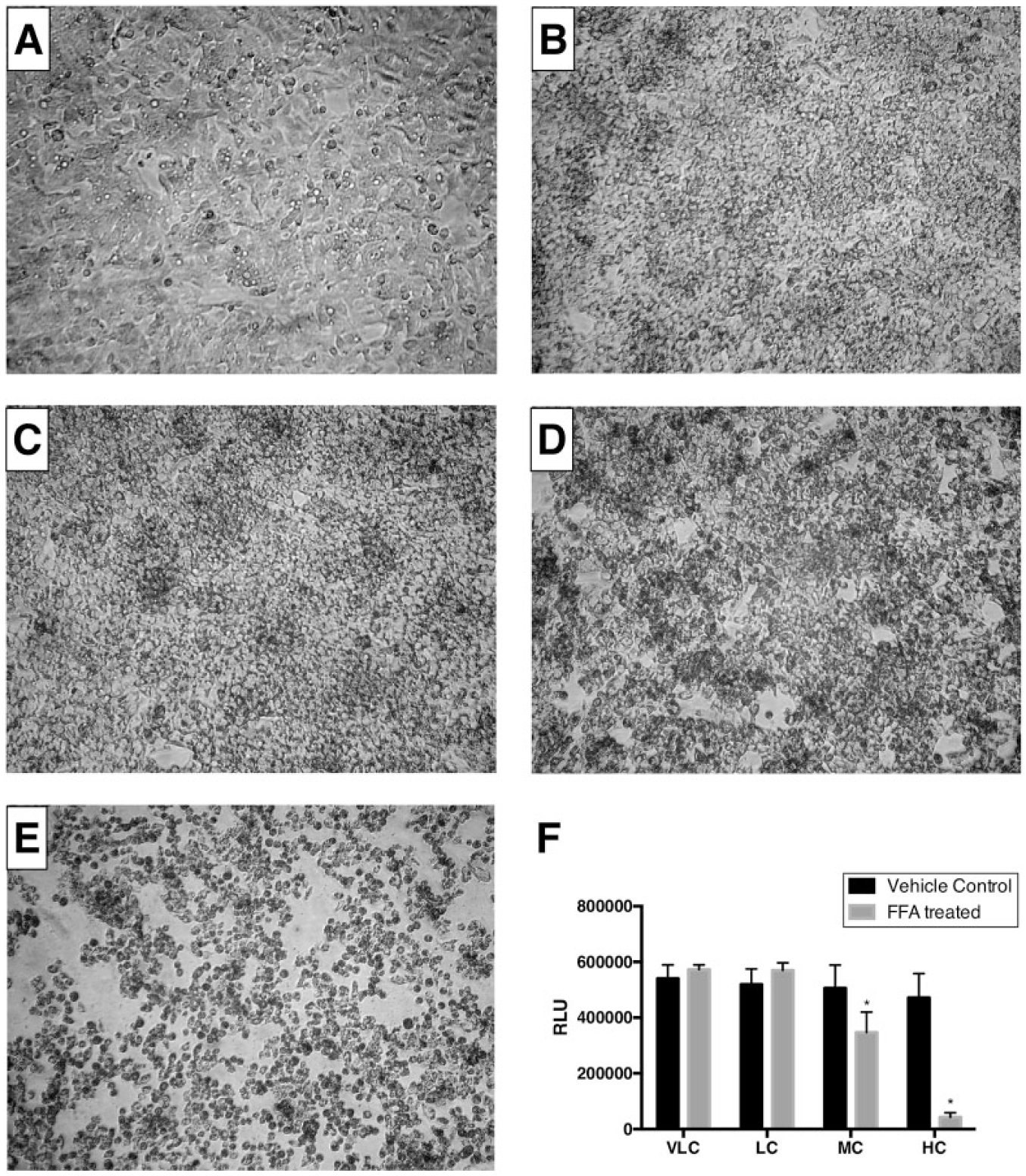
Phase contrast images of HepG2 cells and cell viability counts following treatment with FFAs for 24 h. HepG2 hepatocytes were seeded at a density of 40,000 cells per well in a 96-well plate and incubated at 37 °C overnight as described in Materials and Methods. After a wash with PBS, cells were treated with (
HepG2 Cells Fatten in a Time-Dependent Manner
In order to determine the optimal incubation time required to fatten cells, HepG2 hepatocytes were treated with an LC (1 mM) of FFAs and incubated for 6, 16, or 24 h. The lipid content was determined using the triglyceride assay as described in Materials and Methods. An increase in lipid content was seen as early as 6 h and continued to increase during the 24 h of treatment ( Fig. 3A ). Similarly, lipid accumulation increased when treated with an MC (1.5 mM) of an FFA mixture. At both 6 and 16 h, the lipid levels were higher than those for the corresponding vehicle controls ( Fig. 3B ). Once again, a time-dependent increase in lipid levels was observed. The lipid accumulation was not measured at 24 h because cell viability is dramatically reduced within this period after treatment with FFAs (MC) ( Fig. 3C ). An attempt to identify the optimal duration needed to fatten HepG2 cells with an HC of FFA treatment was not feasible. The cell viability significantly decreased within 6 h of exposure to FFA (HC) mixture (data not shown), suggesting that HepG2 cells do not tolerate an HC of FFAs for even a short duration.
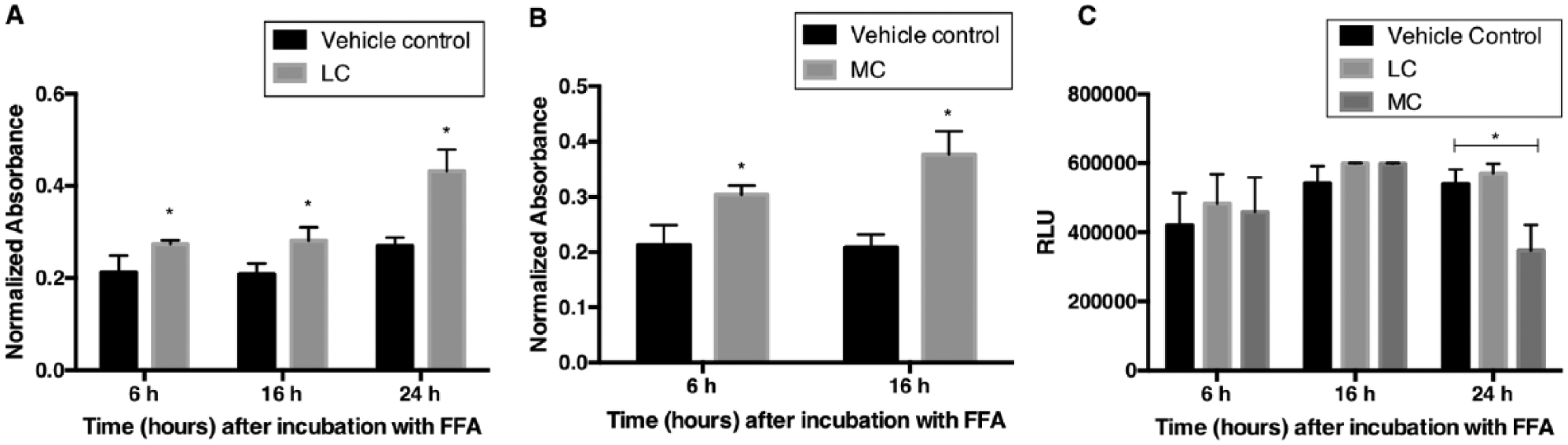
Time course of fatting of HepG2 cells and viability. HepG2 cells were treated with (
AdipoRed Instead of Triglyceride Reagent Is Also Suitable to Measure Intracellular Lipid Levels
In order to establish an assay that is suitable for medium- and high-throughput applications, an assay based on fluorescence using the AdipoRed reagent was evaluated to measure lipid levels. AdipoRed binds to intracellular neutral lipids and fluoresces when excited in the red wavelength range. This process does not require cell lysis or extraction of lipids before quantifying the levels. The procedure merely involves adding AdipoRed reagent to PBS-washed cells and measuring fluorescence emission. By following this procedure (described in Materials and Methods), a standard curve was generated using a serially diluted synthetic triglyceride mixture with concentrations between 3 and 125 mg/dL. The plot shows a linear detection of triglyceride levels within this range ( Fig. 4A ). A comparison with the standard curve generated using the same synthetic triglyceride mixture and quantified using the triglyceride reagent showed a high Pearson coefficient (r = 0.9986) between the absorbance and fluorescence methods of detection (data not shown). Hence, AdipoRed is suitable as an assay reagent to quantify intracellular lipid levels.
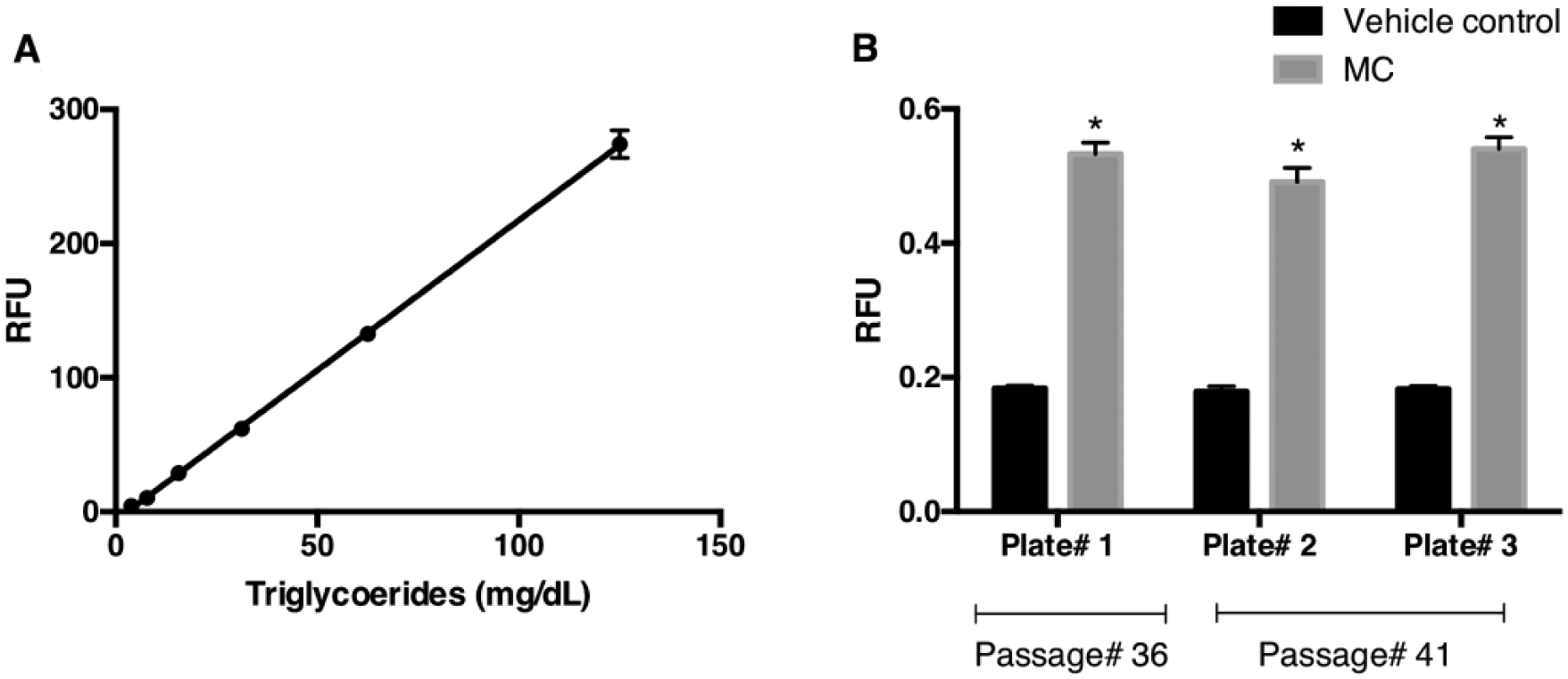
Fluorescence-based method to measure intracellular lipid levels. (
Previous results showed HepG2 cells fatten by 6 h when exposed to an MC of an FFA mixture ( Fig. 3B ). This experiment was repeated to assess if AdipoRed reagent can be used instead of triglyceride reagent to quantify intracellular lipid levels. In brief, HepG2 cells were grown to confluence in a 96-well plate and treated with FFAs (MC) for 6 h as described in Materials and Methods. The cells were then washed with PBS and AdipoRed reagent was added. The fluorescence emission was read using a multiwell plate reader after incubating the cells at room temperature for 15 min. To control for well-to-well variability, cells were stained with Hoechst 33342 dye and fluorescence emission read once again to estimate the amount of nuclear DNA per well. The results once again showed that HepG2 hepatocytes fatten within 6 h of addition of the FFA (MC) mixture ( Fig. 4B ). Interestingly, the percentage change between controls and treated samples was more pronounced in fluorescence readouts (percent change, 164%; mean RFU; vehicle controls vs. FFAs [MC] treated, 0.181 vs. 0.521, respectively) than in absorbance measurements (percent change, 43%; mean relative absorbance; vehicle controls vs. FFAs [MC] treated, 0.213 vs. 0.304, respectively). This suggests that a greater degree of sensitivity can be achieved by using AdipoRed reagent to measure lipid levels. The Z′ factor calculation also revealed that the fluorescence-based assay was of higher quality (>0.5) than the absorbance-based assay (0.15). The SWs, coefficient of variation (CV), and S/B for the fluorescence readouts are all in acceptable ranges ( Table 1 ). An ANOVA showed that a statistical difference (p < 0.05) is seen only between treatment and controls and not between plates or passages. Taken together, this dataset suggests that the AdipoRed-based assay is suitable for screening therapeutic agents.
Results Showing Lipid Levels Measured Using AdipoRed Fluorescent Reagent.
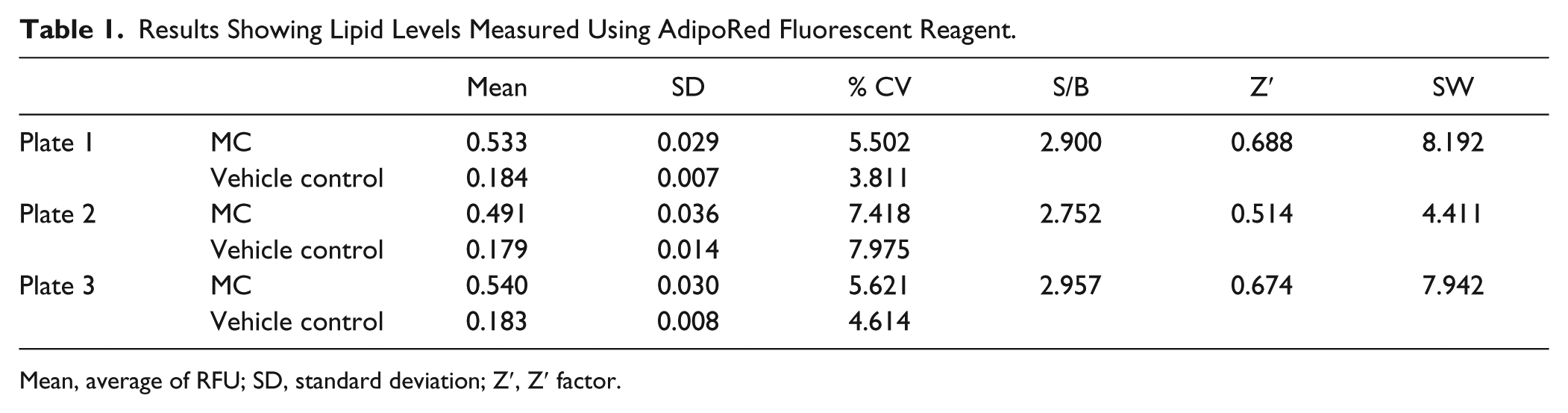
Mean, average of RFU; SD, standard deviation; Z′, Z′ factor.
Treatment with Fenofibrate, a Hypolipidemic Drug, Reduced Fat Levels in a Dose-Dependent Manner
Fenofibrate is a proliferator-activated receptor alpha (PPARα) agonist that has been explored as a therapeutic drug to treat NAFLD 22 and has been shown to possess hypolipidemic effects. To test the utility of the in vitro steatosis cell model to investigate the lipid-lowering effects of therapeutic drugs, HepG2 hepatocytes were treated with an MC of FFA mixture and various concentrations of fenofibrate for 6 h. The lipid levels were then measured using AdipoRed and normalized using Hoechst 33342 staining to control for well-to-well variability. As seen previously, treatment with an MC of FFA mixture led to a significant increase in fat content in HepG2 cells ( Fig. 5 ). In the presence of fenofibrate, the fat levels were lower and decreased in a dose-dependent manner. The maximum dose of fenofibrate tested was 150 µM because concentrations above this dose reduced cell viability (data not shown). The results from this experiment clearly demonstrate the utility of using the HepG2 steatosis cell model and an assay based on AdipoRed to investigate the hypolipidemic effects of drugs developed to treat NAFLD.
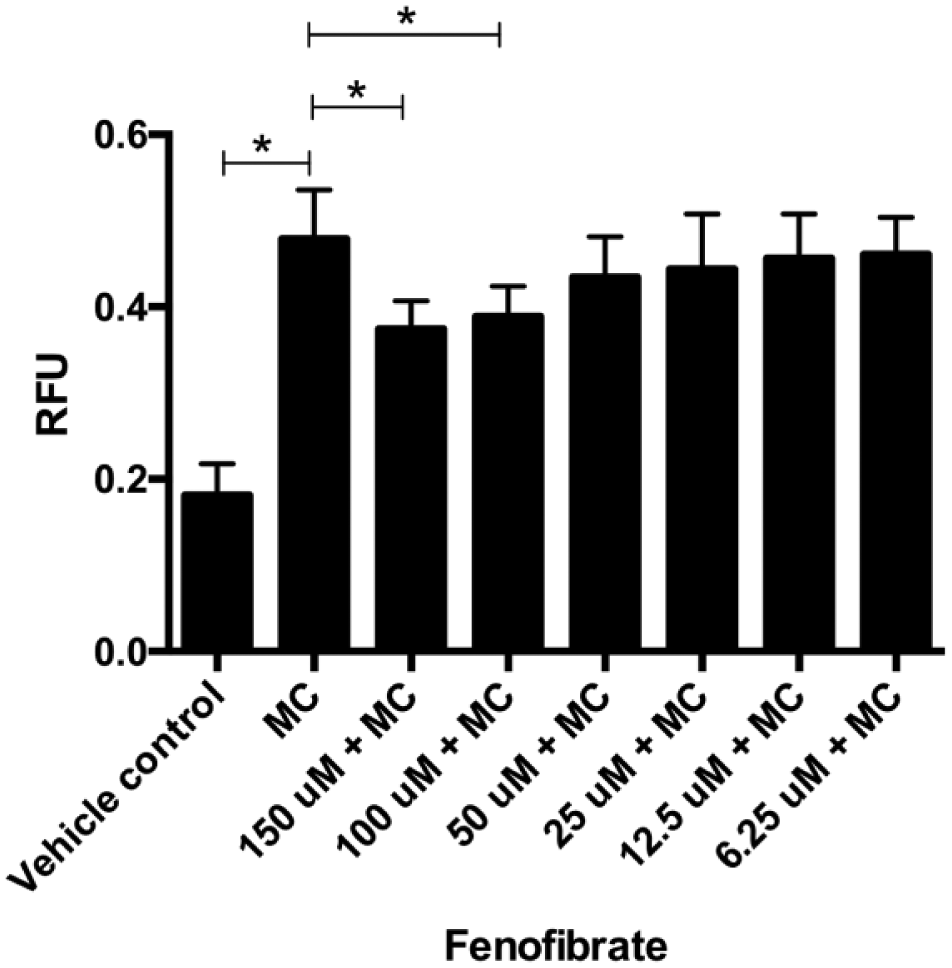
Hypolipidemic effects of fenofibrate. HepG2 cells grown to confluence in a 96-well plate were treated with vehicle control or an MC of FFAs or an MC along with fenofibrate for 6 h. The drug was tested in triplicate, with concentrations ranging from 6.25 to 150 µM. The relative fluorescence observed from three independent experiments using AdipoRed reagent was normalized using Hoechst 33342 fluorescence, and the mean and standard error are plotted. The statistical significance between the grouped means was determined using one-way ANOVA, followed by Sidak’s multiple comparison test (Prism 6, Graphpad Software). *p < 0.05.
Discussion
Given the large number of discovery programs aimed at developing novel NAFLD/NASH therapeutics, 15 the need for cellular models of human steatosis and high-throughput assays to measure intracellular lipid levels is ever increasing. In humans, fatty liver disease is characterized by the presence of >5% of hepatocytes being steatotic or >5.6% of volume fraction of fatty material in liver when measured by either proton magnetic resonance spectroscopy or quantitative fat/water selective magnetic resonance imaging. 23 In addition, the diagnosis of NAFLD requires the elimination of secondary causes and the excessive consumption of alcohol above the daily limits. 24 This essentially translates into a steatotic condition wherein a low-grade inflammatory state persists without any overt cytotoxicity. Previously, it has been reported that hepatocytes fattened in the presence of mixture containing a low proportion of palmitic acid (oleate/palmitate, 2:1 ratio) leads to a steatotic state that is associated with minor toxic and apoptotic effects, thus representing a cellular model of fatty liver that mimics benign chronic steatosis. 9 A high proportion of palmitic acid (oleate/palmitate, 0:3 ratio), on the other hand, represents a cellular model of steatosis in which saturated FFAs promote an acute harmful effect of fat overaccumulation in the liver.10,12 This cellular toxicity and viability has been directly associated with hepatic steatosis induced by a mixture of palmitic and oleic acid.25 –29 Moreover, toxicity was time and concentration (of exogenously added FFAs) dependent. 28 Taking these findings into consideration, we carefully selected the conditions required to fatten HepG2 cells while minimizing cytotoxicity using a fatty acid mixture (oleate/palmitate, 2:1 ratio). In published reports, there are differences in procedures while fattening cells.9,30 In some instances, FFAs were added directly to the growth media, while in other reports FFAs were initially complexed with BSA prior to addition to media. In our experience, we found that complexing FFAs with BSA before adding to the media precluded the formation of fatty acid aggregates and significantly improved solubility in the media. As a consequence, we believe that HepG2 cells took up FFAs more efficiently and fattened within 6 h instead of 24 h, as previously reported. 9 An increase in the concentration of FFA mixture (oleate/palmitate, 2:1 ratio) from 0.75 mM (VLC) to 2 mM (HC) led to a concomitant increase in intracellular lipid levels. The fattening of the cells was also time dependent, with increasing levels of intracellular lipids seen progressively building up for a period of 24 h following incubation with the FFA mixture. Although longer incubation periods were not tested, a previous report indicated that the maximum fatting occurs within 24 h of incubation with oleate/palmitate at a 2:1 molar ratio, irrespective of the concentration of the fatty acid mixture used. 10 The time-course fatting of HepG2 cells showed that cell viability decreased after incubating cells with 1.5 mM (MC) FFAs for 24 h. At 2 mM FFA, cytotoxicity was observed as early as 6 h of incubation. In the case of VLC and LC FFA treatments, the signal difference between the treatment and vehicle control is not large enough during any time period to assess the efficacy of therapeutic molecules. Hence, MC is the most suitable concentration to conduct the assays. The HepG2 cells fattened significantly when incubated with an MC for either 6 or 16 h, making the argument that each of these incubation periods is suitable while choosing assay conditions. However, there are two compelling reasons to fatten the cells for a shorter duration of time while screening a large number of test agents. First, it reduces the number of days required from seeding the cells to testing the efficacy of the drugs. Second, the potency of therapeutic drugs tends to decrease with time, and hence a shorter incubation time in the media is more desirable. In our experiments, sufficient fatting was observed even after 6 h with an MC of FFA mixture. The proportionate change between “no fat” and “FFA added” conditions was even more pronounced when AdipoRed reagent was used instead of triglyceride reagent to measure intracellular lipid levels. Hence, a 6 h fatting paradigm is also suitable as a steatosis model to study the hypolipidemic effects of drugs. As an example, we show that fenofibrate, 22 a drug previously developed to treat NAFLD/NASH, had demonstrable lipid-lowering effects under this paradigm, thereby validating the utility of the hepatic steatosis model for screening therapeutic drugs. Fenofibrate is a small molecule that binds to the nuclear receptor, peroxisome PPARα, leading to its translocation to the nucleus and the transcription of several genes that lead to beta-oxidation, and inhibition of triglyceride synthesis.22,31,32 The hypolipidemic effects of fenofibrate have been previously demonstrated in several hepatic cell lines, rodents fed with high-fat diet, and humans.32 –35 Similar to these observations, fenofibrate showed about a 20% fat reduction in our experiments. While this may not appear to be a substantial reduction in fat levels, it is important to note that this was a single-dose treatment and only for a short duration (6 h). Cornu-Chagnon et al. showed that fenofibrate takes about 24 h to be completely metabolized in cell cultures. 36 Hence, a treatment for a prolonged duration of time or multiple doses could potentially lead to greater lipid reduction.
Traditionally, intracellular lipid levels are quantified using oil-red-o staining 37 or biochemical estimation of free glycerol levels released following lipase digestion of lipids. 38 These methods, however, are not suitable for medium- or high-throughput assaying platforms. To overcome this limitation, we developed an assay based on fluorescence readout that requires minimal handling of cells and does not involve the laborious steps, such as cell lysis and lipid extraction. An assay based on fluorescence also has several advantages over absorbance assays due to their high sensitivity, diverse selection of fluorophores, ease of operation, and various readout modes (fluorometer or microplate reader). 39 Typically, fluorescence- based assays improve the signal one to two orders of magnitude without increasing the background. This is true for the assay described in this article as well. A meaningful and transformative improvement in signal-to-noise ratio provides lower limits of detection, which is critical while differentiating molecules that have a narrow efficacy range. While AdipoRed was used in these studies, it is possible that other fluorescent reagents, such as LipidTox 40 and Bodipy, 41 can also improve the sensitivity of the triglyceride assay. However, conditions need to be specifically optimized for each of the dyes because the S/B, limits of detection, sensitivity, and stability, to name a few, are different for each of these fluorophores.
The results presented in this report show that HepG2 cells fattened using a mixture of oleate/palmitate can effectively be used to reveal the hypolipidemic effects of therapeutic molecules. Along with the fluorescence-based assay, drug screening can be performed under medium- and high-throughput conditions.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was partially funded by National Institutes of Health/National Institute of Diabetes and Digestive and Kidney Diseases grant no. 5R43DK092122. The content is solely the responsibility of the authors and does not necessarily represent the official views of the National Institutes of Health.
