Abstract
Oral and intestinal mucositis is a debilitating side effect of radiation treatment. A mouse model of radiation-induced mucositis leads to weight loss and tissue damage, reflecting the human ailment as it responds to keratinocyte growth factor (KGF), the standard-of-care treatment. Cultured intestinal crypt organoids allowed the development of an assay monitoring the effect of treatments of intestinal epithelium to radiation-induced damage. This in vitro assay resembles the mouse model as KGF and roof plate-specific spondin-1 (RSPO1) enhanced crypt organoid recovery following radiation. Screening identified compounds that increased the survival of organoids postradiation. Testing of these compounds revealed that the organoids changed their responses over time. Unbiased transcriptome analysis was performed on crypt organoid cultures at various time points in culture to investigate this adaptive behavior. A number of genes and pathways were found to be modulated over time, providing a rationale for the altered sensitivity of the organoid cultures. This report describes an in vitro assay that reflects aspects of human disease. The assay was used to identify bioactive compounds, which served as probes to interrogate the biology of crypt organoids over prolonged culture. The pathways that are changing over time may offer potential targets for treatment of mucositis.
Introduction
Mucositis is a clinical condition caused by inflammation and ulceration of the epithelial lining of the intestinal tract and is a common side effect of radiation and chemotherapy used to treat certain malignancies (with a prevalence between 10% and 100% depending on the cancer treatment). 1
Recently, a number of in vitro organoid systems, derived from tissue-resident stem cells, have been described for intestine, stomach, kidney, liver, pancreas, and mammary glands.2–7 Continuously dividing stem cells within the crypt compartment of the intestine are responsible for the self-renewal capacity of the intestinal epithelium. These stem cells divide and provide descendants that differentiate into the four specialized intestinal cell types: enterocytes, goblet, enteroendocrine, and Paneth cells.6,8–10 Such in vitro culture systems have been shown to faithfully recapitulate hallmarks of tissue homeostasis and disease. Moreover, in vitro cultivated colon organoids were able to functionally engraft into superficially damaged mouse colon. 11
Although the small intestine has been used as a means to monitor a number of biological responses to pathophysiological insults or treatments in vivo, the in vitro intestinal crypt organoids were used as model systems in only a limited number of screening applications. One of these models made use of organoids derived from CFTR patients to screen for compounds that would help rectify the CFTR channel dysfunction. 12 This model is limited by the need to monitor the response of individual organoids to compound and challenge, making such an assay system difficult to implement when screening large compound libraries that are arrayed in a plate format. Other screening applications included modeling of the infection of organoid cultures by Salmonella and Helicobacter pylori13,14 or monitoring the effect of compounds on the differentiation of different cell types in the intestinal epithelium. 15
This report describes the miniaturization of culture conditions for intestinal organoids into a 384-well format aimed to measure cell viability. This assay was used to monitor the effect of compound treatment following irradiation of the intestinal crypt organoids, which resulted in the identification of small molecules that increased the recovery of intestinal crypt organoids following irradiation. Surprisingly, these tool compounds showed an altered efficacy after repeated passaging of the intestinal crypt organoids and vastly different effects on two-dimensional (2D) cell cultures exposed to radiation. Gene expression profiling was used to characterize the transcriptional changes that occurred in crypt organoid cultures over time. A number of gene expression changes were identified that could help explain these differences and also be used to identify targets that modulate radiation sensitivity.
Materials and Methods
Tissue Culture Reagents
Components for organoid culture medium (Advanced DMEM/ F-12 containing penicillin, streptomycin, L-glutamine, gentamycin, 75 ng/mL Fc-RSPO1, 50 ng/mL epidermal growth factor [EGF], and 100 ng/mL Noggin) were purchased from the following providers: Advanced DMEM/F-12, penicillin, streptomycin, L-glutamine, and gentamycin were all purchased from Gibco Invitrogen (Carlsbad, CA). EGF and mouse Noggin were purchased from Peprotech (Rocky Hill, NJ), and recombinant human Fc-tagged RSPO1 was prepared in house. 16 EDTA was purchased from Fluka (St. Louis, MO), Matrigel was purchased from Corning (Corning, NY), and Versene and phosphate-buffered saline were purchased from Gibco Invitrogen. HEK293T cell culture medium was composed of DMEM, high-glucose, 10% fetal bovine serum, pyruvate, penicillin, streptomycin, and GlutaMAX (all purchased from Gibco Invitrogen).
Isolation of Small Intestinal Crypts
A detailed description of the protocol used for the isolation of mouse intestinal crypts is given in the supplemental materials and methods. Briefly, small intestines from C57Bl/6 mice were dissected and washed, and fat was removed before the intestine was cut into small pieces, which were again washed. The intestine was then incubated with 2-mM EDTA for 30 min on ice before villi were released by agitation. Villi were further released from the intestine by repeated washing using 2-mM EDTA. Subsequently, crypts were released from the intestine by strong agitation in 2-mM EDTA and separated from intestine fragments using a 70 µm cell strainer (BD Falcon). The purified crypts were collected on a 40 µm cell strainer (BD Falcon) and washed with organoid culture medium without RSPO1, EGF, and Noggin. The crypts were flushed off from the 40 µm filter using organoid culture medium without RSPO1, EGF, and Noggin and pelleted by centrifugation at 1800 g for 5 min. Finally, the pellet was carefully resuspended in 3 mL Matrigel, and 1.5 mL Matrigel-crypt mix was plated per 6 cm dish. After the Matrigel was allowed to solidify in a 37 °C incubator for 15 min, 5 mL organoid culture medium was added for further incubation.
Growth and Passaging of Small Intestinal Crypt Organoids
A detailed description of growth and passaging of the small intestinal crypt organoids is given in the supplemental materials and methods. It usually took 2 weeks for the crypts to recover at first, but then organoids could be passaged once a week, diluting the culture 1:3.
Screening of Annotated Compound Sets
Crypt organoids were individually collected by centrifugation from ten 6 cm dishes as when organoids were passaged. The pelleted organoids were irradiated with 6 Gy using a BIOBEAM 8000 (Gamma-Service Medical GmbH, Leipzig, Germany). Each pellet was resuspended in 4.5 mL Matrigel. Ten microliters of this organoid/Matrigel mix was transferred into all wells of a 384-well plate using a handheld Matrix multichannel pipette, and the plates were briefly centrifuged to distribute the material to the bottom of the plate. The plates were placed in an incubator for 15 min to let the Matrigel solidify. Fifty microliters of organoid medium was added to each well. Using the Echo 550 (Labcyte, Sunnyvale, CA), 15-nL of compound (10-mM stock solution in 100% DMSO) was transferred to each well, yielding a final compound concentration of 2.5 µM. Plates were incubated at 5% CO2, 37 °C, and 95% humidity for 7 days with media changes on day 2 and day 5. On day 7, 5 µL tetrazolium dye solution was added to all wells, and fluorescence signals were measured after 4 h of incubation. Results were transferred to an Excel sheet and analyzed using Spotfire (Tibco, Palo Alto, CA).
Preparation of mRNA from the Crypt Organoid Samples
RNA samples were extracted from crypt organoids using Trizol and column-purified using a Direct-zol TN RNA MiniPrep kit (Zymo Research, Irvine, CA). A detailed description of this preparation is given in the supplemental materials and methods.
mRNA Expression Analysis
Details of the microarray and next-generation sequencing experiments monitoring the effect of keratinocyte growth factor (KGF) and long-term cultivation of the crypt organoids, respectively, are given in the supplemental materials and methods.
Quantitative PCR Gene Expression Analysis
RNA was isolated from untreated and KGF-treated crypt organoid cultures and purified using the RNeasy Plus Mini kit (Qiagen, Hilden, Germany). After the RNA concentration and quality were assessed, cDNA was prepared using the High Capacity cDNA Reverse Transcription Kit (Applied Biosystems, Foster City, CA). The TaqMan PCR protocol (Applied Biosystems) was used for quantitative PCR (qPCR) analyses, all cDNA samples were analyzed in triplicates using the TaqMan PCR kit (Applied Biosystems), and ΔΔCt values were calculated by normalizing to Gapdh.
Gene Set Enrichment Analysis
Gene set enrichment analyses were obtained using Python and R scripts that conduct a series of weighted Kolmogorov-Smirnov tests for the contrast of interest. 17 Gene sets were tested based on their average fold change, and the obtained p values were adjusted using the Benjamini-Hochberg method. Gene sets were then ranked based on the negative log-adjusted p values. Enrichment of genes up- and down-regulated upon KGF treatment in crypt organoids was tested for separately, followed by a manual inspection of the top-ranking gene sets. The gene sets used for this analysis were part of the MetaBase collection (data licensed from ThomsonReuters).
Results
Establishment of an In Vitro Model of Radiation-Induced Mucositis
To identify molecules that protect intestinal epithelium from radiation damage, we established an in vitro model closely resembling the physiology of the mouse small intestine epithelium by adapting a published protocol. 6
Usually, the viability of the intestinal crypt organoids was monitored using the reduction of water-soluble tetrazolium dyes (such as resazurin or alamarBlue; typically, we used PrestoBlue). 18 As crypt organoid cultures did not display autofluorescence, a serial dilution of crypt organoids, plated into 96-well plates, was used to show that the reduction of tetrazolium dyes was proportional to the number of crypt organoids present. Plating of 30 to 35 crypt organoids per well yielded an optimal detection window while maintaining organoid viability (data not shown). The original publication describing this culture method used 500 ng/mL of RSPO1 in the culture medium. 6 We used an Fc-tagged version of RSPO1 that was available in house. 16 To determine the optimal concentration of this RSPO1 version to maintaining robust crypt growth while avoiding excessive Wnt pathway stimulation, a titration of this RSPO1 protein was conducted. Crypt organoids were cultivated for up to 7 days in the presence of increasing concentrations of the Fc-tagged RSPO1, and 75 ng/mL of RSPO1 proved to be the optimal concentration ( Fig. 1A ), supporting crypt organoid growth while maintaining a normal physical organoid appearance.
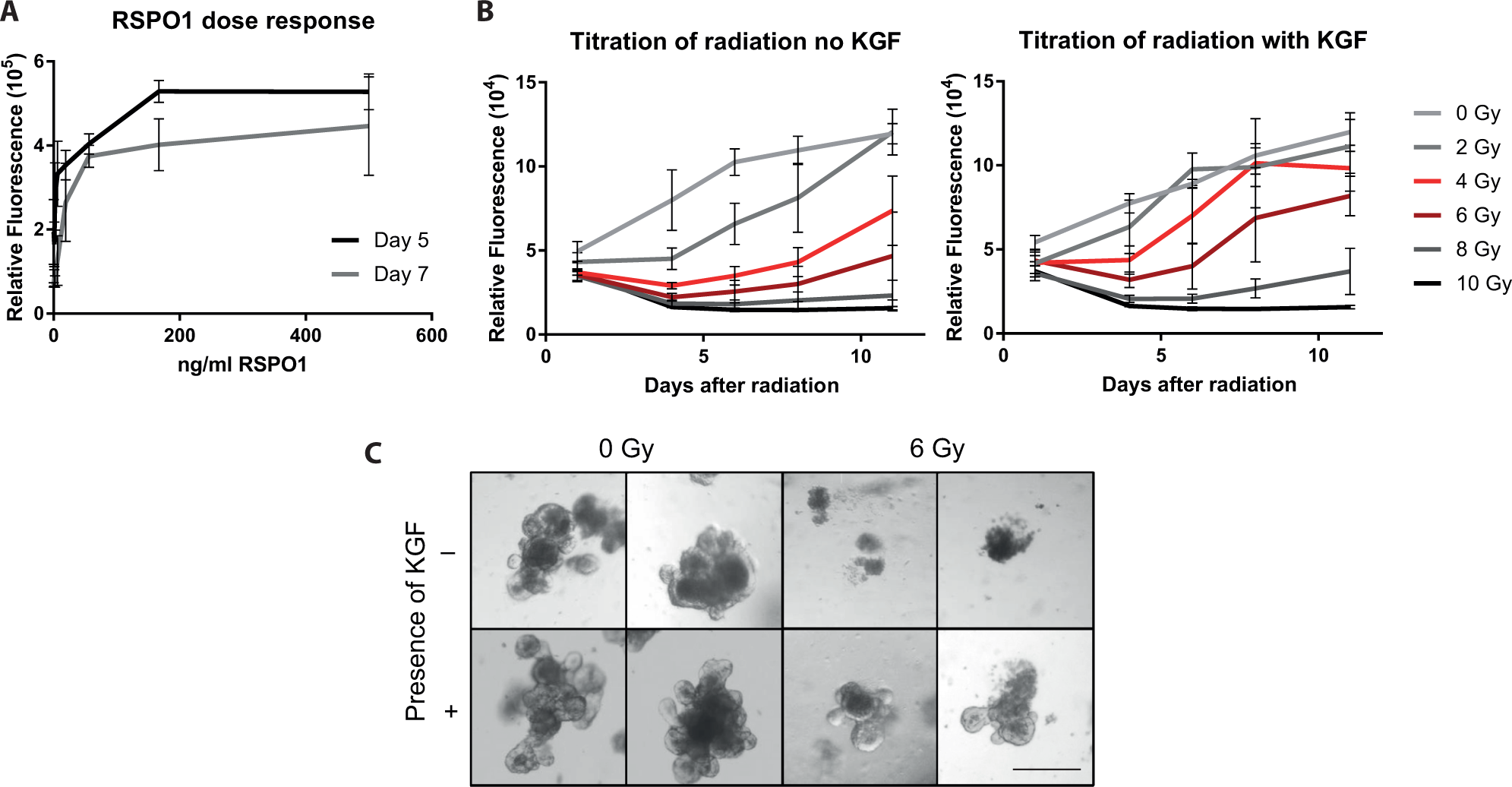
Setup of an in vitro organoid culture system for compound screening. (
To mimic damage induced by radiation therapy in patients, fresh cultures of the intestinal crypt organoids were subjected to different doses of gamma radiation (0–10 Gy), and proliferation/recovery was monitored for up to 11 days. Doses of 4 or 6 Gy initially decreased cell viability before cultures regained viability at ≥6 days; in contrast, nonirradiated cultures continued to proliferate immediately after plating (
Fig. 1B
). At lower radiation doses, the effect on viability was less pronounced, whereas organoid survival was barely or not achieved at higher doses. Because of the limited dynamic range of the assay, viability values typically reached a plateau 9 to 11 days postplating in nonirradiated cultures, whereas irradiated cultures reached this plateau about 2 weeks postradiation or even later (
Fig. 1B
;
To validate this assay system, we reasoned that an approved drug used for the treatment of radiation-induced mucositis should score positively. Palifermin (KGF) is the only approved therapy for the prevention of mucositis in patients undergoing hematology stem cell transplantation. 19 Therefore, KGF was tested in this assay system for up to 11 days. Nonirradiated cultures were analyzed in parallel with and without KGF treatment to determine the effect of radiation. Whereas irradiated, nontreated cultures lost viability until day 4 after irradiation before they regained viability thereafter in dependency of the radiation dose (4 and 6 Gy), the viability of KGF-treated cultures was much more pronounced a few days after irradiation, as evidenced by higher fluorescence values for KGF-treated cultures at 4 and 6 Gy on days 6, 8, and 11 ( Fig. 1B ). For example, the relative fluorescence values for cultures irradiated with 4 Gy in the absence of KGF were 30% and 56% on days 8 and 11, respectively, compared with nonirradiated cultures without KGF, whereas these values were 95% and 79% for the corresponding cultures treated with KGF. Similarly, cultures irradiated with 6 Gy in the absence of KGF were 16% and 30% on days 8 and 11, respectively, compared with nonirradiated cultures, whereas these values were 59% and 63% upon KGF treatment ( Fig. 1B ). The viability of nonirradiated cultures remained high following plating, irrespective of KGF treatment, demonstrating the robustness of the assay. Cell viability data correlated with microscopic assessment; that is, doses ≥6 Gy caused extensive cell death, whereas doses <6 Gy caused some cell death while retaining viable and proliferating crypt organoid structures. Treatment with KGF protected from cell death and supported the formation of normal crypt structures on the organoids ( Fig. 1C ). These results were confirmed using additional readout systems, such as the 3-[4,5-dimethylthiazol-2-yl]-2,5-diphenyl tetrazoliumbromide or CellTiter-Glo luminescent cell viability assay (Promega), as well as image-based readouts. Because of its reproducibility and simplicity, the tetrazolium reduction–based fluorescence readout was used for all subsequent experiments. In summary, these experiments revealed a partial protection of the organoid cultures by KGF following radiation in vitro, mirroring the therapeutic effect of palifermin, thus validating this assay system for the identification of bioactive molecules.
Cellular Pathways Contributing to KGF-Mediated Protection from Radiation-Induced Damage
Characterization of the gene expression changes in response to a known protective agent would highlight the molecular pathways and processes leading to radiation protection. Thus, we analyzed and compared the mRNA expression of sublethally irradiated (6 Gy) crypt organoid cultures that were treated with 250 ng/mL KGF to untreated, irradiated cultures. These treatments were made for 6 days because at this time point, untreated, irradiated organoid cultures started to recover, and hence, the effects of a protective agent on organoid viability would be most pronounced (
Fig. 1B
). mRNA was isolated from six replicate samples of each condition and subjected to microarray analysis.
Figure 2A
presents the differences in gene expression when comparing KGF-treated cultures with untreated cultures. Whereas most RNA species did not significantly change upon KGF treatment (–1.0 < log[fold change] < 1.0), the expression of some genes was increased (210 genes) or down-regulated (276 genes;
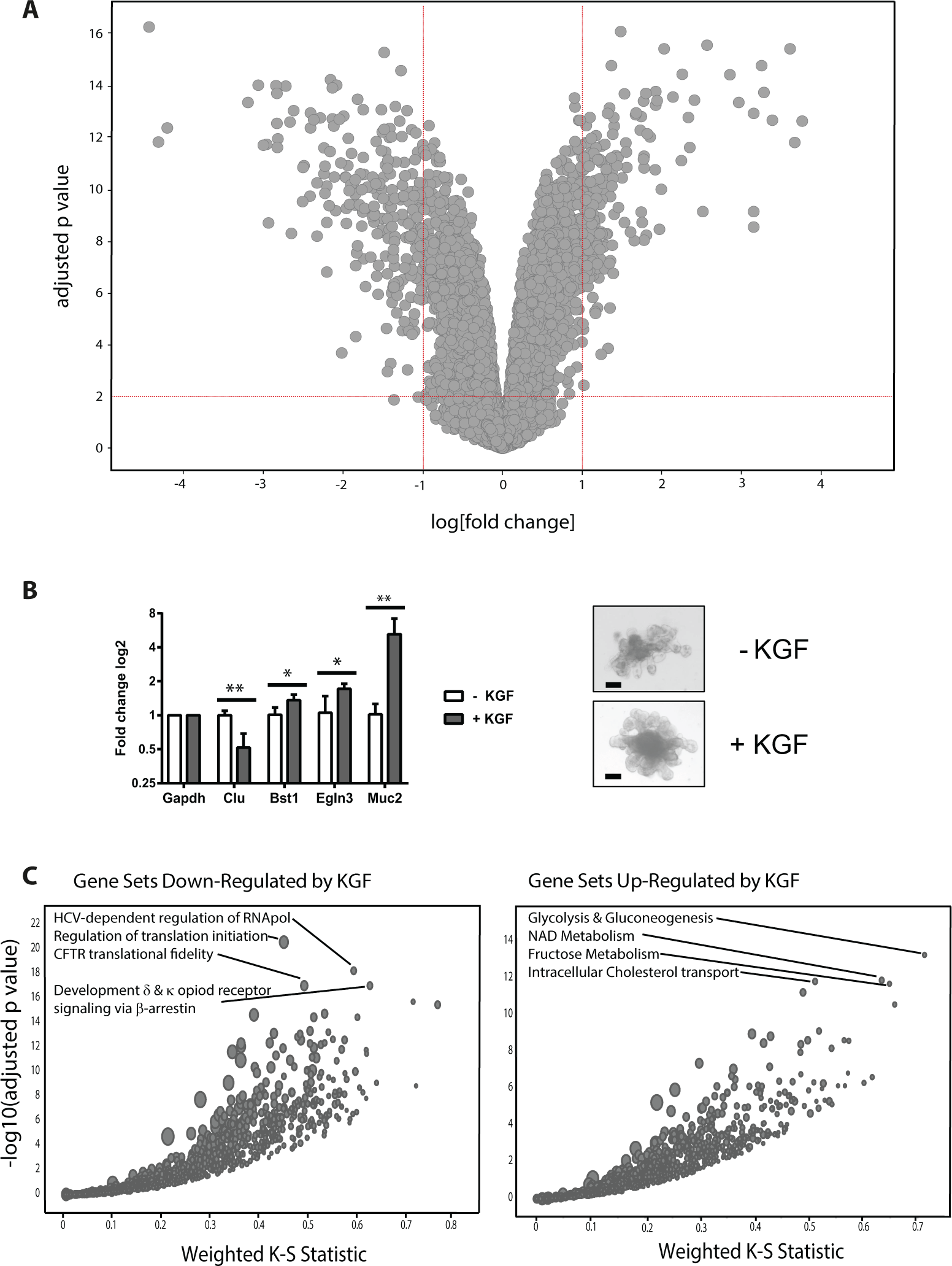
Differential gene expression in irradiated crypt organoid cultures that were treated with or without keratinocyte growth factor (KGF). (
Development of a Miniaturized In Vitro Assay of Mucositis
Once a reproducible assay to measure the viability of crypt organoid cultures in 96-well plates had been developed, the assay was miniaturized to a 384-well format, facilitating throughput and constraining cost. Typically, one 6 cm dish containing confluent crypt organoids was mechanically passaged and the crypts were resuspended in a total volume of 4.5 mL Matrigel. This organoid/Matrigel mixture was then kept on ice while 10 µL per well was added manually to a 384-well plate using a multichannel pipette. The Matrigel/crypt plates were briefly centrifuged (while at 4 °C) to force the crypts to the base of the plate, resulting in a homogeneous organoid distribution per well and a good plating consistency (
Compounds were transferred using the Echo 550 (LabCyte) such that the DMSO concentration was 0.05% for all the compounds tested. To guarantee that the fluorescence values obtained were due to the reduced tetrazolium dye and not due to any components of the assay, we determined fluorescence values in wells with 32 different compounds and a mock control upon incubation in medium alone, Matrigel plus medium, and Matrigel plus medium plus crypt organoid cultures. Fluorescence above background could be measured only in wells containing crypt organoids, demonstrating that fluorescence signals were due to the reducing activity of organoid cultures and not due to autofluorescence of Matrigel, medium, or compounds (
Screening for Radioprotective Compounds on Early and Late Passage Organoids
A library of 2534 compounds, each annotated with biological activity, was used to screen for pathways that could modulate the organoids’ response to irradiation. The assay typically yielded a robust Z′ value of 0.3 (comparing DMSO and CHIR-99021 as the neutral and positive control, respectively
22
;
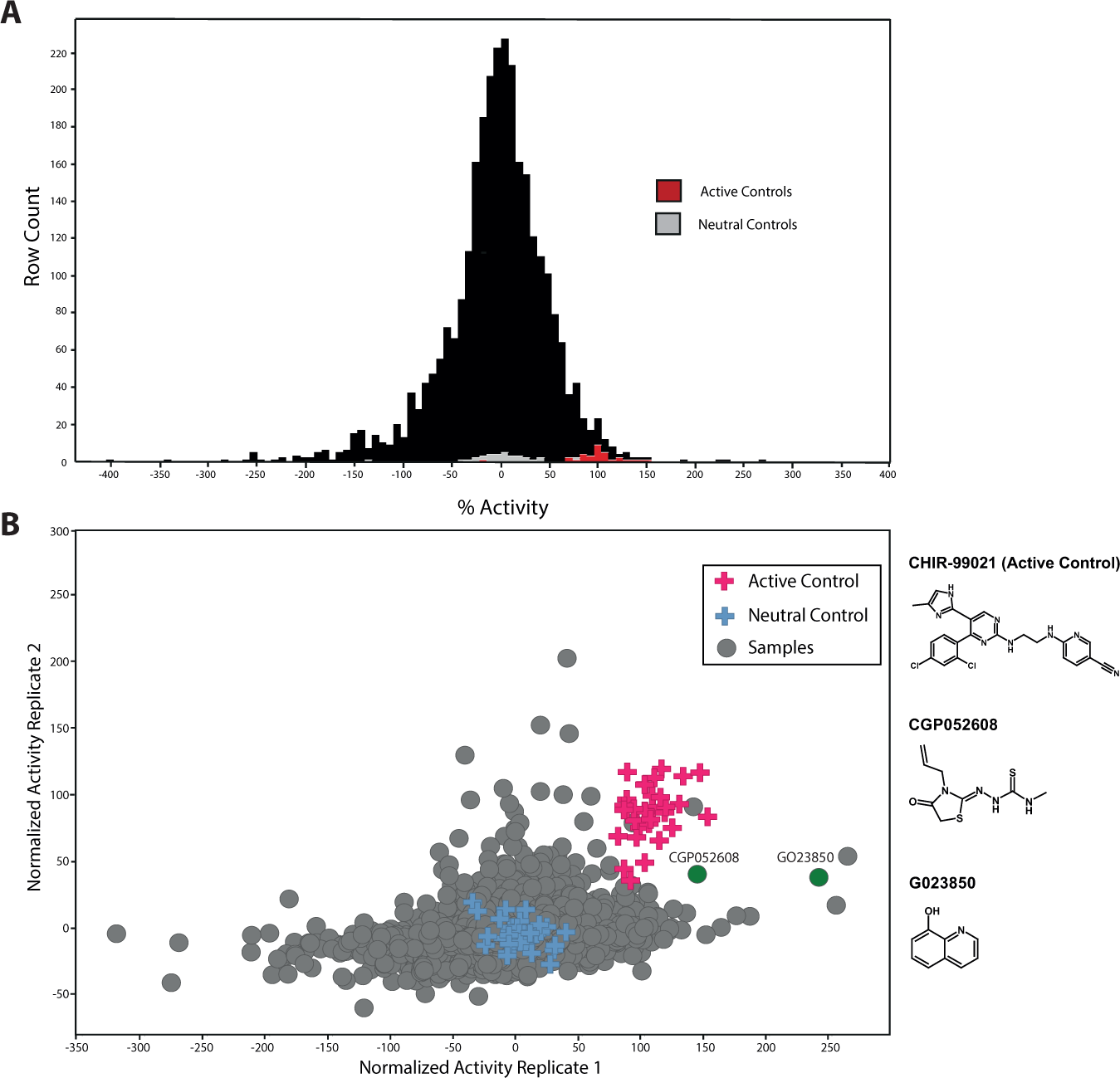
Screens for compounds with radioprotective activity on crypt organoid cultures. Early and late passages of organoid cultures were treated with 2534 compounds with annotated biological activity at a fixed concentration of 2.5 µM per compound and well (n = 1). Organoid viability was determined 7 days after radiation and compond addition. All individual DMSO (neutral control) and CHIR-99021 (active control) compound values are displayed to visualize the robustness of the assay. The average values for DMSO and CHIR-99021 were set to 0% and 100%, respectively. (
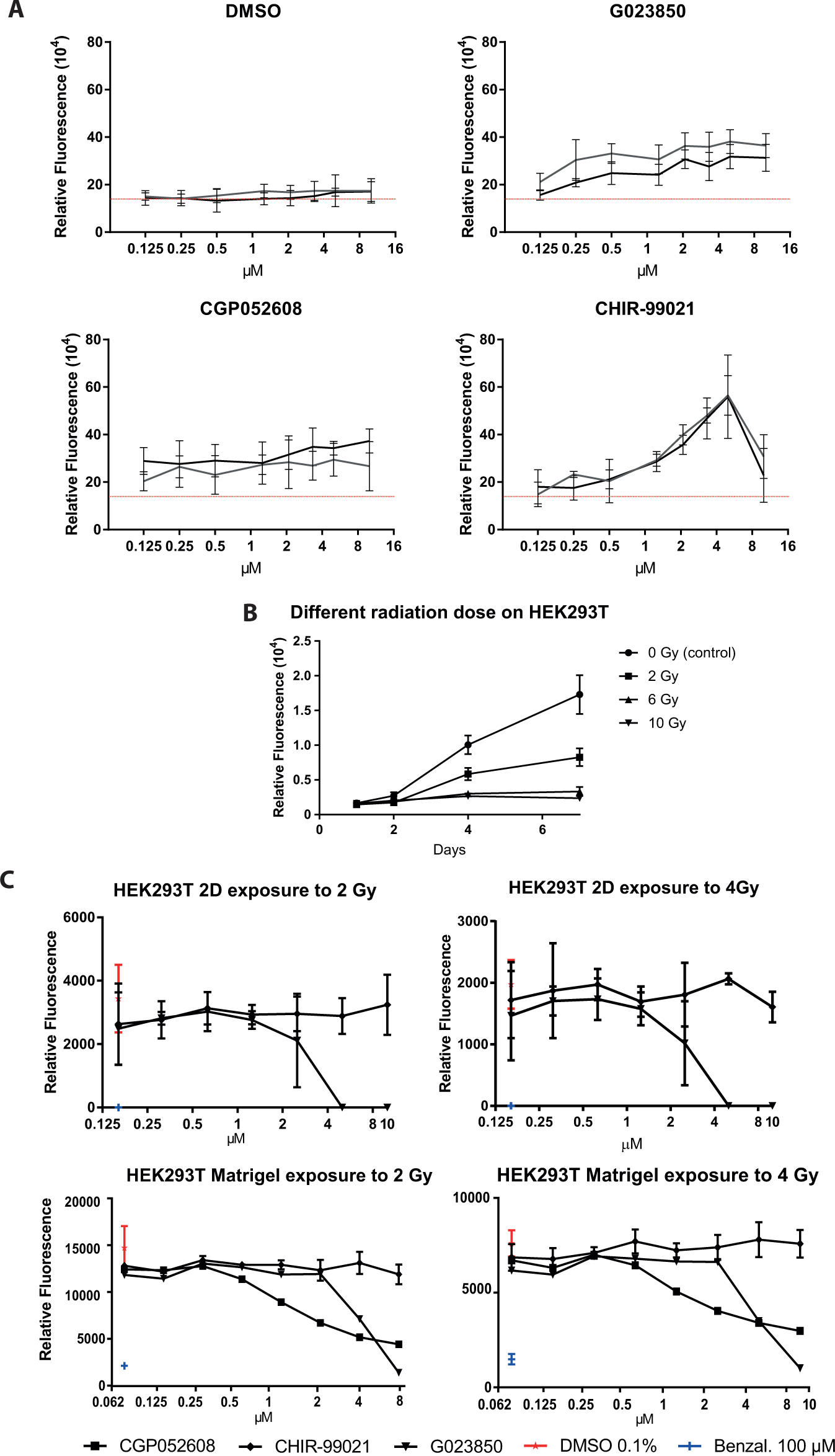
Assessing compound hits in cell-based assays. (
Comparing organoid cultures of differing passage number allowed compound activity to be assessed on two independent cultures of different maturity. The consistent behavior of the positive and negative controls reflected the robustness of the assay, whereas most compounds were not able to rescue viability ( Fig. 3B ). However, a few compounds were able to improve the viability of both early and late organoid cultures, emphasizing the robust activity of those compounds irrespective of organoid age in culture. Using an arbitrary cutoff of 1.8-fold over DMSO in both screens, a number of the active compounds were selected for further validation and characterization. Priority was given to two compounds with mechanisms that had not been mentioned in the context of radiation protection before (CGP052608 and G023850). Some compounds were able to only rescue viability of either early or late passage cultures, revealing an apparent dependency on organoid age. This could be a hallmark of molecular changes that organoids experience during cultivation. A fraction of compounds revealed strong toxicity under both organoid passage conditions, as they may possibly just act by general cytotoxic effects on gut epithelial cells.
Hit Characterization
Following conformation of their identity and purity by liquid chromatography–mass spectrometry, several hits from the screen were characterized by concentration-response experiments ranging from 0.75 to 20 µM, using crypt organoids derived from different mice to ensure reproducibility. Typically, these compounds showed bell-shaped dose-response curves, as is often observed for active compounds in phenotypic screens.23,24 Most active compounds displayed maximal activity between 2 and 10 µM. The data for two hits are depicted in Figure 4A .
To assess complex 3D versus 2D cell cultures as well as cell type and possibly species specificity of compounds identified as radioprotective agents, a number of the active compounds from the crypt organoid screen were tested in a similar assay format using an unrelated cell type, human embryonic kidney (HEK293T) cells, cultivated under the conditions specified for HEK293T cells. First, we determined survival curves of HEK293T cells after different radiation doses to identify the optimal dose. HEK293T cells were trypsinized and irradiated with 0 to 20 Gy before plating. Cell viability was determined up to 10 days following irradiation. HEK293T cells were slightly more sensitive toward radiation than mouse crypt organoids. Doses of 2 to 4 Gy were found to be optimal, because cells slowly recovered viability following irradiation ( Fig. 4B and data not shown). We then plated HEK293T cells that were irradiated with 2 or 4 Gy into 96-well plates and added compound hits at increasing concentrations immediately after plating. Cell viability was determined at 5 days after radiation and compound addition. A number of compounds (including GO23850 and CHIR-99021, as well as DMSO and benzalkonium chloride as controls) that showed activity in the crypt organoid assay were tested, but none revealed any protective effects on HEK293T cells ( Fig. 4C , top two panels, and data not shown). Interestingly, one of the radioprotective hits, G023850, was severely toxic on HEK293T cells. To exclude that the lack of radioprotection of the compounds on the irradiated HEK293T cells was due to the lack of the proper growth supplements, (i.e., the ones present in the crypt organoid cultures), we repeated the HEK293T cell experiment under conditions closely mimicking the organoid system. Thus, following irradiation (2 Gy and 4 Gy) and overnight plating of HEK293T cells in wells of 384-well plates using HEK293T cell culture medium, the medium was removed and Matrigel was added on top of cells and allowed to solidify. Subsequently, crypt culture medium was added, followed by the addition of increasing concentrations of compounds (including GO23850, CGP052608, CHIR-99021, and the DMSO and benzalkonium chloride controls) and incubation for 4 days. As for HEK293T cells grown under HEK293T cell culture conditions, none of the compounds revealed any radioprotective effect ( Fig. 4C , bottom two panels, and data not shown), and both GO23850 and CGP052608 showed toxicity. We concluded that crypt organoid growth supplements were not able to confer radioprotective activity to GO23850 and CGP052608 on HEK293T cells. These data provided strong support to the notion that phenotypic screens toward a particular application need to be carried out using the appropriate cellular system.
Organoid Cultures Undergo Changes in Gene Expression in Culture over Time
As noted above, compounds showed differing radioprotective activity dependent on the passage number of the organoid cultures used. We hypothesized that this was due to organoid-intrinsic changes in culture over time. The availablity of bioactive compounds allowed us to track organoid responses over time in a functional manner.
To investigate whether these functional changes were due to marked changes in gene expression, we conducted a study using five independently generated organoid preparations over the course of nine serial passages, from passage 2 to 10. At each passage, organoid samples were separated and subjected to the following analyses: one sample at each time point from nonirradiated organoids was harvested for RNA expression analysis, and a parallel sample, each, was used to monitor the radioprotective efficacy toward 44 of the most active compounds on irradiated crypt organoids as described above. Interestingly, the radioprotective effects of the compounds changed over time in a passage-dependent manner ( Fig. 5A shows the data for GO23850 and CGP052608 in relation to control treatments; for other compounds, data not shown). The apparent reason for the alteration in radioprotective efficacy was due to increased viability of the organoid cultures, reaching a maximum between passages 6 and 10.
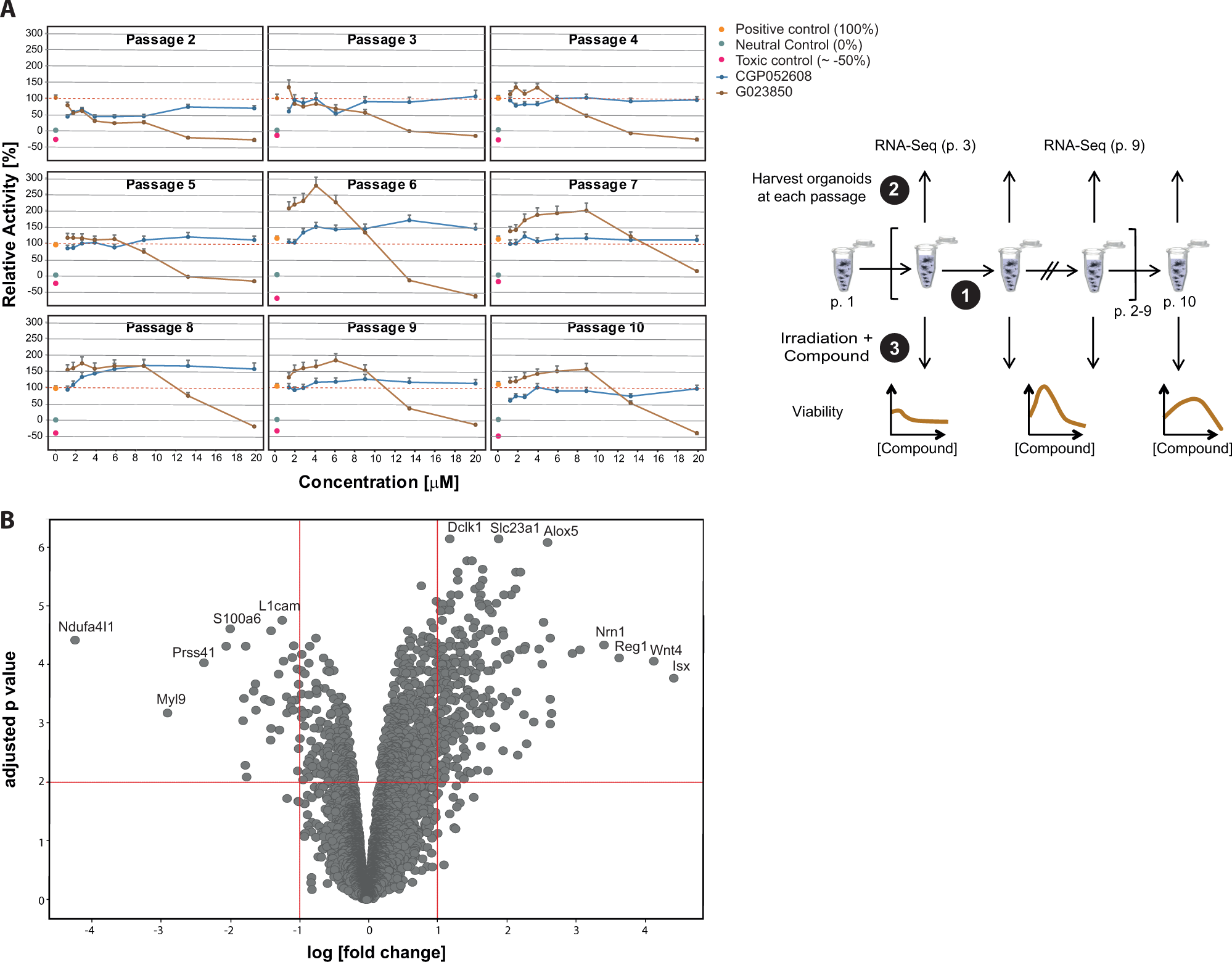
Radiation sensitivity and mRNA expression in organoid cultures change over time. Five independent crypt organoid cultures were established and serially passaged up to passage 10. Changes in radiosensitivity in culture over each passage from 2 to 10. At each passage, the radiosensitivity of cultures toward compound hits from the radiation screen was determined. (
Therefore, global, unbiased mRNA sequencing analysis was conducted on nonirradiated organoid cultures harvested at passage 3 and 9. As shown in
Figure 5B
, multiple mRNA transcripts underwent significant changes in expression levels when comparing organoids from both passages, demonstrating that the organoid cultures had undergone adaptions in culture over time (
Discussion
This report describes a simple endpoint assay in a 384-well format that was used to screen a focused library of compounds with known biological activity for their ability to increase organoid viability following gamma irradiation. Furthermore, gene expression analysis of irradiated crypt organoids treated with KGF, a radioprotective factor, allowed the identification of molecular pathways that may mediate protection from radiation.
Several genes were up-regulated in response to KGF treatment. Muc2 expression is strongly increased by KGF in irradiated gut organoids. This is consistent with the suggestion that KGF may enhance protection in part by increased mucus production in a rat colitis model. 25 One of the most highly up-regulated genes by KGF is bone stromal antigen 1 (Bst1), which codes for an ectoenzyme that converts NAD+ to the paracrine effector cyclic ADP ribose. 26 Bst1 in Paneth cells mediates augmentation of stem cell function in the mammalian intestinal niche in response to calorie restriction. 27 Therefore, Bst1 may positively modulate stem cell activity and thereby augment organoid growth following irradiation. Also up-regulated is prolyl hydroxylase domain protein 3 (Egln3), which has been implicated in the DNA damage response by modulating the ATR/CHK1/p53 pathway, possibly safeguarding genome integrity. 28 Conversely, several genes were down-regulated in irradiated crypt organoids upon KGF treatment. One such gene encodes thrombospondin 1 (Thbs1), which encodes an adhesive glycoprotein involved in cell-to-cell and cell-to-matrix interactions and which had previously been reported to limit recovery from irradiation. 29
This screen identified two compounds that increased recovery/protection from gamma irradiation. One of the compounds, CGP052608 ((Z)-2-(3-allyl-4-oxothiazolidin-2-ylidene)-N-methylhydrazine-1-carbothioamide), a synthetic thiosemicarbazone, had been reported to bind and activate the retinoic acid receptor–related orphan receptor alpha (RORA). 30 This pathway had been reported to be required to bring about pulses of p53 expression in response to gamma irradiation, so modulating this pathway is a potential mechanism by which this compound acts. 31
G023850 (quinolin-8-ol) had been reported to inhibit human methionine aminopeptidases-1 and -2(MetAP-1 and -2). 32 This compound is also reported to interact with RORgamma (https://www.ebi.ac.uk/chembldb/index.php/compound/inspect/CHEMBL451260), and mouse RORgamma had been implicated in DNA repair, possibly explaining the effect of G023850 on organoid survival postradiation. 33 This compound had also been reported to interact with histone-lysine N-methyltransferase, H3 lysine-9 specific 3 (EHMT2), highlighting a potential role of epigenetic alterations in radioprotection (https://www.ebi.ac.uk/chembl/target/inspect/102672).
One compound, GVK04294866 ((R)-8-chloro-4-(3-(methylamino)pyrrolidin-1-yl)benzofuro[3,2-d]pyrimidin-2-amine; https://www.ebi.ac.uk/chembldb/index.php/compound/inspect/CHEMBL1091874), was active only on early but not late passage organoids. This compound has been described as a potent and selective histamine H(4) receptor antagonist. 34 This is of interest because of a recent report that an oral mouse rinse of doxepin improves oral mucositis pain in patients receiving head and neck radiation treatment. 35 Doxepin is a tricyclic antidepressant and is also known, inter alia, as a histamine receptor antagonist (http://www.drugbank.ca/drugs/DB01142).
We could speculate that early and late organoid passage numbers correspond to young and old organoid cultures. Therefore, compounds identified on the latter might be of preferred interest when studying radioprotective agents in adult patients. Nevertheless, we believe that compounds identified under both culture conditions are of highest relevance for future drug discovery approaches.
Small intestinal crypt organoids are composed of multiple cell types, including stem cells and cells comprising the niche. 6 It would be challenging to develop an assay that monitors the full complexity of this complex biological system. Assessing phenotypic changes of organoids by imaging techniques would be a valuable alternative, necessitating the need to plate organoids within thin Matrigel layers. One approach to address the changes in cellular complexity in response to compound treatment is to quantitate mRNAs specific for individual cell types by qPCR, and future studies will make use of this approach. Although it may be possible to develop simple single readout assays to monitor caspase induction as a marker for apoptotic cells, this was not part of this study.
The identification of tool compounds that elicit a phenotypic response in organoid cultures allowed functional interrogation as a function of time in culture. Thanks to these compounds, we were able to detect that the sensitivity of the cultures toward the compounds following radiation changed over time, and these changes were paralleled by changes in global changes in mRNA expression. These findings indicate that, despite uniform culture and passaging conditions, organoid cultures have the propensity to change over time. These are important learnings, because they allow us to define conditions suitable to conduct screens (i.e., applying passages 6–10 for radioprotection). Furthermore, the genes whose expression changed over time may encode targets for recovery from radiation damage. Future studies will be directed to address the molecular basis for these cellular adaptations.
Footnotes
Acknowledgements
We would like to acknowledge the contributions of Joëlle Sprunger, Jennifer Lürding, and Jan Tchorz to experiments during the development of this assay system; Sarah Hevi, Sina Mohammadi, and Sue Menon for providing reagents; and Neil Press and Gabriele Hintzen for intellectual input and contribution to the work.
Supplementary material is available online with this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
