Abstract
Uniform, stable, one-step dyeing of textile fabrics made from a mixture of natural and synthetic fibers using traditional methods remains problematic to this day. As an alternative to traditional methods, a one-step method for dyeing mixed cotton and polyester fabrics with a new natural dye—a complex of collagen and
Introduction
Currently, the most popular items on the textile market are fabrics and products made from mixed synthetic and natural fibers. This is facilitated by a number of advantages of such materials in design, physical, mechanical, and operational properties. In particular, polyester–cotton blended fabrics are universally used in clothing and household textiles due to the combination of the excellent moisture absorption properties of cotton and the dimensional stability of polyester. 1 Unfortunately, the issues of dyeing mixed materials still remain problematic. Due to the differences in the mechanism of interaction of natural and synthetic constituent materials with dye molecules, it is very difficult to select a universal dye. In addition, the dyeing process for polyester and cotton fabrics is complex and consumes a large amount of water. 2 Often, the intensity and stability of color on different fibers turn out to be different, and the uniformity of coloring is disrupted.
Generally, cotton fibers are well dyed with reactive dyes due to its brightness of shade, variety of colors, moisture resistance, and high applicability. However, some problems such as insufficient dye utilization, excessive use of electrolytes, and large volumes of wastewater discharge are always present when using reactive dyes. 3 Disperse dyes are used to dye polyester fibers. 4 For dyeing mixed materials, as a rule, a two-stage method is used: first, cotton fibers are dyed with reactive dyes, and then, polyester fibers are dyed with disperses dyes. 5 At the same time, the synthetic dyes used have a negative impact on the environment and health. 6 Waste valorization is the only solution to reduce the amount of toxic waste released into the environment. 7 Traditional pre-treatment, dyeing, and finishing of textiles have a high environmental impact due to the high consumption of energy, water, and chemicals, as well as the generation of wastewater containing residues of harmful dyes and chemicals. Different wastewater streams can be separated during filtration and during reuse. 8 Considering the environmental impact, environmental issues related to the dyeing industry must be addressed immediately. 9
Nowadays, natural dyes are becoming a trend when used for textile fibers. Environmental sustainability, resource availability, and cost-effectiveness are driving forces in the search for natural sensitive dyes to replace synthetic ones. 10 This is explained by the fact that natural dyes are more environmentally friendly and reduce the demand for synthetic dyes, which make up the bulk of textile dyes. Almost all commercially produced natural fibers and basic synthetic fibers can be dyed and printed using natural dyes.11,12 Researchers propose various natural dyes based on Fuji apple peel, 13 pumpkin peel, 14 chitosan or chitin, carminic acid and purpurin, 15 eucalyptus, mudder, annatto, true indigo and woad, 16 and others.
Preliminary modification of the surface of cotton–polyester fabrics by ultraviolet (UV) irradiation, 4 ultrasonic, plasma, microwave treatment,17–19 and natural polyelectrolytes 5 provides good dyeing quality. Cationic polyelectrolytes improve sorption and fixation of dye on cellulose fibers. 3 Replacing toxic chemicals with less toxic and harmless ones helps to minimize the use of water, chemicals, and wastewater in a textile factory. 20 Harmless reagents include biopolymers, which are characterized by safety, environmental friendliness, biodegradability, and low price with commercial availability. Biopolymers can be involved in various processes of the textile industry, like fiber production, as an alternative to hazardous chemicals used in various textile processing and finishing steps. 21
The presence of metal in the dye molecule makes it easier to apply to the fibers, resulting in a more efficient dyeing process. The dye molecule is usually a monoazo structure containing additional groups, such as hydroxyl, carboxyl, or amino groups, which are capable of forming strong coordination complexes with transition metal ions. Chromium, cobalt, nickel, and copper are commonly used. 22 Their colors cover the entire spectrum, although, as described earlier, they tend to produce dull shades. They can be 1:1 dye:metal complexes or 2:1 complexes and contain mainly one or two azo groups. 23 Metal complex dyes are synthesized by coordinating di- or polyvalent transition metal ions with selective acid dyes. They are mainly applied to wool, silk, and nylon to achieve better wash fastness of dyed fabrics compared with those obtained using acid dye parent. 24
Synthesized chelate complexes
Transition metal ions have good affinity for protein macromolecules, including collagen. In recent decades, collagen biomaterials have received special attention due to their excellent properties, such as low biodegradability, biocompatibility, hydrophilicity, immunogenicity, and ease of processing. 29 Collagen is one of the most common natural proteins and makes up the bulk of animal skins. A review devoted to the production of collagen from animal skins and its physicochemical properties was made by Tang et al. 30 Isolation and study of the properties of collagen show its high reactivity in the reactions of oxidation, 31 hydrolysis, 32 hardening, 33 and grafting. 34 Collagen is known to be used to produce a polymer membrane, 35 to form fibers, 36 and in tissue engineering 37 as a framework for nanoparticles. 38 Porous carbon fibers with diameters ranging from 30 to 50 μm have been produced by wet spinning of collagen materials. 39 Partial hydrolysis of collagen leads to the formation of gelatin. 40 In the textile industry, a solution of hydrolyzed collagen is also used for sizing 41 and fire-retardant treatment of cotton yarn before weaving 42 and as part of a composition to impart fire resistance to the material. 43 The good compatibility of collagen with cotton 41 allowed the use of hydrolyzed collagen in dyeing cotton and leather. 44 Good results were obtained when dyeing leather, where the synergistic effect of hydrolyzed collagen was evident. Both cotton and leather behave differently toward hydrolyzed collagen at different pH values. The color fastness of dyed fabric samples is noticeably affected by the pH of the dye bath solution. 45
The interaction between metal ions and peptides has attracted interest due to the important role of metal ions in many biological processes and potential applications in various fields. 46 Metal ions perform an important function in the action of antibiotics and are involved in specific interactions with proteins and nucleic acids.47,48 Metal ions participate in several biological processes, forming complexes with protein substances.49,50 Collagen membranes in combination with gold and nucleic acid complexes serve as materials for corneal restoration. 51 The formation of protein complexes with metal ions promotes their crystallization, and advances in macromolecular crystallization have rapidly multiplied in recent years with the availability of practical, easy-to-use kits. 52
Thus, complex compounds of proteins, including collagen, are becoming very important in various fields, especially in medicine. Meanwhile, while studying them as a matrix for medicines, sorbents of various ions, catalysts, or inhibitors of chemical processes, not enough attention is paid to the possibility of using them as dyes for textile materials. Time-stable complexes of collagen with transition metal ions are environmentally friendly unique colored substances. The purpose of this work is to determine the possibility of using collagen complexes with chromium (III) and copper (II) ions as a dye for textile materials. Of particular, relevance is finding a convenient, one-step method for dyeing a mixture of cotton and polyester fibers. To achieve the goal, complexes of hydrolyzed collagen with
Experimental part
Materials
Waste cattle skins cleaned of wool were provided by a tannery in Tashkent (Uzbekistan). A collagen-containing solution was obtained by dissolving crushed skins in a sodium hydroxide solution according to the method described by Rafikov et al. 41 To remove electrolytes and low molecular weight substances, a collagen-containing solution was dialyzed using a dialysis bag made from horse intestine. The time and completeness of dialysis is controlled by the specific electrical conductivity of the solution. The studies used a collagen solution after 24 h of dialysis, the electrical conductivity of which is (35–57)× 10−4 ohm−1 cm−1, pH = 6.8–7.0, and the mass fraction of hydrolyzed collagen in it is 9–10%.
For the synthesis of metal complexes, crystalline hydrates of salts
A nonionic emulsifier was used as a surface-active substance (SAS)—fatty alcohol ethoxylate—which are alkyl polyglycol ethers obtained from lauryl-myristyl fatty alcohols (C12–C14), mixed with varying numbers of repeating ethoxy groups.
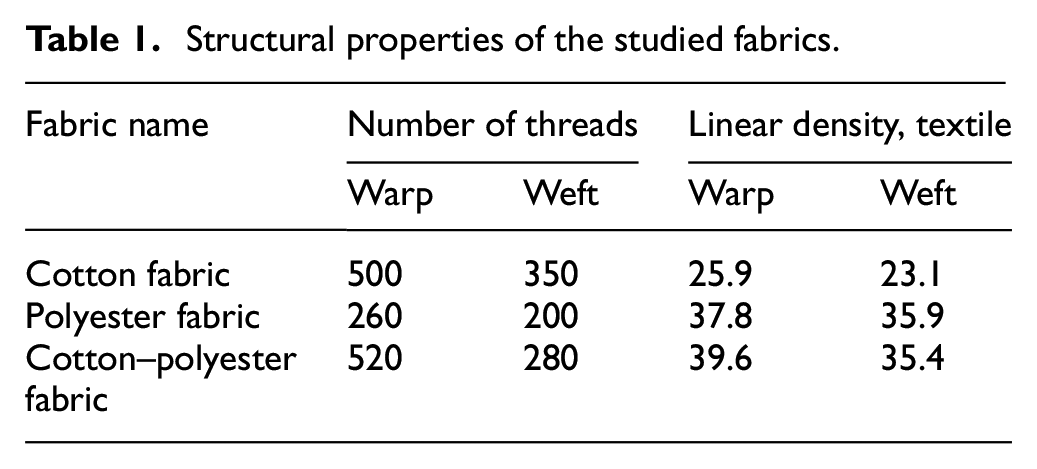
Three types of fabrics were selected for research: cotton fabric with a surface density of 230 ± 5 g/m2, polyester fabric with a surface density of 160 ± 5 g/m2, and mixed fabric made of cotton (20%) and polyester (80%) with a surface density of 250 ± 5 g/m2. The structural properties of the fabrics are given in Table 1.
Structural properties of the studied fabrics.
Methodology for the Synthesis of Collagen Complexes
To obtain collagen coordination complexes, a 10% solution of hydrolyzed collagen and 20% solutions of crystalline hydrates
Method of Dyeing Fabrics with Complex Dye
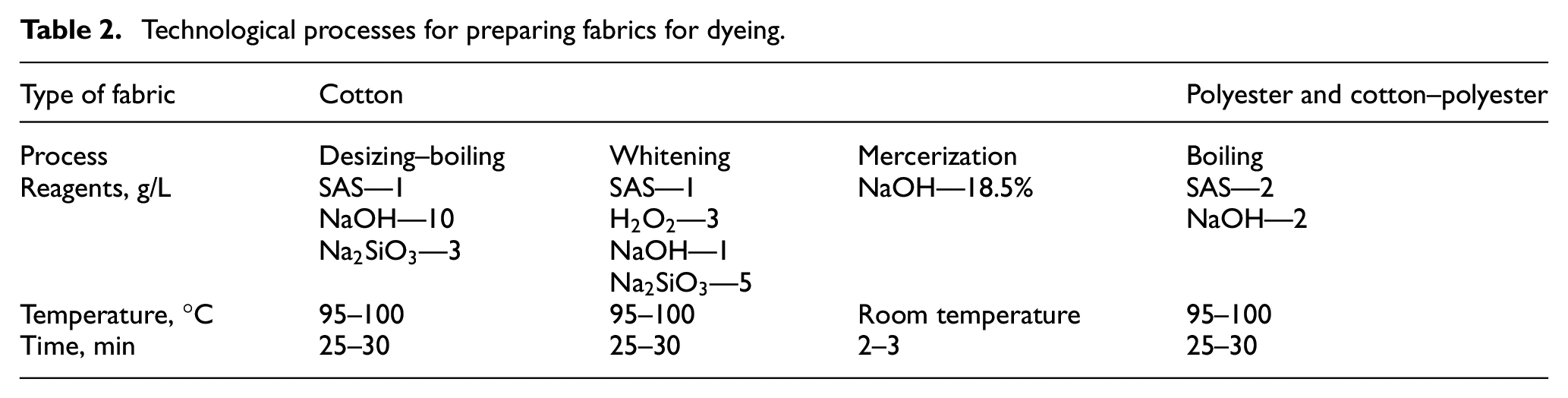
To remove sizing and fatty-wax substances from the surface of cotton yarn, improve the wettability and sorption capacity of the fibers, desizing, boiling, and mercerization of the raw fabric are carried out. For polyester and mixed fabrics, boiling is sufficient to remove fatty-waxy substances and improve the wettability and sorption capacity of the fibers. The fabric is prepared for dyeing by boiling and bleaching in a solution containing reagents at a bath modulus of 30:1 (Table 2). Then, the fabric was thoroughly washed in running water and dried to a constant weight.
Technological processes for preparing fabrics for dyeing.
Fabric dyeing was carried out at room temperature, and the dye bath module was 5:1. Dyeing technology: impregnation in a dye solution contains 180 ± 2 g/L of collagen complex for 2–3 min, squeezing, second impregnation, squeezing, drying at room temperature, heat setting, washing and drying (Figure 1).
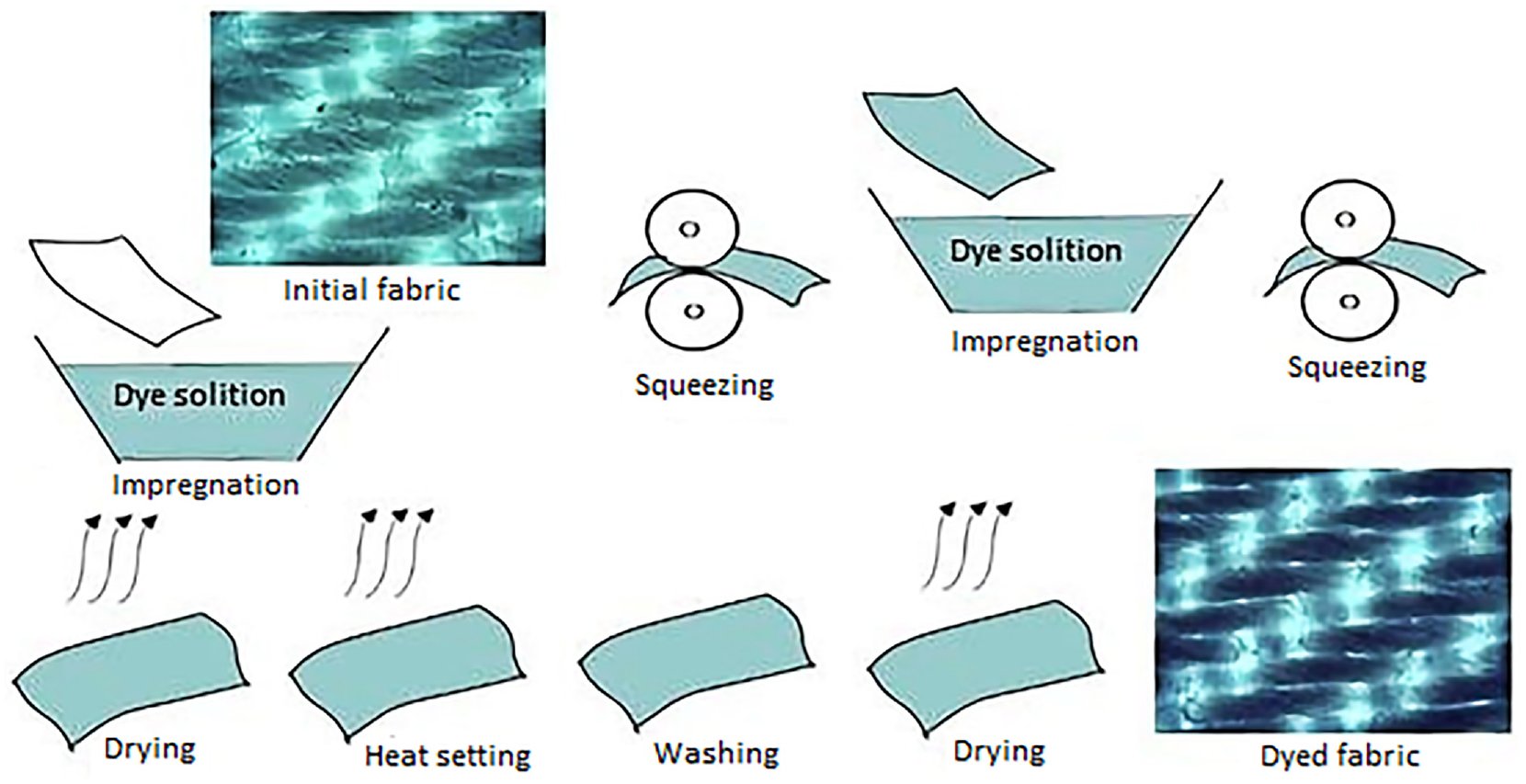
Technology of fabric dyeing with collagen metal complex.
After heat setting, the impregnated samples were washed first in cold water at room temperature for 5–10 min, then in warm water at a temperature of 50–60°C for 5–10 min, then in a soap–soda solution at a temperature of 70–80°C for 15–20 min, and finally in running water.
The mass fraction of the fixed dye (

where
Each experiment was repeated at least three times, and the average value of these experiments was taken as the final result.
Characterizations
The color coordinates of the dyed samples after heat fixation and after washes were determined using the X-Rite iMatch for Color iControl (version 9.7.50). The selection of samples, their preparation, and testing were carried out according to the GOST R ISO 105-J01-99 “Textile materials. Determination of color fastness.” Using the color coordinates, the difference between the color of the dyed fabric before and after washing, expressed in
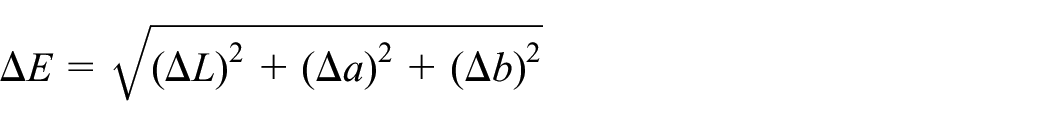
where
The structure of the samples was determined using spectroscopy with Fourier transform infrared (FT-IR) Nicolet IN10 Company Thermo Fisher Scientific (USA) in the scanning range of 500–4000 cm−1.
The study of surface morphology and elemental analysis of samples was carried out using a scanning electron microscope “JSM-IT200LA InTouchScope” (Japan) with the application of SEM-EDS energy dispersive analysis.
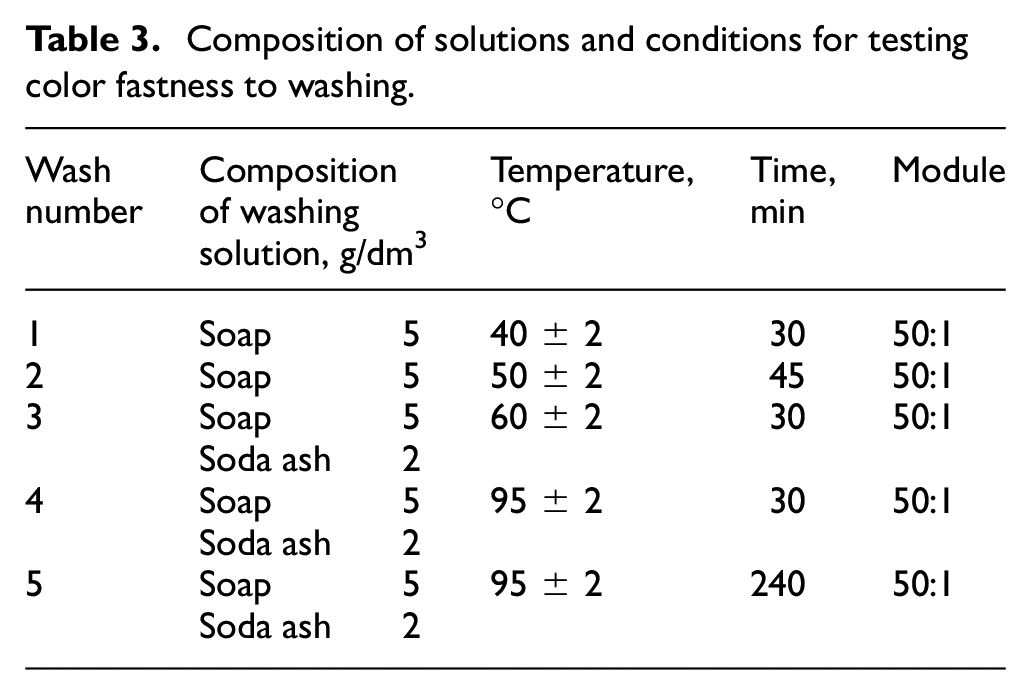
Tests of color fastness to washing under conditions from soft to hard were carried out according to the GOST 9733.4-83 standard. The method is based on mechanical mixing of the working sample along with adjacent tissues in a washing solution at a certain temperature and time. The composition of washing solutions and test conditions are indicated in Table 3.
Composition of solutions and conditions for testing color fastness to washing.
Testing the light fastness of dyed fabric samples was carried out using a color fastness tester for weather (XENON ARC TESTER) equipment in accordance with GOST R ISO 105-B04-2010 “Textile materials. Tests for color fastness.” Tissue samples were exposed to xenon arc lamp light and moisture for 20 h and graded against the blue standard.
Results and Discussion
Mass Ratio of Collagen and Salt in the Complex
Dyeing of cotton, polyester, and cotton–polyester fabrics was carried out in a solution of a complex dye synthesized at three mass ratios of collagen and metal salt: 25:75, 50:50, and 75:50. For each case, the mass fraction of absorbed dye during the dyeing process, fixed dye after heat fixation and after wet treatments were determined (Table 4).
Mass of absorbed, fixed dye on fabric samples.
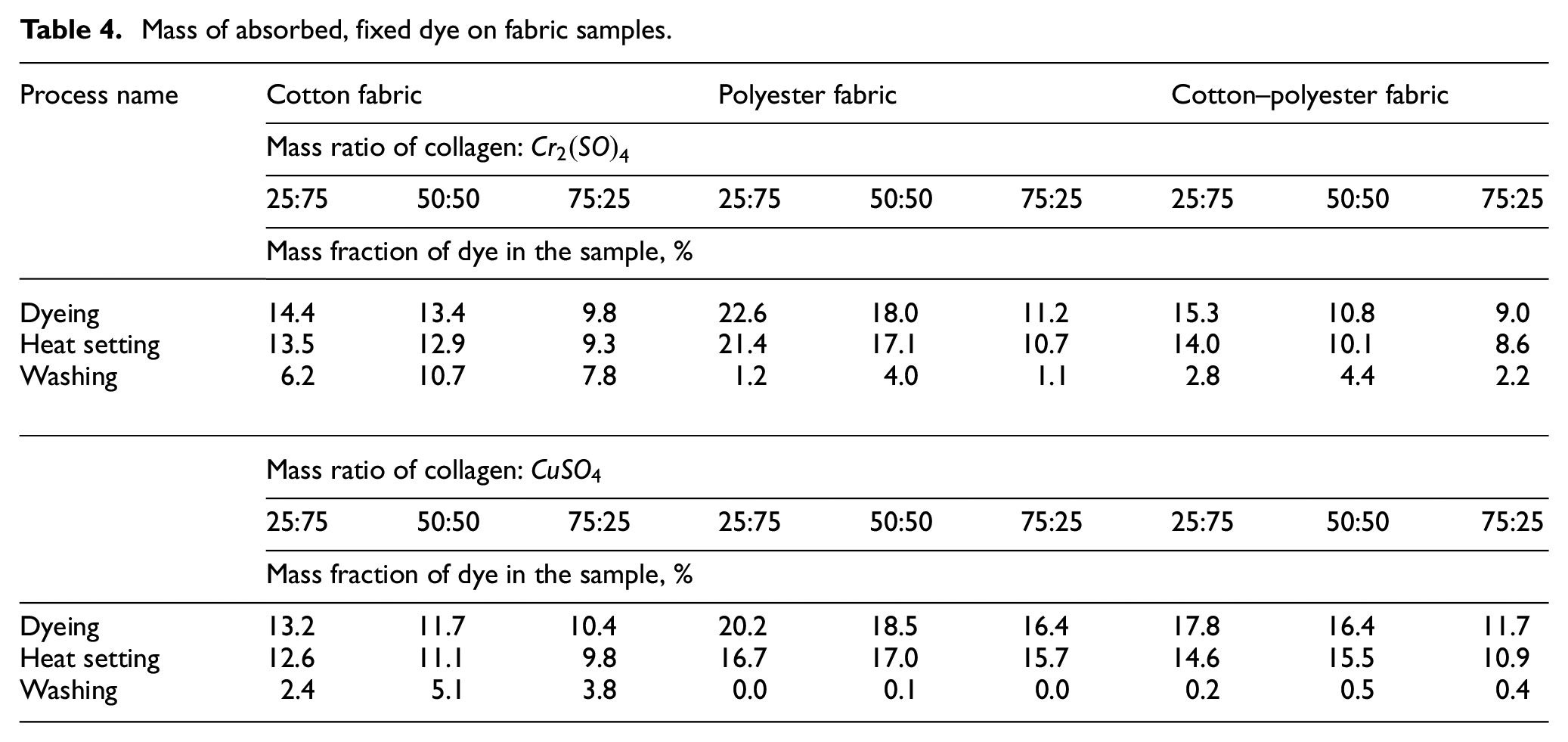
The data in Table 2 convincingly show the advantages and disadvantages of each type and composition of complex dyes. The polyester fabric absorbs both types of dyes very well, while cotton fabric absorbs to a lesser extent. During thermal fixation processes, a slight decrease in the mass of all samples was detected, apparently due to the desorption of bound water and the release of low molecular weight polycondensation products. The most interesting results were shown by the washing process. After all washes, from 17% to 53% of the collagen–
The following studies to determine optimal dyeing regimes were carried out using complexes obtained at a mass ratio of 50:50.
Heat Setting and pH of the Dye Solution
A very important technological parameter of dyeing, especially for mixed materials, is the temperature at which the dye is fixed on the fabric fibers. Apparently, the temperature of short-term heat exposure on the sample should be as close as possible to the melting point of polyester (250–255°C). In this case, fixation of the dye is ensured by the joint melting of the polyester and collagen of the dye. Unfortunately, at temperatures above 230°C, even with short-term exposure, the appearance of the sample noticeably changed, which indicates partial destruction and charring of cotton and collagen fibers. Therefore, the effect of temperature on the dye fixation strength was studied in the range of 120–230°C (Figures 2 and 3). In this case, a solution of a complex dye was studied without adjusting the pH of the medium.
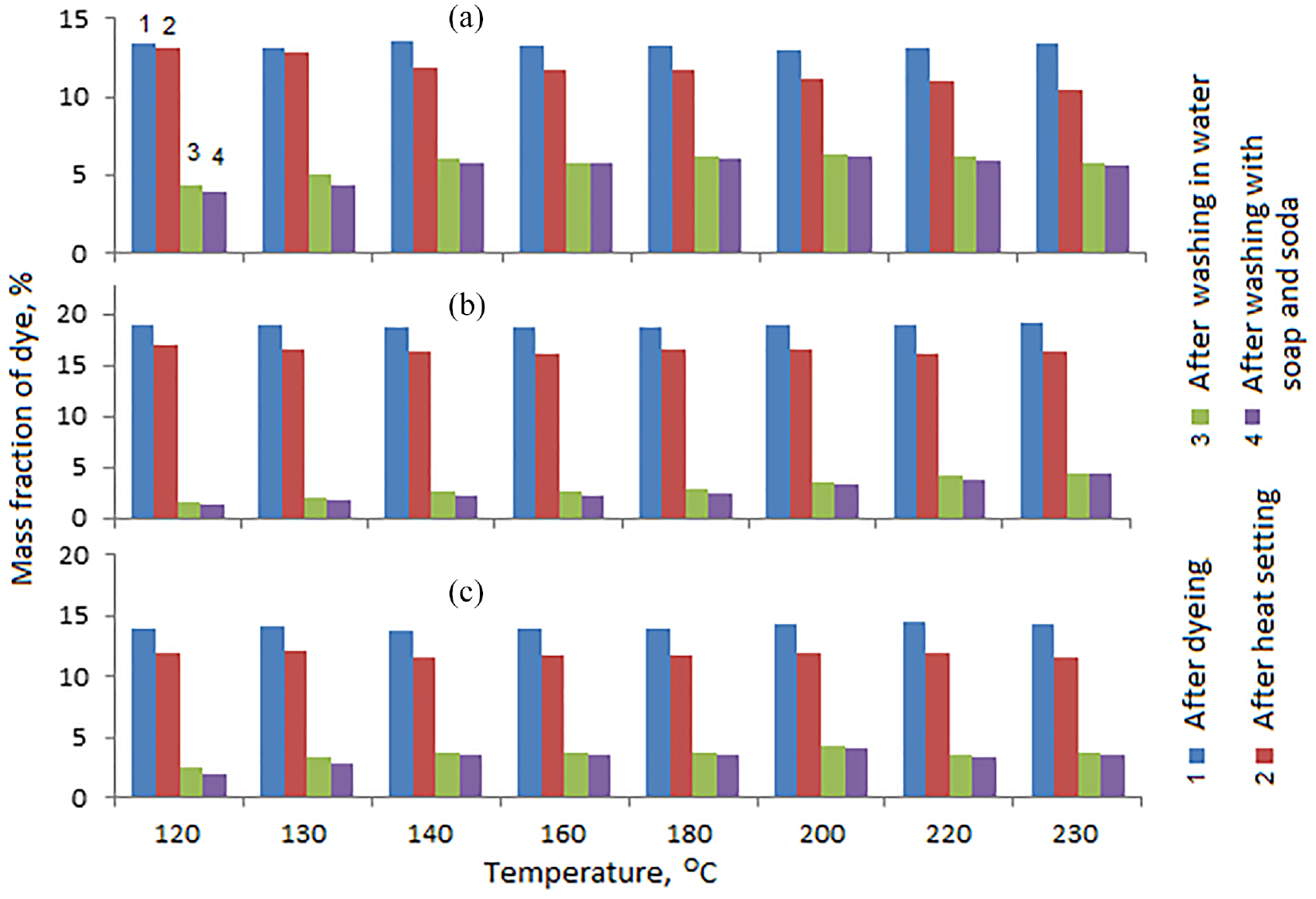
Dependence of the mass fraction of collagen–
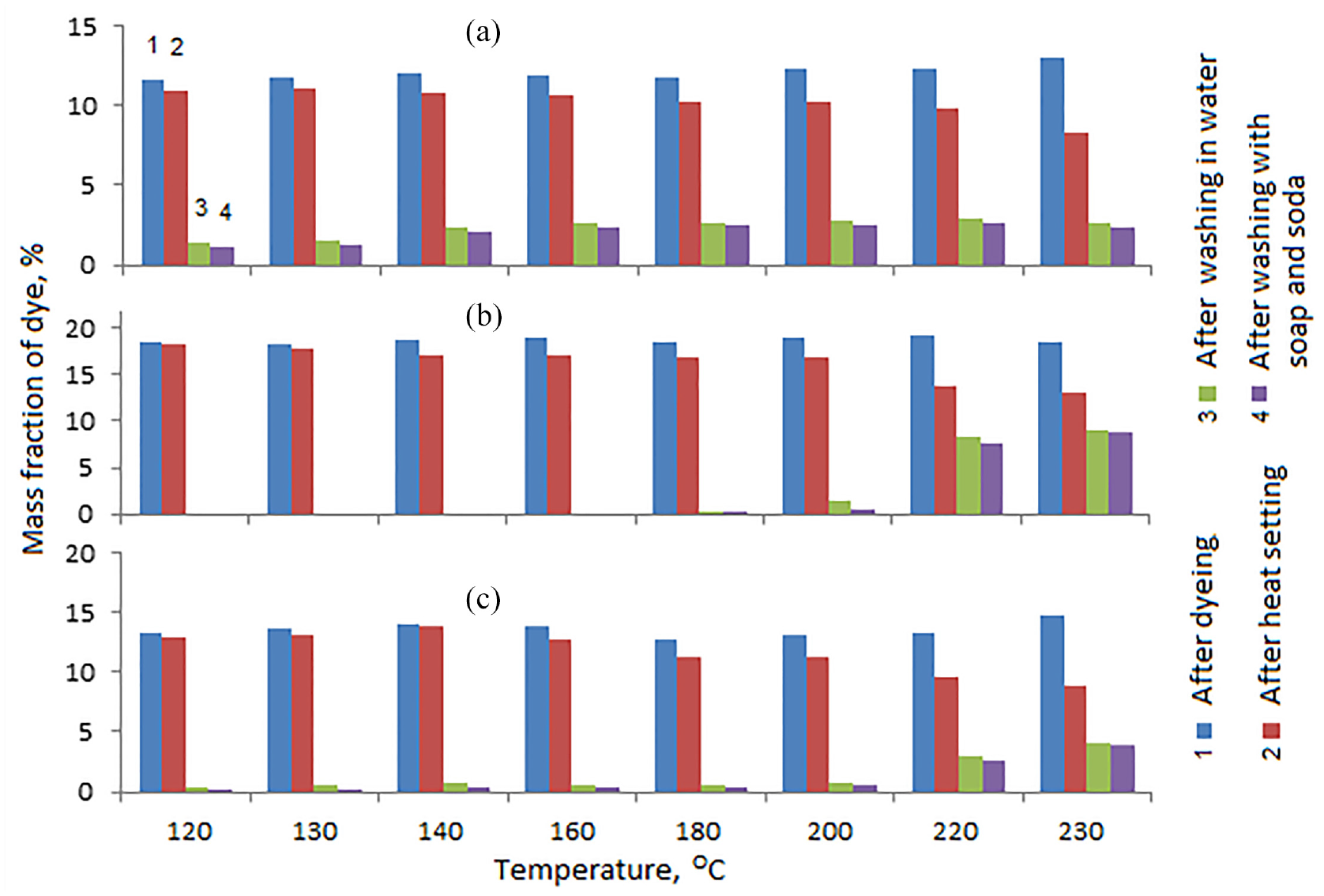
Dependence of the mass fraction of collagen–
According to Figure 2, it is clear that for all tissues, the fixation of the complex collagen–
The collagen–
As is known, when protein macromolecules interact with trivalent chromium ion, chromium passes into the hexavalent state. The data obtained indicate that when collagen interacts with Cr3+, the formation of a donor–acceptor bond predominates, and when interacting with
The following studies were carried out to determine the effect of pH on the degree of fixation of dyes in tissues (Figures 4 and 5). In these studies, the samples were heat-fixed at a temperature of 140°C.
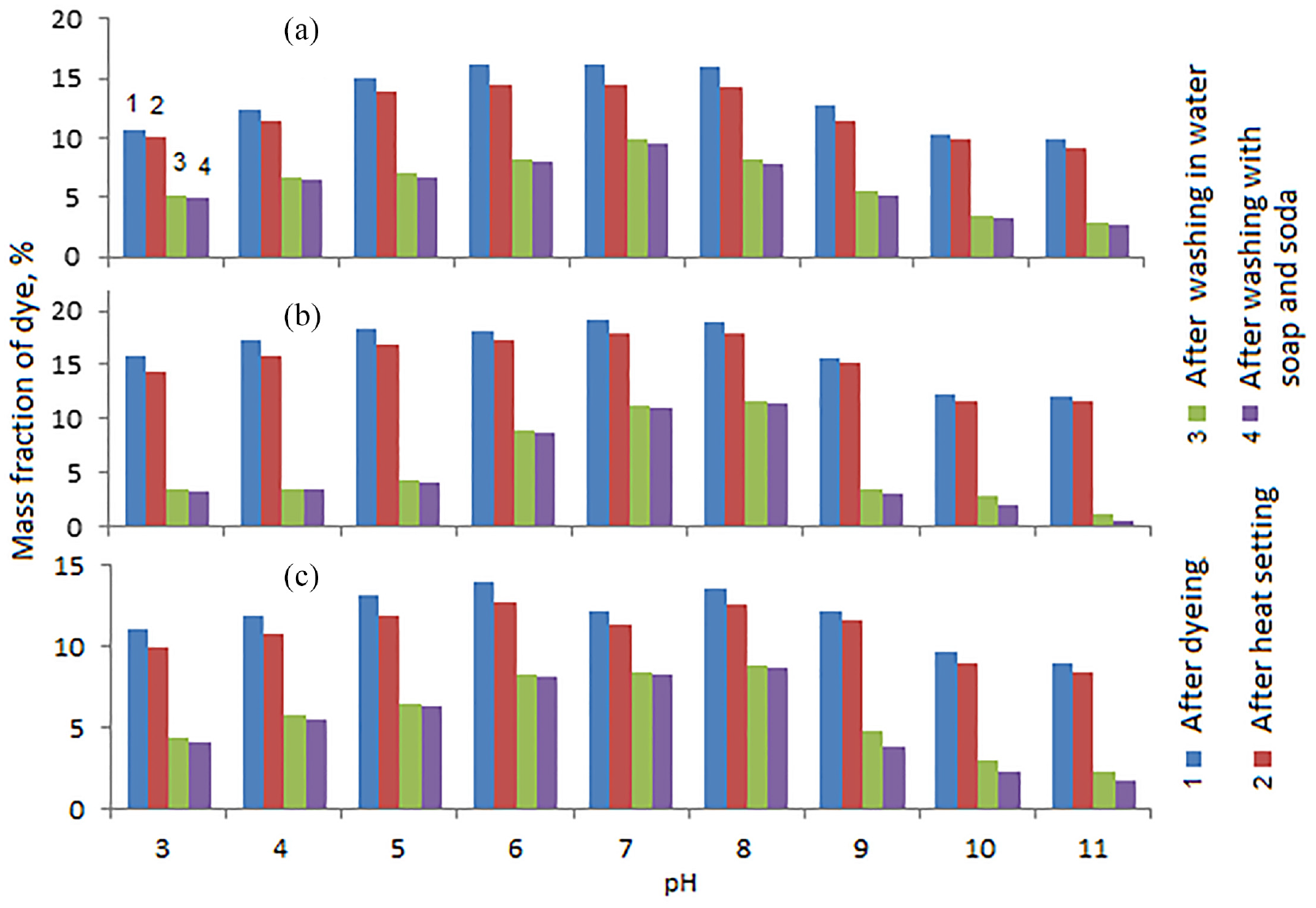
Dependence of the mass fraction of collagen–
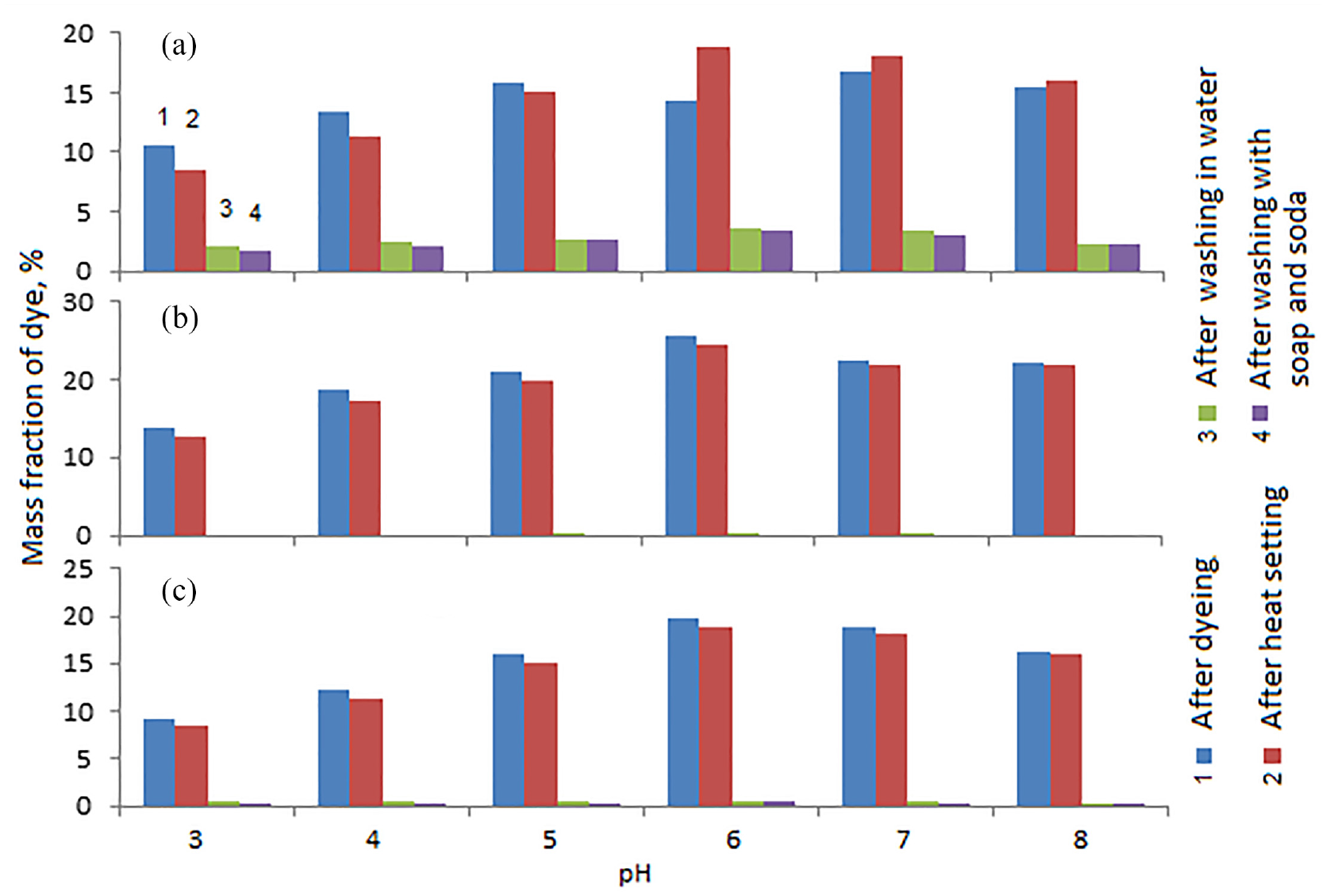
Dependence of the mass fraction of collagen–
It turned out that the solution environment has the most significant influence on the degree of fixation of the complex dye on the fibers of the material. High rates of absorption and fixation after thermal exposure, washing in cold and hot water, in a soap–soda solution of the collagen–
Unsatisfactory results were obtained when dyeing fabrics with the collagen–
To carry out further studies to determine the properties of the samples, cotton, polyester, and cotton–polyester fabrics were selected and dyed with a complex dye at a mass ratio of collagen:
Color Coordinates
The results of determining the color coordinates of dyed fabrics are shown in Figure 6 and Table 5.
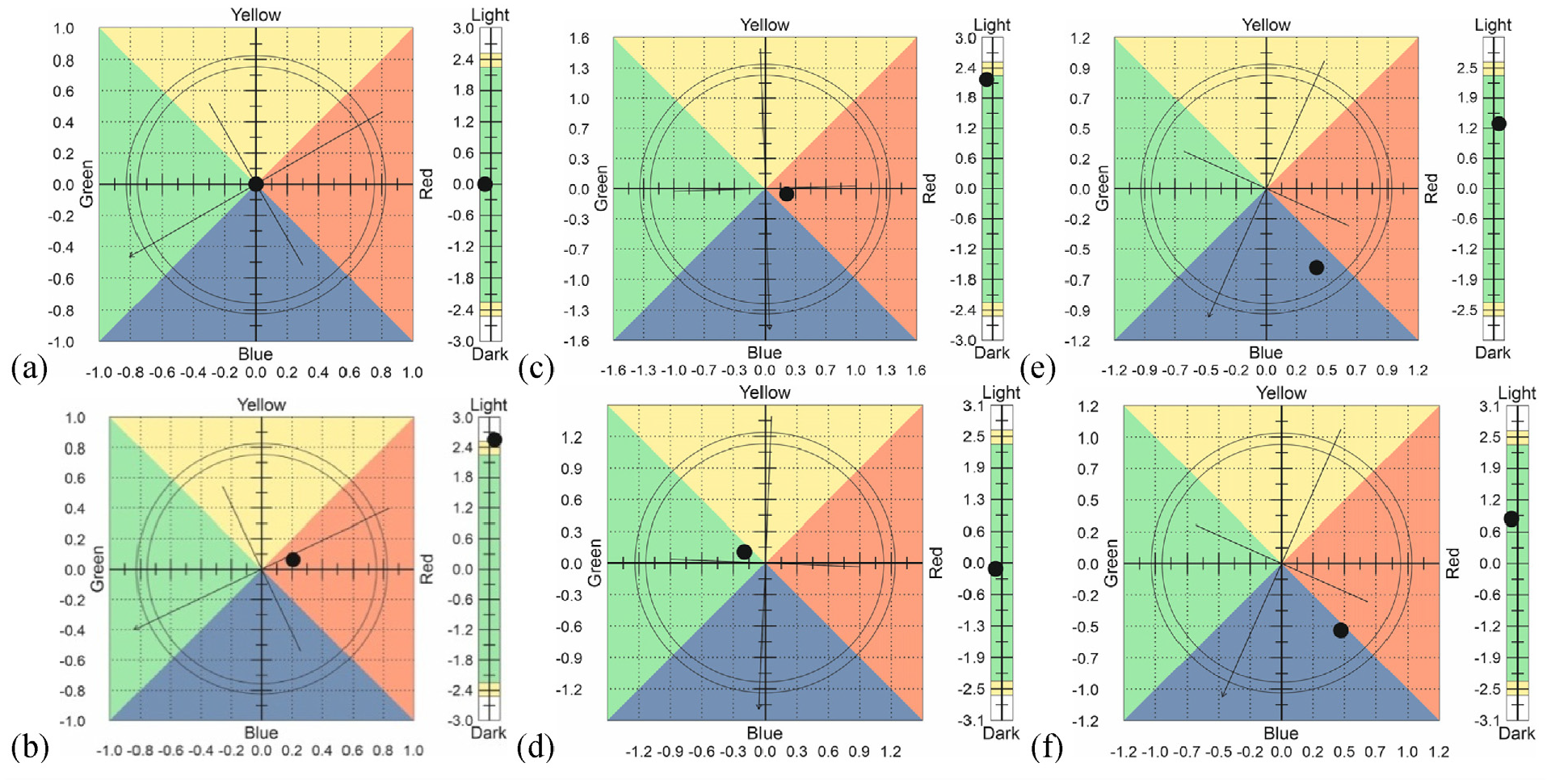
Color coordinates of cotton (a and b), polyester (c and d), cotton–polyester fabrics (e and f), after heat setting (a, c, and e), and after washing (b, d, and f).
Color coordinates values for painted samples, numerator—after heat fixation—and denominator—after washings.
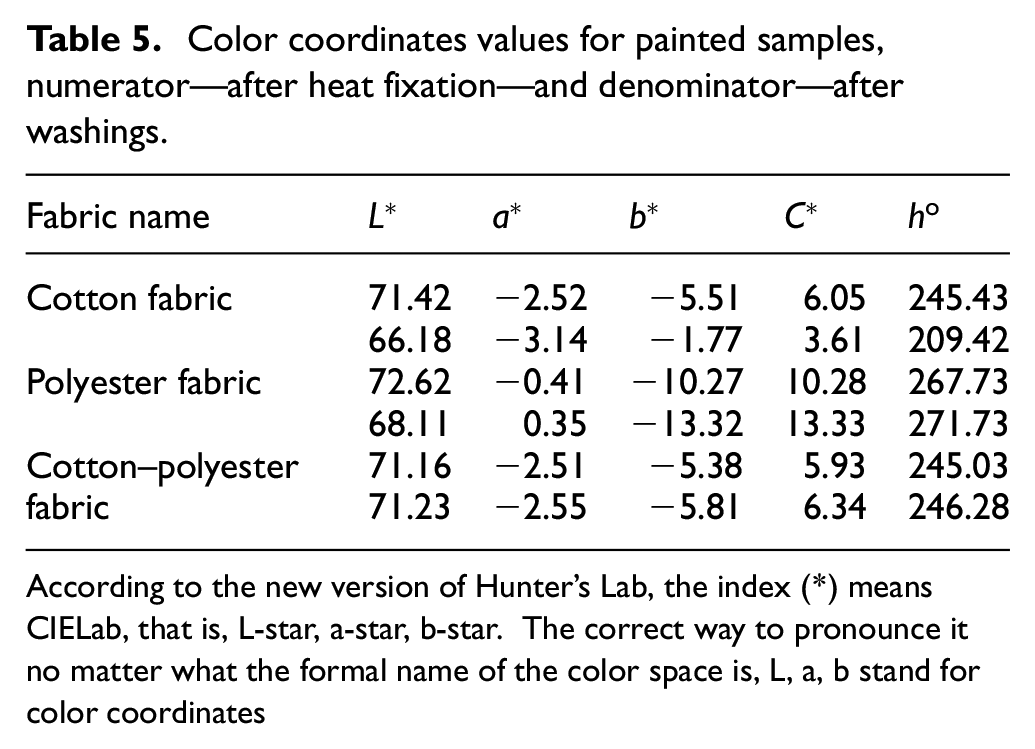
According to the new version of Hunter’s Lab, the index (*) means CIELab, that is, L-star, a-star, b-star. The correct way to pronounce it no matter what the formal name of the color space is, L, a, b stand for color coordinates
In appearance, all three dyed fabrics are approximately the same gray-blue color. As tests have shown, the color coordinates of the samples differ slightly, which means that different shades of colors appear on different fabrics. Apparently, the color coordinates are influenced by the different nature of dye binding to cotton and polyester fibers. After washings, mixed results were obtained. The values for
The following studies were carried out to clarify, using FT-IR, the possible mechanism of interaction of the collagen–
FT-IR
In the FT-IR of cotton fabric, absorption bands characteristic of stretching (
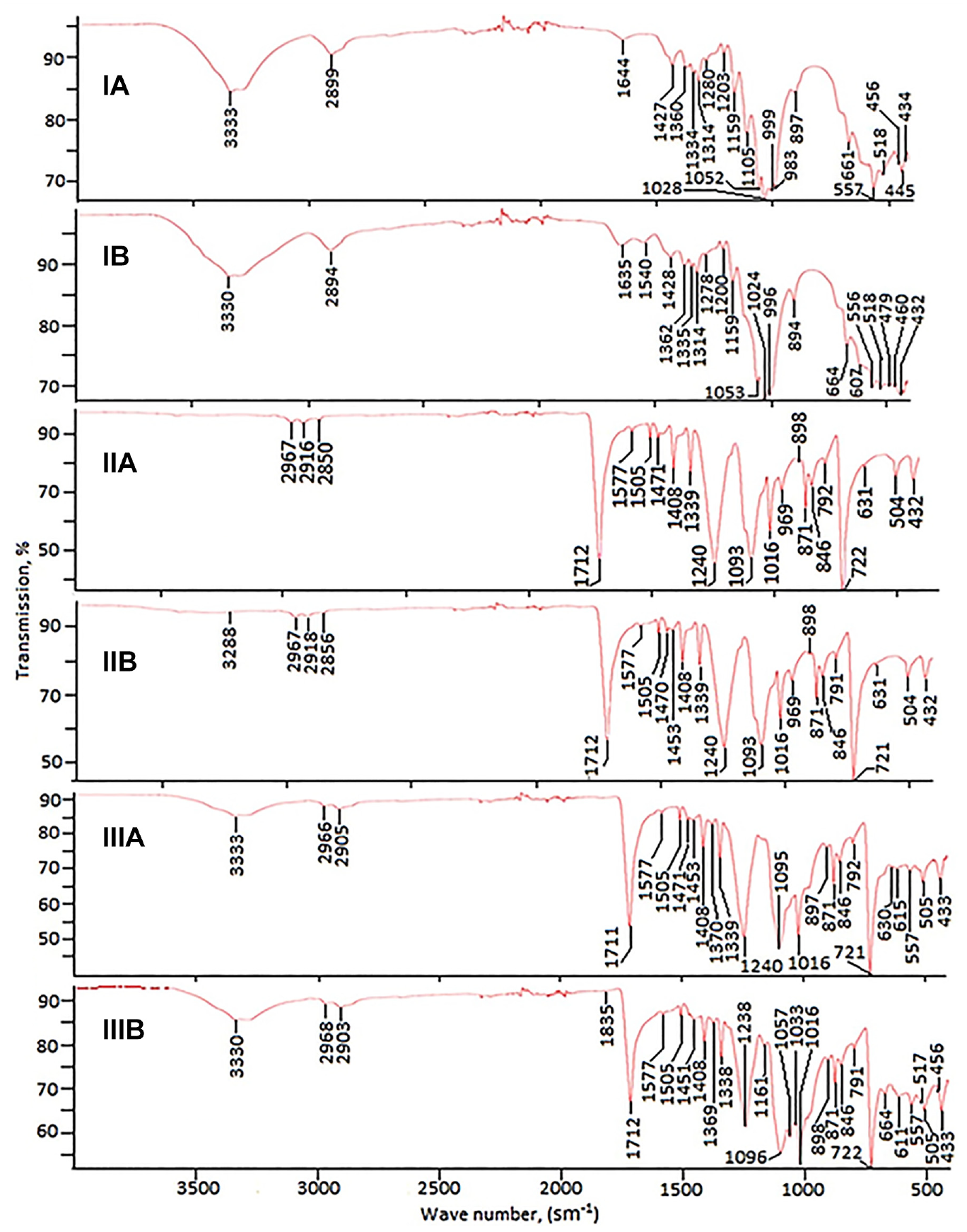
FT-IR of cotton (I), polyester (II), and cotton–polyester (III) fabrics before dyeing (a) and after dyeing (b) with collagen–
In the dyed sample, the main proportion is cotton fabric, so the FT-IR of the collagen metal complex-dyed tissue differs slightly, but still differs from the spectrum of undyed fabric. A new absorption band appears at 1540 cm−1, which relates to the
The following characteristic absorption bands of polyethylene terephthalate were found in FT-IR of polyester fabric: at 2967, 2916, and 2850 cm−1—
In the FT-IR of fabric made from a mixture of cotton and polyester fibers, absorption bands of both cellulose, to a lesser extent, and polyethylene terephthalate, to a greater extent, are observed (Figure 9). Minor changes were detected in the FT-IR of a dyed cotton–polyester fabric sample, including the appearance of subtle absorption bands in the regions of 1161–1033 cm−1 and 664–456 cm−1. Apparently, the collagen metal complex binds to the mixed fabric due to coordination with the cotton component and mutual melting with the polyester component.
Further studies were carried out to determine the uniformity of dye distribution on the surface of the fibers using SEM-EDS.
SEM-EDS
As can be seen from Figures 8–10, dyed fabrics contain mainly 5–6 elements: carbon, oxygen, nitrogen, chromium, sulfur, and sodium (as is known, hydrogen is not recorded in SEM-EDS spectra). Carbon and oxygen are found in cotton, polyester, and collagen; nitrogen and oxygen—in collagen; chromium, oxygen, and sulfur—in chromium (III) sulfate; and sodium—in alkali and table salt, which were used in the processing of raw leather.
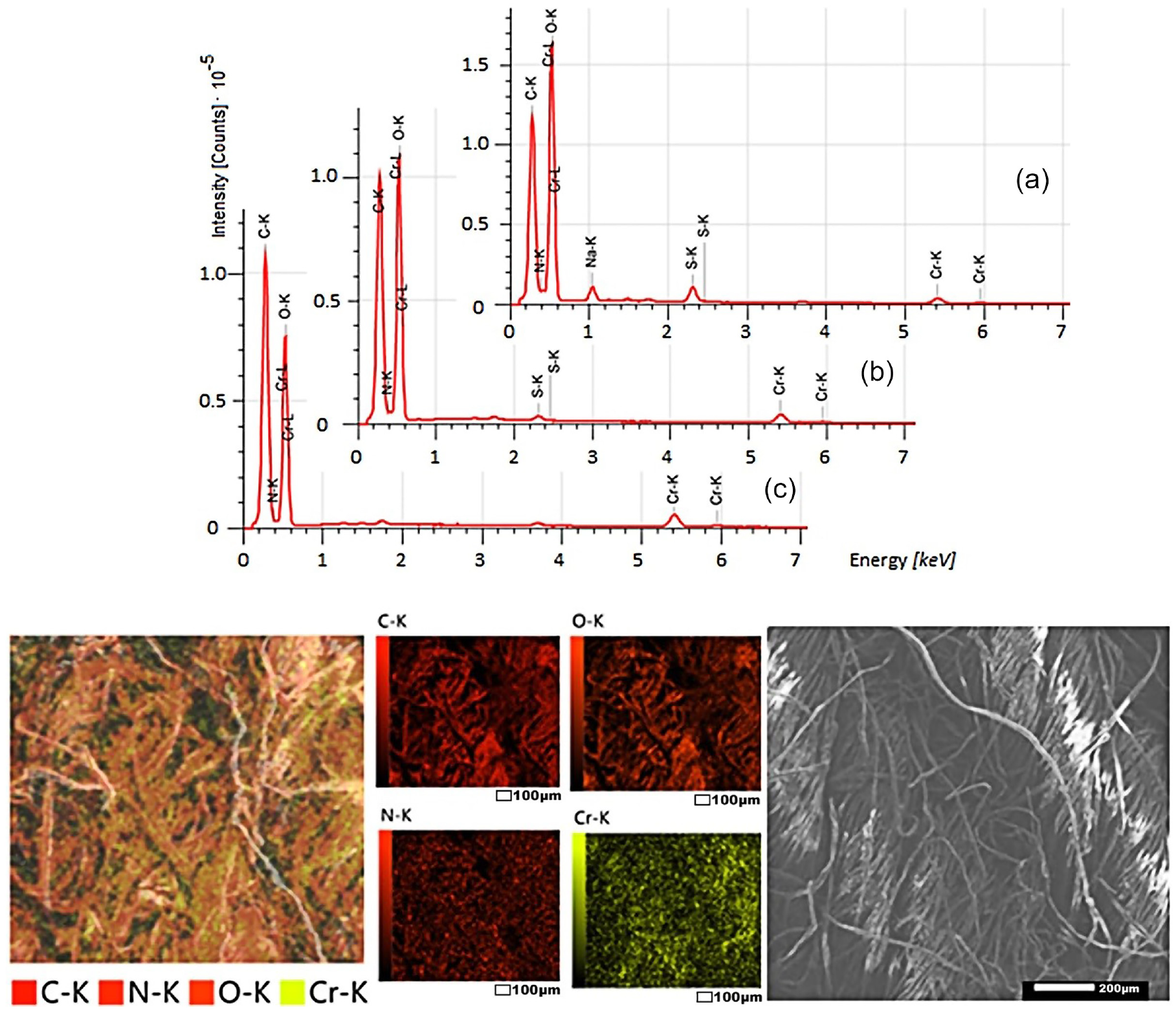
SEM-EDS: elemental composition of the surface of cotton fabric dyed with the collagen–
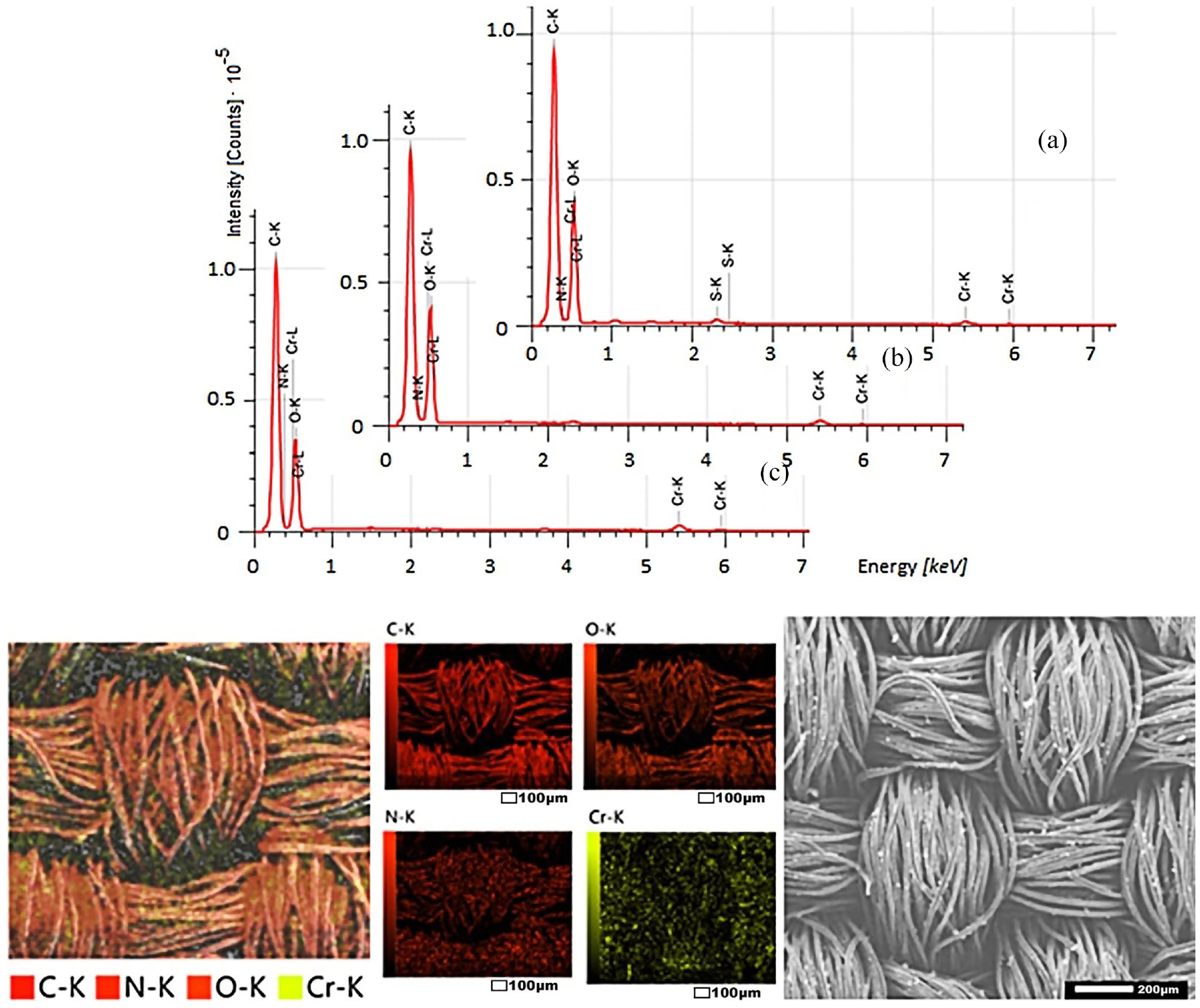
SEM-EDS: elemental composition of the surface of polyester fabric dyed with the collagen–
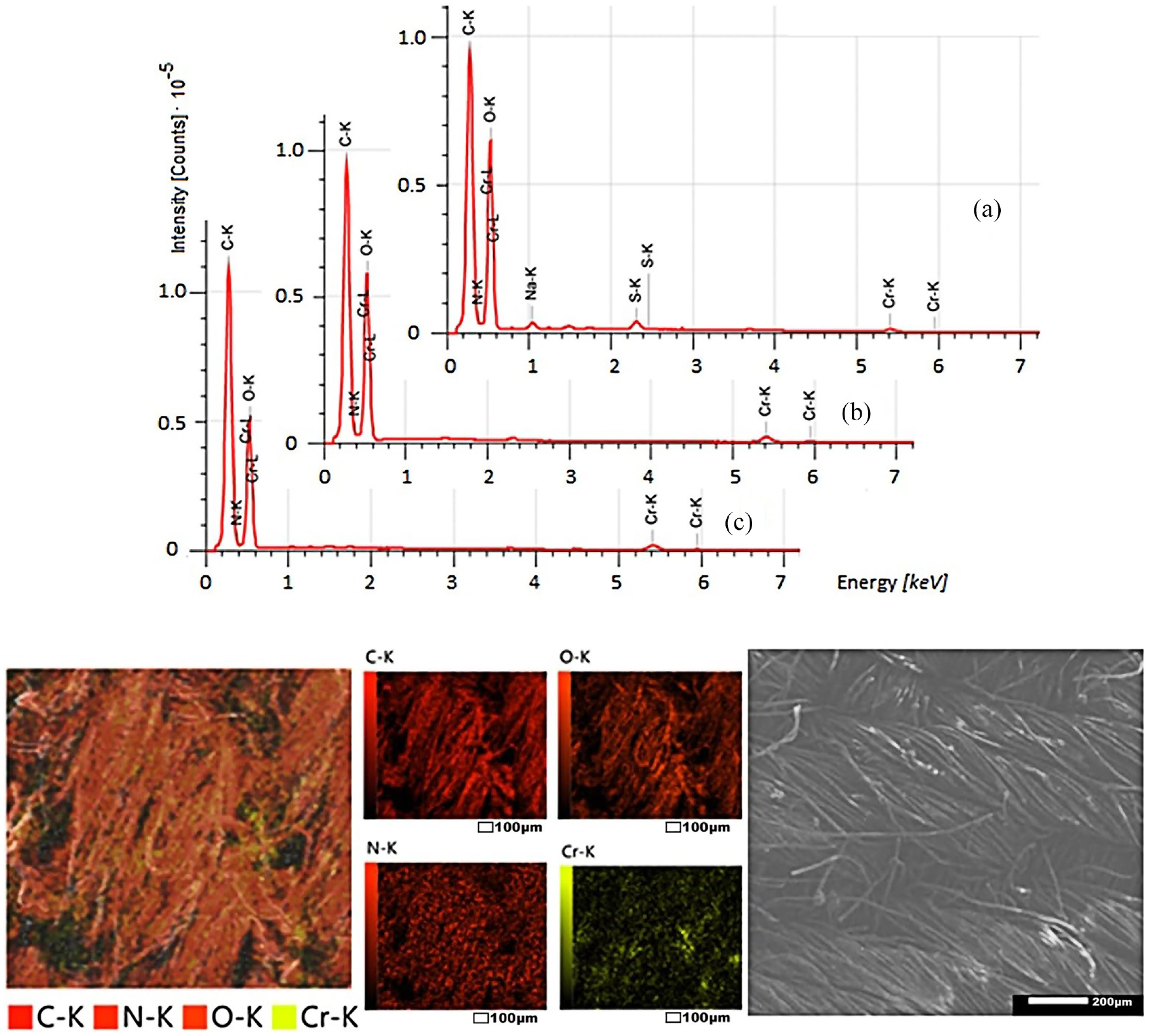
SEM-EDS: elemental composition of the surface of cotton–polyester fabric dyed with the collagen–
After washing dyed samples, the relative amounts of sodium, sulfur, and oxygen are reduced by removing by-products and unbound salt. After all the washings, only four elements remain: carbon, oxygen, nitrogen, and chromium. Most importantly, the nitrogen and chromium of the complex dye are evenly distributed in the pores of the fibers of the materials.
The surface morphologies of different fabric samples are noticeably different. The fibers of cotton and cotton–polyester fabrics look monotonous; the presence of other substances is not visible. On the surface of polyester fibers, the presence of other substances was detected in the form of small dots several microns in size. Apparently, the dye binds to cellulose through coordination bonds, forming a common chemical compound. The formation of coordination bonds between the polyester and the collagen metal complex is possible, but in this case, the complex dye is retained in the micropores of the fibers due to joint melting during heat treatment.
Thus, SEM-EDS studies confirm the results of studies on the influence of processing temperature on the degree of dye fixation on fabrics and FT-IR.
Wash Fastness and Light Fastness
The washing fastness of dyed fabric samples was assessed using a gray standard using a 5-point system (Table 6). Three ratings are given: the first shows the color change of the dyed material itself, the second shows the degree of dye transfer to an identical undyed material, and the third shows the transfer of dye to another undyed material. The light fastness of the dyed fabric was also assessed using a 5-point system (Table 6).
Wash fastness and light fastness of fabrics dyed with collagen–

The data show that cotton–polyester blend fabric had the most wash fastness and most light fastness. The light fastness of cotton fabric is also of high importance. When washed, the dye from cotton fabric does not transfer to undyed fabrics, but the shade of the color changes slightly. The color fastness to washing and light fastness of polyester fabric turned out to be lower, although the colors do not transfer to undyed fabrics when washed. The obtained result confirms a fairly strong binding of the complex dye to fabric made from a mixture of fibers: metal ions with cotton fibers due to ionic and coordination bonds and collagen with polyester fibers due to mutual melting.
Conclusion
Collagen metal complexes with transition metal ions can be a worthy alternative to traditional dyes in the chemical finishing of cotton, polyester, and especially cotton–polyester blend fabrics. Collagen is obtained from untanned animal hide waste that accumulates in tanneries; a simple way to obtain its complex with transition metal salts is carried out in an aqueous medium. A simple and one-bath method for dyeing fabrics is proposed by dipping into an aqueous solution of a complex dye, followed by heat fixation of the dye on the fibers of the materials. The use of production waste, the availability of reagents, and convenient finishing technology with minimization of energy costs help solve some of the environmental and economic problems of the industry. Decent results were obtained when dyeing fabrics with the collagen–
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Ministry of Higher Education, Science and Innovative of the Republic of Uzbekistan (grant no. IL-4821091581).
Data availability statement
The data that support the findings of this study are available from the corresponding author upon reasonable request.
