Abstract
In order to enhance the performance of cotton textiles in generating negative ions (NAI), germanium and pearl powder were utilized as additives, employing a straightforward “ultrasonic impregnation and drying” method to functionalize the cotton fabric surface. For practical application, a functional silane coupling agent (KH-560) was used to modify the fabric’s surface, thereby improving the binding force between the fabric and the functional powders. The study examined the morphology of the functional cotton fabric, NAIs release into the air, thermal stability, and moisture absorption capabilities. Experimental results demonstrated a significant enhancement in NAIs release performance, with a 15-fold increase compared to untreated cotton fabric when functional powders were added. Incorporating germanium powder led to a 2.3-fold augmentation in pearl NAI release, effectively addressing the low pearl NAI release issue. Additionally, the presence of pearl powder, when combined with germanium powder, synergistically enhanced moisture absorption and thermal stability performance, surpassing those of control samples. The developed functional cotton fabric exhibits significant potential for applications in household, healthcare, and clothing textiles, marking a promising advancement in textile functionalization for enhanced performance.
Introduction
Negative air ions (NAI) are oxygen atoms in the air that carry additional negative charges, such as O2- (H2O) n , OH- (H2O) n , and CO4- (H2O) n . These ions are abundant in pristine natural environments like forests, waterfalls, and lakes, especially after heavy rain. Additionally, specific minerals can release NAIs when subjected to external stimuli like heat or electricity. NAI are known as “vitamins” in the air due to their positive impact on human health.1–3 Research has shown that NAI have remarkable effects on various health issues, such as boosting the immune system,4,5 anti-inflammatory effects, 6 and combating bacteria and viruses.5,7 NAI have also been associated with improvements in mental health conditions like depression and insomnia and have shown potential in cancer treatment.4,8,9 Mira’s 10 study on urogenital infections revealed that long-term exposure to NAIs resulted in significant symptom improvement, showcasing their therapeutic potential. Chen et al. 11 demonstrated that incorporating negative ions into germanium-polyamide six composite fibers enhanced the fibers’ antibacterial properties, providing promising applications in functional fabrics. These findings underscore the importance of NAIs for human health. However, the amount of NAIs in polluted urban environments, closed and conditioned rooms, moving cars and aircraft, and near electronic devices such as TVs and computers falls dramatically to several tens per cm3 or even to a complete absence of ions. 7 Indeed, the development of functional fabrics with NAIs for daily life has become a crucial focus in the textile industry. Therefore, utilizing natural and non-toxic minerals with negative ion-release capabilities to enhance the NAI performance of textiles has emerged as a meaningful direction. Cotton fabrics, widely used daily and in close contact with the human body, are ideal for incorporating negative ion-releasing capabilities. This study proposes a method to enhance fabrics’ negative ion release performance by integrating multiple functions into natural minerals.
Pearl, a natural material composed mainly of calcium carbonate and organic substances (including amino acids, trace elements, vitamins, and peptides), boasts multifunctional properties such as UV resistance, antistatic properties, moisture absorption, and perspiration.12,13 Traditionally, pearl has been widely used in traditional Chinese medicine, beauty, and the treatment of various diseases. However, its potential in manufacturing functional textiles has garnered significant attention.13,14 It is an ideal candidate for producing versatile textile materials. Recent studies by Yu et al. 15 have delved into the functional impact of pearl when incorporated into electrospinning nanofibers made from polyacrylonitrile. The outcomes revealed that pearl confers various beneficial characteristics to polyacrylonitrile fibers, opening up broader applications for these materials. Dai et al. 16 electrospinning PLA/pearl powder nanofibers were explored for their potential in bone tissue engineering. The inclusion of pearl in the nanofibers enhanced the biocompatibility of PLA, leading to improved cell proliferation and showcasing significant promise in bone tissue engineering applications. These findings underscore the immense potential of pearl in advancing the development of multifunctional textile materials.
Moreover, pearl exhibits NAIs release capabilities, potentially enhancing human health when used in wearable fabrics. However, the NAI release performance of pearl textile materials remains limited, and the current performance may not be optimal. Hence, germanium, which possesses excellent NAI properties, augments the deficiency in pearl’s NAI capabilities. Germanium, a non-toxic semimetal element, has garnered significant attention for its exceptional NAIs release capabilities. 17 The atomic structure of germanium involves four unbalanced electrons orbiting irregularly around its nucleus. This distinctive arrangement grants germanium thermoelectric and piezoelectric characteristics akin to tourmaline ones. When subjected to temperatures above 32°C or pressure, it generates many NAIs.11,18,19 Leveraging germanium’s exceptional atomic structure can effectively enhance pearl’s negative ion release capabilities. By harnessing the unique properties of pearl and combining the properties of these two natural materials, researchers and manufacturers can pave the way for innovative and health-promoting textile solutions.
This study used a simple and robust “ultrasonic impregnation and drying” method to prepare functional textiles that effectively release NAIs. In the end, a total of four samples were prepared: cotton, pearl/KH-560/cotton (PKC), germanium/KH-560/cotton (GKC), and pearl/germanium/KH-560/cotton (PGKC). Simultaneously, the influence of germanium powder on the NAI properties of pearl was thoroughly investigated, and the prepared functional fabric’s surface morphology, chemical structure, and wettability were systematically studied.
Experimental Section
Materials
Pearl powder, with calcium carbonate (CaCO3) as its primary component, was sourced from Licheno (Shanghai) New Material Technology Co. Ltd. Germanium powder was obtained from Shanghai Macklin Biochemical Technology Co. Ltd. The silane coupling agent solution (KH-560) with a mass fraction of 99% was also supplied by Shanghai Macklin Biochemical Technology Co. Ltd in China. The substrate used in this study was bleach-white woven cotton fabric, characterized by a plain weave fabric structure, a density of 130 warp threads per centimeter by 130 weft threads per centimeter, and a gram weight of 60 g/m2, which was provided by Yinjing Medical Technology Co. Ltd in China. All reagents utilized in this study were of analytical grade.
Surface Modification on Cotton Fabric
A 15 ×15 cm piece of cotton fabric was initially immersed in an acetone solution for a duration of 2 h, followed by thorough rinsing with deionized water and subsequent drying in an oven at 80°C for 2 h. Subsequently, the fabric was immersed in a KH-560 solution with a material-to-liquor ratio of 1:40 at 70°C for 1 h. This surface modification process was carried out to enhance the adhesion between functional nanoparticles and the fabric surface. The cotton fabric, post-modification with the KH-560 solution, was referred to as “KH-560/Cotton” (KC).
Preparation of Nanoparticle-impregnated Cotton Fabric
To enhance the dispersion characteristics of the nanoparticles while preserving their material activity, a planetary ball mill was employed to grind pearl and germanium nanoparticles. The agate ball mill was filled to 80% capacity, and the grinding process took place at room temperature for 1.5 h. Subsequently, the ground powders with a total mass fraction of 0.5% (see Table 1) were dispersed in deionized water and subjected to ultrasonic stirring for 1 h at room temperature. The modified cotton fabric (KC) was then immersed in the dispersed solution and subjected to ultrasonic stirring for 1 h at 50°C. Finally, the fabric was dried in an oven at 80°C for 1 h. This procedure yielded three distinct types of nanoparticle-impregnated cotton fabrics, denoted as PKC, GKC and PGKC. The preparation process for PGKC is illustrated in Figure 1.
Weight of pearl powder.

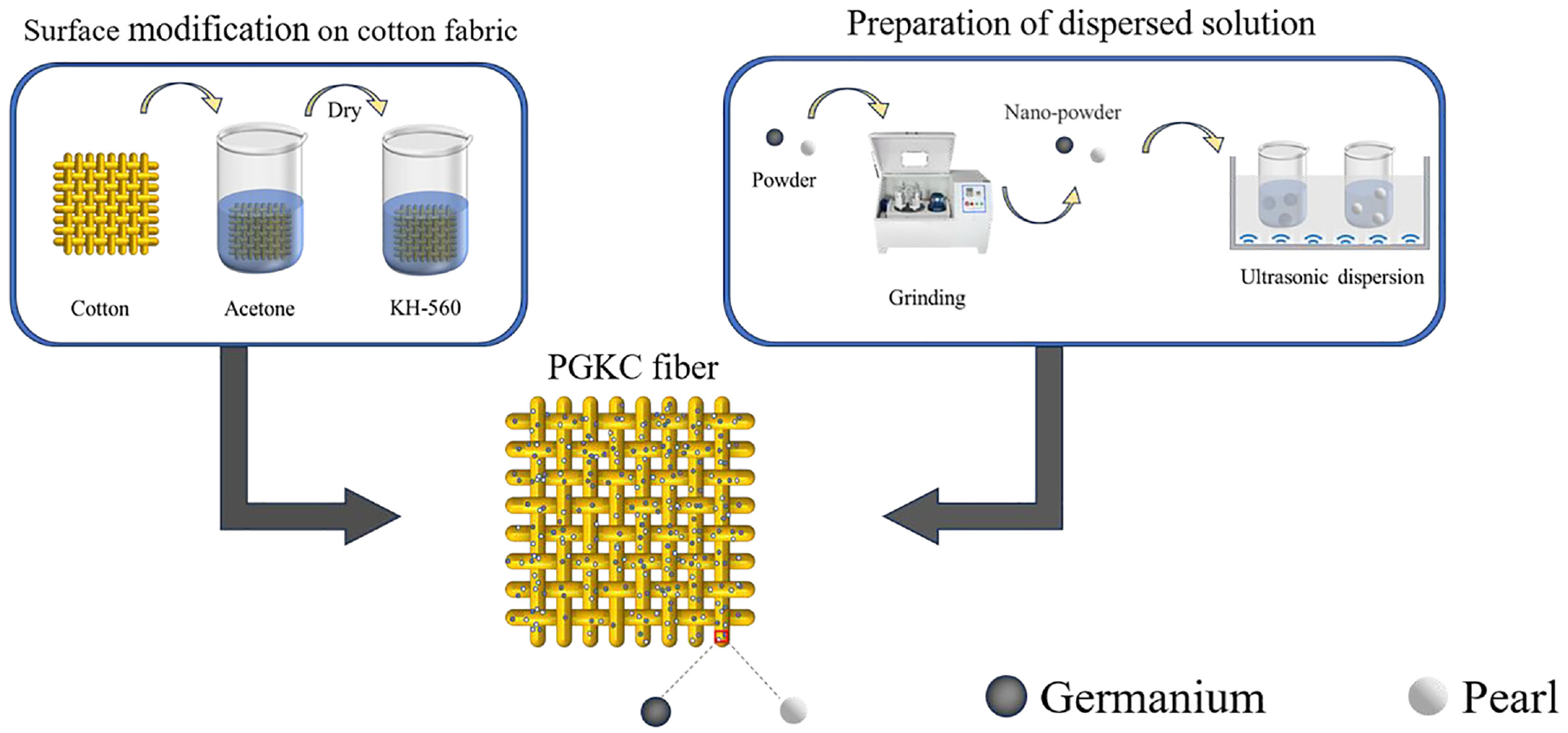
The schematic diagram for the preparation process of PGKC fabric.
Characterization
The samples’ surface morphologies were analyzed using a scanning electron microscope (SEM, Gemini 300, ZEISS, Germany). The samples’ chemical structure was analyzed using Fourier transform infrared spectroscopy (FTIR; a range of 500–4000 cm−1; PerkinElmer Spectrum Two, USA). The samples’ wettability was characterized using an optical contact angle measuring instrument (KRUSS DSA30, Germany). Every sample was measured five times, and the average result was utilized to make judgments. Thermal stabilities of the samples were analyzed using a TGA-Q500 thermogravimetric analyzer (TA, USA) from 30 to 900°C at a heating rate of 10°C/min under a nitrogen atmosphere. According to GB/T 30128-2013, the samples’ negative ion emission quantity was measured using a negative ion detection tester (FFZ471). The testing was conducted at an ambient temperature of 25°C and a relative humidity of 50%. Each sample was tested five times, and the average value was calculated. Additionally, friction was employed as an auxiliary method during the measurement process.
Results and Discussion
Morphology Analysis
The SEM images in Figure 2 show cotton fibers after different impregnation treatments. Figure 2(a) shows untreated cotton fibers appearing as flattened ribbon-like structures with irregular wrinkles, a rough surface, and no impurities. After treatment with KH-560, the fiber surface becomes smooth with regular wrinkles, as shown in Figure 2(b). Compared with Figure 2(a) and Figure 2(b), it can be clearly observed in Figure 2(c) that after impregnation of cotton fibers with pearl powder, the wrinkles on the surface of the fibers disappeared, and the surface of the fibers showed irregular protrusions. This differs significantly from the attachment effect of germanium powder shown in Figure 2(d), where the germanium particles adhere to the cotton fibers in a granular form. This phenomenon may be attributed to pearl particles’ significantly smaller particle size compared with germanium particles. As shown in Table 2, the germanium and pearl particle sizes were decreased from 74 and 2.5 μm to 2 and 0.16 μm after grinding, respectively. Pearl nanoparticles are more prone to forming a dense coverage layer on the fiber surface. At the same time, germanium particles, due to their larger size, tend to have a more scattered distribution on the fiber surface, resulting in uneven and discontinuous coverage. Additionally, because pearls are composed of multiple components, chemical bonds are formed between pearl nanoparticles and the cotton fiber surface and between different pearl nanoparticles during the impregnation process. This can manifest as the formation of agglomerates between the fibers. On the other hand, germanium is a stable single-element material that forms chemical bonds only with the cotton fiber surface. The affinity of germanium for organic substances, iron, copper, and other materials may play a significant role in attracting pearl nanoparticles and causing them to accumulate around the germanium nanoparticles. 17 This phenomenon can be observed in Figure 2(e), where the clustering or accumulation of pearl nanoparticles is prominent near the germanium nanoparticles.

SEM images at varied magnifications: SEM images captured at different magnifications, denoted as (I) 1000× and (II) 20,000×, showcasing the surface morphologies of (a) cotton, (b) KC, (c) PKC, (d) GKC, and (e) PGKC samples.
The particle size of nanoparticles before and after grinding.

Chemical Structure
Figure 3 presents the FTIR spectra of different samples. In Figure 3(a), the FTIR spectrum of the untreated cotton exhibits absorption peaks at 1030 and 3340 cm−1, corresponding to the stretching vibrations of –OH and C–O bonds, respectively. 20 The spectrum of KH-560 displays characteristic peaks at 1169 and 773 cm−1 representing the stretching vibration of Si–O bonds and the bending vibration of C–C bonds, respectively. The 2976 and 2885 cm−1 peaks correspond to −CH2 and −CH3 groups. 21
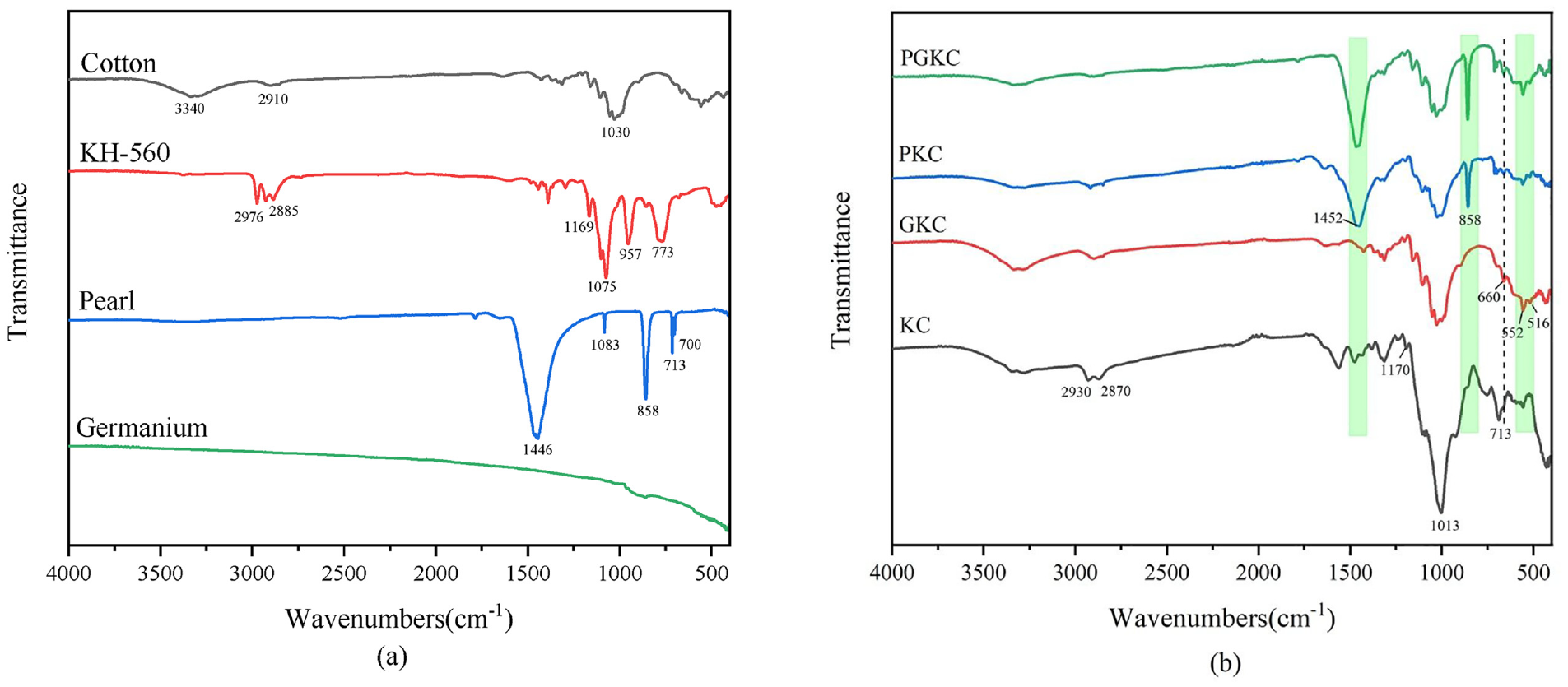
Fourier transform infrared (FTIR) spectra analysis of (a) cotton, KH-560, pearl, germanium and (b) KC (KH-560/cotton), GKC (germanium/KH-560/cotton), PKC (peal/KH-560/cotton), PGKC (pearl/germanium/KH-560/cotton).
Pearl powder consists primarily of calcium carbonate, with the surface layer composed of aragonite-type calcium carbonate and the core consisting of calcite-type calcium carbonate.22,23 The FTIR spectrum of pearl powder exhibits absorption peaks corresponding to the FTIR spectra of aragonite-type CO32−. Precisely, the peak at 1446 cm−1 corresponds to the υ3 antisymmetric stretching vibration, the peak at 1083 cm−1 corresponds to the υ1 symmetric stretching vibration, the peak at 858 cm−1 corresponds to the υ2 out-of-plane bending vibration, and both of the peaks at 713 and 700 cm−1 correspond to the υ4 in-plane bending vibrations, respectively.15,16
In Figure 3(b), the FTIR spectra of PKC and GKC exhibit peaks at 2930, 2870, and 1170 cm−1, resulting from the modification of cotton by KH-560. Compared to KC, the specific peak observed at 516 cm−1 in GKC represents the stretching vibration of Ge. Meanwhile the peaks at 552 and 660 cm−1 correspond to the bending and stretching vibrations of Ge-O, respectively. 24 Similarly, in PKC, the distinctive peaks at 858 and 1452 cm−1 demonstrate the successful attachment of pearl particles compared to KC. In the FTIR spectrum of PGKC, all the observations mentioned earlier can be observed, compared to PKC, with varying degrees of enhancement in the characteristic peaks at 660, 552, and 516 cm−1. All of them can indicate the successful attachment of both pearl and germanium nanoparticles to cotton fibers.
Mechanical Properties
The stress–strain curves of the various samples during the stretching process are shown in Figure 4. These curves are due to the two-step deformation mechanism caused by the weave structure of the fabric: the first deformation involves displacement and sliding of the fibers, and the reduction of the fabric stiffness due to shear deformation of the curl (i.e. at the intersection of vertical fibers), followed by the fibers being stretched until they break. 25 As shown in Figure 4, the fiber strength decreases after the fabric is impregnated with KH-560, while it increases after coating with pearl and germanium nanoparticles. The reduction in PGKC may be due to the degradation of the cotton fibers during repeated drying and reduction at higher temperatures. 26 This is a complex phenomenon, which may be related to the uniform aggregation of nanoparticles on the fiber surface. From the perspective of mechanical analysis, the uneven aggregation of nanoparticles on the fiber surface is considered to be a stress concentration point, which may lead to premature failure of the treated cotton.27,28
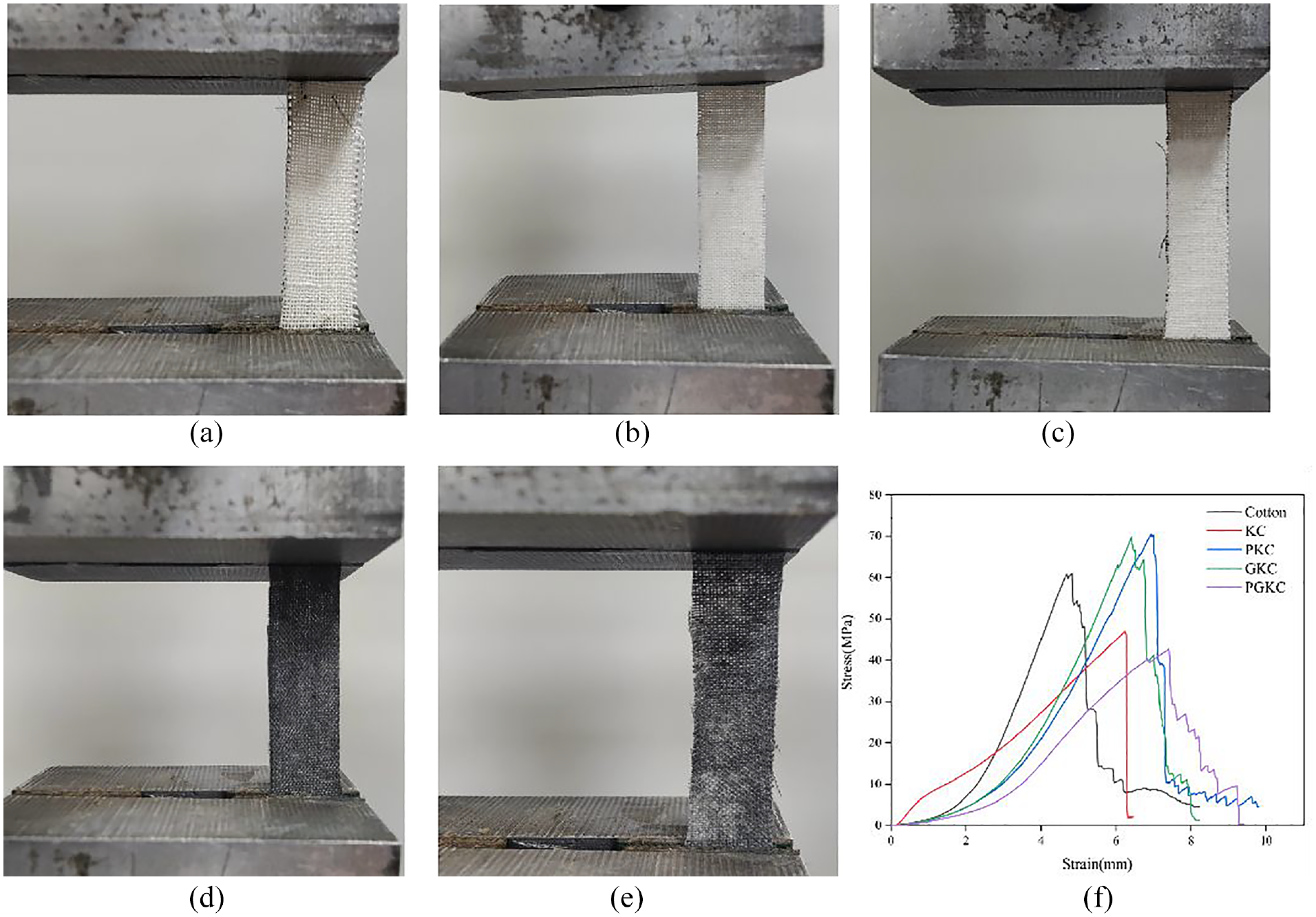
Mechanical properties of (a) cotton, (b) KC, (c)PKC, (d) GKC, (e) PGKC and (f) stress-strain plots.
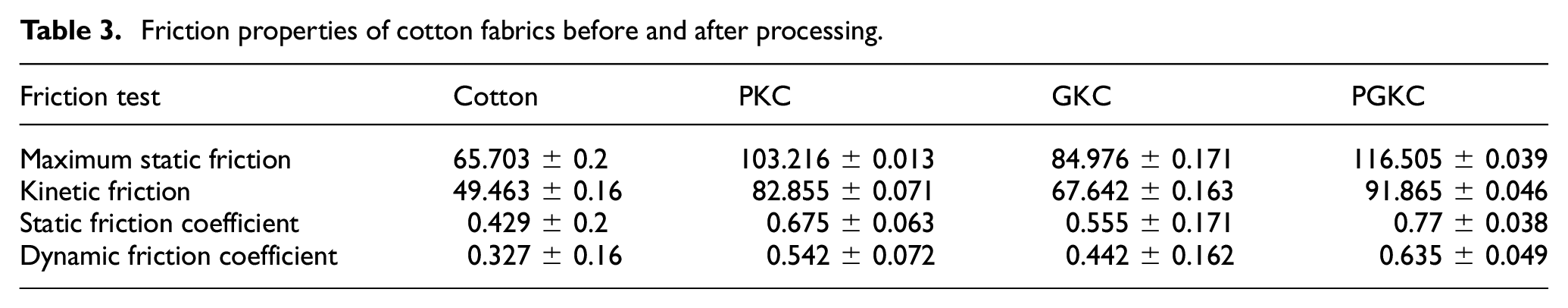
The flexibility of the sample is shown in Figure 5, and the fabric treated with KH-560 is completely in a straight state with a stiff hand feel, which is related to the properties of KH-560. After the attachment process of pearl and germanium nanoparticles, a dehydration condensation reaction occurs between KH-560 and the nanoparticles, resulting in a significant increase in the softness and flexibility of the cotton fabric (Figure 6). In addition, it can be seen from Table 3 that the friction performance of the fabric also increases with the attachment of nanoparticles. This is related to the roughness of the fabric surface. It can be seen from SEM that, compared to other cotton fabrics, PGKC has a higher degree of roughness.
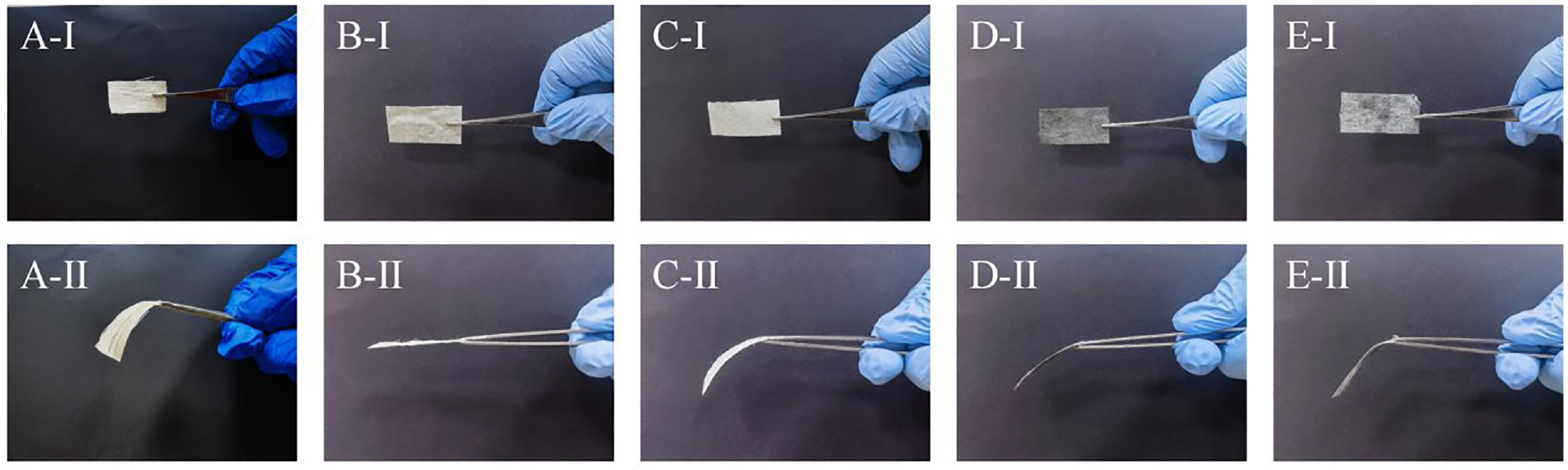
Flexibility of various samples: (a) cotton, (b) KC, (c) PKC, (d) GKC, and (e) PGKC.
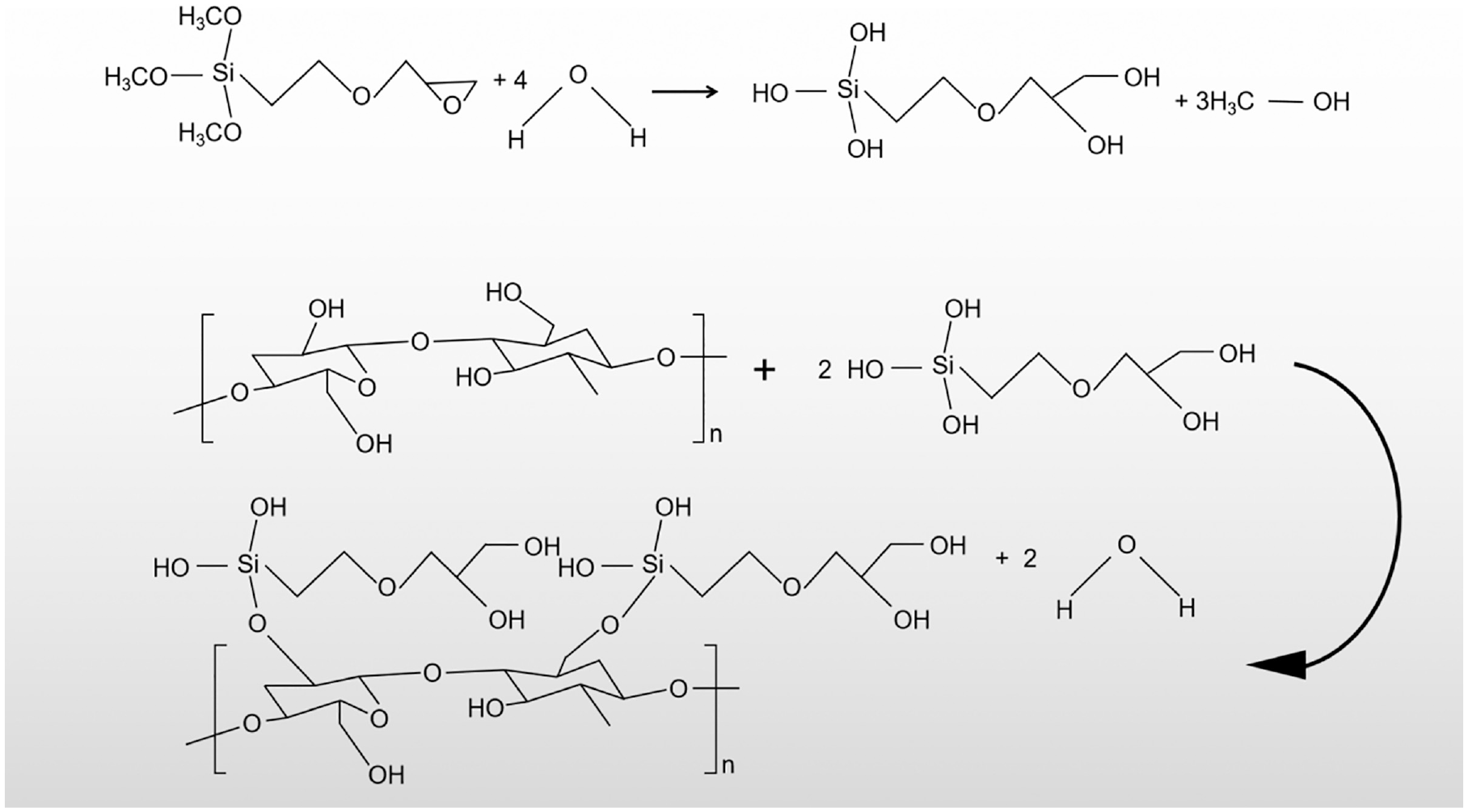
Dehydration condensation reaction between cotton and KH-560.
Friction properties of cotton fabrics before and after processing.
Wettability
Figure 7 illustrates the water contact angles of the different samples at different time intervals. In the case of untreated cotton fibers, the water contact angle remains relatively unchanged within a 30 s interval. This phenomenon is attributed to using a plain weave cotton fabric, exhibiting a smooth and even surface and closely packed and dense inter-fiber structures, which contribute to lower surface energy and consequent impairment of moisture absorption properties. Another critical factor is the melting of cotton wax during the drying process, as cotton wax has a melting point between 60°C and 80°C and exhibits strong hydrophobic properties. When subjected to an 80°C drying process, the melted cotton wax continuously spreads across the fiber surface, forming a cohesive coating that diminishes the wetting capability of the cotton fibers. This hydrophobic layer further impedes water penetration, resulting in the observed absence of a significant change in the water contact angle.
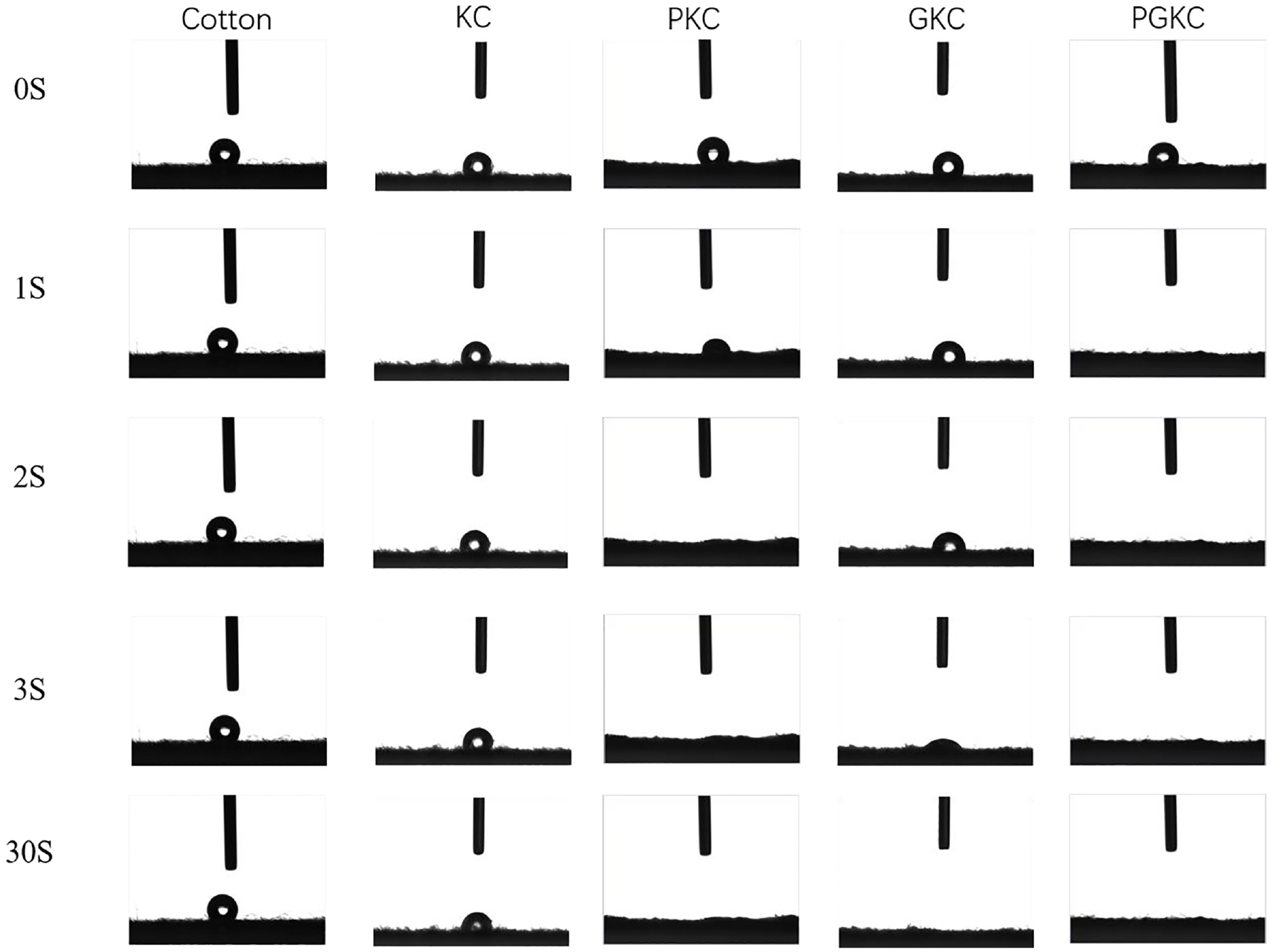
Water contact angle (WCA) analysis: provides the water contact angle measurements for different samples, including cotton, KC, PKC, GKC, and PGKC.
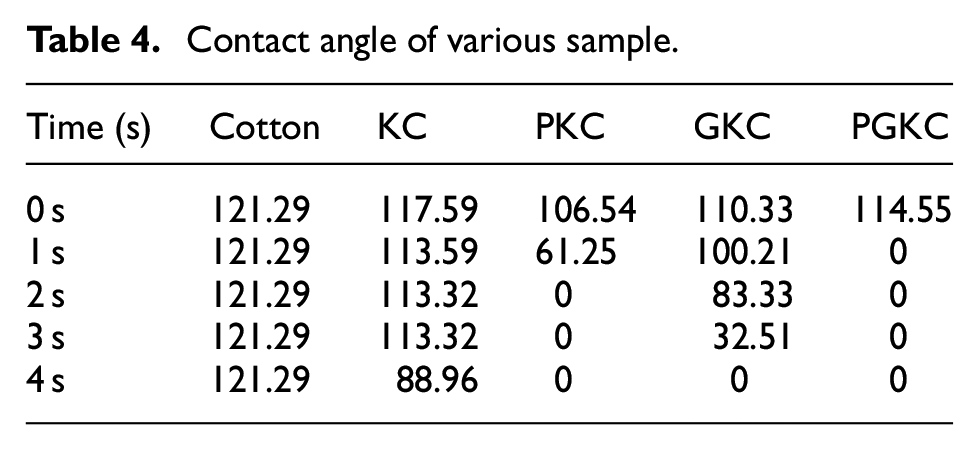
However, after the treatment with KH-560, there is a slight improvement in the moisture absorption performance. This is attributed to the presence of hydrophilic −OH groups on the KH-560 molecules. After the attachment of pearl and germanium nanoparticles, the moisture absorption performance of the fibers undergoes a significant improvement. 29 The PKC sample achieves complete absorption within 2 s, while the GKC sample shows only a small amount of residual water droplets after 3 s. The improvement in moisture absorption is not only attributed to the excellent moisture-wicking ability of the nanoparticles themselves but also likely due to the melting of cotton wax during the ultrasonic impregnation process at 60°C. Additionally, the significant increase in the attachment of nanoparticles on the cotton fiber surface dramatically enhances the roughness of the fiber surface, contributing to the enhanced moisture absorption properties. The moisture absorption performance of PKC is superior to that of GKC, primarily due to the pearl nanoparticles mainly consisting of CaCO3, which exhibit excellent hydrophilic properties, further enhancing the moisture absorption performance of PKC.
Simultaneously, in Figure 7, it is evident that the moisture absorption performance of PGKC is notably superior to those of the other samples. This accumulation of pearl nanoparticles around the germanium nanoparticles can have interesting effects on the overall performance and characteristics of the composite material. It may lead to increased interparticle interactions, surface modifications, or material property changes (Table 4).
Contact angle of various sample.
Washing Durability
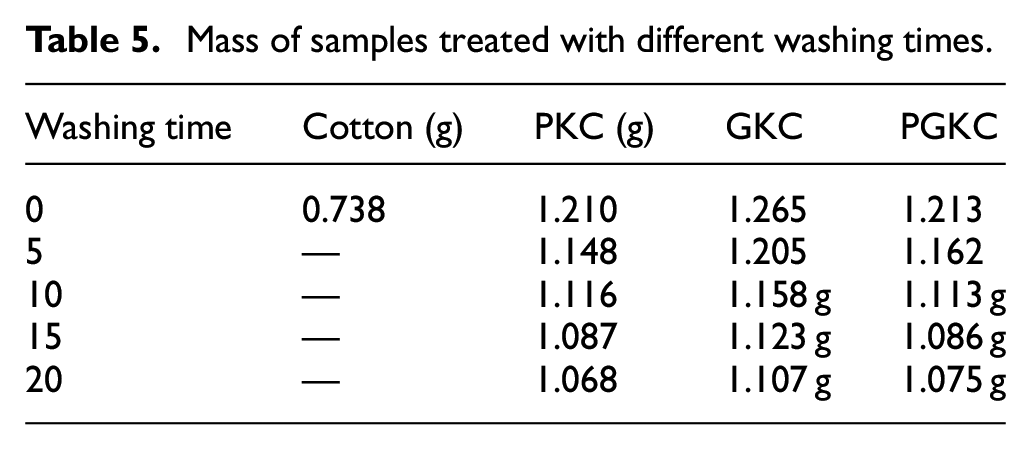
The changes in fabric mass after washing are shown in the table. After 10 washes, the mass of the treated cotton fabric did not change much. As the number of washes increased, the mass of the cotton fabric began to decrease significantly. Even so, it was still more than 70% of the original mass. The rapid reduction may be due to the shedding of nanoparticles that were not firmly attached after washing. After the shedding of this type of nanoparticle, the remaining nanoparticles have a strong chemical bond connection, the mass begins to stabilize, and the mass loss rate begins to decrease (Table 5).
Mass of samples treated with different washing times.
Thermal Stabilities
Figure 8 presents the thermogravimetric (TG) curves of cotton, KC, PKC, GKC, and PGKC. The thermal decomposition behavior of the samples can be divided into three stages.
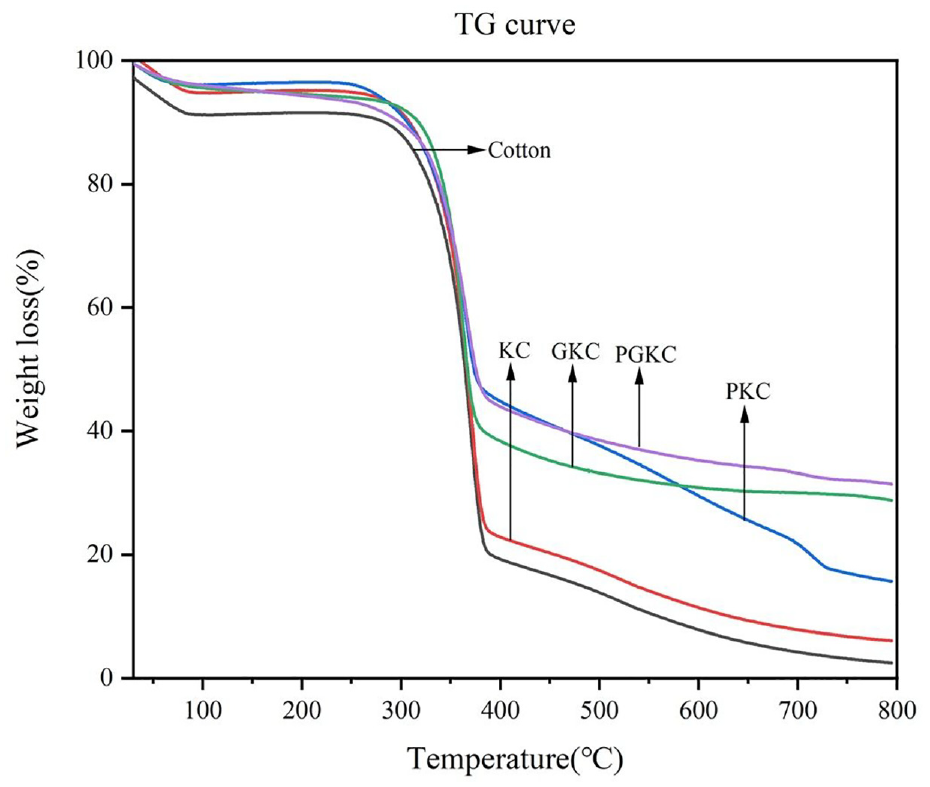
Thermogravimetric (TG) curves of different samples.
The first stage occurs from room temperature to 250°C, and the leading weight loss during this stage is attributed to the evaporation of moisture, the decomposition of bound water between materials, and the decomposition of unstable oxygen-containing functional groups in the samples. 30
The second stage occurs from 250 to 500°C, during which the decomposition of oxygen-containing functional groups such as −COOH, −C=O, and −OH occurs. We can observe rapid thermal decomposition for each sample in the TG curves. 29 The rapid decomposition temperature onset for PKC is around 320°C, lower than that for the other samples. This stage corresponds to the thermal decomposition of organic matter in the pearl, which can be attributed to the overall loss of organic matter
The third stage is the high-temperature carbonization, ranging from 500°C to 800°C. At this stage, most of the samples will be carbonized, and the temperature rise minimally impacts the mass loss of the remaining residues. As shown in Figure 8, at this temperature range, the mass residues of KC, PKC, GKC, and PGKC are higher than those of cotton due to the excellent thermal stability of pearl and Ge, indicating that surface modification effectively enhances the thermal stability of cotton. Additionally, around 600°C, there is a rapid thermal decomposition behavior for pearl, which is caused by the thermal decomposition of CaCO3 in the pearl powder.
Negative Air Ions Releasing Properties and Mechanism
Figure 9 shows the release of NAIs after the cotton fabric was impregnated with different powders. Previous research indicates that an NAI release exceeding 500/cm3 meets health requirements, and reaching 1500/cm3 corresponds to good air quality. 31 After being coated with pearl and germanium particles, the NAI release of the cotton fabric significantly increased, reaching 800 and 1980/cm3, 6.7 times and 16.5 times that of untreated cotton, respectively.
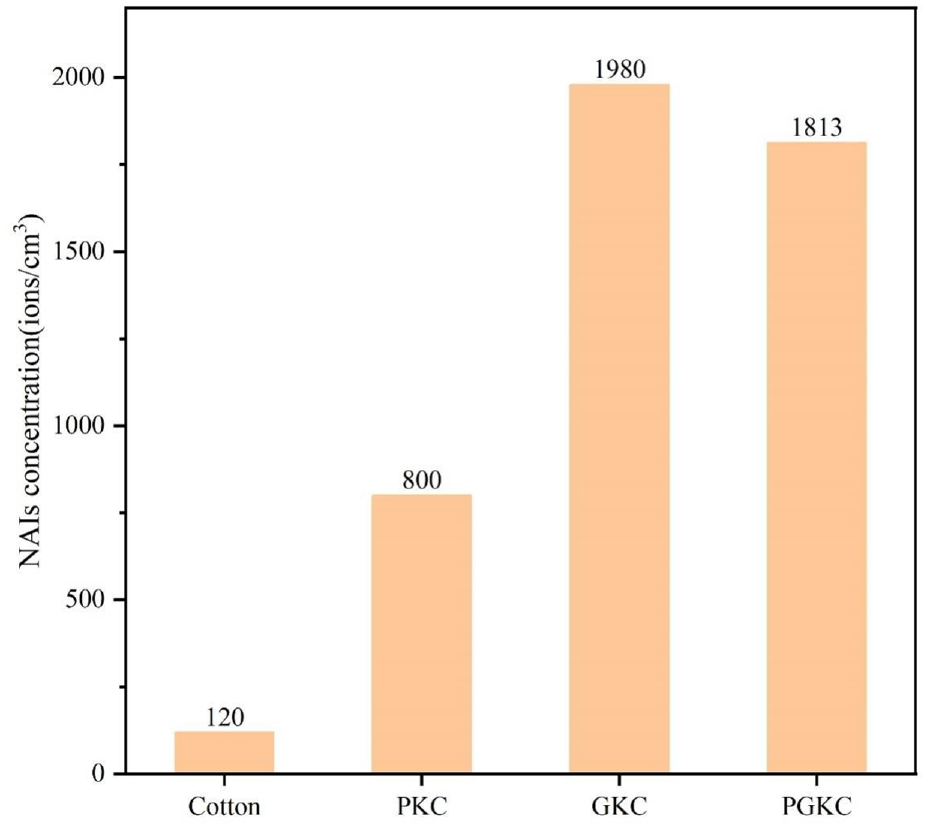
Negative ion emission of cotton fabric.
The exceptional NAI release performance of germanium contributes to the higher NAI release in GKC than in PKC. The majority content of pearl is CaCO3, which results in a slightly lower negative ion release performance, but it still meets the minimum standard for promoting human health. During the preparation of PGKC, some cotton fibers form chemical bonds with pearl nanoparticles, and the remaining part forms chemical bonds with germanium particles. This reduces the amount of germanium particles adhering to the fabric compared to GKC, decreasing NAI release.
Figure 10 illustrates the NAI release mechanism of the particles. When the treated fabrics are subjected to a changing temperature or compression, the electrons outside the nuclei of the nanoparticles escape their original orbits and become free electrons. Consequently, this generates an electrostatic field when many free electrons accumulate. As the electric field intensifies, the electrostatic voltage ionizes the surrounding air molecules and produces numerous negative ions. These negative ions then combine with neutral air molecules in the presence of water molecules to form NAIs. 32
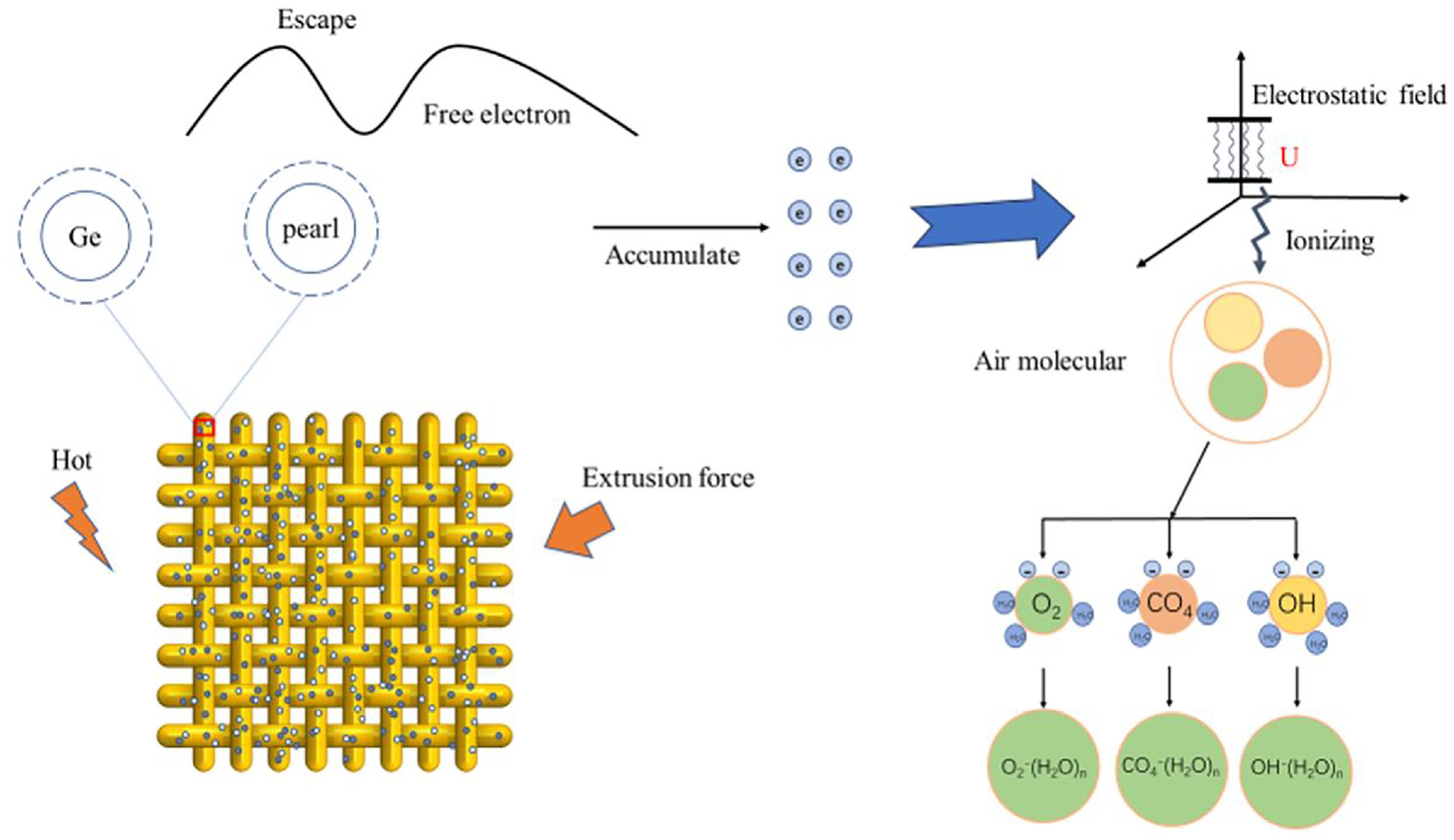
Schematic of the negative air ion release mechanism.
The smaller particle size corresponds to enhanced surface activity, contributing to increased external electrons and electrostatic voltage activity. This, in turn, leads to a higher level of air molecule ionization and NAI generation. 11
Conclusions
In summary, this paper employed a simple loading technique known as the “ultrasonic impregnation and drying method” to create a functional cotton fabric with two natural mineral materials (pearl and germanium) attached to its surface to enhance NAI production. The cotton fibers were modified using KH-560. Scanning electron microscopy analysis confirmed the successful attachment of both types of nanoparticles to the fabric. The introduction of nanoparticles significantly increased the release of NAIs from the cotton fabric. Notably, germanium remarkably enhanced pearl, boosting NAI release from 800 to 1813 ions/cm3 when germanium nanoparticles were added. This represented a substantial increase, 6.7 and 15 times that of the cotton fabric. This outstanding promotion also had a positive impact on the thermal stability of the functional cotton fabric.
Moreover, a synergistic interaction developed between pearl and germanium, effectively enhancing the moisture absorption capabilities of germanium. As a result, PGKC (pearl, germanium, KH-560 coated cotton) exhibited exceptional moisture absorption performance. Consequently, the unique properties of KH-560, pearl, and germanium offer promising opportunities for developing functional textiles in wearable health products, medical applications, and home and outdoor applications.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Performance and Mechanism Study of Multifunctional Pearl Fiber (Grant No. 2021-FZ-011), National Innovative Training Program for College Students (Grant No. 202210856030).
