Abstract
Phage display technology (PDT) is a powerful tool for the isolation of recombinant antibody (Ab) fragments. Using PDT, target molecule-specific phage-Ab clones are enriched through the “biopanning” process. The individual specific binders are screened by the monoclonal scFv enzyme-linked immunosorbent assay (ELISA) that may associate with inevitable false-negative results. Thus, in this study, three strategies were investigated for optimization of the scFvs screening using Tomlinson I and J libraries, including (1) optimizing the expression of functional scFvs, (2) improving the sensitivity of ELISA, and (3) preparing different samples containing scFvs. The expression of all scFv Abs was significantly enhanced when scFv clones were cultivated in the terrific broth (TB) medium at the optimum temperature of 30 °C. The protein A–conjugated with horseradish peroxidase (HRP) was found to be a well-suited reagent for the detection of Ag-bound scFvs in comparison with either anti-c-myc Ab or the mixing procedure. Based on our findings, it seems there is no universal media supplement for an improved expression of all scFvs derived from both Tomlinson I and J libraries. We thus propose that expression of scFv fragments in a microplate scale is largely dependent on a variety of parameters, in particular the scFv clones and relevant sequences.
Introduction
Antibodies (Abs) have been used for many research, diagnostic, and therapeutic applications. 1 A hybridoma tool and different display technologies, in particular phage display technology (PDT) as one of the commonly used approaches, have been applied to generate monoclonal antibodies (mAbs) against a variety of antigens (Ags). 2 PDT is a versatile and straightforward tool to expand scientific capability for the production of recombinant Ab fragments against any Ag based on the phage-Ab library used. 3 The concept underlying PDT is the linkage of the genotype to the phenotype, in which the Ab variable region genes are fused approximately close to the gene coding for a coat protein of a filamentous phage (i.e., often gene 3 minor coat protein) to display as a fusion protein with the desired binding function. 4 Generally, PDT is considered a molecular diversity selection platform, through which Ab fragment gene repertoires are cloned into a phagemid vector to generate a combinatorial Ab library. Functional Ab fragments could be displayed on the surface of filamentous phage particles as a g3p fusion protein. 5 Specific phage-Ab binders are subsequently enriched from the combinatorial library by multiple rounds of affinity selection on any Ag in a process known as biopanning. 6 During this process, recombinant phage-Abs are incubated with immobilized Ags of interest, and then specific phage binders are eluted after washing out unbound phages. The eluted specific binders are amplified through an infection of Escherichia coli cells and prepared for the subsequent panning rounds. 7 Biopanning is an exclusive enrichment process that results in different settings, including (1) background binders (e.g., phage binders to solid surface materials presented by enzyme-linked immunosorbent assay (ELISA) plates or magnetic beads during biopanning), (2) nonspecific phage binders (e.g., bald phages or phage-Ab binders to unwanted Ags existing in a selection environment, such as streptavidin binders in solution-phase biopanning), 3 or (3) even Ag-specific phage-Ab binders. Therefore, a powerful and rapid screening assay after biopanning is a crucial step for the identification of Ab fragments that specifically bind to the respective Ag molecules.
There are a variety of screening methods, such as ELISA, Western blot, flow cytometry, BIAcore, and immune cyto-/histo-chemistry. Microtiter plate (MTP)–based ELISA has been the most extensively used assay during the screening process in PDT.8,9 For the ELISA screening assay, individual scFv clones from a desired round of biopanning are basically grown in a microplate platform and then binding reactivity of scFvs secreted into the culture supernatant is assayed with immobilized Ag or whole cell. The ELISA provides a large number of individual scFv clones to be screened simply and rapidly at the same time without special equipment. 10 However, a major drawback of the ELISA screening approach might be the false-negative results that arise from the low sensitivity of the scFv ELISA and/or the low expression of scFv Abs in the MTP.11,12 The low sensitivity of the scFv ELISA is likely attributed to the low reactivity of scFvs with their own target Ags, in large part because of either their low affinity/avidity or the low density of the target Ag. 13 Monovalent Ab fragments have a single binding site, resulting in a low avidity for the target Ag, in particular when compared with the conventional bivalent mAbs. It has been shown that the yield of Ag-reactive scFv clones can be increased through mixing monovalent scFvs with an anti-tag mAb prior to the utilization for binding assay in order to dimerize scFv fragments with improved avidity. 13 On the other hand, based on individual clones, the level of scFv expression may be varied widely, affecting the overall results of the screening assay. Therefore, several parameters (e.g., media composition and different supplements) should be applied to improve the expression of functional scFv Abs in the MTP platform.
In the current work, to increase the production of scFv Abs in the MTP level, we studied various media supplements, such as sucrose, Triton X-100, glycine, glycerol in different concentrations, and different culture media (e.g., 2xYT, buffered 2xYT, Luria-Bertani [LB], buffered LB, and TB), to improve the yield of positive hits during the high-throughput screening procedure after the biopanning process.
Materials and Methods
ScFv Ab Fragments
The four scFv Ab fragments used in this study were previously isolated using the semisynthetic human single-fold scFv libraries I + J (Tomlinson I + J, Source BioScience, Nottingham, UK), against the second extracellular loop (ECL2) of gastrin/cholecystokinin B receptor (CCKB-R). Two scFv clones, ID3 and IC4, from library I 14 and two other clones, JC1 and JC2, from library J (our unpublished data) were obtained through solution-phase biopanning using a biotin-streptavidin system against a biotinylated peptide equivalent to ECL2. The library I and J is based on a human single framework for VH (V3-23/DP-47 and JH4b) and VL (O12/O2/DPK9 and Jκ1), respectively, with DVT and NNK randomization with about 1.4 × 108 transformants. In this library, scFv Abs are cloned in an ampicillin-resistant phagemid vector (pIT2) close to the gene of minor coat protein III of the filamentous phage.
Production of Soluble scFvs in MTPs
The E. coli HB2151 harboring the defined scFv Abs were cultured using TYE plates supplemented with 2% glucose and 100 µg/mL ampicillin and incubated overnight at 37 °C. The next day, a single colony was inoculated into 96-well MTPs containing 200 µL of 2xYT + 2% glucose + 100 µg/mL ampicillin, and incubated overnight at 37 °C under shaking (1000 rpm) in an MTP shaker (Thermo Shaker PST-60 HL Plus, BOECO, Hamburg, Germany). Then, 4 µL of the culture was transferred into 200 µL 2xYT or an alternative media (supplemented with 2% glucose and 100 µg/mL ampicillin). The culture was grown under shaking (1000 rpm) at 37 °C for 1–2 h to reach the optical density (OD600) value of 0.9 as an indicator to initiation of the induction of the recombinant Ab expression. After centrifugation at 1800g for 10 min, bacterial media were changed with the induction media containing 200 µL of 2xYT or an alternative media supplemented with 100 µg/mL ampicillin + 0.05 mM isopropyl-β-
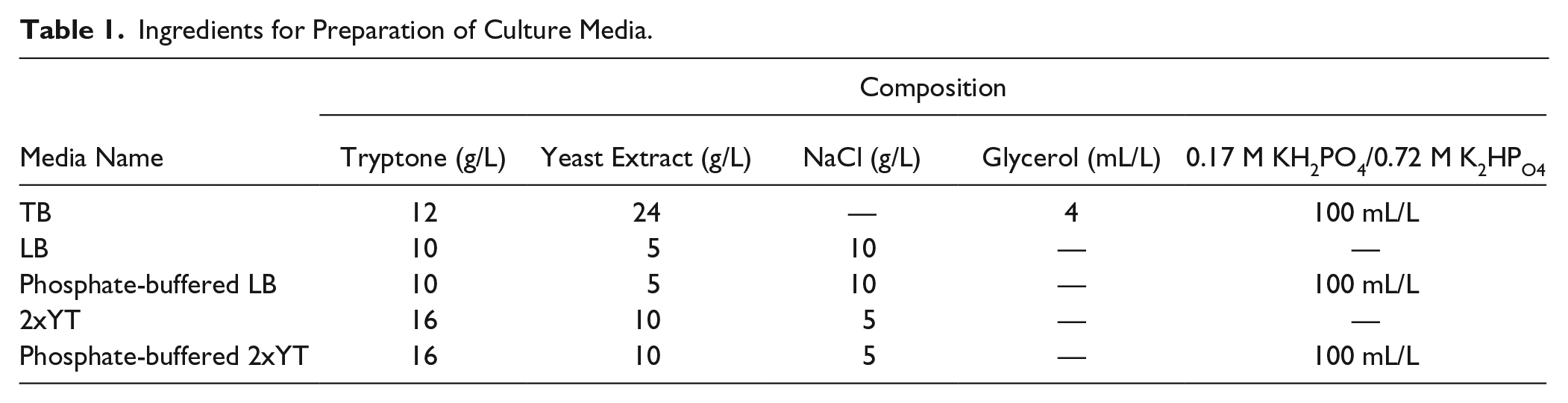
Ingredients for Preparation of Culture Media.
ELISA
High binding ELISA plates (Biomat) were used to coat the biotinylated peptide indirectly. A volume of 100 µL/well biotinylated-BSA (Thermo Fisher Scientific, Waltham, MA) was applied to the plates at a concentration of 2 µg/mL in phosphate-buffered saline (PBS) at 4 °C overnight. After washing (3×) with PBST (i.e., PBS supplemented with 0.1% v/v Tween 20), 100 μL/well streptavidin (Thermo Fisher Scientific) at a concentration of 10 µg/mL in PBS with 0.5% gelatin was added to the wells and incubated under gentle shaking at room temperature for 90 min. Following washing as described above, the plates were incubated with 100 µL/well of the biotinylated peptide(200 nM) at 4 °C for 2 h. After blocking with 2% MPBS (i.e., 2% w/v skimmed milk powder in PBS) at room temperature for 90 min, the plates were incubated with serial dilutions of the scFv-containing supernatants in equal volume of 2% MPBS for 90 min. Otherwise stated, binding of scFvs to the biotinylated peptide was detected by 1:8000 dilution of protein A–HRP (Thermo Fisher Scientific), recognizing the heavy chain variable region of gene 3 family (VH3). Then, the staining reaction was developed by adding 100 µL of 3,3′,5,5′-tetramethyl benzidine (TMB) as a substrate and stopped by adding 50 µL of 5% sulfuric acid. The OD values were measured at 450 nm subtracted from 630 nm using a microtitre plate reader, BioTek ELx800 (BioTek, Winooski, VT).
Statistical Analysis
One-way analysis of variance (ANOVA), followed by Bonferroni multiple comparison post hoc test, was applied to compare differences among the experimental groups using GraphPad Prism 7.0. Statistically significant differences were considered at p < 0.05.
Results
To increase the rate of positive hits during the soluble scFv ELISA screening, three different approaches were implemented: (1) optimizing the expression of functional scFvs, (2) improving the sensitivity of ELISA, and (3) preparing different samples containing scFv in the assay. In this study, we used four scFvs that were screened previously against ECL2 of CCKB-R: two scFvs from the library I (D3 and C4) and two scFvs from the library J (JC1 and JC2).
The production of functional scFvs with phage display vector pIT2 was optimized using different conditions, including expression temperatures (20, 25, and 30 °C), media supplements (glycine, sucrose, Triton X-100, and glycerol), and culture media (TB, LB, phosphate-buffered LB, 2xYT, and phosphate-buffered 2xYT).
Impacts of Temperature
In all scFv clones, the cultivation temperature displayed a strong influence on the production of scFvs with optimal expression at 30 °C ( Table 2 ). Noticeably, lowering the temperature to 25 °C and 20 °C resulted in a significant decrease in the production of functional scFvs.
Summary of the MTP Production of Soluble ScFvs.
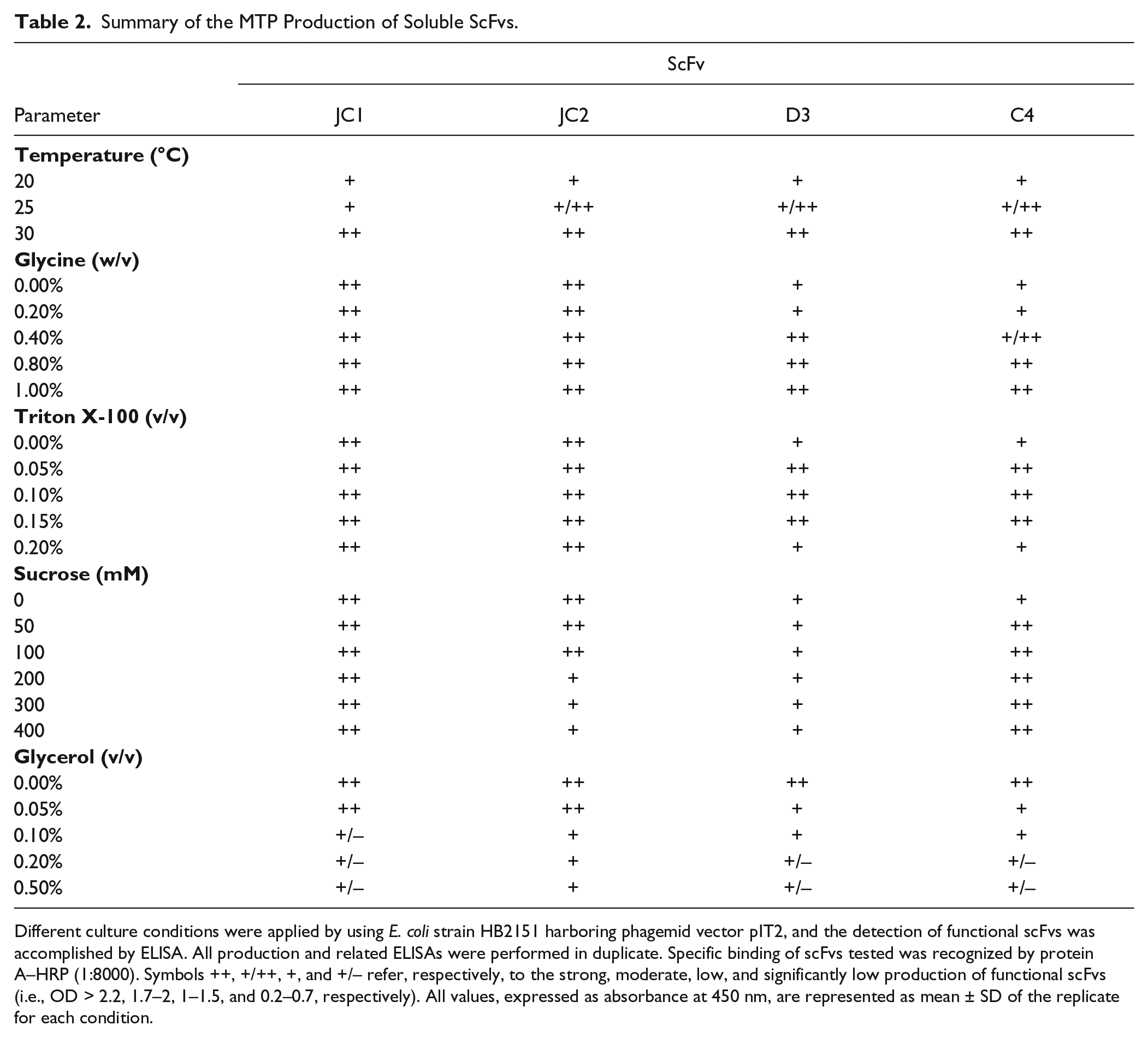
Different culture conditions were applied by using E. coli strain HB2151 harboring phagemid vector pIT2, and the detection of functional scFvs was accomplished by ELISA. All production and related ELISAs were performed in duplicate. Specific binding of scFvs tested was recognized by protein A–HRP (1:8000). Symbols ++, +/++, +, and +/– refer, respectively, to the strong, moderate, low, and significantly low production of functional scFvs (i.e., OD > 2.2, 1.7–2, 1–1.5, and 0.2–0.7, respectively). All values, expressed as absorbance at 450 nm, are represented as mean ± SD of the replicate for each condition.
Influence of Media Type and Supplements
The effect of media supplements was analyzed using 2xYT medium at 30 °C overnight. Glycine at 0.8% and 1% concentrations increased the functional yield of two scFv clones (D3 and C4), but not the other two scFv clones (JC1 and JC2). As shown in Table 2 , the use of Triton X-100 at designated concentrations (i.e., 0.05%, 0.1%, and 0.15% v/v) as media supplement resulted in an enhanced functional production of two scFvs (i.e., D3 and C4). The addition of 50 or 100 mM sucrose increased the functional production of one of the four scFv clones without any effect on the production of the other three ( Table 2 ). The introduction of glycerol to the media surprisingly decreased the expression of all four scFv clones. The composition of the medium is another significant parameter that should be evaluated for the production of functional scFvs. Based on our findings ( Fig. 1 ), we found the TB media to be significantly influential among all other culture media tested, resulting in an increased yield of all scFvs. Thus, TB media was considered as the most suitable media for the expression of functional scFvs during screening.
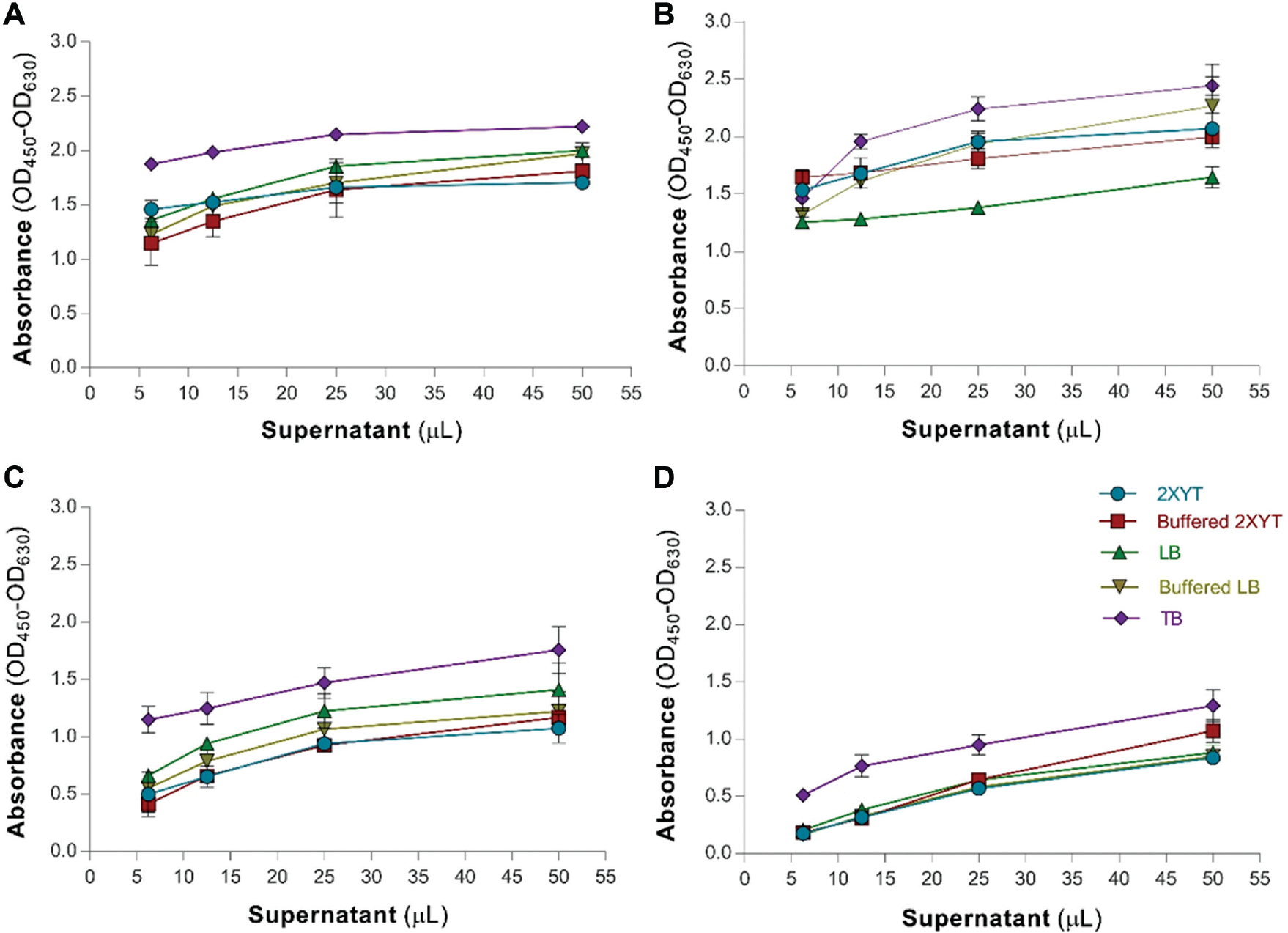
Influence of different culture media on scFvs yield. Supernatants of four scFvs designated JC1 (
Effect of Different Detection Methods
To circumvent the low sensitivity of soluble scFv ELISA as a major bottleneck during screening, we aimed to enhance the ELISA signals via different detection methods using protein A–HRP and no mixing approach (i.e., anti-c-myc tag and goat anti-mouse HRP conjugated as primary and secondary Abs, respectively) as well as a mixing approach (incubation of monovalent scFvs with anti-c-myc tag mAb; for detailed protocols, refer to the work conducted by Wang et al. 13 ). As shown in Figure 2 , with all scFvs tested, the maximal binding reactivity was obtained by using protein A–HRP as the detection reagent, while the mixing method failed to provide high ODs.
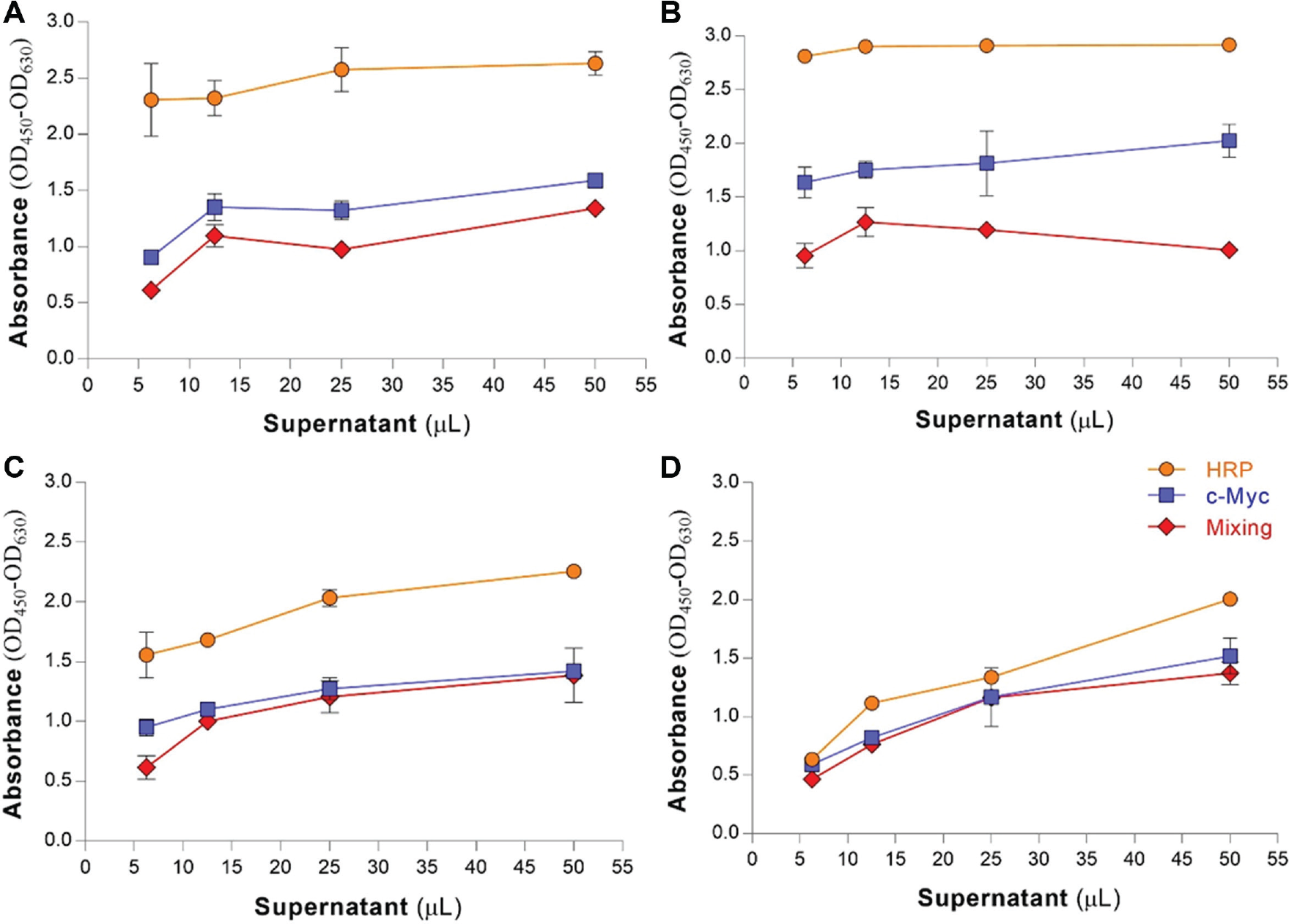
Effect of different detection methods applied in the binding assay. Supernatants of four scFvs designated JC1 (
Effect of Different Sample Preparations
Another strategy to enhance the signal intensity during binding assay was to use different scFv sample preparations, including (1) supernatant, (2) periplasmic, and (3) supernatant plus periplasmic samples. As shown in Figure 3 , among samples prepared for the binding assay, the supernatant preparations and the periplasmic extracts were found to produce the maximum and minimum signals, respectively.
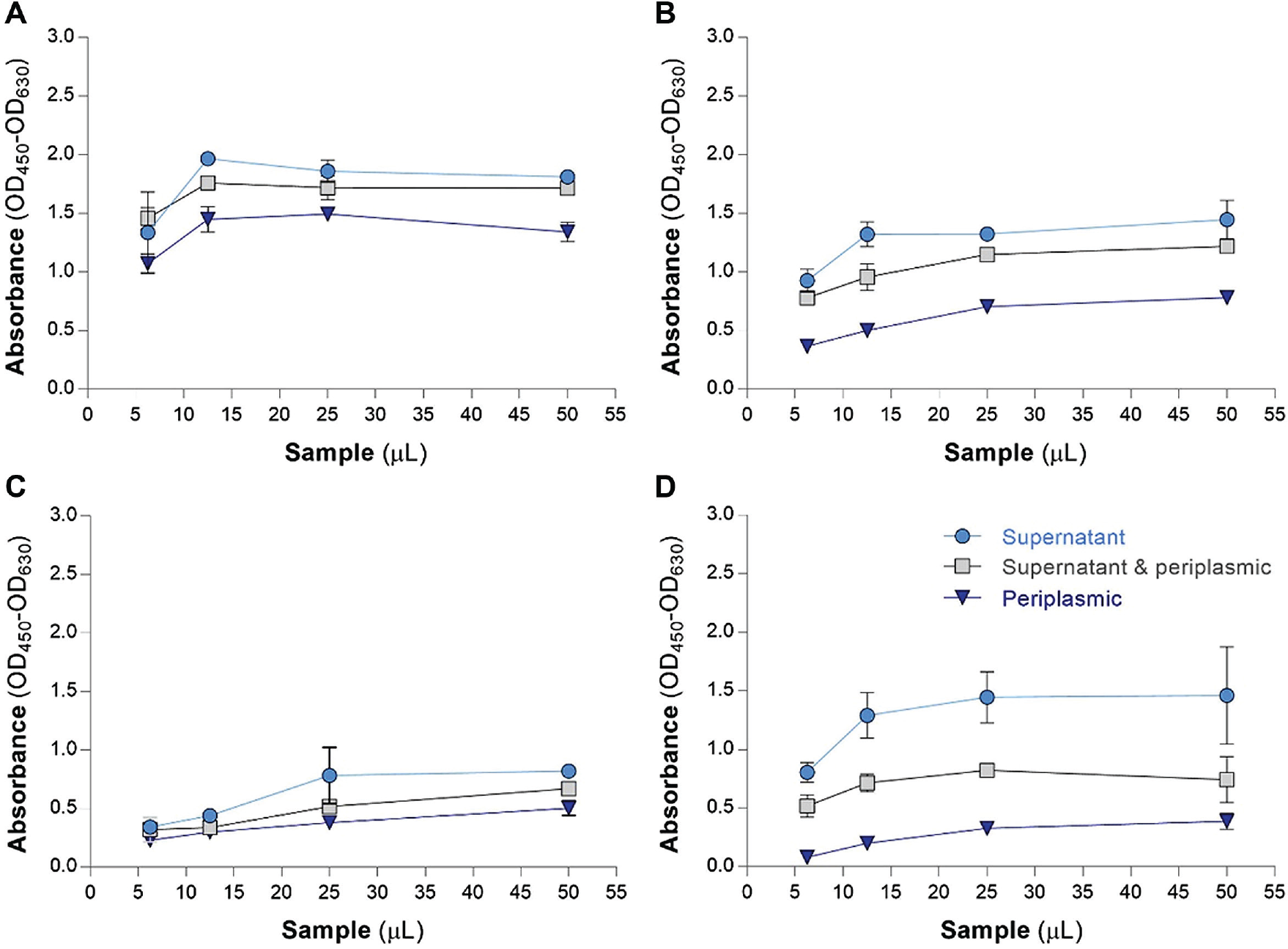
Effect of different sample preparations on ELISA signal improvement. Different sample preparations of four scFvs designated JC1 (
Discussion
To date, a large number of Abs have been produced as specific detection reagents and used in different applications, such as high-throughput proteomics studies, to analyze various undefined Ags. Considering about 40,000 genes in the human genome in combination with different splice variants and their various post-translational modifications, an extremely huge number of proteins (i.e., at least 100,000 different protein products) can be expected for the biological functions,15,16 for whose analyses the use of arrays of Abs appears to be inevitable. However, many Abs are yet to be produced for such analyses. In contrast to the traditional hybridoma technology, PDT provides a combinatorial in vitro approach for the generation of Ab fragments (e.g., scFv, Fab, and Fv) specific to any designated Ag. Using PDT, Ab fragments can be generated within a short period of time, in large part because of avoiding animals’ immunization and use. 17 In addition, once coupled with certain modifications for large ligand-based selection (e.g., through a biotin-streptavidin system), PDT provides great capacity for automated and robotic drug discovery. This unique features enable PDT as a high-throughput Ab screening technology to be well suited for proteomic analysis in the postgenomic research era. 18 In general, such endeavors require high-throughput screening procedures via phage ELISA to identify individual phage binders. Phage ELISA is a fast and robust screening assay because it allows many clones to be screened in parallel. However, it may be associated with some inadvertent false-positive signals, arising from scFv fragments that are functional merely when fused to the phage coat protein pIII. 19 Therefore, individual binders should be screened as secreted soluble scFv Abs. To this end, phagemid vectors with the dual purpose of the monovalent display of Ab fragments and the production of soluble Ab fragments have been developed by the insertion of an amber codon between the Ab and pIII genes to speed up the screening procedures through avoiding the Ab fragment genes to be subcloned. 20 In this work, we considered different strategies to improve the functional scFv production in MTPs and to augment the ELISA signals to facilitate the screening process after phage biopanning.
It has been well documented that the expression of recombinant proteins can be affected by the cultivation temperature. The aggregation of proteins at low temperatures (i.e., 30 and 25 °C) is prevented, in large part, by increasing the ratio of soluble fraction to insoluble fraction, thereby improving the production of functional recombinant proteins.21,22 Consistent with this line of reasoning, the cultivation temperature needs to be optimized during the screening of soluble scFv binders in order to improve the production of functional scFvs. Based on our experiments, the cultivation at 30 °C appeared to be the best temperature for screening scFv clones from the Tomlinson I + J libraries. It should be noted that a further decrease in the cultivation temperature (down to 25, 20, and 18 °C) can improve the solubility of recombinant proteins; however, the overall production of functional scFvs was shown to be substantially reduced because of a possible decrease in the bacterial growth and metabolic enzyme activity.23,24
It has already been reported that glycine and Triton X-100 are able to dramatically increase the production and excretion of exogenous proteins in the culture medium, in large part by virtue of enhanced permeability of the cell wall and/or the induction of bacteriolysis.25,26 As a proof of concept, a research group showed an increased yield of scFv-TNF-α fusion protein in E. coli K802 up to 14- and 38-fold, respectively, by addition of 2% glycine and 1% Triton X-100 into the induction media. 24 Similarly, our findings revealed that use of 1% glycine and 0.1% Triton X-100 could increase the functional production of at least two scFvs in E. coli HB2151. However, based on our findings, a further increase in the concentration of glycine (>1%) and Triton X-100 (>0.2%) led to a significant decrease in the production of functional scFvs (data not shown). Therefore, we propose that an optimized concentration of glycine and Triton X-100 is an essential factor for increasing the functional yield of recombinant proteins, which is largely dependent on E. coli host strain and species and the sequence of the recombinant protein. Sucrose is another culture media additive used to improve the functional yield of scFvs in MTPs. Several studies have shown that the addition of nonmetabolized sugars, such as raffinose (a trisaccharide consisting of galactose, glucose, and fructose) or sucrose, could increase the efficiency of protein folding, and hence the amount of soluble recombinant protein. Presumably, these sugars diffuse into the periplasmic space and increase the osmotic pressure, which results in enlargement of the periplasmic space and also reduction of the local concentration of secreted protein, thereby preventing the protein aggregation. 27 In a systematic effort using four different scFv Abs, 100 mM sucrose was shown to efficiently improve the functional yield of two scFvs in MTPs. 8 Kipriyanov et al. showed a 15- to 150-fold increase in the functional yield of scFv-phOx specific to hapten 2-phenyloxazol-5-one using 0.4 M sucrose. 28 However, another research group highlighted that the addition of 0.4 M sucrose in small-scale culture media could result in an increased ratio of soluble anti-c-Met scFv to insoluble fraction without any effect on the functional yield. 23 Our results revealed that addition of 100 mM sucrose into the induction culture media could improve the functional yield of only one scFv, upon which we speculate that the effect of sucrose on the productivity of either soluble or functional scFvs is conditional, depending on amino acid sequence. Glycerol can be used as a carbon source for the enhanced protein expression, perhaps because of the increased cell densities. 29 Accordingly, the functional expression of anti–Burkholderia pseudomallei exotoxin scFv in XL1-Blue as the host was shown to be increased by using 0.2% glycerol. 30 In our study, the addition of glycerol to the culture media imposed negative impacts on the expression of all scFvs. We speculate that a possible explanation for such impact can be the fast accumulation of dihydroxyacetone phosphate that is harmful for the host cell.
The composition of bacterial culture media is another important parameter influencing the expression of scFv Ab. In a study conducted by Hust et al., TB and 2xYT supplemented with 10% phosphate buffer were shown to be the most suitable media for the expression of different functional scFv Abs, perhaps because of an increased cell density in MTPs. 8 Likewise, in the current study, we found TB to be the best medium for the expression of four scFv clones obtained from Tomlinson I + J libraries. We believe that the composition of the TB medium is enriched with the greater amount of yeast extract that helps the growth of bacteria despite the presence of glycerol and phosphate. It should be highlighted that the other three types of culture media containing phosphate buffer resulted in decreased expression of functional scFvs compared with the related media without phosphate buffer.
In addition to the low expression of scFv ELISA, the low sensitivity of scFv ELISA could result in another limitation during the screening process. Thus, we examined the effects of three different detection reagents (i.e., protein A–HRP, anti-c-myc Ab, and mixing) to improve the detection signal. Among these reagents, protein A–HRP and mixing were found to produce the strongest and weakest signals, respectively. These results are contrary to the previous findings, 13 in which it was reported that the mixing method that involves the incubation of scFv fragments expressing a c-myc epitope tag with anti-tag mAb 9E10 prior to their use in binding assays could provide a two-fold increase in the binding reactivity of scFv fragments with the corresponding Ag. 13 The reason for this difference may be attributed to the use of detection reagents produced by different companies, in which the type and amount of HRP used for conjugation are different.
Finally, to achieve scFvs with the highest expression, different scFv samples (i.e., supernatant, periplasm, and supernatant plus periplasm samples) were used in our study. Although in a study conducted by Hust et al. the supernatant plus periplasm samples provided stronger signal than the culture supernatant, 8 our findings showed that the culture supernatant, supernatant plus periplasm, and periplasm samples provided the highest binding reactivity, consecutively. This can be explained through the use of different bacterial hosts for the expression of scFvs, that is, E. coli HB2151 in our study and MRF XL1-Blue in the study directed by Hust et al. 8
Footnotes
Acknowledgements
The authors are very grateful for the technical support provided by the members of the Research Center for Pharmaceutical Nanotechnology at Tabriz University of Medical Sciences.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The authors would like to acknowledge Tabriz University of Medical Sciences for the funding (grants 93003 and 95004).
