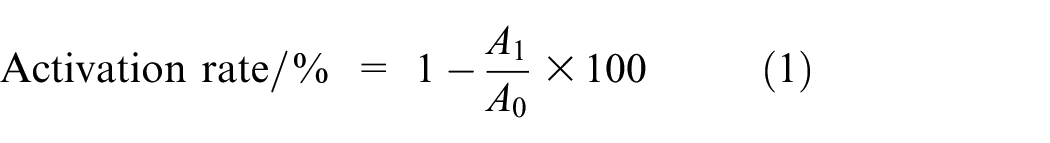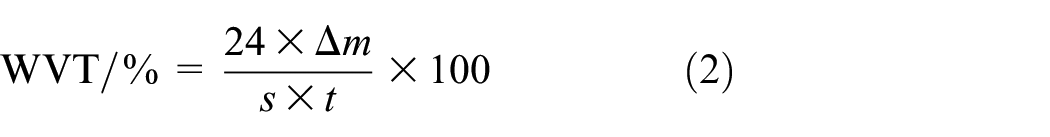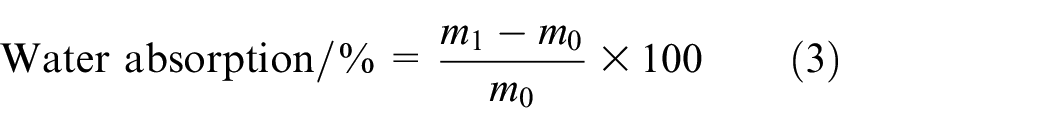Abstract
Waterproof moisture-permeable films with excellent water resistance and breathability are employed in functional clothing to improve the comfort of wearers. Although polytetrafluoroethylene microporous membranes exhibit outstanding water resistance and moisture permeability, their application is limited because of the environmental impact of fluorinated compounds. For this reason, herein, we aimed to improve the hydrophobicity of thermoplastic polyurethane (TPU) microporous membranes while maintaining their moisture permeability. We employed silane coupling agents to modify nanosilica particles, which were then coated on TPU microporous membranes to enhance their waterproof performance. The optimized parameters for the modification process of silica nanoparticles are γ-aminopropyltriethoxysilane and hexadecyltrimethoxysilane (silane coupling agents) concentrations of 8% relative to the mass of silica, a modification time of 2 h, and a temperature of 70°C. The water contact angle of the particles modified under these conditions reached 138.58°. The modified SiO2 particles were used to treat the TPU film, with an optimal mass fraction of the modified silica of 3% and a modification time of 4 h. The water vapor transmission of the modified TPU film was 5661.92 g/(m2·24 h), with enhanced hydrophobicity, and its water absorption rate was 12.41%, which is 16.49% lower than before modification. Additionally, under simulated heavy rain conditions, the water absorption rate of the composite fabric with modified TPU film was lower by 14.7% than that of the composite fabric with unmodified film.
Introduction
In recent years, with the increasing popularity of outdoor activities, there has been a surge in sports such as skiing, road running, trail running, and camping. 1 These activities require outdoor sportswear fabrics to simultaneously exhibit excellent moisture permeability (to remove sweat produced by the wearer) and outstanding water resistance to ensure the comfort of the wearer. 2 Currently, this challenge is addressed by applying waterproof moisture-permeable films to the fabric, with the most commonly used being polytetrafluoroethylene (PTFE) microporous membranes and nonporous thermoplastic polyurethane (TPU) films.3,4 However, fluorinated compounds can degrade into perfluorooctanoic acid or shorter-chain fluoroalkanes, which adversely affect the environment and living organisms.5,6 At the same time, conventional TPU films, owing to their high hydrophilicity, absorb water, leading to decreased durability and significantly limiting their application in highly moisture-permeable waterproof fabrics. Therefore, to improve the performance of waterproof and highly moisture-permeable TPU-based fabrics, the hydrophilicity of the TPU films must be reduced while maintaining their high moisture permeability. Studies have shown that although TPU microporous membranes can effectively enhance the moisture permeability of fabrics, they suffer from a rapid decline in waterproof performance over time, primarily because of the hydrophilic nature of TPU membranes. Therefore, improving the hydrophobicity of TPU microporous membranes is key to enhancing their overall performance.
To address the issue of excessive water absorption in films, three approaches have been explored: (1) dissolution of the film and recasting with added fillers; 7 (2) introduction of hydrophobic segments during synthesis; 8 and (3) increase in the surface roughness of the film. 9 Among these, hydrophobic modification of the surface of the films using SiO2 particles has shown promising results, with studies demonstrating that the treated TPU films exhibit improved hydrophobicity. For instance, researchers have hydrophobized SiO2 particles and used them to spray-coat TPU films, resulting in superhydrophobic coatings with enhanced hydrophobic properties. 10 Additionally, Guo et al. 11 have reported a superhydrophobic TPU film with good wear resistance, corrosion resistance, and self-cleaning properties obtained by spraying hydrophobic SiO2 nanoparticles onto a semi-cured TPU film. Inspired by these research findings, the present study aimed to further improve the performance of TPU films for highly moisture-permeable waterproof fabrics through the modification with SiO2. Our objective was to enhance the hydrophobicity of the films while maintaining their moisture permeability.
For this purpose, γ-aminopropyltriethoxysilane (KH550) and hexadecyltrimethoxysilane (HDTMS) were selected to modify the surface of silica particles. KH550, with its –NH2 groups, can react with esters, carboxylic acids, and many alkyl groups, thereby improving the adhesion of SiO2 particles to the film surface. 12 HDTMS, with its long-alkyl-chain substituent group, forms low-surface-energy siloxanes after hydrolysis, leading to an increase in the hydrophobicity of SiO2 particles through dehydration condensation reactions between silanol groups and the hydroxyl groups on the SiO2 surface. The modified SiO2 particles were used to treat the TPU film. The effects of the dosage of silane coupling agents KH550 and HDTMS on the hydrophobic properties of the particles were investigated to determine optimal modification parameters. Further, the modification process of the TPU film was investigated, and the relationship between the structure of the modified TPU film and its water absorption and moisture permeability was examined. Ultimately, this study aims to solve the problem of high water absorption and poor waterproof performance of TPU films and provides guidance for the partial replacement of microporous PTFE membranes with TPU films.
Experimental
Experimental Materials and Instruments
Material: The TPU film, characterized by a thickness of 0.025 mm and a grammage of 18.5 g/m2, the water-resistant polyester plain weave fabric (with a yarn fineness of 167 dtex, warp density of 300 ends/10 cm, weft density of 376 picks/10 cm, weight of 0.683 g/cm2, and a water resistance rating of level 5), and the wet-cure polyurethane hot-melt adhesive DJ1102B (PUR) were all supplied by Zhejiang Dongjin New Material Co., Ltd; nanosilica, with an average particle size of 7–40 nm, was purchased from Shanghai Een Chemical Technology Co.
Drugs: KH550 silane coupling agent (Shanghai Aladdin Biochemical Technology Co., Ltd.); hexadecyltrimethoxysilane (HDTMS) (McLean Chemical Reagent Co., Ltd.); ethanol (Hangzhou Gaocrystal Fine Chemical Co., Ltd.); and 0.5 mol/l sodium hydroxide standard solution (Shenzhen Xijing Biotechnology Co., Ltd.) were used.
Instruments: An HD101B-1 electric heating blast drying oven (Nantong Hongda Experimental Instrument Co., Ltd.); a Zetasizer Lab laser nano-particle sizer (Malvern Instruments Ltd., UK); a Nicolet iS20 Fourier Infrared Spectrometer (Thermo Fisher Scientific (China) Co., Ltd.); an Ultra 55 field emission scanning electron microscope (Zeiss, Germany); TG 209 F1 thermogravimetric analyzer (NETZSCH Scientific Instruments Trading Co., Ltd.); a Kino SL200B Video Contact Angle Measuring Instrument (Kono Industries, Inc., USA); a CMT4202 Z020 Electronic Universal Testing Machine (ZWICK, Germany); a LAB-W80 Moisture Transmission Performance Tester (Ruidong Inspection Instrumentation Co., Ltd., Taiwan); a Bundesmann Tester (Silea Telas Co., Ltd.); and a multi-functional pilot production machine TC040EH (Taiwan Xinqiang Advanced Machinery Co., Ltd.) were used.
Experimental Methods
Preparation of Hydrophobically Modified Silica Particles
A certain amount of silicon oxygen coupling agent KH550 and cetyltrimethoxysiloxane at a certain mass ratio were added to a 250 ml three-necked flask. Aliquots of 50 ml of ethanol and 10 ml of deionized water were added, and the pH was adjusted to 7. Then, 5 g of silica was dispersed in 50 ml of ethanol using ultrasonication and added to the flask. The reaction was carried out at 70°C for 2 h. After completion, the reaction mixture was dried in an oven at 110°C for 2 h and then stored in a sealed bag.
Processes for Hydrophobic Modification of TPU Films
Different amounts of modified silica powder were dispersed in 50 ml of ethanol solvent and ultrasonically dispersed for 1 h to prepare silica dispersions with varying mass fractions. Subsequently, TPU films were cut into circular samples with a diameter of 15 cm and immersed in the silica dispersion for 1–8 h. Finally, the films were removed using tweezers and dried in an oven at 80°C to obtain the silica-modified thermoplastic polyurethane films.
Preparation of Laminated Composite Fabric
In the preparation process of laminated composite fabrics, the adhesive tank is first heated to the set temperature. After 20 min, the knife gap is adjusted. Subsequently, the thermoplastic polyurethane (TPU) adhesive is heated to its molten state and injected into the adhesive tank. Following this, the entire production line is initiated. The molten TPU adhesive is extruded by a doctor blade into the holes of a rotating engraved roller, and through pressurization, the adhesive within the holes is transferred onto the film. Upon passing through the lamination rollers, the film adheres tightly to the fabric. Throughout this process, strict control over the operational parameters is maintained: an application rate of 17.6 g/m2 for the adhesive, a lamination temperature of 110°C, a compounding clearance of −0.1 mm, and a line speed of 3.0 m/min. Under these conditions, it is ensured that the TPU adhesive cures adequately in a controlled environment with suitable temperature and humidity levels. Finally, under a certain tension, the material is wound up for subsequent use.
Testing and Characterization
ATR-FTIR Test and Thermogravimetric Analysis (TGA)
Attenuated total reflection Fourier transform infrared (ATR-FTIR) spectroscopy was performed using a Nicolet iS20 FTIR spectrometer. The spectra were recorded in the wavenumber range of 500–4000 cm−2 with a resolution of 4.0 cm−2.
Thermogravimetric analysis was conducted in accordance with ASTM E1131-20 using a thermogravimetric analyzer (TG 209 F1). Nitrogen was utilized as the carrier gas at a flow rate of 50 ml/min. The sample was heated from ambient temperature to 800°C at a heating rate of 10°C/min.
SEM Test
Before imaging, the materials were sputter-coated with a thin layer of gold to enhance surface conductivity. The surface morphology of the materials was then observed using a desktop scanning electron microscope (Ultra 55).
Activation Rate Test
The activation rate test for modified particles is conducted in accordance with the procedures specified in GBT 19281-2014. This test utilizes the hydrophobic characteristics of the particle surfaces to quantitatively characterize the extent of surface coating. A sample weighing A0 grams is added to a beaker containing 200 ml of distilled water and subjected to reciprocal shaking at a frequency of 120 cycles per minute for 1 min. The mixture is then allowed to stand undisturbed for 30 min. Once distinct stratification is observed, the supernatant floating layer is removed, and the settled fraction is dried to a constant mass in an oven set at 105°C, and subsequently weighed as A1 grams. The calculation is performed according to formula (1).
Particle Size Test
To measure the particle size of a silica dispersion, the procedure was conducted in accordance with GB/T 19077.1-2008, “Particle Size Distribution—Laser Diffraction Method—Part 1: General Principles.” An appropriate amount of silica dispersion was taken and diluted with deionized water to a mass fraction of 0.5%. The particle size of the dispersion was measured using a laser nanoparticle size analyzer (Zetasizer Lab) at a temperature of 25°C.
Mechanical Performance Test
The mechanical properties of the SiO2-TPU films were tested using a universal testing machine (CMT4202 Z020) according to the international standard ISO 1184-1983, “Determination of tensile properties of plastic film.” The SiO2-TPU films were cut into dumbbell-shaped specimens (20 × 5 mm) using a standard mold. After measuring the thickness, the tensile properties were tested.
Contact Angle Test
To evaluate the hydrophobicity of the modified silica nanoparticles, the modified nano-SiO2 particles were ground and an equal amount of the sample was placed in a small container. The samples were then pressed into tablets using a tablet press. According to standard GB/T 30693-2014, the sample was placed flat, then, a 2 μl water droplet was suspended from the needle tip. The sample stage was raised to bring the sample surface into contact with the hanging water droplet, after which the sample was retracted to complete the droplet transfer. The static contact angle of the tablets was measured using a video contact angle meter (Kino SL200B).
Water Vapor Transmission (WVT) Test
According to the GB/T 12704.2-2009 standard, water vapor transmission (WVT) was tested using a water vapor transmission tester (model: LAB-W80). The test was conducted under controlled conditions of 38°C temperature and 50% relative humidity. The WVT was calculated based on the change in mass of the moisture cup (including the sample and water) over a specified period, as shown in equation (2):
where WVT is the water vapor transmission (g/(m2·24 h)),
Water Absorption Test
The water absorption test for TPU films was conducted in accordance with the GB/T 1034-2008. Initially, dried TPU films were cut into squares measuring 60 × 60 mm. The specimens were then weighed to determine their initial mass. Following this, the films were immersed in distilled water at a controlled temperature of 23°C for a period of 48 h. Afterward, the surface of the films was wiped dry, and the mass of the absorbed water was measured. The water absorption of TPU and SiO2-TPU films was calculated using equation (3).
AcutEye High-speed Camera Test
High-speed photography was conducted using an AcutEye high-speed camera (Cyclone-5-700-C-RT) under the illumination of a cold light source. The images of water droplets falling onto the film were captured at a frame rate of 450 frames per second and transmitted to a computer for analysis.
Heavy Rain Resistance Test
The heavy rain resistance of the fabric was evaluated following the “Textiles-Determination of Water Repellency of Fabrics by the Bundesmann Rain-Shower test” (ISO 9865-1991). Initially, the sample was weighed to determine its initial mass, denoted as m1, with a precision of 0.01 g. The fabric specimen was positioned on the sample holder with the test surface facing upward, ensuring it was flat and free from tension, and was securely held in place with a suitable clamping ring. The rain shield was retracted, and the water repellency of the wetted sample was visually assessed at 1, 5, and 10 min using a reference sample for comparison. The wetting condition of the unexposed side of the sample was also noted. After a 10 min exposure period, the volume of water that had penetrated through the fabric was measured, quantified by the volume of water collected in the sample container, reported in milliliters (ml). The sample was subsequently subjected to centrifugation for 15 s to remove excess water, and its mass was immediately reweighed to obtain the final mass, denoted as m2, again with a precision of 0.01 g. The water absorption rate (W) was expressed as a percentage of the initial mass and calculated using equation (4).
where: m1 is the mass of the sample before the test (g) and m2 is the mass of the sample after the test.
Results and Discussion
Optimization of the Hydrophobic Modification of Silica
To systematically optimize the hydrophobic modification process of silica, we employed a controlled variable method to adjust three key parameters: the amount of the modifying agent, the blending ratio of the modifying agents, and the reaction temperature. This approach was used to investigate the effects of these factors on the hydrophobic properties of the silica particles.
Effect of KH550 Concentration on Particle Hydrophobicity
To systematically investigate the effect of the concentration of KH550 (silane coupling agent) on the hydrophobicity of modified silica, the modification temperature was fixed at 70°C and the modification time was set to 2 h. Under these conditions, the concentration of KH550 was varied stepwise. Hydrophobicity was evaluated by measuring the water contact angle and activation rate of the modified silica particles.
Figure 1 shows the effect of the KH550 concentration on the hydrophobic properties of the modified silica particles. The water contact angle gradually increases with the KH550 concentration, reaching a maximum of 128.43° at 8% concentration. With a further increase in concentration to 10%, the contact angle decreases. This suggests that at 8% KH550, the coating process of silica with KH550 reaches saturation, with further increases in the KH550 dosage being detrimental to the modification process. 13 This observation is corroborated by the trend in the activation rate (Figure 2), where the activation rate exceeds 95% at 8% KH550, but decreases at higher concentrations, tending toward equilibrium. This suggests that at 8% concentration, KH550 effectively coats the nanoparticle surface and that 8% is the optimal KH550 dosage.
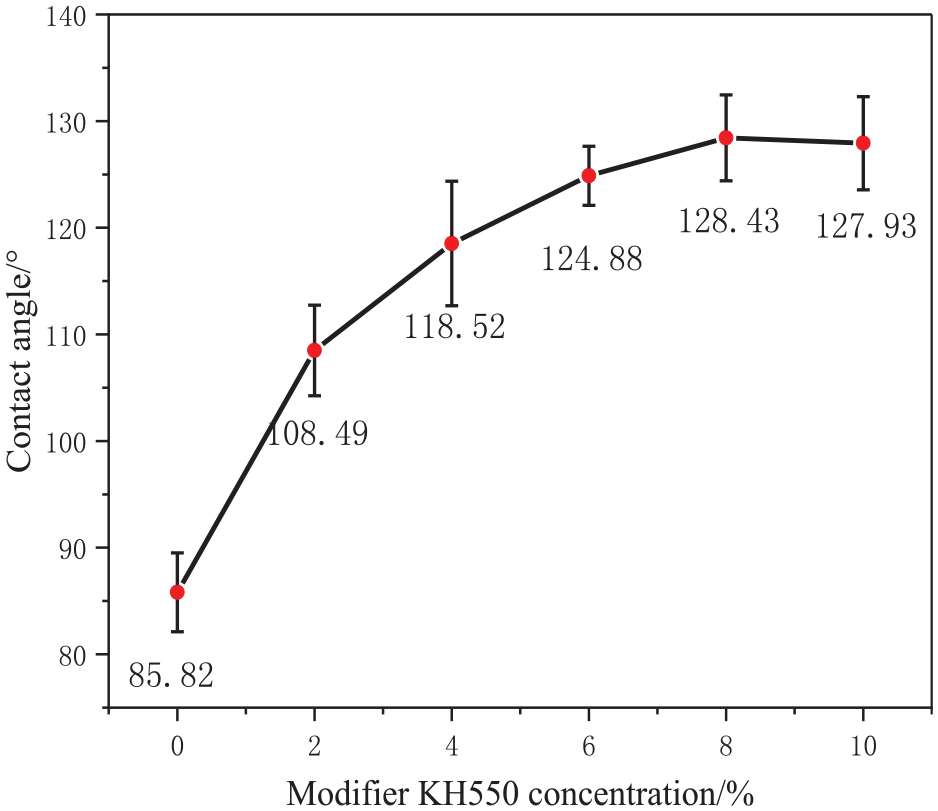
Effect of the KH550 concentration on the contact angle ofT-SiO2.
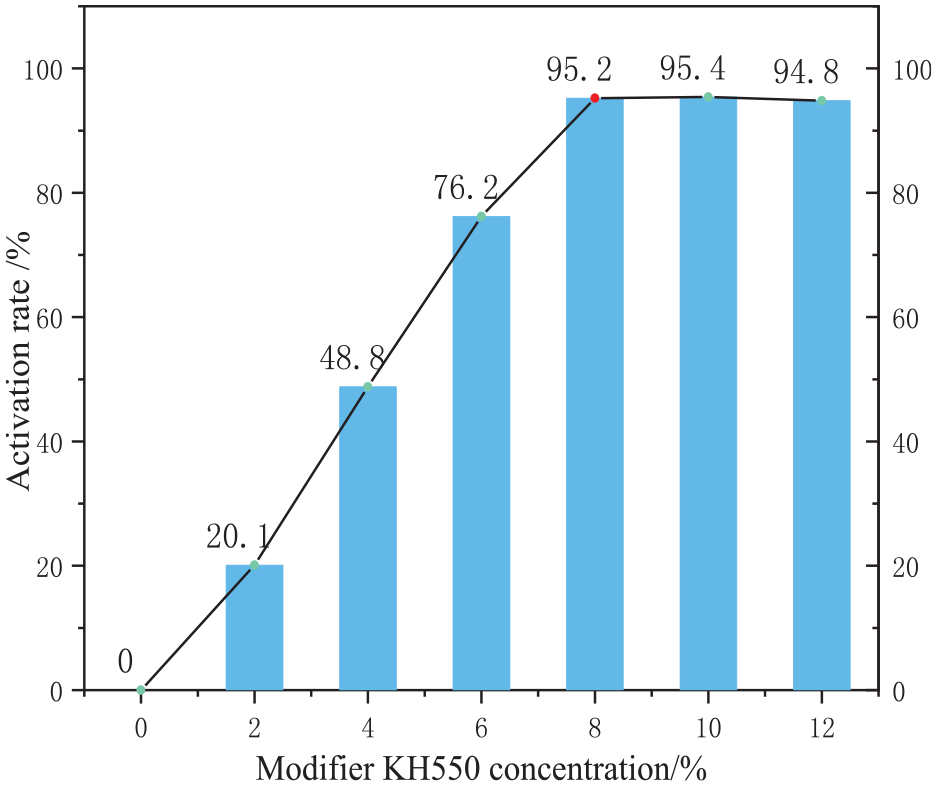
Effect of KH550 concentration on the activation rate ofT-SiO2.
Effect of HDTMS Concentration on Particle Hydrophobicity
To further enhance the hydrophobicity of the T-SiO2, HDTMS was added as a second silane coupling agent. The effect of HDTMS concentration on the water contact angle of the modified particles was systematically investigated by varying the concentration of HDTMS while maintaining the KH550 concentration at 8% and modification temperature at 70°C. The test results are shown in Figure 3.
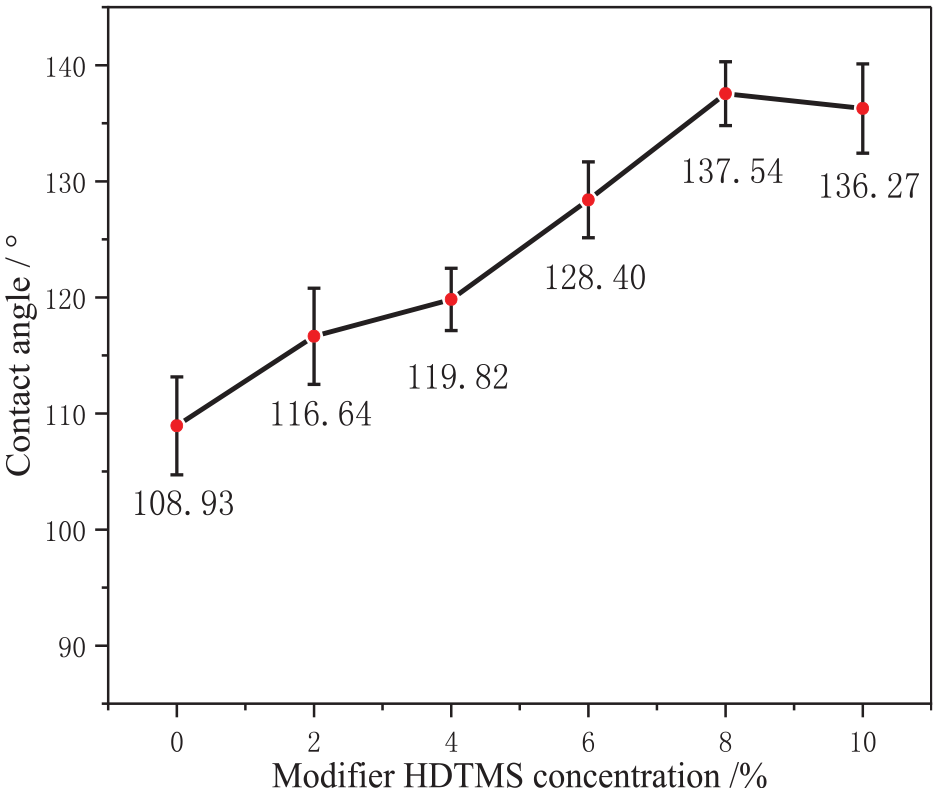
Effect of HDTMS concentration on the contact angle of T-SiO2.
As shown in Figure 3, with increasing HDTMS concentration, the water contact angle of T-SiO2 initially increases and then decreases. The maximum water contact angle (137.54°) is observed at an HDTMS concentration of 8%. The increase in hydrophobicity was attributed to the hydrolysis of HDTMS to silanol, which underwent a dehydration and condensation reaction with hydroxyl groups on the surface of silica, forming a low-surface-energy silicone. 14 This led to the formation of a two-dimensional ordered hydrophobic thin-film layer on the surface of the particles. At the same time, the increase in HDTMS concentration above 8% did not improve the hydrophobicity of T-SiO2. Thus, 8% was considered the optimal dosage of HDTMS (1:1 ratio with KH550).
Effect of the Modification Temperature on the Particle Hydrophobicity
To investigate the relationship between the hydrophobicity of T-SiO2 particles and modification temperature, we performed modification at different temperatures, with the concentrations of KH550 and HDTMS both maintained at 8%. The water contact angle of T-SiO2 obtained at different temperatures was measured. The test results are shown in Figure 4.
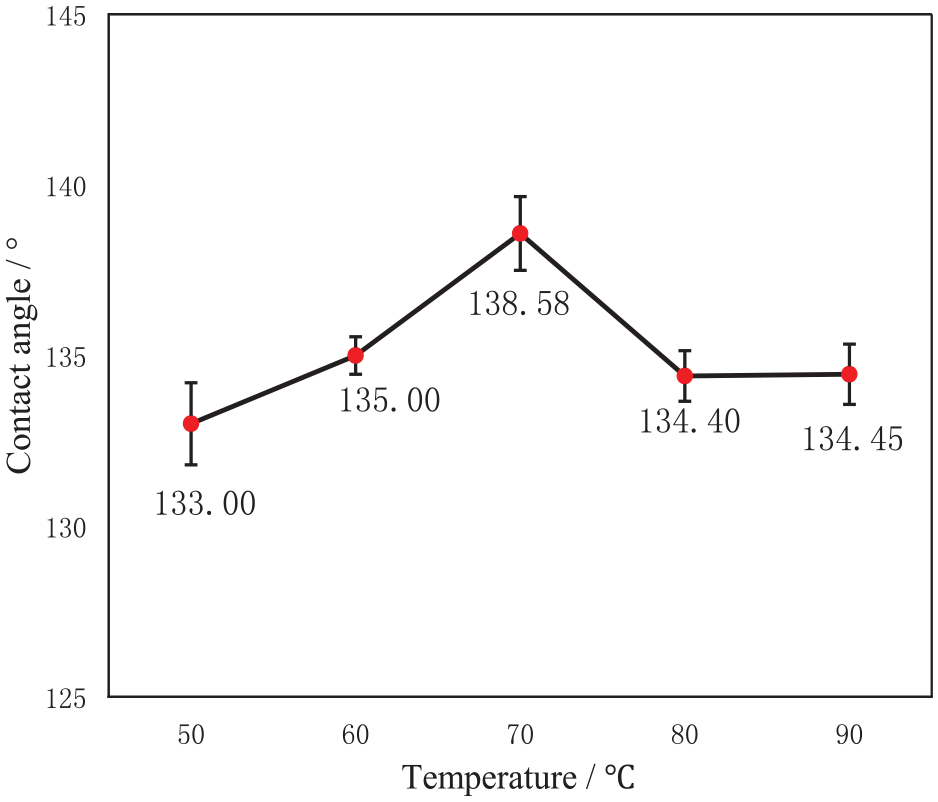
Effect of the modification temperature on the contact angle of T-SiO2.
As shown in Figure 4, with increasing temperature, the water contact angle of T-SiO2 initially increases and then decreases, reaching a maximum of 138.58° at 70°C. This was attributed to the chemical energy of the reactants being insufficient to allow complete bond breakage at low temperatures. With increasing temperature, molecular motion intensifies and the probability of collision increases, increasing the grafting ratio. 15 However, with the increase in temperature beyond 70°C, the water contact angle decreases, possibly because of the excessive probability of collision among silane coupling agent molecules, leading to self-polymerization and impeding the reaction progress. Consequently, 70°C was selected as the optimal modification temperature.
Particle Size Analysis
After optimizing the hydrophobic modification process, the properties of T-SiO2 particles were characterized, starting with particle size. SiO2 nanoparticles exist in two forms: monodisperse primary particles and agglomerated secondary particles. The primary particles exhibit high reactivity because they are excited, while the secondary particles are relatively stable. 16 Owing to the abundance of hydroxyl groups on their surface, SiO2 nanoparticles possess high surface binding energy, rendering them prone to agglomeration. Therefore, in the preparation of modified nanocomposites, the dispersion of SiO2 nanoparticles should be improved through surface treatment. The particle size distribution of T-SiO2 and unmodified silica was assessed using a laser nano-particle sizer, with the results shown in Figure 5.
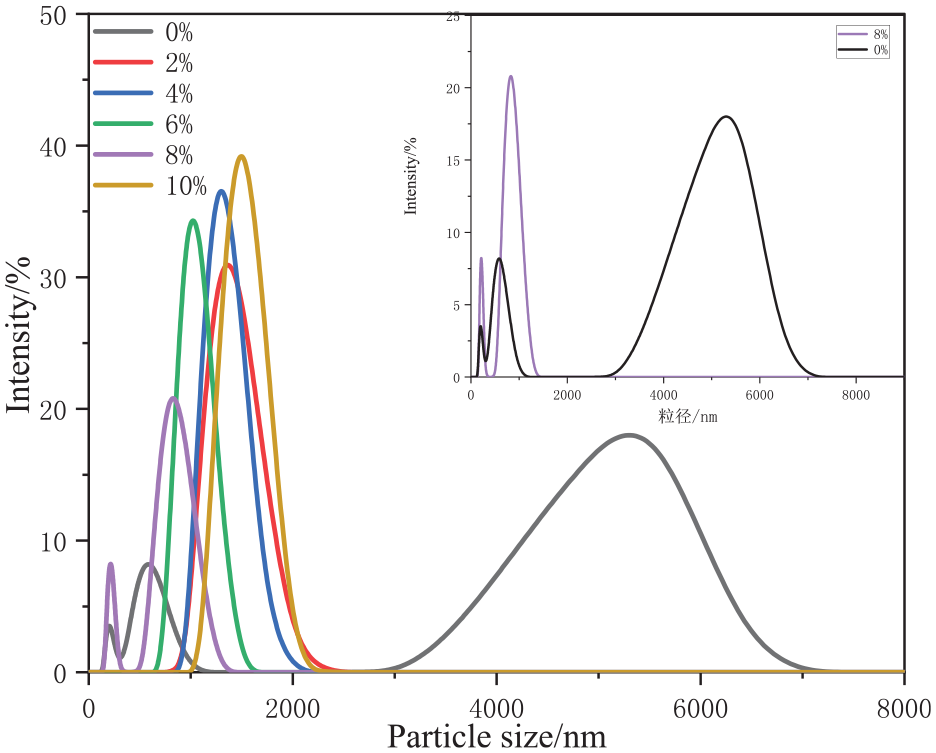
Particle size distribution of T-SiO2 at different KH550 concentrations.
As shown in Figure 5, before modification, SiO2 nanoparticles are mainly in the form of agglomerates, with the distribution modes (bimodal distribution) of 5000 and 800 nm, whereas the modes of the particle size distribution of the modified nanoparticles are 1000 and 100 nm. The effect of the KH550 concentration on the particle size of T-SiO2 is presented in Figure 5. With increasing dosage of KH550, the size of agglomerates noticeably decreases, accompanied by considerable changes in particle size distribution. Specifically, at a KH550 dosage of 8%, the sizes of both types of modified SiO2 agglomerates are notably lower than in unmodified nanoparticles. This decrease was attributed to the chemical bonding between the hydroxyl groups on the surface of SiO2 and the silane coupling agent, which improved the dispersion of particles and effectively prevented agglomeration. 17 However, a further increase in the modifier amount to 10% results in an increase in particle size. This was attributed to the excess of the silane coupling agent generating oligomers and exerting a bridging effect, thereby impeding the coupling reaction and causing particle agglomeration, ultimately resulting in an increase in particle size.
Structural Characterization of T-SiO2
To further verify whether the silane coupling agents were successfully grafted onto the nanosilica surface, detailed structural characterization was performed. This section presents the results of the Fourier transform infrared spectroscopy (FTIR) and thermogravimetric analysis (TGA) for both unmodified and modified silica particles.
ATR-FTIR Analysis
The properties of silica can be altered by grafting the hydrolysis products of silane modifiers on its surface, which reduces the number of hydrophilic hydroxyl groups present on the surface of silica, lowers surface energy, and diminishes the polarity of silica, thereby enhancing the dispersibility of silica particles. 14 Herein, ATR-FTIR was employed to characterize the surface functional groups of nanosilica particles before and after modification, with the results shown in Figure 6.
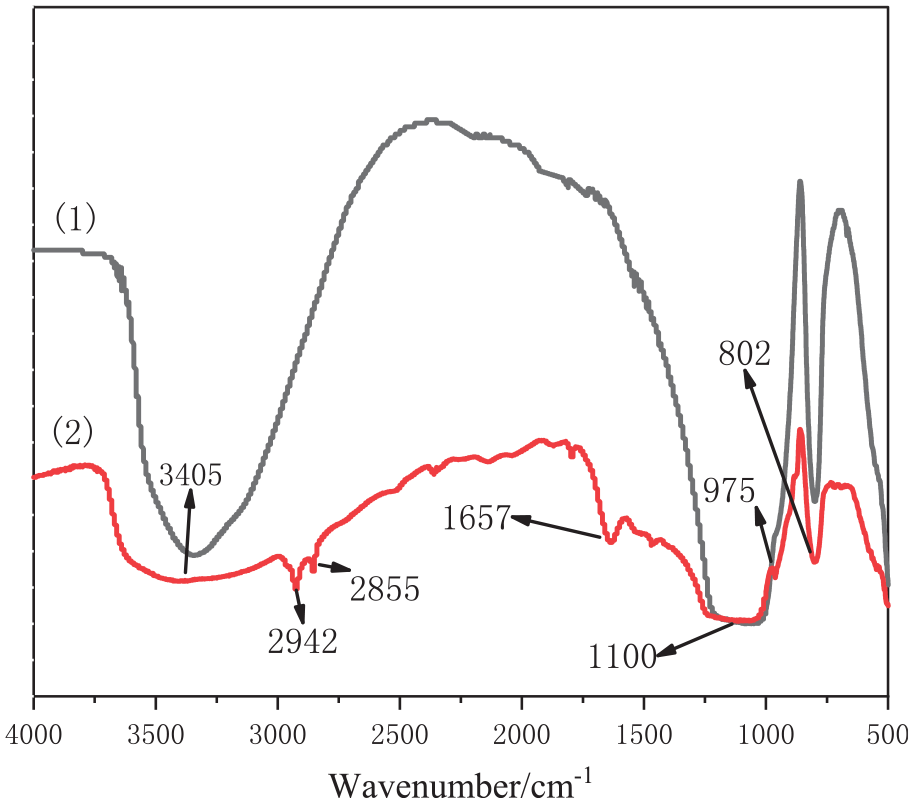
ATR-FTIR spectra of SiO2 before (1) and (2) after modification.
Figure 6 reveals that after the surface modification, a weak C-H stretching vibration peak and a characteristic absorption peak of -CH3 emerge at 2942 and 2855 cm−1 respectively. Additionally, the peak at 1657 cm−1 appearing after modification is a characteristic absorption peak of the deformation vibration of N-H in the N-H plane in -NH2. 18 Furthermore, the absorption peak at 802 cm−1 corresponds to the Si-O-Si symmetric stretching vibrations. The appearance of characteristic peaks of -NH2 and -CH2 in the spectrum of T-SiO2 confirms successful grafting modification.
TGA
To further confirm the modification of silica, TGA was performed using a differential scanning calorimeter. The weight loss of the T-SiO2 particles over a specific temperature range was measured to confirm the successful grafting modification.
As shown in Figure 7, the mass of both unmodified and modified silica decreases with increasing temperature in the range of 30–200°C. This was attributed to the decomposition of residual ethanol and moisture adsorbed on the silica particles. 19 Below 200°C, the weight loss of T-SiO2 is lower than that of unmodified silica, which is associated with the increased hydrophobicity of T-SiO2. The modified silica particles exhibit weight loss between 200°C and 400°C, possibly because of the cleavage of bonds on the silica surface and grafted modifier macromolecules, resulting in the detachment of the latter from the silica particle surface. With a further increase in temperature, substantial weight loss occurs between 400°C and 550°C, corresponding to the decomposition of the silane coupling agent coating on the silica particle surface. Above 600°C, the weight loss curve becomes relatively flat, suggesting that the silane coupling agent has completely decomposed. 20
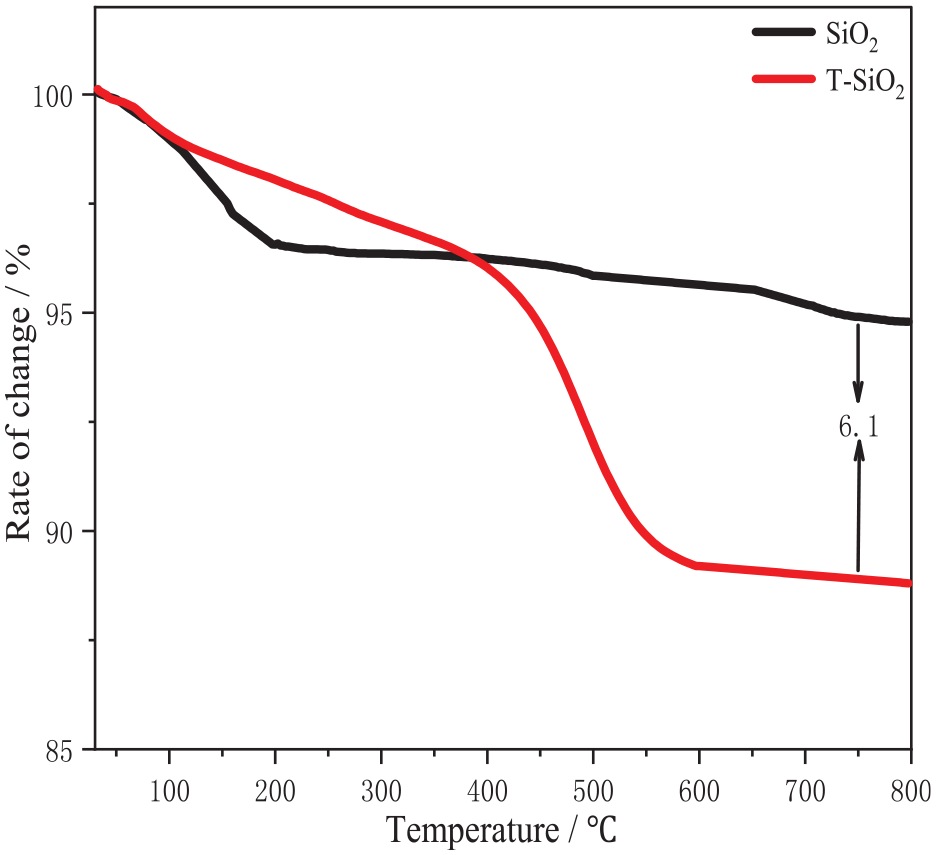
TGA curves of SiO2 before and after modification.
Hydrophobic Modification of TPU Films
We further investigated the hydrophobic modification of microporous TPU films with T-SiO2. This section explores the effects of T-SiO2 concentration and modification time on the hydrophobicity of TPU films to determine optimal modification conditions.
Effect of T-SiO2 Mass Fraction on the Film Properties
First, the effect of T-SiO2 mass fraction on the film properties was examined, with the results presented in Figures 8 and 9.
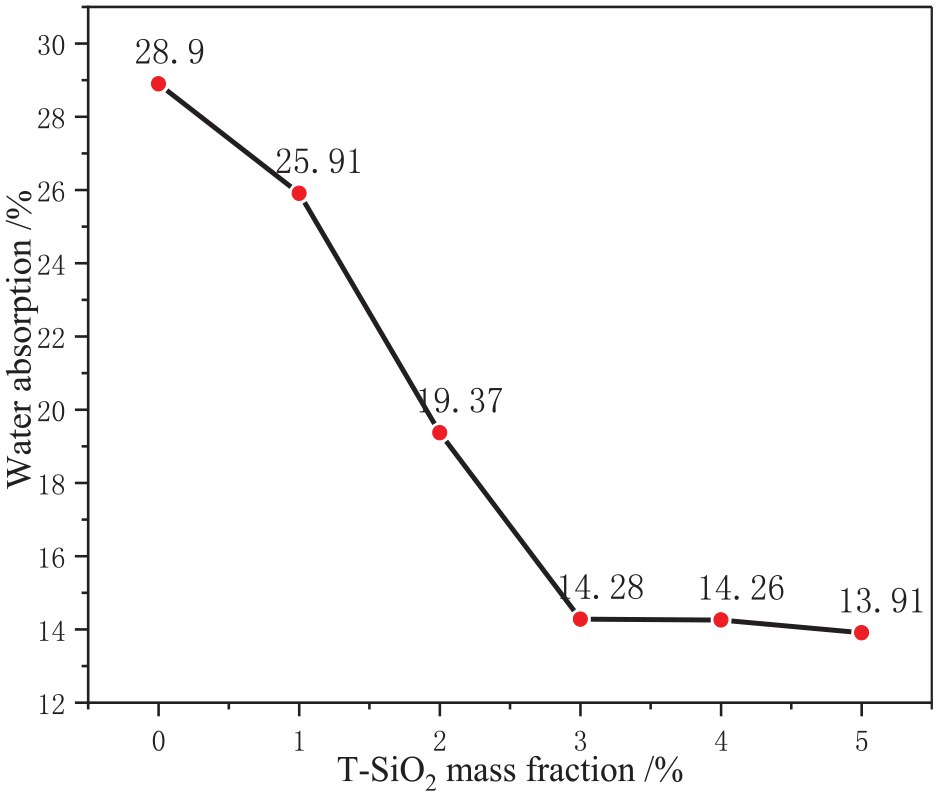
Effect of T-SiO2 mass fraction on the water absorption of the film.

SEM images of TPU films: (a) unmodified and (b–d) modified with 1%, 3%, and 5% T-SiO2.
As shown in Figure 8, with increasing mass fraction of T-SiO2, the water absorption rate of the film notably decreases. At a mass fraction of 3%, the water absorption rate reaches equilibrium, stabilizing at approximately 14%. This was primarily attributed to the increase in the hydrophobicity of the film surface with T-SiO2 mass fractions, leading to reduced water absorption. However, the excess of T-SiO2 results in the blockage of pores within the film because of the large specific surface area and small size of T-SiO2 particles, 21 as illustrated in Figure 9. Consequently, excessive T-SiO2 impedes the water vapor transmission of the film. To investigate this aspect in greater detail, the impact of the T-SiO2 mass fraction on the water vapor transmission was examined, with the findings presented in Figure 10.
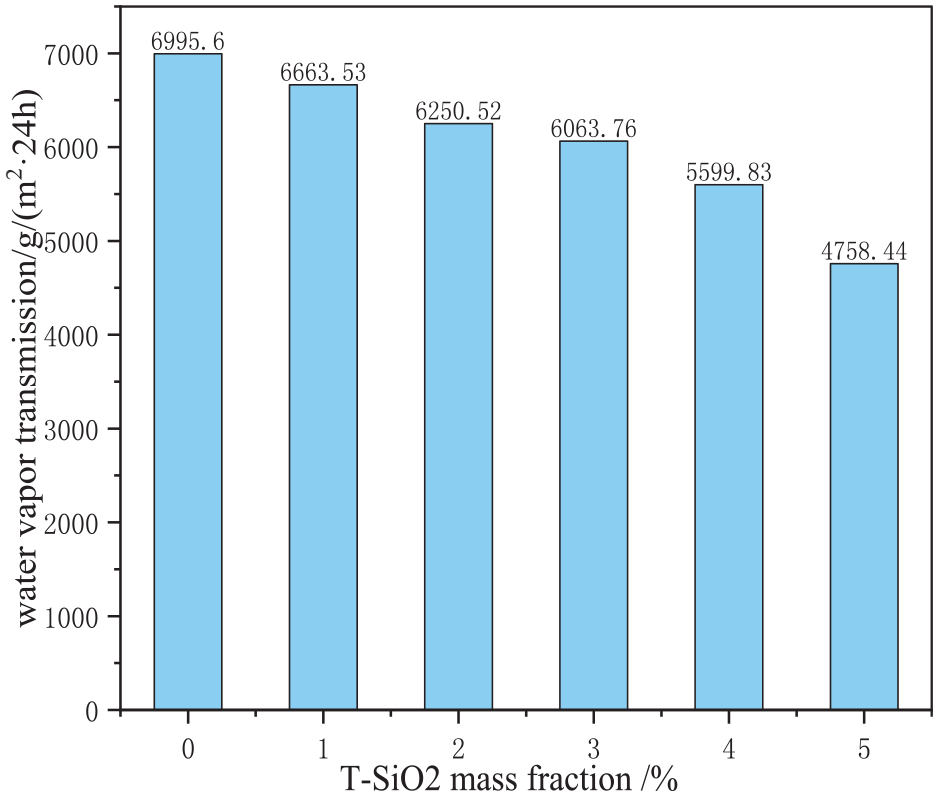
Effect of T-SiO2 mass fraction on the water vapor transmission of the films.
As shown in Figure 10, with increasing mass fraction of T-SiO2, the water vapor transmission of the film decreases, comprising 6063.76 g/(m2·24 h) at a mass fraction of 3%. Water molecules migrate through the TPU film, from the high-humidity side to the low-humidity side, through the adsorption–diffusion–desorption mechanism relying on hydrogen bonding and other intermolecular forces. 22 At low mass fractions, silica particles minimally affect the film permeability. In particular, at a T-SiO2 mass fraction of 3%, the permeability of the film is 6063.76 g/(m2·24 h). To a certain mass fraction of T-SiO2, the pores in the film remain permeable as T-SiO2 is deposited only on the surface of the film. However, the increase in T-SiO2 mass fraction beyond the saturation concentration substantially worsens water vapor transmission. Thus, a mass fraction of 3% was considered optimal.
Effect of Modification Time on Film Properties
After determining the effect of T-SiO2 mass fraction on the properties of the TPU film, we investigated the impact of modification time on the water absorption and moisture permeability of the films.
Figure 11 shows that with the extension of modification time, the water absorption rate of the film notably decreases. At a modification time of 4 h, the water absorption rate stabilizes at approximately 12%. This stabilization was primarily attributed to the attachment of T-SiO2 particles to the film surface, enhancing its hydrophobicity and reducing the absorption rate. However, prolonged modification time can lead to the blocking of the film pores by T-SiO2 particles (Figure 12), thereby compromising the water vapor transmission of the film. Hence, we also investigated the effect of the modification time on water vapor transmission, with the results presented in Figure 13.
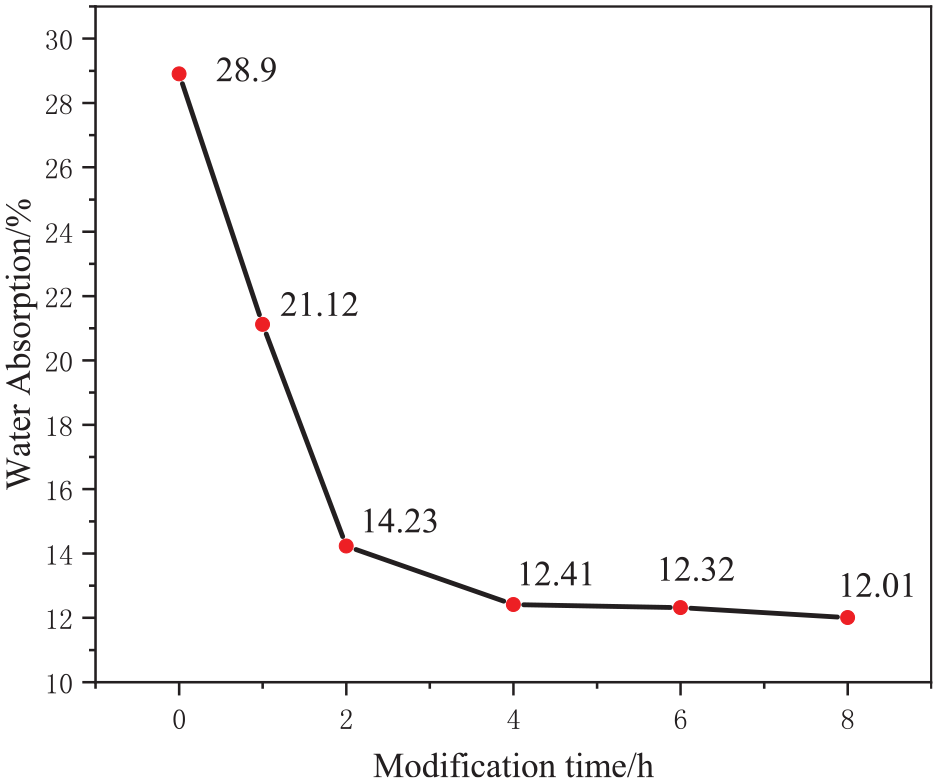
Effect of modification time on the water absorption of the film.

SEM images of films: (a) unmodified and (b–d) modified with T-SiO2 for 2, 4, and 8 h.
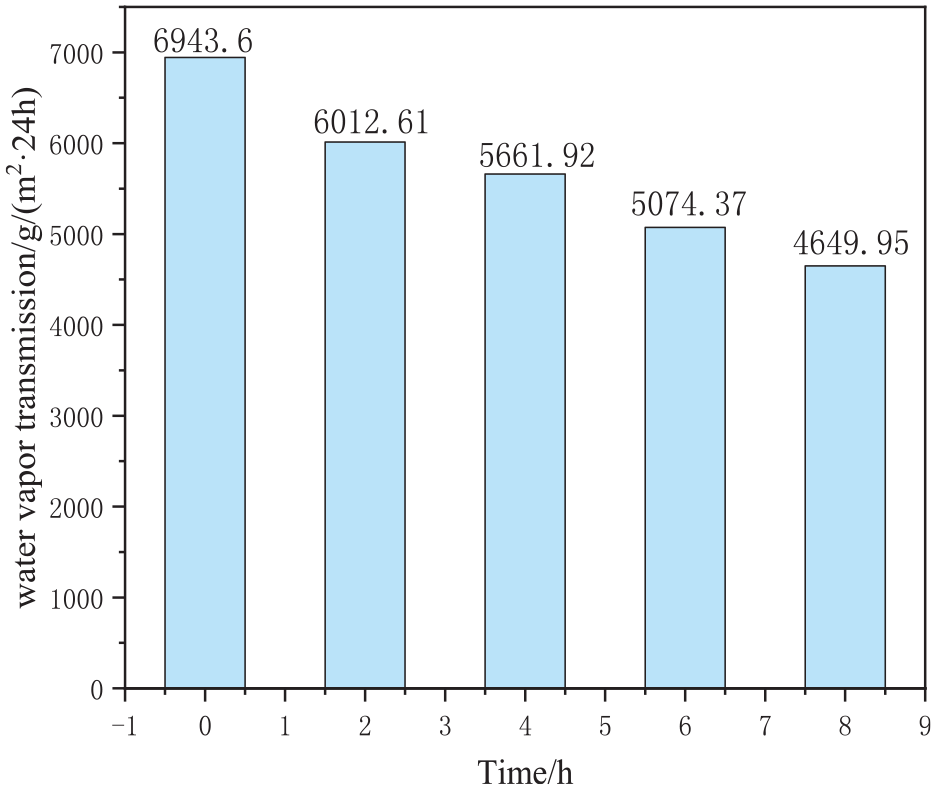
Effect of modification time on the water vapor transmission of the films.
As shown in Figure 13, the water vapor transmission of the films decreases with the extension of modification time. This decrease was primarily attributed to the reduction in the pore space within the film caused by prolonged modification. Specifically, at a modification time of 4 h, the water vapor transmission of the film is 5661.92 g/(m2·24 h). Thus, a modification time of 4 h was considered optimal.
Analysis of Other Properties of Modified TPU Films
Surface Hydrophobicity of TPU Films
To comprehensively evaluate the hydrophobicity of the modified TPU films, we conducted several experiments, including water drop dynamic impact performance tests and contact angle measurements.
The changes in the shape of water droplets and water flow on the film were captured using a high-speed camera, 23 with the test results shown in Figures 14 and 15.

Photographs of water droplets: (a) unmodified film, (b) modified film before contact, (c) during contact, (d) bouncing up, and (e) resting on modified film.

Photographs of water flow at 45° on: (a) unmodified and (b) modified films.
Owing to the surface structure and properties of hydrophobic materials, energetic water droplets infiltrate the surface, displacing air pockets beneath the surface layer and becoming pinned. 24 Figure 14 shows the morphological alterations in 5 μl water droplets dropped onto the film surface from a height of 5 cm. The droplets adhere to the surface of the unmodified film (Figure 14(a)). At the same time, water droplets do not adhere to the surface of the modified film; instead, they rebound, eventually settling in a spherical shape on the film surface after multiple bounces (Figure 14(b)–(e)), indicating reduced surface adhesive forces.
Figure 15 shows a continuous flow of water shot at an angle of 45° onto the surface of the unmodified and modified films. Prior to modification, water adheres to the film surface, whereas upon contacting the modified film, it swiftly recoils, rapidly advancing forward. Eventually, only a few spherical droplets remain on the film surface. This observation suggests that the modified film exhibits enhanced waterproof performance against continuous water flow. This improvement was attributed to the micro and nanostructure formed through the crosslinking of silica particles bonded with a silane coupling agent and the uncrosslinked particles. The hydrophobic surface of the film, generated by numerous hydrophobic groups on the surface of the silane coupling agent, contributes to the enhancement of the waterproof performance.
To quantify the hydrophobicity of the modified film, contact angle measurements were conducted. As shown in Figure 16, the water contact angle of the unmodified film is approximately 79.08°, whereas the contact angle of the modified film is 110.16°. This higher contact angle further confirms the enhanced hydrophobicity of the modified film, consistent with the dynamic impact performance observed in the high-speed camera tests.

Water contact angle on the surfaces of: (a) unmodified and (b) modified films.
Mechanical Properties
Figure 17 presents the stress-strain curves of the unmodified and modified TPU films. The stress–strain curves do not substantially differ, suggesting a minimal impact of the modification on the mechanical properties of the TPU film. This was primarily attributed to the physical nature of modification with silica, which preserved the film structure. 23
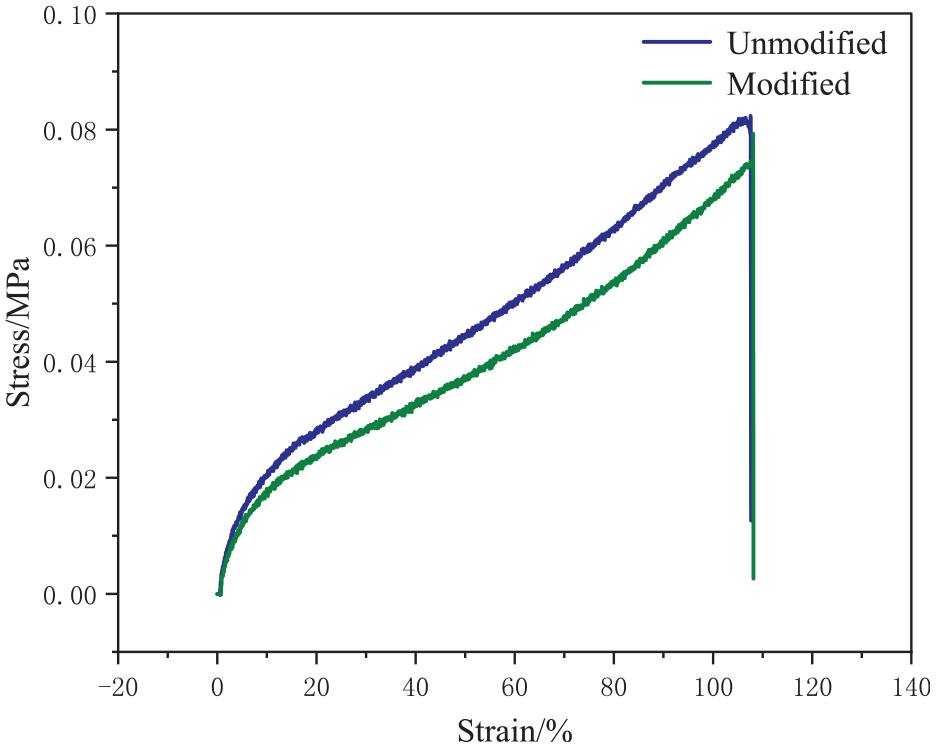
Stress–strain curves of the unmodified and modified films.
Comparative Analysis of the Waterproof Performance and Moisture Permeability of the Fabric Before and After Modification
The effect of film modification on the heavy rain resistance of breathable fabrics was investigated using the Bundestest apparatus. The water repellency of the samples was rated based on a reference sample at 1, 5, and 10 min. The water resistance grade and water absorption rate are presented in Table 1.
Storm protection rating and water absorption of the fabrics before and after film modification.
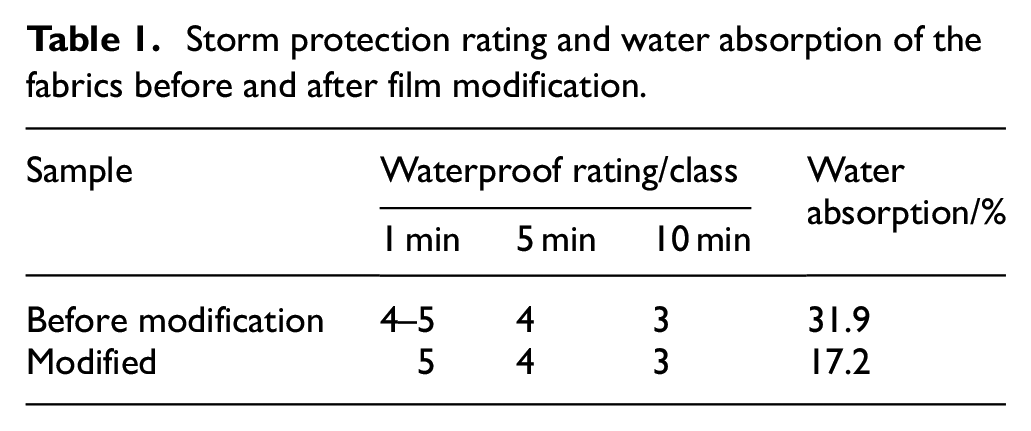
As indicated in Table 1, the waterproof grade of the fabric before and after film modification remains largely unchanged. This is because the waterproof grade of waterproof, breathable composite fabrics is primarily determined by the waterproof performance of the fabric substrate. At the same time, the water absorption rate of the modified composite fabric is lower by 14.7% than before the modification. This reduction demonstrates a significant improvement in the water absorption rate of the modified waterproof and moisture-permeable fabric under heavy rain conditions.
Conclusion
In this study, nanoscale silica particles were hydrophobically modified using silane coupling agents, and the modification process was optimized to prepare nanoparticles with excellent hydrophobic properties. These nanoparticles were then successfully applied to TPU films to significantly reduce their water absorption. The result revealed that under conditions of 8% KH550 and HDTMS concentrations, a temperature of 70°C, and a modification time of 2 h, the silane coupling agents successfully grafted onto the SiO2 surface, resulting in a substantial enhancement of the hydrophobicity of T-SiO2 nanoparticles, with a water contact angle reaching 138.58°. Subsequently, the TPU film was modified with T-SiO2 nanoparticles, with the optimal modification conditions being a T-SiO2 nanoparticles mass fraction of 3% and a modification time of 4 h. SEM images confirmed the successful attachment of T-SiO2 nanoparticles onto the TPU film. The modification process had minimal impact on the mechanical properties but its water absorption rate was lower by 16.49% than that before modification. Additionally, the water vapor transmission of the modified TPU film was 5661.92 g/(m2·24 h). Furthermore, when the modified TPU film was laminated with fabric, the water absorption rate of the composite fabric decreased by 14.7% compared to the unmodified film, demonstrating a significant improvement.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.