Abstract
In this study, a novel way for modification of wool fabrics by extracted feather protein is introduced. The present investigation aims to establish an environmental friendly approach with feather waste to achieve shrink-resistant wool. For this purpose, chicken feathers were hydrolyzed with a commercial proteolytic enzyme accompanied by a reducing agent. This process was optimized to achieve maximum extraction yield. The extracted feather protein was then applied to modify the wool fibre surface in the presence or absence of a cross-linking agent, glycerol diglycidyl ether. The obtained results showed that treatment of wool fabric with feather protein led to a considerable improvement in anti-felting properties, and application of the cross-linking agent enhanced the shrink-resistant properties of wool further. Besides, the bending length of wool fabrics increased and their tensile strength was apparently unaffected in comparison with the untreated one. The scanning electron micrographs indicated that wool fibers were still intact and were not severely damaged by the applied treatment. The Fourier transform infrared spectra also approved the formation of cross-linked protein on wool fabric.
Introduction
In the absence of any preventive treatment, almost all types of woven and knitted wool products will shrink after washing. Felting shrinkage is a non-reversible change in fabric dimensions and is caused by the progressive fiber entanglement of wool products subjected to mechanical action, most particularly during laundering. This tendency of wool to felt prevents the use of untreated wool materials as machine-washable textiles. The physical structure of the scaly cuticle layer of the wool fibre, i.e. the differential frictional effect (DFE), is considered to be the main reason for felting [1,2]. Either scales removal or smoothing the edges would reduce the felting tendency. For this purpose, three different processes have been commercially practiced: subtractive (oxidation, reduction), additive (synthetic resin layer), and a combined process (e.g. chlorine/Hercosett process). Despite imparting excellent and robust shrink resistance to wool, these processes cause severe ecological problems or make the fibers handle harsh [3].
Because of such environmental concerns, much research effort has gone into searching for environmentally friendly processes for wool shrink resistance. Extensive research has been undertaken to develop an enzyme-based shrink-resistant finishing treatment of wool [4–8]. During the enzymatic treatment with conventional proteases, wool fiber was damaged by enzyme penetration into the interior of fibers and breaking down the cell membrane complex. It is difficult to limit enzymatic degradation to the cuticle scales and to achieve machine-washable wool without significant fiber damage [9,10]. An alternative solution is to enlarge the molecular size of the protease by covalent coupling with a polymer to limit the proteolytic attack to the fiber surface, thus controlling the damage to the wool. However, the enzyme modification processes are expensive and commercial standards for machine washability were difficult to meet especially for knitted wool fabrics [11–15].
In the present work, application of extracted feather protein on wool fabrics has been attempted to achieve shrink-resistant wool. World-wide poultry processing plants produce millions of tons of feathers as a waste product annually, which consist of approximately 90% keratin [16]. Chicken feathers are approximately half feather fiber (barbs) and half quill (rachis) by weight, the quill being the stiff central core off which soft, interlocking fibres branch [17]. Chemically, feather keratins are small proteins, uniform in size, with a molecular weight around 10 kDa. They are rich in hydrophobic residues and have β-sheet conformation [18]. Keratin is regarded as a three-dimensional polymer interlinked by inter-molecular bonding of disulfide cystine amino acid and inter- and intra-molecular bonding of polar and non-polar amino acids, which are responsible for its high stability and distinctive physical properties [19]. There are a couple of procedures to hydrolyze keratin. Keratin hydrolysates can be prepared by acid, alkali, enzymatic, or reduction methods. Moreover, due to the non-reactive characteristic of keratin, hydrolysis is difficult, and as a result it is better to combine these methods to boost the yeild of hydrolysis [20,21]. In this study, feather was hydrolyzed with a commercial protease, Savinase, along with a reducing agent. Several parameters such as enzyme concentration, treatment time, and feather concentration were evaluated to find the optimum hydrolysis condition. Then, the wool fabric was treated with the extracted feather protein with or without a cross-linking agent. The physical properties of treated fabrics such as felting shrinkage due to washing, tensile strength, alkaline solubility, and bending length were tested. The microstructure of treated fibers was also characterized by scanning electron microscopy (SEM) and Fourier transform infrared (FTIR) spectroscopy.
Materials and methods
Materials
White chicken feathers were collected from a slaughterhouse located in the south east of Tehran, Iran. The wool fabric with plain weave (2.268 g/cm2 weight and 42 end per inch × 39 picks per inch) was provided by Pars textile company (Tehran, Iran) and used throughout this study.
The proteolytic enzyme was the alkaline serine endoprotease, Savinase 16.0LEX (EC.3.4.21.14), supplied by Novozymes A/S (Bagsvaerd, Denmark). The cross-linking agent, glycerol diglycidyl ether (GDE), and the reducing agent, sodium sulphite, were supplied by Sigma-Aldrich (St. Louis, USA) and Loba Chemie Inc. (Mumbai, India), respectively. All other chemicals were of analytical grade and purchased from Merck Co (Darmstadt, Germany).
Pre-treatment of feathers and wool fabric
The pretreatment was performed in a solution containing 2 g/l of a non-ionic detergent and 1% sodium carbonate at liquor to goods ratio of 40:1 for 30 min at 60℃. After washing, the samples were rinsed thoroughly with distilled water and air-dried.
Protein extraction from feathers
Cleaned chopped feather (10, 20, and 30 g/l) was incubated with Savinase (0.5, 1, 2, and 4% v/v) in sodium borate buffer solution (50 mM, pH = 8.5) containing 10 g/l sodium sulfite and 1 g/l sodium dodecyl sulphate (SDS) at 55℃ for different treatment times (30, 60, 90, 120, and 150 min). The reducing agent, sodium sulfite, and the anionic surfactant, SDS, were used to break down the disulfide bonds in combination with the protease and to reduce the surface tension between feather and solution, respectively. After enzymatic hydrolysis, 5 ml acetic acid (0.5 M) was added to deactivate the enzyme while raising the temperature to 75℃ for 15 min. Each supernatant was then filtered through Whatman #41 filter paper to separate any fibrous material from the solution. The released soluble protein in each supernatant was measured by Lowry method using bovine serum albumin as standard [22]. The effects of enzyme concentration, treatment time, and feather concentration on protein extraction were evaluated to find out the optimum hydrolysis conditions.
In order to determine the molecular weight of the extracted protein from feathers, the electrophoretic separation of the released protein was performed by SDS-PAGE according to the Laemmli method [23] using the Mini-PROTEAN 3 Cell system from Bio-Rad. The 12% polyacrylamide gel was processed at 100 V in running buffer, Tris–glycine SDS. Proteins were stained with silver nitrate and analyzed based on a wide range molecular weight marker (Fermentas, USA, MW 10–170 kDa).
Treatment of wool fabric using extracted feather protein
Treatment conditions for different samples.
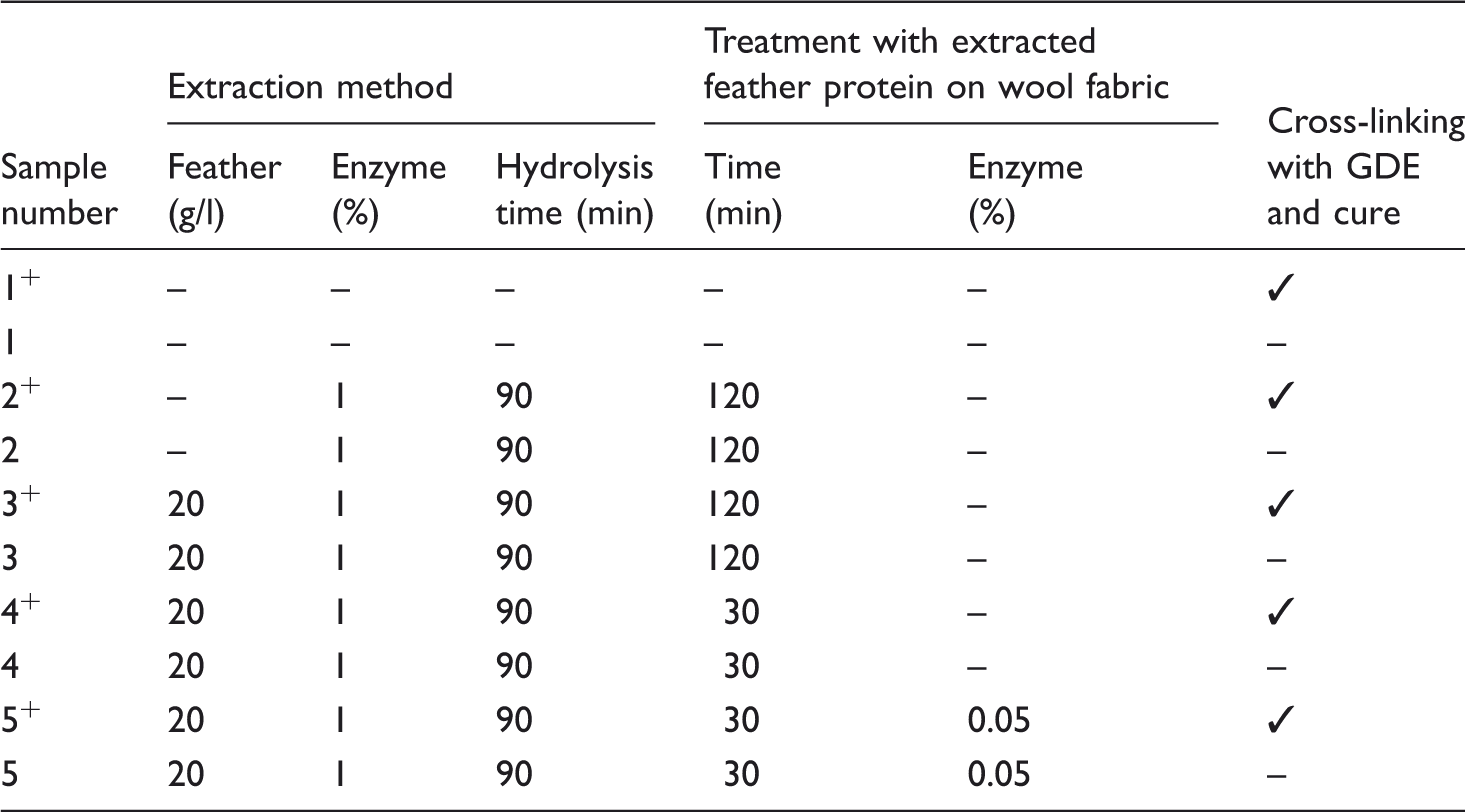
Characterization of treated wool fabrics
Scanning electron microscopy
The surface morphology of the samples was examined with scanning electron microscope (SEM, KYKY-EM3200, China) at 24 kV acceleration voltage after gold coating.
FTIR spectroscopy
To study the chemical changes during the processing of wool fabrics, the FTIR analysis was also performed by Thermo Nicolet Nexus 670 Spectrophotometer. The characteristic spectra were scanned in the wave number range of 4000 to 400 cm−1 at a resolution of 4 cm−1 using KBr pellets.
Washing test to assess fabric shrinkage
Felting shrinkage of wool fabrics due to washing was measured according to ISO 6330 5A. The fabrics were washed three times in solution containing 1% non-ionic surfactant and 20 metallic ball bearings at a liquor to goods ratio of 40:1 for 45 min at 55℃. After each washing cycle, the fabrics were dried in a 50℃ oven for 4 h, conditioned at 60% relative humidity and 25℃ for 24 h, and then weighted. The weight loss as a result of the aforementioned treatment was calculated from the following equation:

Tensile strength
Tensile strength of treated wool fabrics was measured by a universal testing machine Instron 5566 according to ISO5081. The samples were balanced at 25℃ and a relative humidity of 60% for 24 h before testing. The samples were in dimension of 25 mm × 150 mm after drawing out the yarns of 5 mm at both sides. The test was performed only in the warp direction of the fabrics and the results were given as the arithmetic means of three different samples [25].
Bending length
The bending length of the treated and non-treated wool fabrics was measured according to ASTM D1388. The dimensions of the samples were 2.5 cm × 15 cm.
Alkaline solubility
The solubility of wool in alkali may provide a useful index of the extent of the change in its chemical properties brought about by reagents like cross-linking agents. The weight reduction of wool fabrics in sodium hydroxide (0.1 N) was calculated and the solubility in alkaline was measured according to ASTM D1283-05.
Results and discussion
Effect of process parameters on yield of extracted feather protein
The effect of enzyme and feather concentration on released protein when hydrolysis time was kept constant at 90 min is shown in Figure 1(a). The results reveal that maximum protein can be extracted when 20 g/L feather was incubated with 1% enzyme. It is known that a constant substrate concentration requires a certain amount of enzyme to reach adsorption saturation for hydrolysis and further increase in enzyme loading would result in the presence of more free protease in the reaction mixture which might cause a hindrance for proteolytic attack. Thus, there is an optimum limit for enzyme loading to attain the maximum efficiency. In addition, based on the literature, high substrate concentration resulted in low hydrolysis yield due to product/substrate inhibition as well as enzyme inactivation [26,27]. Therefore, more protein is released when hydrolysing 20 g/l feather comparing to 30 g/l feather.
Effect of (a) feather and enzyme concentration and (b) processing time on released protein. Error bars correspond to standard deviation.
In order to evaluate the effect of treatment time on hydrolysis yield, enzymatic procedure was carried out with the optimized amount of enzyme and feather concentration for different hydrolysis times. The given results in Figure 1(b) show that there is an evident increase in released protein with rising hydrolysis time up to 90 min. In reverse, prolonging incubation time more causes a decline in this response. As the protease catalyzes the hydrolytic cleavage of feather into smaller peptide chains, more soluble proteins are liberated in the reaction mixture. However, after a certain time such as 90 min in this study, the free protease molecules might hydrolyze the released protein in the mixture resulting in a decrease in the amount of this component. As a consequence, the optimized condition was found to be as follow: enzyme concentration—1% (v/v), hydrolysis time—90 min, and feather concentration—20 g/l.
SDS-PAGE electrophoresis
Figure 2 shows the electrophoresis separation patterns of the extracted protein from feather under the optimum conditions. A formulation of high molecular weight standard between 10 and 170 kDa was applied to lane (a) for calibration. As for feather protein in lane (b), protein fractions around 10 kDa can be observed. According to Woodin et al., feather keratin is composed of fairly homogeneous protein, mainly consisted of β-keratin with an average molecular weight of 10 kDa [28].
Electrophoretic separation patterns (SDS-PAGE) of samples; lane (a): MW standard and lane (b): extracted feather protein.
Characterization of treated wool fabrics
Felting shrinkage
Felting shrinkage resistance of different wool fabrics after three washing cycles.
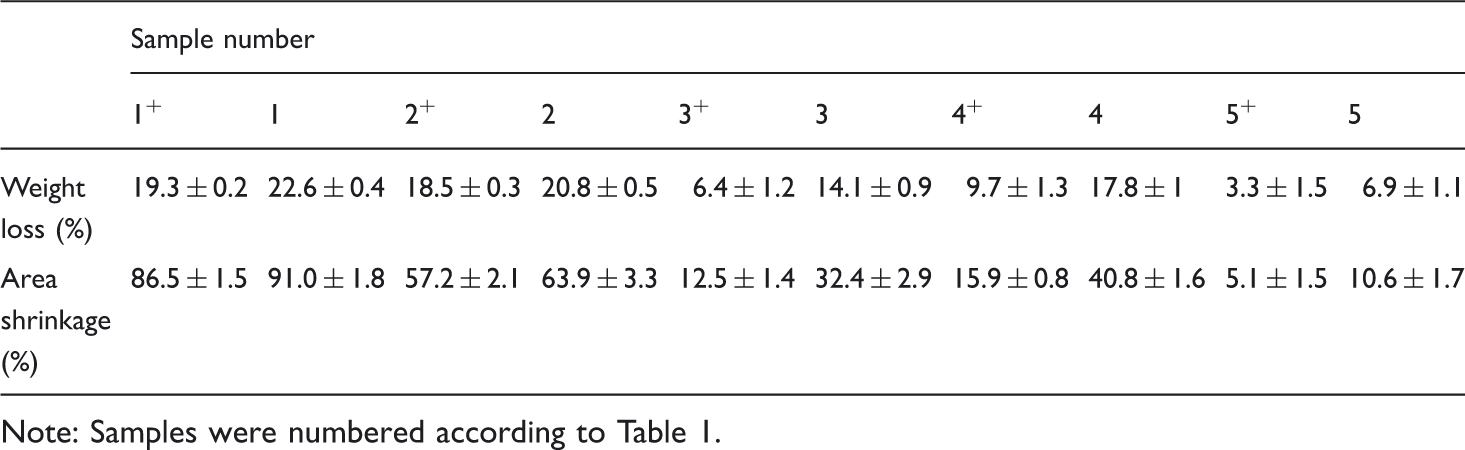
Note: Samples were numbered according to Table 1.
It should be mentioned that 2 h treatment on wool fabrics with additional enzyme would be too long causing excessive fiber damage due to the penetration of the protease into wool fibers; thus, the treatment time was reduced to 30 min in this case (sample 5). As can be seen, the presence of 0.05% enzyme in the treatment solution greatly decreases area shrinkage from 40.8% (sample 4) to 10.6% (sample 5) without cross-linking treatment. This enhancement in shrink resistance of wool fabric could be explained by the partial removal of cuticle scales during the proteolytic reaction.
The cross-linking agent was used to fix the extracted feather protein to the fabrics surface. According to the obtained results, shrink resistance of the samples has been improved further using GDE after three times 5A washing. Indeed, the reported percentages of area shrinkage as well as weight loss of the cross-linked samples decrease with respect to those treaded with extracted protein but not cross-linked. The obtained results reveal that cross-linking is required to fix the protein onto the fiber surface to achieve a dimensionally stable wool fabric after washing. In other words, the extracted feather protein is bound to the wool fabric by permanent cross-links which are fast to washing. Control samples show that the applied cross-linking agent has minimal effect on felting shrinkage of samples that have not been treated with extracted protein (samples 1 and 2).
Tensile strength and bending length
The results of tensile strength and bending length of the samples are given in Figure 3 implying that cross-linking agent increases both the strength and bending length of all samples whether they were treated by feather protein (samples 3, 4, and 5) or not (samples 1 and 2). The decrease in flexibility of wool fabrics can be related to forming cross-links between wool polypeptide chains. In addition, curing treatment at 140℃ can increase the bending length further. According to the obtained results, it can be concluded that the applied process (surface modification of wool with feather protein) did not affect tensile strength of wool fabrics significantly except for samples treated with additional enzyme (sample 5) which led to a noticeable decrease in the fabric strength.
(a) Tensile strength and (b) bending length of different wool fabrics. Error bars correspond to standard deviation.
Alkaline solubility
The damage in the wool fabrics could be estimated by alkaline solubility. The effect of the modification treatment on alkaline solubility of wool fabrics is depicted in Figure 4. The alkaline solubility of treated samples decreases in comparison with the control sample, which can be related to the attachment of feather protein on wool surface. Besides, application of cross-linking agent results in a significant decline in the solubility due to the crosslinks formed in the fabric [29]. This hypothesis is in harmony with the results of anti-felting properties of the cross-linked fabrics after washing.
Alkaline solubility of different samples. Error bars correspond to standard deviation.
Surface analysis
The treated samples were subjected to SEM analysis to investigate whether any morphological changes occurred during the treatment, which would lead to the changes in physical properties detected above. The surface of the untreated wool fiber in Figure 5(a) consists of a fine network of small, overlapping cuticle scales which are responsible for felting of wool during aqueous washing processes.
SEM micrographs of wool fibers: (a) untreated, (b) and (c) 120 min treatment with feather protein followed by cross-linking and cure, and (d) 30 min treatment with feather protein in the presence of 0.05% enzyme followed by cross-linking and cure.
For treated fabric in Figure 5(b) and (c), the ridges at the base of the projecting scales have been filled in with feather protein and smoothed to prevent the scales from interlocking which results in a more dimensionally stable wool fiber. These micrographs validate the obtained results of felting shrinkage test. The images show that wool fibers are intact and are not severely damaged by the applied treatment. However, for the sample treated with extracted feather protein in the presence of 0.05% Savinase (Figure 5d), fiber degradation and fibrillation have occurred which confirms the significant decrease in tensile strength.
FTIR analysis
The FTIR spectra of wool fabric treated with feather protein followed by cross-linking with GDE and then cured (sample 3+) were compared with the untreated sample (sample 1) in Figure 6 indicating no significant changes in the chemical structure of fibers after the treatment. The spectra show characteristic absorption bands assigned mainly to the peptide bonds (CONH). The broad absorption band region from 3500 to 3200 cm−1 can be attributed to the stretching vibration of N–H and O–H bonds. The bands in the 3000–2800 cm−1 region are related to C–H stretching modes. The vibrations in the peptide bonds originate bands known as amide I, II, and III. The amide I band is connected mainly with the C = O stretching vibration and it occurs in the range of 1700–1600 cm−1, while the amide II, which falls in the range of 1580–1480 cm−1, is related to N–H bending and C–H stretching vibration. The amide III band occurs in the range of 1300–1220 cm−1 and it results from in-phase combination of C–N stretching and N–H in-plane bending [30].
FTIR spectra of raw and treated wool fabrics.
As can be seen in Figure 6, the principal absorption differences are in the amides regions. The underlying area in treated fabric has increased which provides evidence for the presence of feather protein. In addition, the new peak at 1100 cm−1 for treated wool originates from –C–O– stretching vibration in –CH2–O–CH2– of the cross-linking agent, GDE [29].
Conclusion
The present study investigated the feasibility of wool fabric surface modification with extracted feather protein as an environmental friendly approach. For this purpose, feathers were subjected to enzymatic hydrolysis accompanied by a reducing agent with the aim of protein extraction. The effects of different parameters such as enzyme loading, substrate concentration, and hydrolysis time were evaluated in order to optimize the extraction process. It was found that the highest amount of protein could be released by hydrolysing 20 g/l feather with 1% Savinase for 90 min.
Therefore, feather protein with an average molecular weight of 10 kDa, which was extracted under the attained optimum condition, was applied on wool fabrics to achieve shrink-resistant wool. According to the obtained results, there was a significant improvement in shrink resistance of the fabrics treated with feather protein and the cross-linking agent enhanced the anti-felting properties further. Therefore, fixation of the extracted protein to the wool fabric by the applied cross-linking agent led to a high level of shrink resistance which can be maintained after three washing cycles. Besides, tensile strength of treated fabrics was comparable to the raw one. However, the alkaline solubility and flexibility decreased. SEM micrographs showed the presence of the extracted feather protein on the surface of wool fibers. FTIR spectroscopy also confirmed the formation of cross-links between feather protein and wool. As a consequence, feather waste as a cheap well-accessible protein source could be effective for eco-friendly shrink-resist finishing of wool fabrics.
Footnotes
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
