Abstract
Angiogenesis is a promising target for anticancer therapies, but also for treating other diseases with pathologic vessel development. Targeting the vascular endothelial growth factor (VEGF) pathway did not proof as effective as expected due to emerging intrinsic resistance mechanisms, as well as stromal contributions leading to drug insensitivity. Therefore, alternative strategies affecting the interaction of endothelial cells (ECs) with other stromal cells seem to be more promising. Human preclinical in vitro angiogenesis models successfully recapitulating these interactions are rare, and two-dimensional (2D) cell cultures cannot mimic tissue architecture in vivo. Consequently, models combining three-dimensionality with heterotypic cell interaction seem to be better suited. Here, we report on an improved human fibroblast–EC coculture assay mimicking sprouting angiogenesis from EC-covered microbeads resembling existing endothelial structures. Culture conditions were optimized to assess pro- and antiangiogenic compounds. Important characteristics of angiogenesis, that is, the number of sprouts and branch points, sprout length protrusion, and overall vessel structure areas, were quantified. Notably, the endothelial sprouts display lumen formation and basal membrane establishment. In this model, angiogenesis can be inhibited by genetic interference of pro-angiogenic factors expressed in the fibroblasts. Moreover, bona fide antiangiogenic drugs decreased, whereas pro-angiogenic factors increased vessel formation in 24-well and 96-well settings, demonstrating the applicability for screening approaches.
Keywords
Introduction
Angiogenesis is defined as the expansion of the preexisting vascular network that can occur by different mechanisms, of which sprouting and branching of new capillary vessels is the most prominent. Angiogenesis plays a major role in development and reproduction, as well as in wound healing, but is also a critical process in a variety of pathological disorders, including intraocular neovascular disorders and inflammatory diseases. 1 Regarding cancer, angiogenesis contributes to tumor progression and metastasis. 2 On the other hand, malfunctions in vascular growth can be causative for ischemic events such as myocardial infarction, diabetic ischemia, and stroke or neurodegenerative disorders. 3 Under normal conditions, vessels and endothelial cells (ECs) are in a quiescent state, but ECs have a dynamic capability to quickly respond to pro-angiogenic signals, for example, in case of injury. Angiogenesis is driven by the spatial release of pro-angiogenic factors such as vascular endothelial growth factor (VEGF), fibroblast growth factor (FGF), and platelet-derived growth factor (PDGF) when new blood supply is needed.4,5 The angiogenic process includes basement membrane breakdown and extracellular matrix (ECM) remodeling, EC migration/invasion, proliferation, lumen formation, and finally, vessel stabilization and perfusion. Beside ECs, the main cellular players involved in angiogenesis are pericytes, smooth muscle cells, and fibroblasts. Vessels are embedded in a basal membrane and are surrounded by connective tissue. During the angiogenic process, fibroblasts are predominantly responsible for ECM maintenance and providing regulatory factors.6,7
As excessive, inadequate, or compromised vessel outgrowth is involved in a multitude of pathologic events, targeting angiogenesis either negatively or positively is a promising therapeutic approach. To date, neither therapeutic stimulation by pro-angiogenic factors nor inhibition of angiogenesis could meet the expectations in the clinics. Most approaches to block excess angiogenesis focused on the VEGF or related pathways. Emerging intrinsic resistance mechanisms, such as VEGF-independent vessel growth or genetic variations in VEGF receptors, can lead to loss of efficacy of these drugs. 8 Apart from that, stromal cells such as myeloid cells, 9 pericytes,10,11 and fibroblasts 12 are implicated to modulate drug responses and to help, for example, cancer cells to escape the angiogenic blockage.8,13
Thus, there is urgent need for improved antiangiogenic therapeutic concepts. However, human preclinical in vitro angiogenesis models faithfully recapitulating the in vivo situation, such as heterotypic cell–cell interactions and tissue architecture, are rare. Moreover, there is compelling evidence that three-dimensional (3D) cell cultures are better suited to predict the efficacy of a specific drug in vivo. 14 Consequently, better models are required that include other cell types than ECs in a 3D setting to gain more information on the molecular mechanisms of angiogenesis. Novel molecular players involved in angiogenesis, as well as the context-dependent activity of specific targets, remain to be evaluated.
A commonly used 3D assay for screening of pro- and antiangiogenic factors is the Matrigel tube formation assay. Matrigel is a complex mixture of basement membrane proteins and several growth factors derived from Engelbreth–Holm–Swarm mouse sarcoma cell 15 and allows formation of capillary-like structures in short time. However, despite the fact that stromal interactions are missing, network formation is not restricted to ECs. Other cell types, such as fibroblasts or cancer cells, also form interconnected cellular nets on Matrigel. Thus, comparability to the in vivo situation has to be questioned. 16 Other 3D assays try to mimic the in vivo situation by embedding ECs in an ECM, that is, collagen I or fibrin gels with optional addition of other stromal cells. 17 These 3D assays are difficult to analyze due to the structural network in the matrix that needs to be measured in more than one optical plane.
As an alternative, Bishop et al. developed a powerful method to obtain vessel-like structures by directly cocultivating ECs with fibroblasts. 18 No addition of an external matrix is needed, as the ECM that is required for EC tube formation is provided by densely seeded fibroblasts on a two-dimensional (2D) surface. Nevertheless, three-dimensionality is provided in addition to direct cell–cell contact. However, the cultures display reduced thickness and therefore are easy to analyze. In angiogenic tube formation assays, generally tubule length, number of tubules, number of branch points, and in some cases, the tubule area are determined. 17 Performing the original fibroblast coculture assay, drawing a conclusion on all parameters is difficult, as an initial structure imitating a preexisting vessel is missing. In the fibrin gel bead assay, first described by Nehls and Drenckhahn, 19 ECs are seeded onto microcarrier beads before insertion into a fibrin gel matrix. In this setting, the microcarrier beads are representing the preexisting vascular structure, from where angiogenic sprouting occurs. With the following method, we combined the effective formation of mature EC vessel structures in fibroblast cocultures with the simulation of angiogenic sprouting from existing endothelial structures mimicked by EC-covered microbeads. Although being a human cellular 3D test system, analysis of the most important characteristics of angiogenesis is facilitated.
Material and Methods
Cells
Primary human umbilical vein endothelial cells (HUVECs, ATCC no. PCS-100-013) were grown in EGM-2-MV (no. CC-3202, Lonza, Basel, Switzerland). HUVECs not older than p4 were used. Mouse skin fibroblasts obtained from p19ARF–/– mice (a gift of Professor W. Mikulits, Medical University of Vienna), IMR-90 lung fibroblasts (ATCC CCL-186), and hTERT-immortalized foreskin fibroblasts (hTERT-BJ1, cat no. 4001-1, Clontech, Mountain View, CA) were cultured in Dulbecco’s modified Eagle’s medium (DMEM, no. 21969-035, Gibco, Life Technologies, Carlsbad, CA) supplemented with 10% fetal bovine serum (FBS; no. 10500-064, Gibco, Life Technologies). Normal human colonic fibroblasts (NCFs) were established as reported previously 20 and were propagated in EGM-2-MV. Cells and cocultures were cultivated in a humidified incubator with 5% CO2 under normoxic conditions at 37 °C.
Microcarrier Bead Fibroblast Coculture Assay
Collagen-coated Cytodex 3 microcarrier beads (no. 17-0485-01, Amersham, GE Healthcare, Buckinghamshire, UK) were covered with HUVECs according to the protocol described previously by Nakatsu et al. 21 the day before the coculture assay was started. In brief, 400 ECs per bead were used. In total, 2500 beads were mixed with 106 cells in a 5 mL tube in 1.5 mL complete EC growth medium. The tube was placed vertically in the incubator and incubated at 37 °C for 4 h. The suspension was mixed up every 20 min by shaking the tube gently for efficient coating. Afterwards, the bead–cell suspension was transferred into a 10 cm tissue culture plate in 8 mL of EGM-2-MV and incubated at 37 °C overnight to remove cells not attached to the beads. The next day, beads were washed three times in a 15 mL tube by gravity sedimentation and resuspension in 1 mL of fresh growth medium. For counting, 20 µL of the bead suspension was pipetted onto a coverslip and bead number was determined using a microscope. The number of beads per milliliter was calculated using the following formula: (number of beads in 20 µL × 1000)/20. Beads were resuspended in fresh EC growth medium at a concentration of 100 beads per milliliter. Twelve-, 24-, or 96-well plates were coated for at least 1 h with 50 µg/mL collagen I (no. 354236, Corning, Corning, NY) in 20 mM acetic acid and washed rigorously with ddH2O. For the microcarrier bead fibroblast coculture angiogenesis assay, 4 × 105 fibroblasts were seeded into each well of a 24-well plate (8 × 105 in 12-well plate, 6 × 104 for 96-well plate) in 1 mL of DMEM/10% FBS. Bead suspension (200 µL) was added and the plates were placed in a 37 °C incubator overnight. The next day, medium was removed and replaced by 1 mL of angiogenesis standard medium (EGM-2-MV:DMEM/10% FBS in a ratio of 1:10). The cocultures were incubated for 14 days, when BJ1 fibroblasts were used, or 7 days for all other fibroblasts, changing the medium every other day. A schematic outline of the assay is given in Figure 1A .
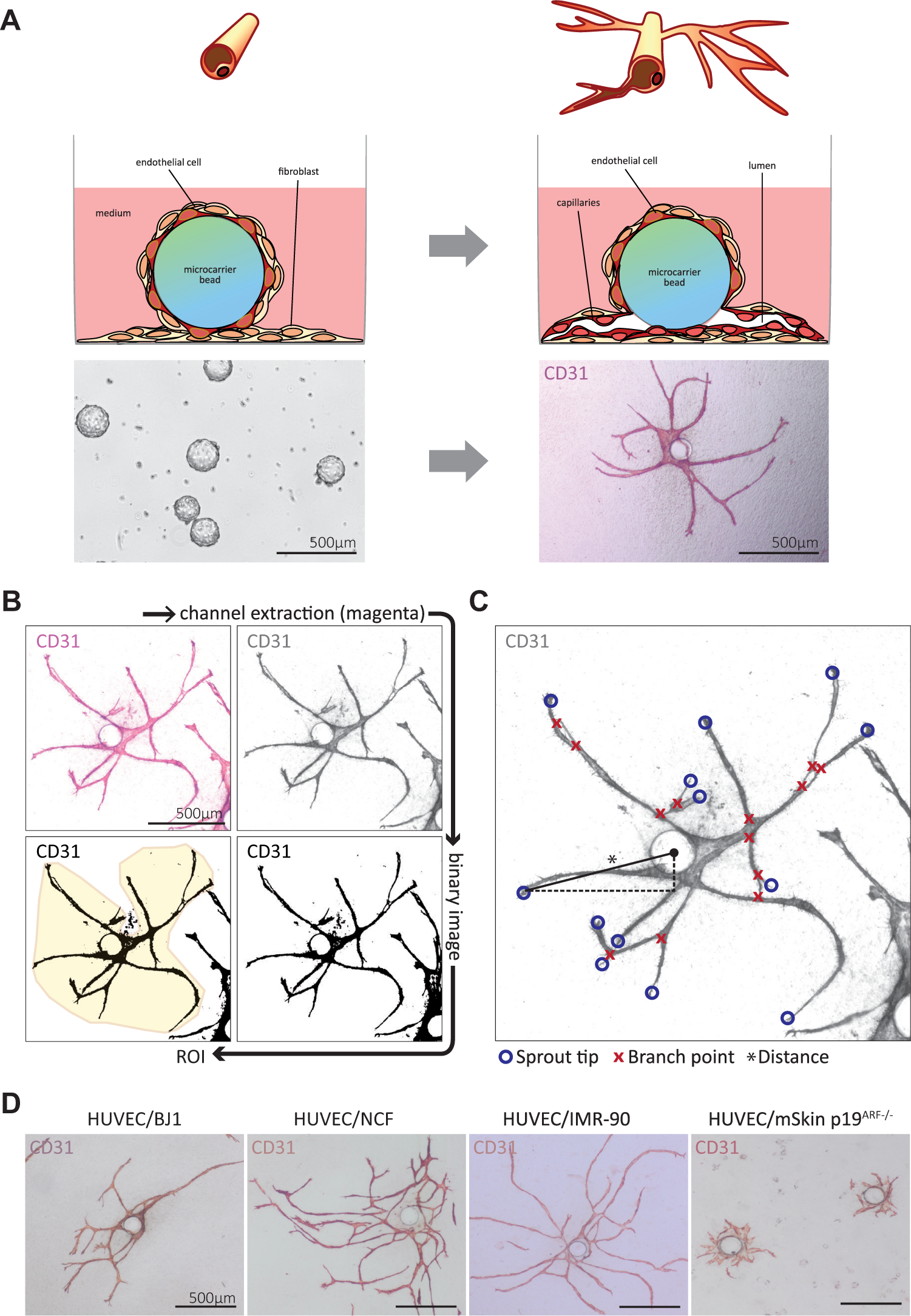
Setup and quantitative assessment of the novel angiogenesis assay. (
Testing of Pro- and Angiogenic Agents in the Angiogenesis Coculture Model
Cocultures with NCFs were treated with 10 ng/mL of recombinant human-VEGF165 (no. 100-20, Peprotech, Rocky Hill, NJ), 10 ng/mL of recombinant human-b-FGF (no. 100-18B, Peprotech), 500 ng/mL bevacizumab (Avastin, PZN 03159646, batch B8502H30, Roche, Basel, Switzerland), or 25 nM of nintedanib (no. S1010, Selleckchem, Houston, TX). Standard medium containing fresh factors/inhibitors was exchanged every other day.
Immunofluorescence and Immunohistochemistry Staining
EC sprouts or ECM components were visualized by immunofluorescence or immunohistochemistry (IHC) staining. EC–fibroblast cocultures were fixed with Roti-Histofix 4% (Roth, Karlsruhe, Germany) for 15 min at room temperature and afterwards permeabilized and blocked in 1× phosphate-buffered saline (PBS)/0.5% Tween 20/1% bovine serum albumin (BSA) (no. BE-17-512F, Lonza/no. A1389.1000, Applichem, Darmstadt, Germany/Albumin Fraktion V pH 7.0; no. A1391.0100, Applichem). The cocultures were incubated with primary antibodies against CD31 at a dilution of 1:500 (JC70A, no. M0823, Dako, Glostrup, Denmark), laminin B1 at 1:500 (LT3, no. GTX72366, GeneTex Inc., Irvine, CA), and collagen IV at 1:700 (no. ab6586, Abcam, Cambridge, UK) and visualized with Alexa Fluor 488 and 647 secondary antibodies (nos. A11034 and A31571, Life Technologies) at a dilution of 1:500. DAPI (1 µg/mL; no. 32670, Sigma-Aldrich, St. Louis, MO) served as a nucleic counterstain. Confocal fluorescence images were recorded on a Leica-SP8 with a 20× immersion objective (NA = 1.3). IHC staining of angiogenic sprouting was conducted using the same primary anti-CD31 antibody, which was further incubated with a biotinylated anti-mouse secondary antibody (no. BA-2000, Vector Laboratories, Burlingame, CA) and a streptavidin-conjugated horseradish peroxidase reagent (no. RE7104-CE, Leica Biosystems, Nussloch, Germany). The reaction was visualized using an AEC+ substrate chromogen (no. K3461, Dako). For bright-field or epifluorescence image capturing, an Olympus IX51 microscope (Olympus GmbH, Hamburg, Germany) and an Olympus camera XC50 were used. IHC overview images were taken using a CL 1500 ECO stereomicroscope (Carl Zeiss, Oberkochen, Germany) and a ToupCam camera (UCMOS, ToupTek Photonics, Zhejiang, China).
Image Processing and Analysis of Angiogenic Parameters
Angiogenesis was quantified by measuring the EC structure area, the number of sprout tips and branch points, and the distance of the individual sprout’s tips to the center of the bead. Before semiautomated analysis using ImageJ (Version 1.42q, U.S. National Institutes of Health, Bethesda, MD), all pictures were processed in Adobe Photoshop CS6 (Version 13.0.6, Adobe Systems Incorporated, San José, CA), in order to extract the magenta channel. The magenta channel was converted into a binary in ImageJ, and EC structure area was measured by marking the region of interest (ROI) with a polygon to ensure that only the areas of structures emerging from one single bead were measured. A micrometer scale was set using a picture containing a scale bar with a known distance. The number of sprouts and branch points were counted using ImageJ’s plugin “Cell Counter.” With this tool, the center of the bead and the position of the sprout tips were also marked. The coordinates of these points were used to measure the distance from sprout tips to the center of the bead as an approximation for sprout length by determining displacement vectors from the bead center to the individual sprout tips. For a graphical representation of the image processing and analysis workflow, see Figure 1B , C .
siRNA-Mediated Knockdown
To silence VEGF-A and angiogenin (ANG) expression in NCFs, siRNA-mediated knockdown was conducted as described in Rosner et al. 22 using Lipofectamine RNAiMAX Transfection Reagent (no. 13778075, Life Technologies) and SMARTpool ON-TARGETplus siRNAs (VEGF-A siRNA, no. L-003550-00-0005; ANG siRNA, no. L-011206-00-0005; Dharmacon, Lafayette, CO). Cells transfected with ON-TARGETplus Non-targeting Pool (no. D-001810-10-05, Dharmacon) served as control.
Western Blot
Total cell lysates of NCF-NTC, NCF-siANG, and NCF-siVEGFA were prepared by cell lysis in radioimmunoprecipitation assay (RIPA) buffer. Protein lysates were separated on a 12.5% sodium dodecyl sulfate–polyacrylamide gel electrophoresis (SDS-PAGE) gel and blotted onto a nitrocellulose membrane. The membrane was probed with primary antibodies against ANG (C-1, no. sc-74528, Santa Cruz Biotechnology, Dallas, TX) at a dilution of 1:500 and VEGF-A (no. ab46154, Abcam) at a dilution of 1:1000, followed by incubation with horseradish peroxide (HRP)–conjugated secondary antibodies (nos. A120-101P and A90-116P, Bethyl Laboratories, Montgomery, TX) at 1:10,000. α-Tubulin (DM1A, no. CP06, Merck Millipore, Darmstadt, Germany) at 1:1000 served as loading control. Chemiluminescence signals were detected using Pierce ECL Western Blotting Substrate (no. 32209, Thermo Fisher Scientific, Waltham, MA) and developed in a Kodak Medical X-Ray Processor 2000 (Eastman Kodak Company, Rochester, NY).
Statistical Analysis
Data were collected in Microsoft Excel 2013 and further analyzed in GraphPad Prism. Comparisons between differentially treated, or genetically modified groups and control groups were calculated using a t test for independent samples (two tailed, unpaired) provided the data were normally distributed. Data not following a Gaussian distribution were compared with a nonparametric Mann–Whitney u test. p values of ≤0.05 were considered statistically significant. The data were graphically displayed in dot plots with the median depicted for the sprout tip’s distance to the bead center, and box and whisker plots for all other parameters. The box represents the interquartile range (IQR) between the 25th and 75th percentiles, and the horizontal line indicates the median; the ends of the whiskers display 1.5 times the IQR. In all statistics of the angiogenesis assay, only data of single beads were used. Beads, whose structures were intersecting each other, were excluded from all analyses. A minimum of 10 beads was analyzed per condition to rule out differences due to the varying size of the microcarrier beads.
Results
Optimization of Culture Conditions for a Novel 3D Angiogenesis Coculture Model
As angiogenesis implies sprouting and branching from existing vessels to form new ones, the main parameters to quantify the angiogenic response are sprout number, sprout length, the number of branch points, and vessel density emanating from preexisting vessels.
17
In the fibroblast coculture assay published previously,
18
EC structures can be counted and the length of single sprouts can be determined, but no information about vessel outgrowth and branching capability from preexisting structures can be achieved, as there is no initial structure from where angiogenic sprouting is started. In the fibrin gel bead assay
19
with the EC-covered microcarrier bead, an equivalent to an established vessel in vivo is provided. However, the setup of the model is technically demanding. To gain more information on the influence of direct cell–cell contact on angiogenesis and to facilitate data acquisition and analysis, we combined the major advantages of these two assays and cocultured HUVECs attached to microcarrier beads with hTERT-immortalized skin fibroblasts (BJ1) to set up a novel modified 3D angiogenesis model. These genetically engineered fibroblasts were used for initial optimization of the assay. A schematic representation of the assay is represented in
Figure 1A
. Endothelial structures were visualized by staining the EC marker CD31. First, different growth media were tested to select the optimal medium composition for supporting angiogenic sprouting in this system. When cocultures were kept in DMEM/10% FBS, HUVECs formed angiogenic sprouts. Fibroblast-derived signals were sufficient for HUVECs to form vessels from their starting point, the EC-covered microcarrier bead; however, the development of widely ramified networks was missing. Addition of 10% complete EC growth medium (EGM-2-MV) containing VEGF/b-FGF/EGF/IGF1 to DMEM/10% FBS enhanced EC structure area formation by a factor of 19.5 (p < 0.001), as well as increased branching of the structures (
Next, we demonstrated the suitability of fibroblasts derived from diverse other human organs to induce formation of vessel-like structures in coculture with HUVECs. Lung fibroblasts (IMR-90) and NCFs were even more potent to induce angiogenic structures (
Figure 1D
) than the immortalized skin fibroblasts. Thus, cultivation time could be reduced to 5 or 7 days, with NCFs cocultured with HUVECs as they supported EC sprouting more efficiently (
EC Networks Display Lumen Formation and the Establishment of a Basement Membrane Surrounding the Vessel-Like Structures
Z-stack analysis by confocal microscopy verified the presence of lumina within the EC network ( Fig. 2A ) in the 3D angiogenesis assay, as it was already shown.18,23 Tubular structures emerged from the bead and display further branching. The lumen was devoid of cellular material, as demonstrated by the absence of nuclei. DAPI counterstaining revealed the position of the surrounding fibroblasts, demonstrating that the EC structures were embedded in several dense layers of fibroblasts. In orthogonal views of Z-stack images at a higher magnification, lumen formation within the EC networks was evident in vertical and horizontal sections ( Fig. 2B ). Nuclei were recognizable in the walls of the tube-like HUVEC structures. The cross section diameter of this vessel-like tubular structure was about 20 µm, big enough for human erythrocytes (approximate size of 7.5 µm) to pass through, further supporting the physiological relevance of the novel angiogenesis model. Confocal analysis of endothelial cocultures with NCF stained for laminin B1 and collagen IV revealed that capillary-like sprouts were surrounded by a basal membrane ( Fig. 2C ). EC tubules were embedded in laminin, and the staining was restricted to the tubular structures. In contrast, collagen IV showed a broader distribution not only encasing EC tubes, but also being efficiently expressed by the fibroblasts.
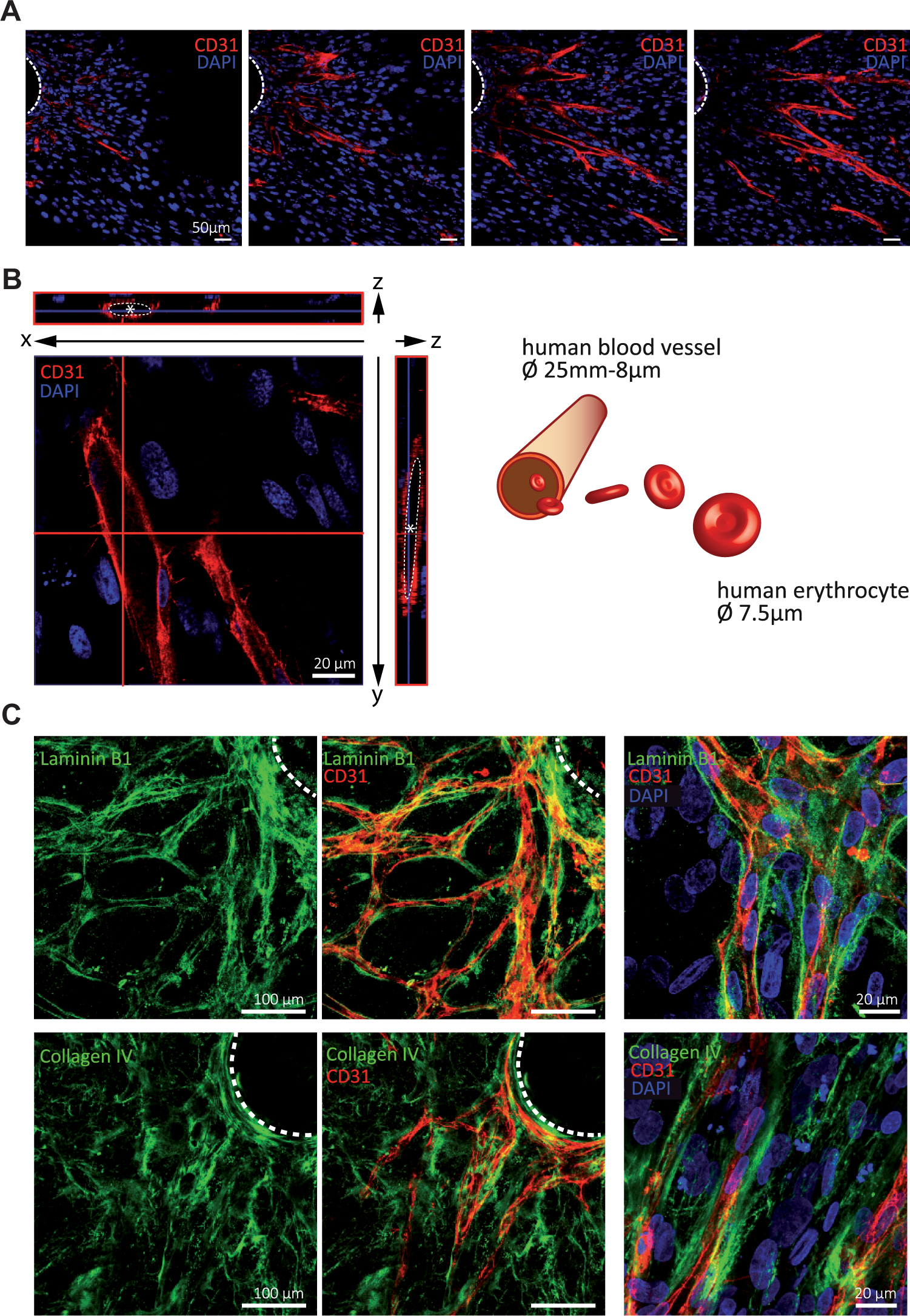
Endothelial structures form 3D tubular networks surrounded by a basement membrane in the fibroblast coculture angiogenesis assay. (
Knockdown of Angiogenin or VEGF-A in Fibroblasts Diminishes Vessel Formation
Apart from getting insight into the molecular mechanisms of angiogenesis, another major advantage of in vitro angiogenesis assays is to evaluate stimulating or inhibitory signaling molecules or compounds for their possible impact on vessel formation. Thus, the function of angiogenic growth factors provided by stromal fibroblasts can be evaluated by genetically silencing their expression in these cells. In a proof-of-principle experiment, we performed a siRNA-mediated knockdown of the potent angiogenic growth factor VEGF-A or ANG in NCF (NCF-siVEGFA, NCF-siANG) and evaluated the angiogenic response when these cells were cocultured with HUVEC-coated beads. After 7 days of cultivation in angiogenesis standard medium, cocultures were fixed and IHC stained for the EC marker CD31 ( Fig. 3A ). Angiogenic parameters were analyzed as described before (see also Material and Methods). Indeed, knockdown of VEGF-A or ANG ( Fig. 3B ) led to a significant reduction of all four angiogenic parameters analyzed. More specifically, silencing ANG decreased the vessel area and mean distance to bead center 1.4-fold and the number of sprouts and branch points by a factor of 1.9 and 2.4, respectively. Knockdown of VEGF-A led to a twofold reduction of EC structure areas. Regarding sprouts and branch points, the numbers were decreased 2.3- and 3.3-fold, whereas the sprout tip distance to bead center was only reduced by 1.3-fold ( Fig. 3C ).
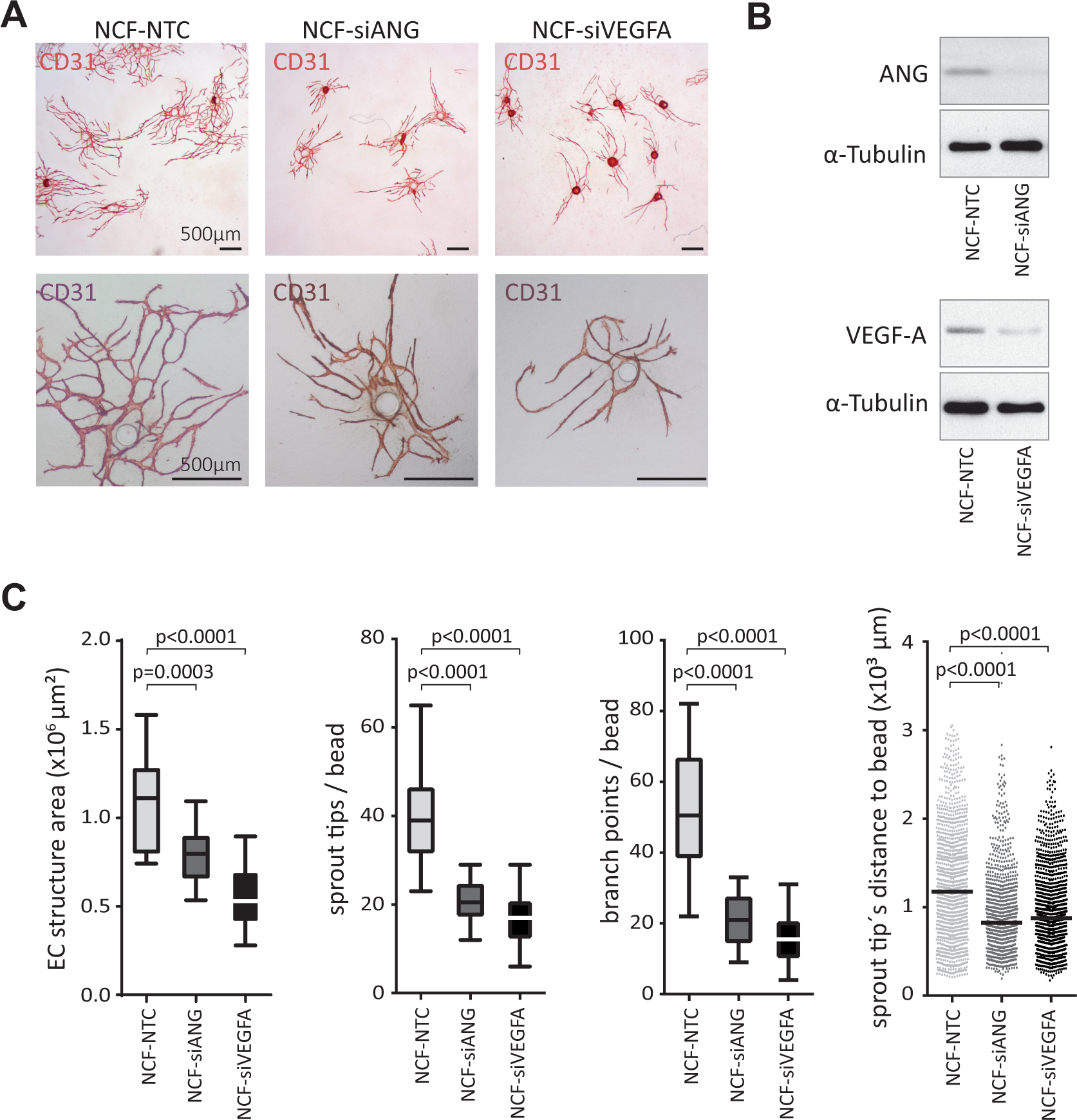
siRNA-mediated knockdown of angiogenic growth factors VEGF-A and ANG in fibroblasts decreased angiogenesis in vitro. (
Pro- and Antiangiogenic Factors Display the Anticipated Induction/Reduction of Angiogenesis in the Coculture Assay
Finally, we aimed to demonstrate that the modified 3D angiogenesis coculture assay is a suitable model for testing and screening of both pro- and antiangiogenic agents in a 24-well plate setting. In order to determine whether the angiogenic process can be further induced in our model, the potent pro-angiogenic growth factors basic FGF (b-FGF) and VEGF165, the most abundant form of VEGF-A, were applied to HUVEC cocultures with NCF at a concentration of 10 ng/mL. For inhibiting angiogenesis, 500 ng/mL of bevacizumab (Avastin) or 25 nM nintedanib was added to standard angiogenesis medium. Bevacizumab is a Food and Drug Administration (FDA)–approved humanized monoclonal antibody against VEGF-A for treatment of several cancers. Nintedanib is a triple tyrosine kinase inhibitor that simultaneously blocks the receptor tyrosine kinase activities of VEGF, PDGF, and FGF receptors. In the clinics, nintedanib is used as antiangiogenic medication for lung cancer patients and for treatment of idiopathic pulmonary fibrosis. In representative pictures of the EC structures formed under the different cultivation conditions, the enhancing and inhibitory effects of the admixed agents on angiogenic sprouting are evident (
Fig. 4A
). Quantitative analysis revealed that in all four angiogenesis parameters, a significant increase was achieved by adding either b-FGF or VEGF165, which displayed a more pronounced stimulatory effect. EC structure areas were 2.6-fold increased when medium was supplemented with VEGF165 and 2.2-fold with b-FGF. Sprout numbers and the numbers of branch points were induced 2- and 2.6-fold, respectively, by addition of VEGF165, or 2.1- and 1.4-fold when b-FGF was added. Mean distance from sprout tips to the bead center was increased by a factor of 1.2 and 1.3 when treated with VEGF165 and b-FGF compared with the controls. Bevacizumab significantly inhibited vessel density, sprouting, and branching ability, as well as vessel outgrowth compared with controls by 2.2-, 2.8-, 4-, and 1.3-fold. Treatment with the triple angiokinase inhibitor nintedanib completely abrogated angiogenic sprouting in the cocultures at low nanomolar concentration (25 nM) (
Fig. 4B
). However, although this inhibitor also has an effect on PDGF and FGF receptor signaling in fibroblasts, they did not differ in their morphology or density compared with those in the other conditions (
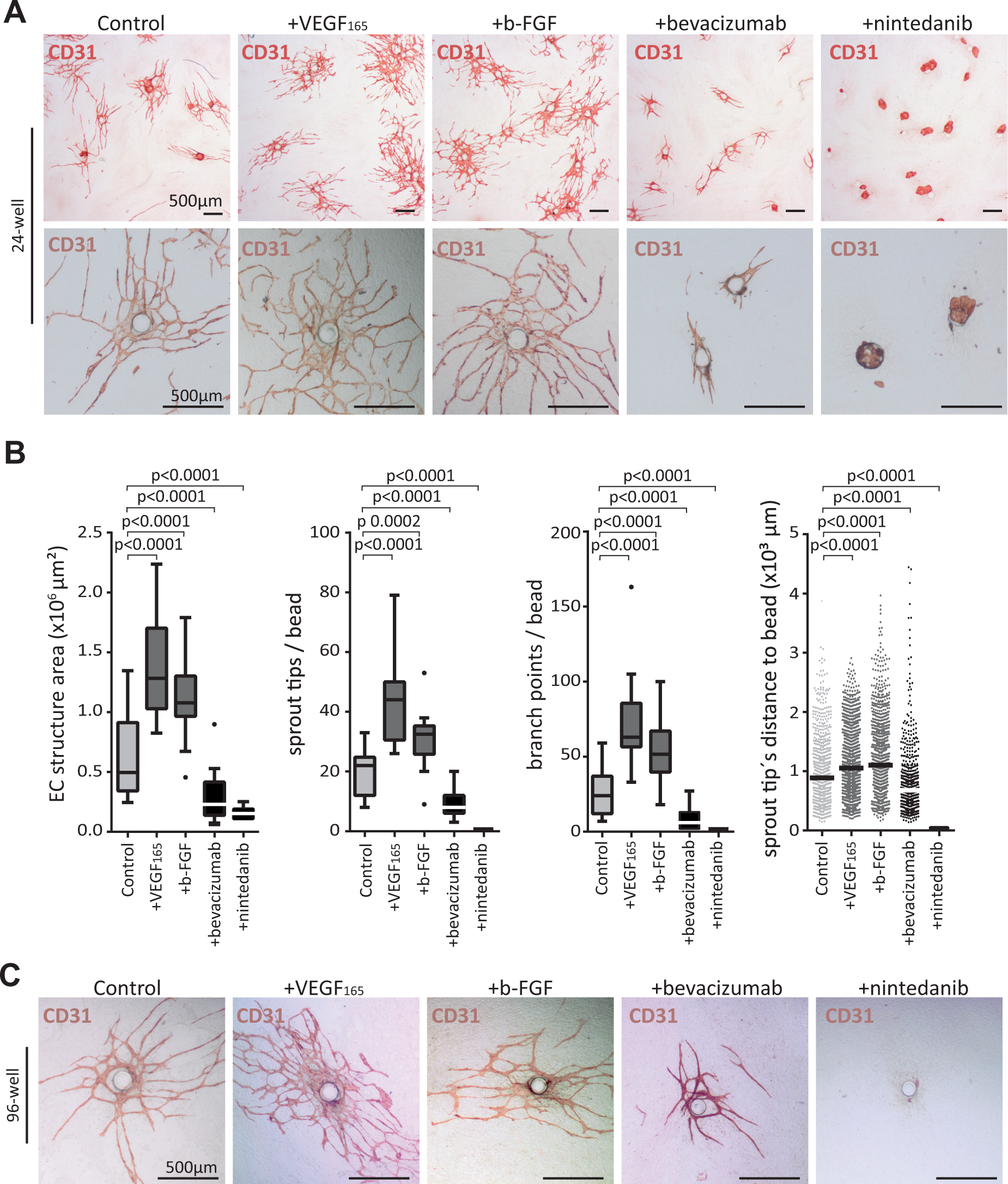
Pro-/antiangiogenic agents increase/decrease sprouting in the microcarrier–fibroblast coculture assay. In 24-well plates, cocultures with NCF were treated with either 10 ng/mL of VEGF165 or 10 ng/mL of b-FGF, or with the antiangiogenic substance bevacizumab at 500 ng/mL or nintedanib at 25 nM, to demonstrate the applicability of the assay to screen for pro-/antiangiogenic agents. Factors or compounds were freshly added with every medium exchange every other day, and cocultures were propagated for 7 days. (
Discussion
Despite moderate efficacy of first-generation angiogenesis inhibitors and pro-angiogenic medication, targeting angiogenesis is still a promising therapeutic concept with high potential. It turned out that the lack of efficiency was mainly due to the acquisition of intrinsic resistance mechanisms, as well as decreased drug responsiveness due to the interaction with the microenvironment.8,13 Hence, novel preclinical models are needed that mimic the increased complexity present in vivo. Studying 3D models employing other stromal cells than ECs alone will help to shed more light on the molecular mechanisms of angiogenesis with a special focus on heterotypic interactions.
Second, a robust, objective, and quantitative description of angiogenic response is of importance to reliably evaluate even subtle changes in case of functional interference. This might be depletion of a gene of interest in any of the involved cell compartments or testing inhibitory or stimulatory drugs. As angiogenesis is defined as the expansion of the vascular network by sprouting and branching from preexisting vessels, the main characteristics for assaying angiogenesis are the number and length of sprouts, the number of branch points and anastomoses, and the vessel density (EC structure area).1,17
We employed a strategy to most closely mimic sprouting angiogenesis and to quantify the above-mentioned parameters accurately. For this, the advantageous parts of two existing angiogenesis assays were combined. The preexisting vessel is simulated by EC-covered microbeads as established by Nehls and Drenckhahn, 19 and the interaction of ECs with stromal fibroblasts is mediated by placing the EC beads within very dense fibroblast cultures. Here, we further report that fibroblasts of different origin can be used, demonstrating the general feasibility of the assay to mimic angiogenesis in different tissues or organs and even organisms (i.e., the use of mouse fibroblasts for HUVEC sprouting analysis). EC–fibroblast cocultures were first described by Bishop et al., 18 and vessel-like structures were emanating over time, which can be counted, and the length of the sprouts and overall vessel area can be measured. However, sprout number, vessel outgrowth, and branching capability from preexisting structures cannot be determined since an initial structure from where the angiogenic process started is missing. Moreover, as single cells are seeded, this assay might rather imitate the process of neovascularization. Consequently, the novel assay employing the EC-covered beads better resembles angiogenesis.
EC spheroids might be an alternative to microbeads. However, they tend to disintegrate with prolonged culture period, making it difficult to identify the position of the initial structure. This does not happen with beads, since they stay intact and the origin of the angiogenic sprouts can be exactly defined.
For EC–fibroblast cocultures, it was recommended to preplate fibroblasts a few days before adding the ECs. 24 However, as we seeded EC-coated microcarriers simultaneously with fibroblasts, there was no detectable difference to preseeding (data not shown). This minimized workload and incubation time. The simultaneous seeding might even be advantageous in this model, as the entire bead is covered with fibroblasts from the beginning.
Compared with the fibrin gel bead assay,19,21 in which EC-covered microcarrier beads are embedded in a fibrin gel droplet and form angiogenic structures, analysis is facilitated in the cocultivation assay due to topologic reasons. The EC structures emerge in a 3D matrix provided by very dense layers of fibroblasts. However, the z dimension is kept to a minimum compared with the fibrin gels. Thus, all structures appear in one level and can be assessed with a conventional inverted microscope without the need of sophisticated high-content imaging techniques. Apart from that, the structures grow substantially big and form highly branched and anastomosing EC structures, closely mimicking the appearance of vascular networks found in vivo. Remarkably, sprouts reach lengths of 600–700 µm in the fibrin gel assay under optimal conditions, 25 whereas sprout lengths up to 3 mm were achieved in the EC microbead coculture assay. EC networks were covered by a basal membrane. The formation of tubular structures, exhibiting cell-free lumen, was demonstrated by Z-stack confocal imaging, as reported earlier.18,23 The lumen diameter was big enough to theoretically carry human erythrocytes underscoring the close recapitulation of vessels in vivo.
The descriptive characterization validated the assay to recapitulate essential features of sprouting angiogenesis in vivo. However, the value of every biological in vitro model is considerably increased by easy functional interference for hypotheses testing. First, in proof-of-concept studies, we showed that angiogenic sprouting could be manipulated by genetic modification of stromal fibroblasts, as demonstrated earlier. 26 We selected two bona fide pro-angiogenic molecules (ANG and VEGF-A), whose depletion in the fibroblasts significantly decreased sprouting angiogenesis. Interestingly, VEGF-A knockdown in the fibroblasts led to a reduction of angiogenesis despite the presence of recombinant VEGF in the culture medium. The recombinant protein could not fully restore angiogenesis, which might be due to either the low amount of VEGF in the standard angiogenesis medium or the need for high local concentrations at the fibroblast–EC interface.
Thus, via siRNA/shRNA-mediated knockdown or CRISPR/Cas9 knockout technology, specific candidate targets can be silenced, and consequently, their effect on sprouting and branching abilities can be evaluated. Moreover, cells isolated from genetically modified animals could be subjected to the angiogenesis assay since we demonstrated that mouse cells were also able to support angiogenesis in vitro. As fibroblasts derived from various tissues of the human (and murine) body were shown to work in the described experimental setup, the assay offers the possibility to investigate the angiogenic process in a context-specific manner. This is of interest not only for further delineating the context-related mechanisms or the response to a specific drug, but also for examining the microenvironmental influence on the activity of possible novel targets. Of note, so far only HUVECs were used as an EC source, in order not to modulate too many parameters at once in the model. However, angiogenesis depends on changes in the microvasculature; thus, it would be more physiologic to use microvascular ECs for in vitro assays. The applicability of microvascular ECs for fibroblast-mediated vessel formation has already been shown. 23 Therefore, there is clear need to expand our studies to ECs isolated from desired target tissues, matching the source of the fibroblasts. From an academic point of view, it will be interesting if these organ-specific assays would alter the angiogenic capacity. If this would be the case, the determination of drug response in tissue-matched EC–fibroblast cocultures would be important. Taken together, the assay might be a powerful tool to further investigate candidate signaling pathways or to perform even genome-wide knockdown/-out screens for identifying novel molecules involved in EC–fibroblast cell interaction–dependent angiogenesis.
A major task of the newly established angiogenesis assay was to set up a robust model to evaluate substances or signaling molecules for their possible impact on angiogenesis and to provide an effective cellular screening platform. In proof-of-concept studies, angiogenesis could be effectively inhibited by two clinically approved antiangiogenesis drugs. The VEGF neutralizing monoclonal antibody bevacizumab (500 ng/mL) and the triple angiokinase inhibitor nintedanib (25 nM) were effectively inhibiting angiogenesis in our system. These FDA-approved drugs were chosen because they proved to work in man.27,28 The complete inhibition of angiogenesis by nintedanib might be due to the simultaneous action of the inhibitor on ECs and fibroblasts.29,30 It is tempting to speculate that despite there being no obvious phenotypical change detectable, nintedanib might cause a change of the fibroblast secretome and ECM production, leading to the effective block of vessel formation. This hypothesis is readily testable in our novel angiogenesis model.
On the other hand, the pro-angiogenic substances VEGF-A and b-FGF augmented vessel formation under standard assay conditions. We speculate that the effects of angiogenesis-inducing agents might be even more pronounced when tested under suboptimal assay conditions. For example, we identified media formulations with less profound pro-angiogenic effects during the course of fine-tuning the assay, which might be selected to test potential angiogenesis inducers. Finally, the 3D coculture method could be successfully adapted to a 96-well format for drug screening purposes and yield equally well-established vascular structures, which might be automatically analyzed by advanced imaging analysis software.
Footnotes
Acknowledgements
We would like to thank Dr. Wolfgang Mikulits for providing the p19ARF–/– mice.
Supplementary material is available online with this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Herzfelder Family Foundation.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
