Abstract
As an important high-tech anti-counterfeiting material, fluorescent fiber has attracted great interest from researchers in the textile field. In order to endow chitosan (CS) with good fluorescence for the preparation of anti-counterfeiting fibers, various amounts of fluorescent molecules—perylene tetracarboxylic acid (PTCA)–were used to label CS to prepare a series of CS-perylene with different degrees of labeling (DL). Then, nanofiber membranes were produced by electrospinning using fluorescent CS/PVA blend spinning solutions. The effects of the DL on application performance of the CS-perylene were studied. It was found that labeling the appropriate amount of perylene unit onto CS was an effective way to endow the CS with good fluorescence properties. With the increase in the DL of the CS-perylene, its fluorescence intensity increased initially, reached a maximum when the DL was 0.1718 mol%, and then decreased. At a DL of 0.1718 mol%, the CS-perylene/PVA nanofiber membrane could emit bright green fluorescence. Meanwhile, the labeling of the perylene unit did not bring about adverse effects on the surface morphology of the electrospun CS-perylene/PVA nanofiber membrane. The fluorescent CS-perylene nanofibers overcome many shortcomings of the commonly used fluorescent fibers and show great potential for use in the anti-counterfeiting field.
Fluorescent fibers have become one of the most commonly used anti-counterfeiting materials today and have been widely applied in products such as banknotes, passports, stamps, securities, and high-grade garment fabrics.1–3 Fluorescent anti-counterfeiting fibers are mainly synthetic ones such as polypropylene (PP), polyethylene terephthalate (PET), and polyacrylonitrile (PAN).4–6 Common preparation methods for fluorescent fibers include the blending of fluorescent particles and fiber-forming polymers, fluorescent dyeing or printing of fibers, and fluorescent-polymer spinning. Li et al. 4 prepared a luminous PP/SrAl2O4:Eu2+, Dy3+ fiber by a melt-spinning method which used triarylsulfonium hexafluoroantimonate (TSHF), organic silicon-modified luminescent materials and PP substrate to produce a new luminous material with blue-light emission in the darkness. Wang et al. 6 prepared three kinds of novel fluorescent Eu(BA)3phen/PAN, Tb(BA)3phen/PAN, and Eu/Tb(BA)3phen/PAN (BA, benzoic acid; phen, phenanthroline) fibers by electrospinning. With the increase in the incorporation of rare earth complexes into PAN, the fluorescence intensities have been significantly enhanced and reached their maximum value at 2.5 wt% for Eu(BA)3phen/PAN fibers and 2.0 wt% for Tb(BA)3phen/PAN fibers.
The previous investigations have played an important role in expanding the types and improving the functions of fluorescent anti-counterfeiting fibers. However, there are many urgent issues that need to be addressed in the selection of both fiber substrate and fluorescent molecules for the preparation of the fluorescent fibers. In terms of the fiber substrate, the fluorescent fiber raw materials are still mainly petroleum-based polymers. The study on fluorescent bio-based polymers is in the primary stage, which cannot meet the requirements of sustainable development of the chemical fiber industry. As for fluorescent molecules, rare earth elements such as europium, terbium, and yttrium have been widely used in the manufacture of fluorescent fibers. However, the radioactivity of their isotopes and associated elements (e.g. uranium, thorium, and radium), as well as the carcinogenicity that may occur when rare earth powders come into contact with the skin or are ingested into the human body, are serious concerns for many medical experts.7–9
Chitosan (CS) has abundant resources and is a non-toxic, biodegradable, skin friendly, anti-inflammatory, typical bio-based polymer. Some investigators10,11 have prepared pure CS electrospinning nanofibers using trifluoroacetic acid (TFA) or acetic acid (HAc) as the spinning solvent. Instead of pure CS, more investigators12–14 usually blend CS spinning solution with common synthetic resin (e.g. PVA and PEO) ones to prepare electrospinning fibers due to the excessive brittleness and rigidity of pure CS fibers. As a kind of commonly used fluorescent molecule, perylene derivatives exhibit many superior properties, such as stable molecular structure, high fluorescence quantum yield and thermal stability.15–17 In addition, most perylene derivatives are inexpensive. Taking perylene tetracarboxylic acid (PTCA) as an example, its price is only about 30 USD/kg. As is well known, molecular chains of CS contain a large amount of reactive groups (e.g. –OH and –NH2), which provide a basis for the chemical modification of CS. For instance, CS can undergo esterification and amidation reactions with carboxylic acids. It can be inferred that the fluorescent CS product obtained by introducing fluorescent units onto the molecular chains of CS through chemical modification will have better fluorescence durability than the product obtained by blending CS with fluorescent small molecules physically. It is both economical and environmentally friendly for the chemical fiber industry to develop such kinds of fluorescent bio-based fibers. However, to our knowledge, there are few reports on the preparation of bio-based textile fibers labeled by organic fluorescent units at present.
In the present study, various amounts of perylene units were labeled onto molecular chains of native CS to prepare a series of fluorescent CS with different degrees of labeling (DL) of perylene. Then, the fluorescent CS spinning solutions were mixed with PVA solution at a fixed volume ratio to prepare CS-perylene/PVA nanofiber membranes by electrospinning, respectively. In terms of fluorescence intensity and thermal stability of the CS-perylene, the surface morphology and tensile properties of the CS-perylene/PVA nanofiber membranes, and the effects of the DL on the application properties of the fluorescent CS were evaluated systematically. Finally, an appropriate DL was recommended for the preparation of CS-perylene to enhance its serviceability as a fluorescent bio-based polymer material for the manufacture of anti-counterfeiting fibers.
Experimental
Materials
CS (degree of deacetylation: 88.2%) was extracted from lobster shell and then hydrolyzed to appropriate apparent viscosity using diluted HCl for electrospinning. The detailed extraction and hydrolyzation processes are described in the Supporting Information (SI). Perylene tetracarboxylic acid (PTCA), dibutyltin dilaurate (DBTDL), dicyclohexylcarbodiimide (DCC), 4-dimethylaminopyridine (DMAP), LiCl, and dimethylacetamide (DMAc) were of analytical grade. PVA resin (degree of polymerization, 1700; degree of alcoholysis, 99%; molecular weight of constitutional unit, 44.4) was chemically pure.
Preparation of CS-Perylene Derivatives
PTCA exhibits poor solubility due to the presence of a condensed ring conjugated system in its structure and being in a plane, which enhances the π-bond interactions between molecules. In order to fully dissolve PTCA, the concentration of PTCA in DMAc was controlled at a low value of 0.214 g/L to maintain the stability of the solution. The feeding amount of PTCA for preparing CS-perylene was altered by changing the volume of PTCA solution in the study.
At first, 0.0428 g of PTCA was dissolved completely in 200 mL of DMAc at 60°C to obtain Solution A. Anhydrous LiCl (9.7770 g) was dissolved into 120 mL of DMAc under vigorous stirring at 60°C to form a LiCl/DMAc compound solvent (weight percentage of LiCl, 8%). Then, 1.6352 g of CS (molar number of constitutional units, 0.01 mol), 0.0244 g of DMAP (catalyst) and 0.2472 g of DCC (dehydrator) were added to the LiCl/DMAc compound solvent to obtain Solution B. In the study, the feed molar ratios of PTCA to constitutional unit of CS were controlled at 0.05%, 0.10%, 0.20%, 0.40%, and 0.80%, respectively. The five fluorescent CS samples prepared were numbered as CS-perylene 1#, 2#, 3#, 4#, and 5#, respectively, according to the order of the feed molar ratio from low to high. Taking CS-perylene 5# as an example, the preparation process of the fluorescent chitosan is described below. Ten milliliters of Solution A was transferred to Solution B. The mixture was refluxed at 100°C for 1.5 h under vigorous stirring using a mechanical stirrer (Talboys Engineering Corp., model T Line 134-1) at 1000 rpm and then cooled to ambient temperature. The solution was precipitated with ethanol, centrifuged at a ratio of 10,000 rpm and washed with ethanol thoroughly. The CS-perylene precipitate was dissolved in dilute HAc (0.1 mol/L) and added to a dialysis bag. The rotation speed of the dialysis bag and the frequency of changing the distilled water outside the dialysis bag were both kept high, which were about 100 rpm and once per 3 h, respectively, in order to make all the unreacted reagents removed via dialysis in distilled water for 24 h. The CS-perylene was freeze dried, pulverized and stored in desiccator. The synthetic route of CS-perylene is shown in Figure 1.
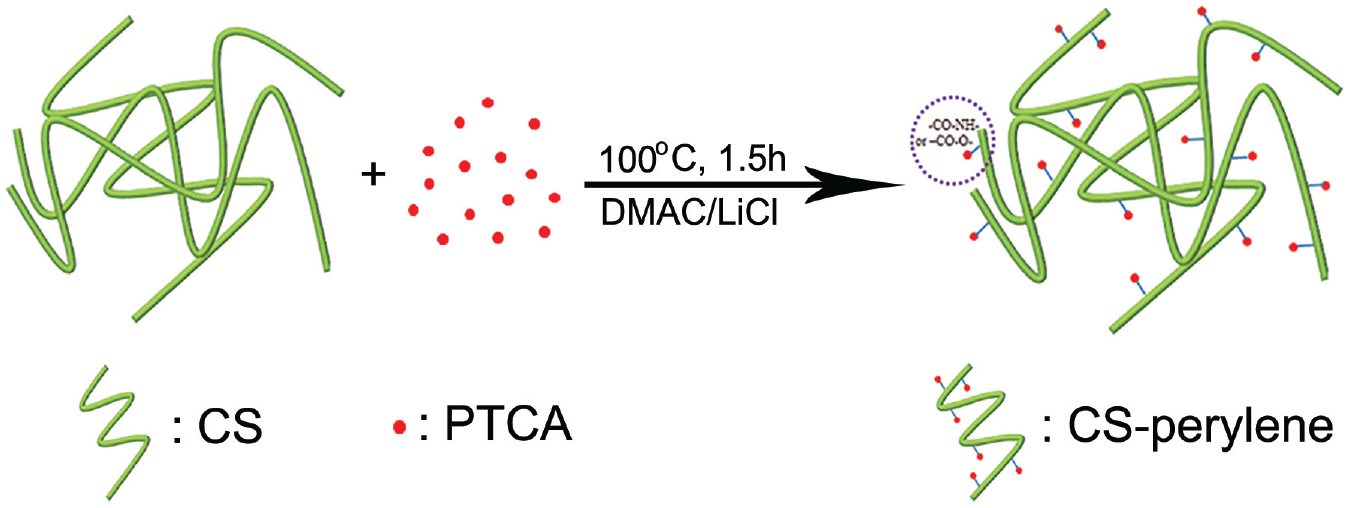
Synthetic route of CS-perylene.
Measurement of Degree of Labeling (DL)
The DL in this study denoted the molar ratio of perylene to constitutional unit of CS and was determined using a UV-2450 UV-visible spectrophotometer (Shimadzu Co., Ltd., Japan). The maximum absorption wavelength (λmax) of PTCA solution was determined to be 475 nm using a UV-visible spectrophotometer. The absorbance of five known different mass concentrations of PTCA solutions at λmax was measured to plot a colorimetric standard curve as shown in Figure S1. The CS-perylene prepared was dissolved in dilute HAc (0.1 mol/L) to form solutions (1000 mg/L). The absorbance of the CS-perylene solution at 475 nm was measured to calculate the DL of each CS-perylene sample according to the colorimetric standard curve.
FTIR Characterization
Fourier transform infrared spectrometer (FTIR) was used to verify the labeling of perylene unit onto molecular chains of CS. All the CS-perylene samples were purified before characterization. The fluorescent CS samples were fully washed using DMAc to remove the unreacted PTCA adhering on the CS-perylene completely. The measurement was taken on Nicolet Nexus spectrophotometer through the diffuse reflectance technique with a spectral resolution of 2 cm–1 for 64 scans.
NMR Characterization
1H-NMR and 13C-NMR were used to confirm the labeling of perylene unit onto the CS. The unlabeled CS and the purified CS-perylene were dissolved in D2O/trifluoroacetic acid-d blended solvent and characterized using an AVAMCE III 400 MHz Digital NMR spectrometer (Bruker Co. Ltd, Switzerland). The concentration of CS solution was about 5% (w/w).
TGA Characterization
The thermal behaviors of the unlabeled CS and the purified CS-perylene were compared using thermogravimetric analysis (TGA). The TG and DTG curves of the unlabeled and the fluorescent CS samples were obtained using a TG/DTA6300 (Seiko Co., Ltd, Japan) integrated thermogravimetric analyzer. About 5 mg of the sample was heated at 10°C/min in a range of 30–800°C under N2 atmosphere.
Measurement of Fluorescence Intensity
The fluorescence intensity of the CS-perylene solutions was measured with a Hitachi F-7000 fluorescence spectrophotometer (Hitachi High-Tech Global Co., Ltd., Japan). The CS-perylene derivatives were dissolved in dilute HAc (0.1 mol/L) to form solutions (1000 mg/L). A fixed excitation wavelength (λex = 475 nm) was set to measure the fluorescence emission spectrum of the CS-perylene solutions.
Observation of CS-Perylene Fluorescence
The fluorescence of the freeze dried CS-perylene was observed using a DM300 fluorescent microscope (Leica Microsystems, Germany). The CS-perylene sections were photographed under bright field and blue excitation light by fluorescent microscope, respectively.
Preparation of Nanofiber Membrane
Seven percent CS solutions (w/w) were prepared by dissolving the CS-perylene with different DL in 5% of diluted HAc at 50°C. Simultaneously, 11% PVA solutions (w/w) were prepared by dissolving PVA in distilled water at 50°C. The CS and the PVA solutions were mixed with equal volumes at room temperature under magnetic stirring for 2 h to form a homogenous solution. The CS/PVA blend spinning solution was obtained after standing for 4 h for defoaming. The detailed electrospinning process is described in the SI.
SEM Observation of Nanofiber Membrane
The surface morphology of the CS/PVA nanofiber membrane was observed using an SU3800 SEM (Hitachi Co., Ltd, Japan). The membrane was mounted on a conductive adhesive tape and sputter coated with gold palladium prior to observation. A 10 kV voltage was used for all the observations. The fiber diameter was measured by Nano Measurer software.
Measurement of Tensile Properties of Nanofiber Membrane
The CS/PVA nanofiber membranes were cut into strips (70 × 10 mm), which were conditioned at 65% R.H. and 20°C for 24 h before testing. The tensile strength and tensile elongation of the nanofiber membranes were measured on a BZ2.5/TNIS Zwick Material Tester. Twenty strips of each nanofiber membrane were measured to obtain the mean value.
Statistical Analysis
The data were analyzed using SAS software (SAS Institute, Inc., Cary, NC, USA). The confidence interval was set at 95% and a p-value smaller than 0.05 was considered to be a statistically significant difference by Tukey’s HSD test. According to the operation results of the analysis software, small letters, such as a, b, c, were inserted above the data points in the figure of tensile properties of the CS/PVA nanofiber membrane. The data points with the same small letter were not statistically significantly different from each other.
Results and Discussion
FTIR Analysis of Unlabeled CS and CS-Perylene
The FTIR spectrum of unlabeled CS and CS-perylene is shown in Figure 2. The main characteristic absorption bands of CS (e.g. the ones around 1660 and 1550 cm−1 corresponding to amide I and amide II, respectively18,19) could be found in all the curves. Two new characteristic peaks at about 1250 and 895 cm−1 could be observed in the curves of all the CS-perylene in addition to the absorption bands of unlabeled CS. The two new peaks at 1250 and 895 cm−1 could be assigned to the asymmetric C-O stretch vibration of carboxyl and the C-H bending vibration of the aromatic ring, respectively.20,21 The two peaks proved the labeling of perylene onto the molecular chains of CS after the acylation reaction between PTCA and CS. Moreover, the intensity of the characteristic absorption band of amide I around 1660 cm−1 increased after the labeling of perylene unit. The increase in the intensity of the absorption band proved the occurrence of amidation between amino groups of CS and carboxyl groups of PTCA. In addition, the intensities of the stretching vibration absorption peaks of C-O (primary and secondary hydroxyl groups) at about 1035 and 1080 cm−1 respectively, both decreased after labeling of the perylene unit. The decreases in the intensities proved the occurrence of the esterification between primary and secondary hydroxyl groups of CS and carboxyl groups of PTCA.
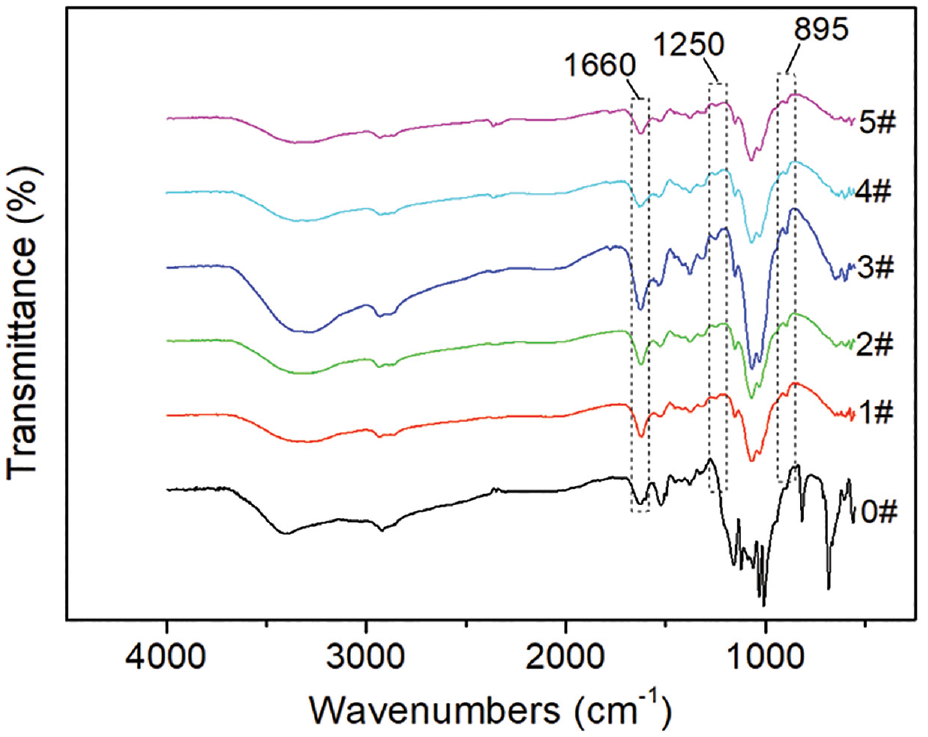
FTIR spectrum of unlabeled CS and CS-perylene.
NMR Analysis of Unlabeled CS and CS-Perylene
The 1H-NMR spectra of unlabeled CS and CS-perylene are shown in Figure 3. The chemical shift at about 4.8 ppm corresponds to the proton peak of the solvent (D2O). Besides the chemical shift of unlabeled CS (Figure 3(a)), such as the one in the range of 2.9–4.1 ppm corresponding to methine and methylene in the backbone of CS, 22 a new chemical shift in the range of 6.5–7.5 ppm was found in the spectra of the CS-perylene derivatives that could be considered the proton of aromatic rings of perylene. 23 Due to the low DL of the CS-perylene, the proton peaks of perylene were too low to observe when the feed molar ratio of PTCA to constitutional unit of CS was lower than 1/500.
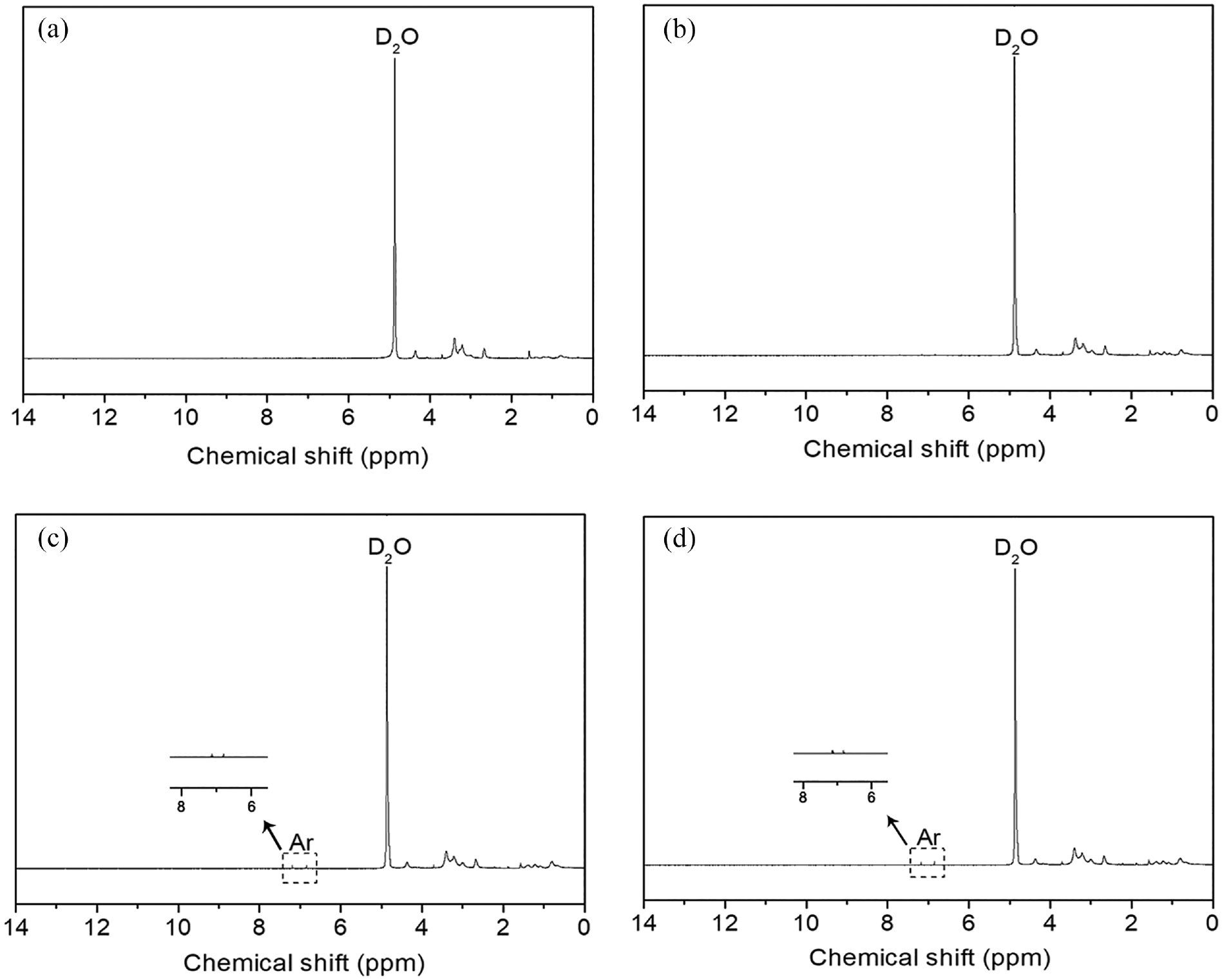
1H-NMR spectra of unlabeled CS (a), CS-perylene 2# (b), CS-perylene 3# (c), and CS-perylene 4# (d).
The 13C-NMR spectra of unlabeled CS and CS-perylene are displayed in Figure 4. The peaks of the carbons in N-acetyl glucosamine and glucosamine units, such as C=O at ∼160 ppm, C1 at ∼109 ppm, C2 at ∼51 ppm, and –CH3 at ∼26 ppm,24,25 could be observed in the 13C-NMR spectra of all the CS. After the labeling, the intensity of the carbonyl carbon peak at ∼160 ppm was enhanced, especially for the CS-perylene samples with high DL (≥0.0762 mol%). The increase in the intensity of the carbonyl carbon peak could confirm the generation of ester and amide groups after the acylation reaction between CS and PTCA.
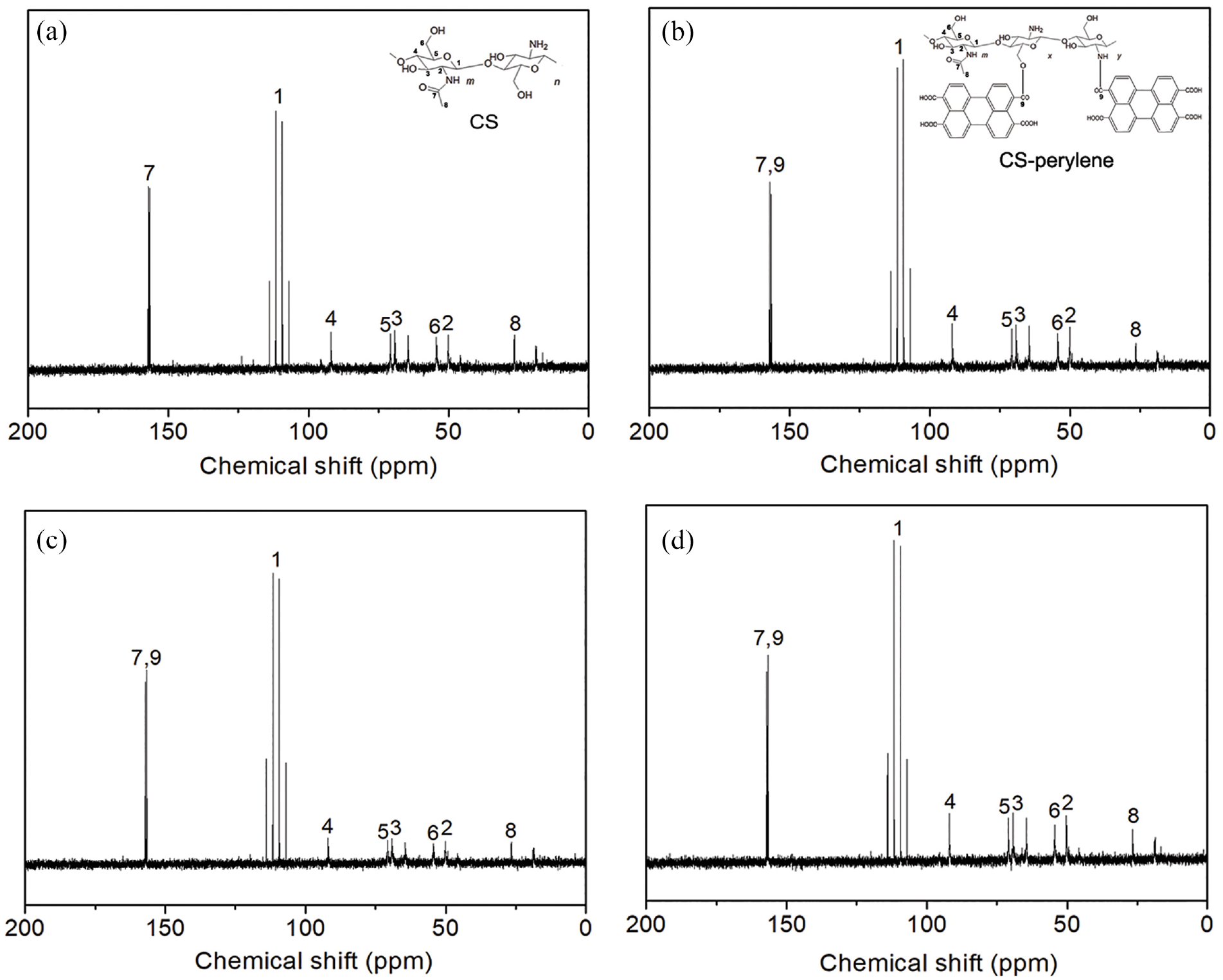
13C-NMR spectra of unlabeled CS (a), CS-perylene 2# (b), CS-perylene 3# (c), and CS-perylene 4# (d).
Characterization of UV-Visible Spectrophotometer
Due to the low DL of CS-perylene in this study, it is necessary to measure the absorbance of the CS-perylene solution to calculate the DL value using a UV-visible spectrophotometer. The absorbance of the CS-perylene solution at λmax, the DL of CS-perylene, and the mass concentration of perylene unit are shown in Table 1. The DL of the CS-perylene increased with the increase in the feed molar ratio of PTCA to constitutional units of CS.
DL of CS-perylene at different feed molar ratios of PTCA to constitutional units of CS.
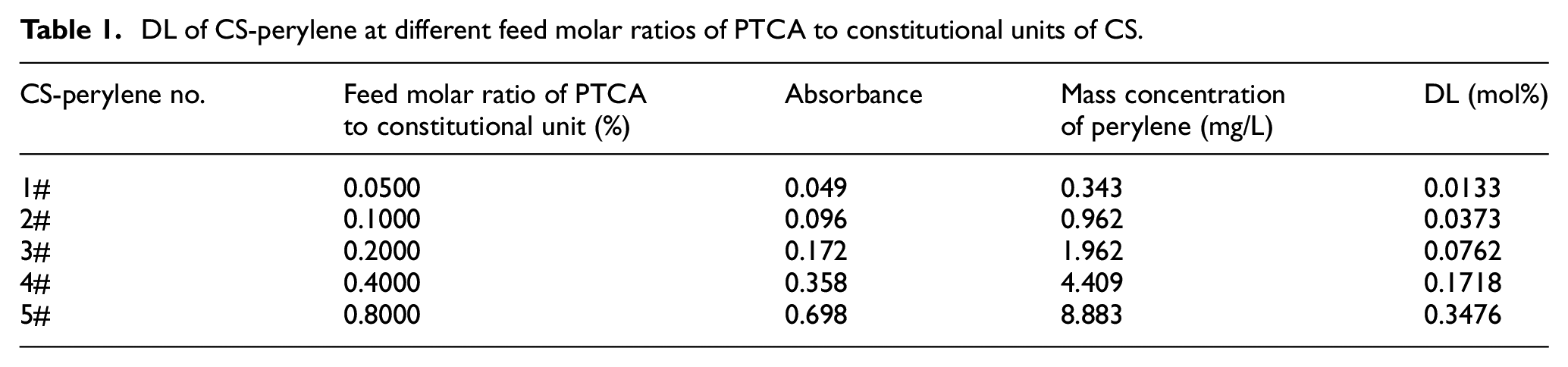
With the increase in the feed concentration of PTCA fluorescent molecules, more carboxyl groups of PTCA were able to undergo amidation and esterification reactions with amino and hydroxyl groups of CS, respectively. Consequently, more perylene units could be labeled onto the CS. In other words, the DL of the fluorescent CS was increased. The absorbance of the CS-perylene solution was also enhanced therewith.
Thermogravimetric Analysis of Unlabeled CS andCS-Perylene
Figure 5 reveals the thermal degradation behaviors of unlabeled CS and CS-perylene. The degradation process of the unlabeled CS could be divided into three main stages: (1) moisture removal from CS (<120°C), (2) thermal degradation of CS (220–500°C), and (3) carbonization of CS (>570°C). In Stage 1, both bound water and absorbed water of CS were removed. The water loss rate reached the maximum at about 70°C. In Stage 2, the molecular chains of CS were decomposed by thermal oxidation, the glycosidic bonds were broken and adjacent hydroxyl groups were dehydrated to ether. The weight loss rate reached the maximum at 300°C. In Stage 3, CS start to be carbonized from 570°C, lost its original characteristics, and finally formed a black coagulum. 26
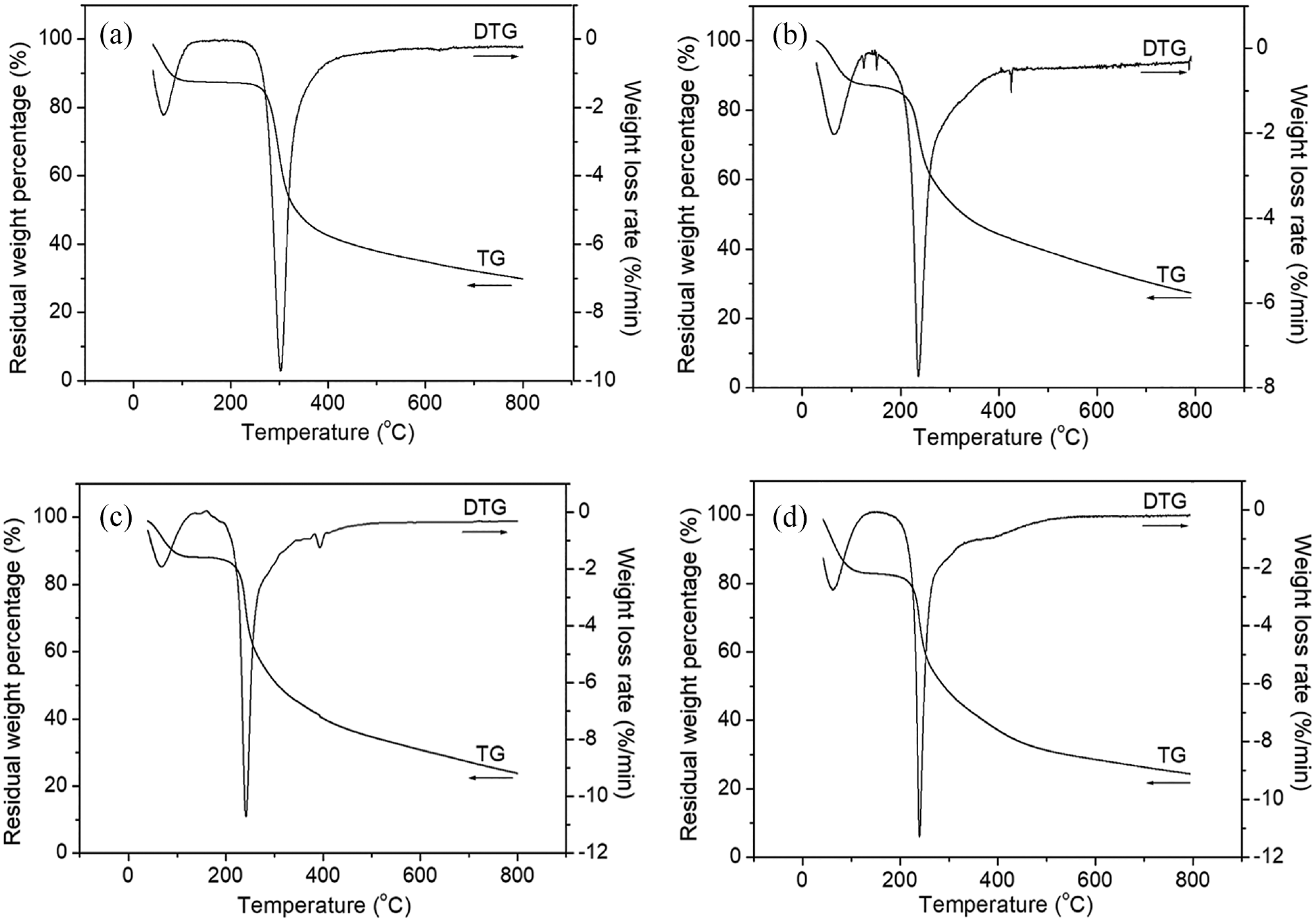
TGA thermograms of unlabeled CS (a), CS-perylene 1# (b), CS-perylene 4# (c), and CS-perylene 5# (d).
Compared with the thermogram of unlabeled CS, the thermal degradation trends of the CS-perylene derivatives were similar in Stage 1. In Stage 2, a remarkable difference between the unlabeled CS and CS-perylene is that starting temperature of oxidative decomposition of the latter decreased to only about 175°C. Meanwhile, the weight loss rate of the CS-perylene reached the maximum at about 240°C. The lower oxidative decomposition temperature in Stage 2 indicated that the CS-perylene exhibited lower thermal stability than the unlabeled CS at high temperature (>170°C). In Stage 3, the thermal degradation trends of the unlabeled CS and CS-perylene were also similar but the carbon residue rate of the former (∼32%) was slightly higher than that of the latter (∼25%). The lower carbon residue rate of the CS-perylene also indicated the decrease in their thermal stability.
As described above, the CS-perylene was synthesized at high temperature (100°C) under vigorous stirring (1000 rpm). High temperature and vigorous stirring for a long time could cause breakages of part of intermolecular hydrogen bonds and glucosidic bonds of CS. The breakages of the hydrogen and glucosidic bonds became the main reason for the decrease in the thermal stability of the CS-perylene. 27 As for the CS-perylene derivatives with different DL in the study, they exhibited similar thermal stabilities due to the use of the same synthesis conditions and low DL values (0.0133–0.3476 mol%). It should be noted that both the preparation and end-use temperatures of the fluorescent chitosan electrospun anti-counterfeiting nanofibers are generally not higher than 120°C. Therefore, the labeling of the perylene onto the CS will not have adverse effects on the production and utilization of the CS electrospun fibers from the perspective of heat resistance.
Effect of DL on Fluorescence Properties of CS-Perylene
The fluorescence intensities of the CS-perylene solutions and the photos of the CS-perylene sections under bright field and blue excitation light are displayed in Figures 6 and 7, respectively. As observed from Figure 6, the fluorescence intensity initially increased with the increase in the DL of the CS-perylene, reached the maximum when the DL was 0.1718 mol% (i.e. CS-perylene 4#), and then started to decrease. The detection result is in good agreement with the variation trend of fluorescence intensity of the CS-perylene section observed by the naked eye under a fluorescence microscope as shown in Figure 7.
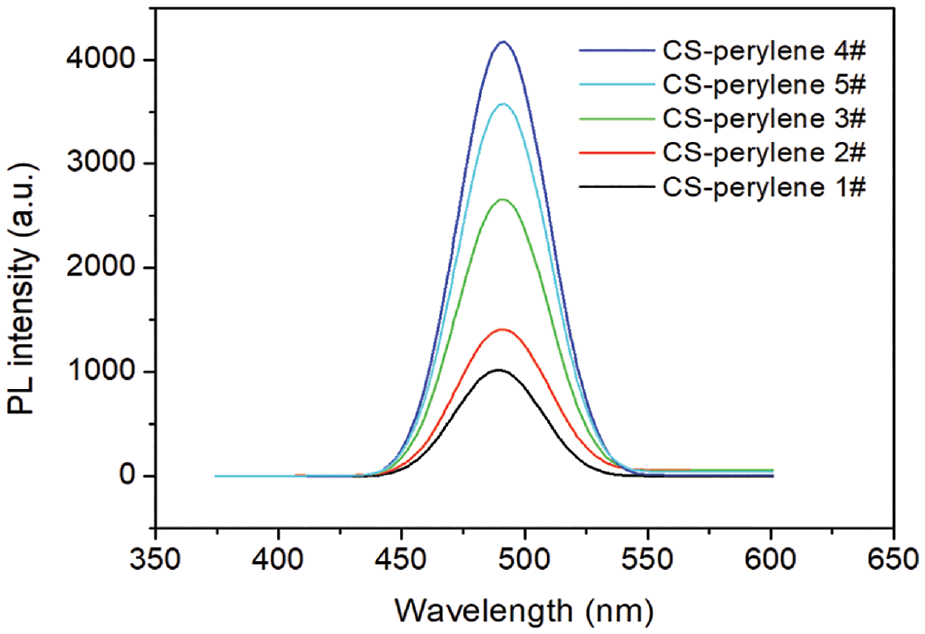
Fluorescence spectrum of CS-perylene solutions with different DL (λex = 475 nm).

Photos of sections of CS-perylene 1#–5# under bright field (a–e) and blue excitation light (f–j) taken by a fluorescence microscope.
Perylene is a kind of aggregation caused quenching (ACQ) luminogen and usually possesses strong luminous efficiency in a dilute solution state. However, its fluorescence will weaken or even disappear in aggregation or solid state.22,28 Therefore, only when appropriate amounts of perylene units are labeled onto the molecular chain of a macromolecule, can the macromolecule be endowed with good fluorescence properties. In this case, the fluorescence intensity of the perylene-labeled macromolecule (e.g. CS) directly relates to its DL. When the DL value of the CS-perylene is in a low range (<0.1718 mol%), the more perylene units are labeled onto the molecular chain of CS, the more the chemical groups are able to fluoresce. Consequently, the initial increase in the amount of the perylene labeled onto the CS contributes to the enhancement in the fluorescence intensity. However, the fluorescence quantum yield decreased instead when the DL of CS-perylene exceeded 0.1718 mol% due to the occurrence of the ACQ phenomenon.
According to detection result of the fluorescence intensity of the CS-perylene, 0.1718 mol% could be regarded as a critical DL from which the perylene started to aggregate. The concentration of the CS-perylene solution for the fluorescence intensity test was 1000 mg/L. In the ideal case of absolutely uniform dispersion of CS-perylene macromolecules in dilute HAc and the arrival at the critical DL (0.1718 mol%), a perylene unit took up about a cubic space 1.61 × 105 nm3 (edge length: 54.42 nm). The major reason for the aggregation of the conjugated ACQ luminogens is the π–π stacking interaction between perylene units. The more intense the π–π stacking interaction, the greater is the degree of fluorescence quenching. In general, the distance of π–π stacking interaction is in the range of 0.34–0.40 nm.29,30 Obviously, the average distance between the perylene units when the DL of the CS-perylene arrived at the critical value was far longer than 0.40 nm. It could be speculated that ACQ had occurred in the CS-perylene solution before the perylene units fell in the range of π–π stacking interaction. The cause for the occurrence of the ACQ phenomenon was the intense thermal motion of the perylene owing to the lack of spatial constraints.
Effect of DL on Surface Morphology of Nanofiber Membrane
Figure 8 depicts the surface morphology of unlabeled CS/PVA and CS-perylene/PVA nanofiber membranes. As shown in the SEM images, the labeling of perylene onto the CS has no obvious effect on the average fiber diameter. All the nanofibers produced by the unlabeled and labeled CS/PVA spinning solutions exhibit a uniform diameter (∼140 nm) and no visible bead-string structure. This indicates that as long as the DL of the CS-perylene is controlled at low value (<0.348 mol%), the labeling of perylene units will not affect the electrospinning process of the CS.

SEM images of nanofiber membranes of unlabeled CS/PVA (a) and CS-perylene 1#–5#/PVA (b–f).
Effect of DL on the Tensile Properties of Nanofiber Membrane
The effect of the labeling of the perylene on the tensile properties of the CS/PVA nanofiber membrane is displayed in Figure 9. The unlabeled CS/PVA nanofiber membranes possessed higher tensile strength but lower tensile elongation than all the CS-perylene/PVA ones. As for the CS-perylene/PVA nanofiber membranes, the DL had no marked effect on their tensile properties.
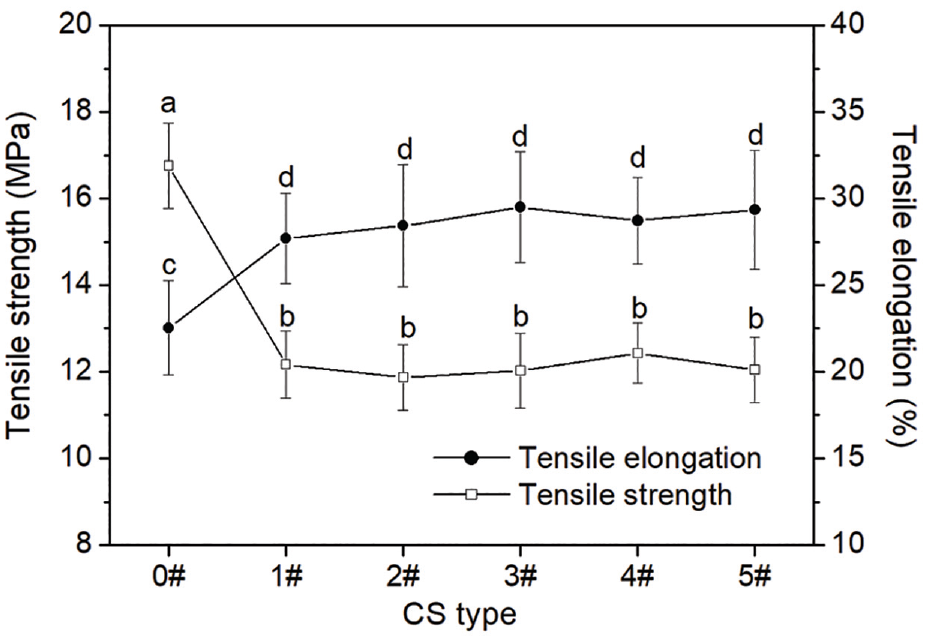
Tensile properties of nanofiber membranes of unlabeled CS/PVA (0#) and CS-perylene 1#–5#/PVA (1#–5#).
As mentioned above, during the synthesis process of the CS-perylene, the high temperature and vigorous stirring led to the breakages of parts of the intermolecular hydrogen bonds and glucosidic bonds of the CS. The breakages of the hydrogen and glucosidic bonds directly caused tensile strength of the nanofiber membrane to decrease. As is well known, CS contains a large number of hydrogen bonds and has a regular macromolecular structure. Thus, the CS fiber exhibits excessively high rigidity and brittleness.10,11 The breakage of the hydrogen bonds is beneficial to the relative slip between molecular chains of the CS.31,32 As a result, tensile elongation of the CS-perylene/PVA nanofiber membrane increased therewith. As far as the CS-perylene derivatives were concerned, the DL brought about an insignificant influence on tensile property of the nanofiber membrane due to the use of the same synthesis conditions and low DL values.
Effect of DL on Fluorescence Property of Nanofiber Membrane
The photos of the CS/PVA nanofiber membranes under sunlight and 365 nm UV light are shown in Figure 10. The CS-perylene/PVA nanofiber membranes exhibited a very light pink color (Figure 10(a)), which was difficult to recognize with the naked eye and to distinguish from the unlabeled CS/PVA nanofiber membrane under sunlight. Thus, the nanofibers possessed good concealment. On the contrary, all the CS-perylene/PVA nanofiber membranes under UV light could emit fluorescence of different brightnesses (Figure 10(b)), which was easily recognizable with the naked eye. Among all the CS-perylene/PVA nanofiber membranes, Sample 4# emitted the brightest green fluorescence, which derived from the high fluorescence quantum yield of CS-perylene 4#. Due to the good fluorescence properties of the nanofiber membrane, it can be used as fluorescence anti-counterfeiting materials in various areas and avoid many end-use problems of the fluorescent fabrics produced by dyeing or printing with fluorescent dyes, such as low washing fastness and friction fastness.

Photos of nanofiber membranes of unlabeled CS/PVA (0#) and CS-perylene 1#–5#/PVA (1#–5#) under sunlight (a) and 365 nm—UV light (b).
Conclusion
Labeling an appropriate amount of fluorescent molecule PTCA onto molecular chain of CS is one of the effective methods for preparing functional polymer materials with good fluorescence properties. With the increase in the feed molar ratio of PTCA to constitutional units of CS from 0.05% to 0.80%, the DL of the CS-perylene prepared can keep increasing. With the increase in the DL, the fluorescence of the CS-perylene initially increases, reaching a maximum at a DL of 0.1718 mol%, and then starts to decrease. Compared with unlabeled CS, the thermal stability of the CS-perylene decreases to some extent but the decrease will not affect the preparation and end-use of the CS electrospun nanofibers. As for the CS-perylene derivatives with different DL values, their thermal stabilities show no marked differences. The diameters of unlabeled CS/PVA and all the CS-perylene/PVA fibers are around 140 nm. The CS/PVA nanofibers have uniform thickness and no visible bead-string structure. In addition, all the CS-perylene/PVA nanofiber membranes possess lower tensile strength but higher tensile elongation than the unlabeled CS/PVA one. Meanwhile, the CS-perylene derivatives-based nanofiber membranes have similar tensile properties. The CS-perylene (DL: 0.1718 mol%)/PVA nanofiber membrane has good concealment under sunlight but can emit bright green fluorescence which is easily recognizable with the naked eye under UV light. The CS-perylene nanofibers have shown great potential to be used as anti-counterfeiting materials for the manufacture of banknotes, passports, stamps, securities, and high-grade garment fabrics, etc. The investigation lays a solid foundation for future research on the preparation of perylene labeled bio-based fluorescent nanofibers.
Supplemental Material
sj-docx-1-aat-10.1177_24723444241295413 – Supplemental material for Preparation and Application Performance of Perylene-Labeled Fluorescent Chitosan Electrospun Anti-Counterfeiting Nanofibers
Supplemental material, sj-docx-1-aat-10.1177_24723444241295413 for Preparation and Application Performance of Perylene-Labeled Fluorescent Chitosan Electrospun Anti-Counterfeiting Nanofibers by Yunhan Li, Shengbin Zhu, Enqi Jin, Chi Shen and Manli Li in AATCC Journal of Research
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The work was supported financially by Technological Research Project for Public Welfare of Zhejiang Province (LGG21E030005; LGG22E030002), Postdoctoral Scientific Research Project of Zhejiang Province (276089), Scientific and Technological Research Project for Public Welfare of Shaoxing (2022A12004), and “Spearhead and Leading Goose + X” Research and Development Project of Zhejiang Province (2024C02007). Financial sponsors do not endorse the views expressed in this publication.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
